Abstract
Human kallikrein 6 (hK6), also known as zyme/protease M/neurosin), is expressed in many normal glandular tissues. The aim of this study was to determine whether hK6 is expressed in salivary gland tissues and salivary gland tumors (both benign and malignant), using an immunohistochemical method. Pleomorphic adenomas (PA), adenoid cystic carcinomas, polymorphous low-grade adenocarcinomas, acinic cell carcinomas, mucoepidermoid carcinomas, and adenocarcinomas not otherwise specified of both minor and major salivary glands were examined. Cells lining duct-like structures and non-duct-like cells were scored. Only in PA of minor salivary gland origin was overall staining higher in duct-like than in non-duct-like cells. In all other tumors exhibiting both types of cells, hK6 staining was similar in both duct-like and non-duct-like cells. Tumors that exhibited non-duct-like cells only also exhibited cytoplasmic staining. Results of this study show that salivary gland tumors express hK6, apparently downregulated in comparison with normal salivary gland tissue, and that this expression is not specific for any of the tumors studied.
H
RT-PCR and other technologies have shown that most, if not all, human kallikreins are expressed in the salivary glands (James et al. 1996; Diamandis and Yousef 2001; Yousef and Diamandis 2001; Petraki et al. 2003). It is thus possible that some members of this family may be valuable biomarkers for differential diagnosis, subtyping, and monitoring of patients with salivary gland carcinomas. Monoclonal and polyclonal antibodies have been developed for some kallikreins and efforts are continuing to complete development and characterization of antibodies for all members of the kallikrein family.
Human kallikrein 6 (hK6), also known as zyme/protease M/neurosin), is expressed in many normal glandular and non-glandular tissues including salivary gland, brain, uterus, heart, thymus, prostate, liver, breast, thyroid, spleen, placenta, trachea, testis, and kidney (Anisowicz et al. 1996; Little et al. 1997; Yousef et al. 1999; Petraki et al. 2001). hK6 was dramatically down-regulated in metastatic breast carcinoma cells in comparison to primary carcinoma cells or normal breast epithelial cells (Anisowicz et al. 1996; Diamandis and Yousef 2001). This is the first report on hK6 expression in salivary gland tumors.
The aim of this study was to determine whether hK6 is expressed in salivary gland tissues and salivary gland tumors (both benign and malignant) and if this expression can be used for differential diagnosis. hK6 expression of cells lining duct-like structures, ductal, non-ductal, and non-duct-like cells was assessed to determine if there were any differences among apparently different cell types.
Materials and Methods
Archival formalin-fixed, paraffin-embedded tumor tissues from the Division of Oral Pathology, Department of Pathology, University of Western Ontario were cut in 5-μm sections and stained using a standard immunoperoxidase technique. Twenty six pleomorphic adenomas (PA), 23 adenoid cystic carcinomas (ACC), 13 polymorphous low grade adenocarcinomas (PLGA), 7 acinic cell carcinomas (ACI), 24 mucoepidermoid carcinomas (MEC), 8 adenocarcinomas not otherwise specified (A, NOS), and 62 normal gland controls were used in the study. Appropriate positive and negative (tissue slides with primary antibody omitted) controls were used.
An hK6 rabbit polycolonal antibody raised against fulllength hK6 produced recombinantly in a mammalian stable cell line system was used at a dilution of 1/2000 (Anisowicz et al. 1996). The recombinant hK6 was produced and purified by HPLC as described previously (Diamandis et al. 2000b). This antibody is highly specific and has no detectable cross-reactivity with other kallikreins on Western blots (Petraki et al. 2001).
Staining procedures included deparaffinization in xylene for 13 min with two changes of xylene at room temperature (RT) followed by transfer through graded alcohols and rehydration. Endogenous peroxidase activity was blocked with fresh 3% H2O2 in methanol for 5 min. Sections were rinsed in PBS for 10 min on a shaker. Antigen retrieval was achieved by immersing the slides in boiling citrate buffer (pH 6.0) for 10 min at high power and 10 min at 50% power in a microwave oven. They were then rinsed in water and PBS for 5 min, blocked in 10% horse serum for 30 min at RT in a humidified chamber, and incubated with the hK6 primary rabbit polyclonal antibody for 1 hr at RT. After two washes in PBS, the biotinylated goat anti-rabbit secondary antibody (1/200 dilution prepared in 10% horse serum; Vector Elite Kit, Vector Laboratories, Burlington, Ontario) was applied for 30 min at RT. After two rinses with PBS, the freshly prepared ABC reagent was applied for 30 min at RT. The enzymatic reaction was developed in a freshly prepared solution of 3,3′-diaminobenzidine tetrahydrochloride (DAB; Sigma - Aldrich, Oakville, Ontario) for 5 min. The sections were then rinsed with water, counterstained with hematoxylin for 3 min, dehydrated, cleared with xylene, and mounted.
A proportion score and intensity score using a well-documented system were used to assess hK6 immunostaining (Allred et al. 1993; Tuck et al. 1998). The proportion score represents the estimated fraction of positively staining normal gland or tumor cells (where 0 = none; 1 < 1/100; 2 = 1/100–1/10; 3 = 1/10–1/3; 4 = 1/3–2/3; 5 = 2/3). For staining intensity, the score is represented by the estimated average staining intensity of positively staining normal gland or tumor cells (where 0 = none; 1 = weak; 2 = intermediate; 3 = strong). Overall amount of positive staining was then expressed as the sum of the proportion and intensity scores (ranges = 0 for negative staining and 2–8 for positive staining). Scores for ductal or duct-like, non-ductal, and non-duct-like elements were added and an average obtained for each entity. In normal salivary gland tissue, ductal and acinar cells were scored separately. In tumor tissue, cells lining both duct-like structures and non-duct-like cells were scored for extent of positive and intensity of staining. For purposes of this study, duct-like cells are regarded as those cells that line the lumens of duct-like structures within tumor tissue, whereas non-duct-like cells are any cells in the tumor tissue that are not obviously lining ducts. For MEC, squamous cells, mucous cells, and intermediate cells were scored. Non-epithelial cells were not scored. Staining was assessed by two separate examiners to achieve consistency by comparison and correlation of assessments to reduce interexaminer variability.
For statistical analyses, Mann-Whitney, Kruskal-Wallis, and Tukey-Kramer Multiple Comparisons Tests were used where appropriate.
Results
hK6 immunoreactivity was localized in the cytoplasm of cells that stained. Staining was present in all tumors studied, in both duct-like and non-duct-like cells (see Table 1). The degree of positive staining and intensity of staining is also shown. MEC are presented separately because of the different cell types seen (squamous, intermediate, and mucous cells) (Table 2). Tumors that exhibited non-duct-like cells also exhibited only cytoplasmic staining.
Staining in ductal cells and non-ductal cells and total scores for immunostaining of salivary tumors for human kallikrein 6 (average staining, SD a )
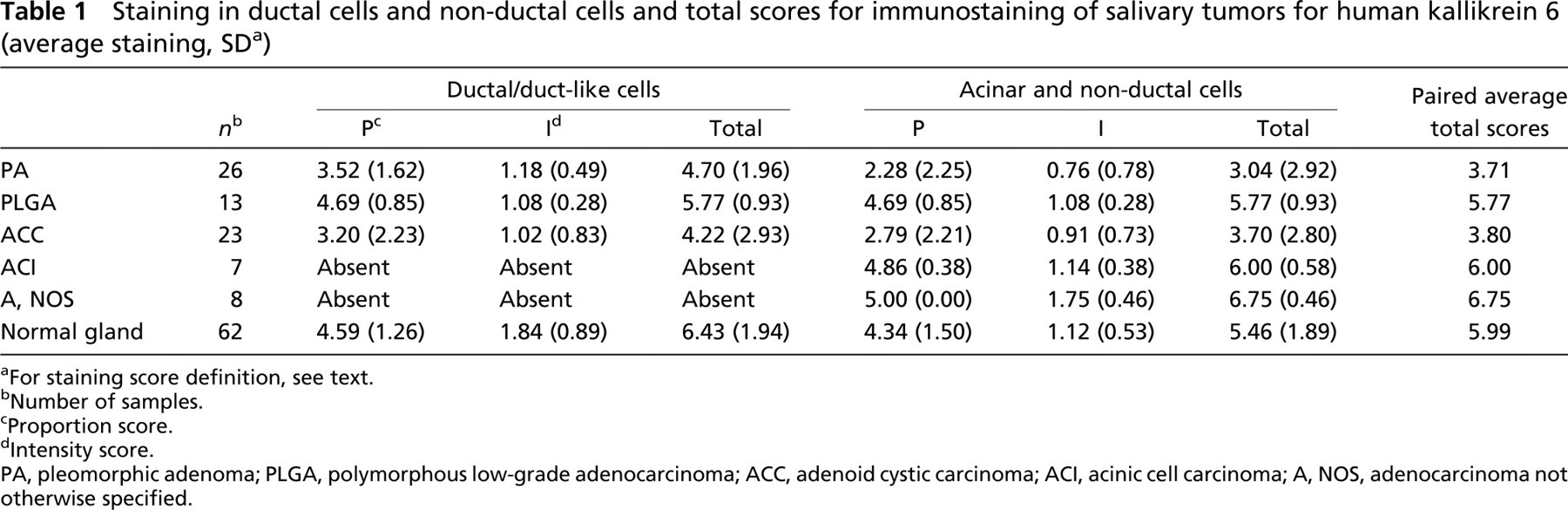
aFor staining score definition, see text.
bNumber of samples.
cProportion score.
dIntensity score.
PA, pleomorphic adenoma; PLGA, polymorphous low-grade adenocarcinoma; ACC, adenoid cystic carcinoma; ACI, acinic cell carcinoma; A, NOS, adenocarcinoma not otherwise specified.
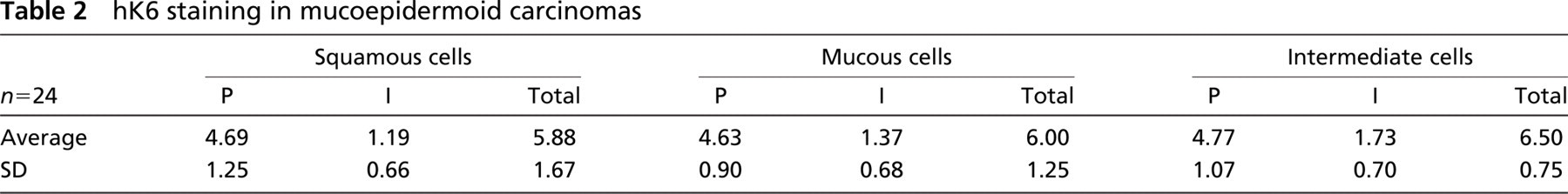
hK6 staining in mucoepidermoid carcinomas
Average staining in normal glands (Figures 1A and 1B) and tumors is shown in Table 3. Interestingly, major glands showed higher average staining than normal minor glands (6.78 vs 5.69; p=0.007). PA (Figure 1) stained significantly less than normal glands (3.71 vs 5.99; p<0.0001). Malignancies also stained less than normal glands (5.27 vs 5.99; p=0.0027). PA stained significantly less than malignancies as a group (3.71 vs 5.27; p=0.007). Of the individual malignant tumors, only ACC (Figure 1D) stained significantly less for hK6 than did normal glands (average 3.80 vs 5.99; p=0.0002). Differences between tumors were as follows: PA less than PLGA (Figure 1E) (3.71 vs 5.77; p=0.0151) and MEC (Figure 1F) greater than ACC (6.12 vs 3.80; p=0.0027).
Differences of staining between cell types (duct-like vs non-duct-like) were also evaluated. Staining in myoepithelial cells in normal salivary glands was not detected. Cytoplasmic staining around mucous globules in mucous acinar cells was assessed (see Figure 1); hence, the high positivity score (Table 1). PA duct-like cells stained significantly lower than normal gland ducts (4.70 vs 6.43; p<0.0001), and malignant tumor duct-like cells also stained significantly lower than normal gland ducts (4.78 vs 6.43; p<0.0001). Individual malignant tumors, which showed significantly decreased duct-like cell staining when compared with ductal cells of normal glands, included ACC (4.22 vs 6.43; p=0.0003) and PLGA (5.77 vs 6.43; p=0.005). PA, the malignant tumors as a group, and ACC all showed significantly less staining of non-duct-like cells than acinar cells of normal glands (3.04, 4.44, and 3.70 vs 5.46; p=0.0007; 0.0355 and 0.0071, respectively). We interpreted the weakly intense but generalized staining of squamous cells in MEC to be positive.
In PA only, overall staining was much higher in duct-like than in non-duct-like cells, although this difference was not significant. In all other tumors exhibiting both types of cells, hK6 staining was similar in both ductlike and non-duct-like cells. Generally, there were no significant differences in the same tumor between ductlike and non-duct-like cells. Some differences occurred between different tumors when the same cell type was compared: PA non-duct-like cells stained less than PLGA non-duct-like cells (3.04 vs 5.77; p=0.0102) and ACC non-duct-like cells less than PLGA non-duct-like cells (3.70 vs 5.77; p=0.0380). hK6 staining in A, NOS is shown in Figure 1G and in ACI is shown in Figure 1H.
The Kruskal-Wallis test revealed that the groups of cells in MEC were significantly different in their expression of hK6 (p=0.0488; see Table 2), intermediate cells having a higher total score than mucous and squamous cells.
Discussion
Little et al. (1997) isolated hK6 mRNA from salivary gland tissue, and Petraki et al. (2001) reported the presence of hK6 in ductal and acinar cells of normal salivary glands. In this paper we detail the presence in terms of extent and intensity of staining, as well as the site of hK6 staining in normal salivary gland tissue and describe its presence in numerous salivary gland tumors.
hK6 expression was clearly greater in ductal cells of normal salivary gland tissues, less in acinar cells, and apparently not expressed in myoepithelial cells. Results of this study indicate that most of the studied salivary gland tumors showed downregulated levels of expression of hK6. hK6 staining in all tumors, although diffusely positive in most cells, was of uniformly low intensity in all tumors of both major and minor glands. We considered that hK6 was expressed by cells of apparent myoepithelial differentiation in PA (as well as in duct-like cells) (Figure 1) and also in cells of apparent squamous differentiation in MEC, albeit at low levels. This may represent aberrant expression in tumor cells.
hK6 is a trypsin-like serine protease, and its physiological role is unknown. It is upregulated in female genital and in gastrointestinal carcinomas (Yousef et al. 2004) but is dramatically downregulated in metastatic breast carcinoma cells, in comparison to primary carcinoma cells or normal breast epithelial cells (Anisowicz et al. 1996; Diamandis and Yousef 2001). It can degrade extracellular matrix proteins and may contribute to the ability of tumor cells to invade and metastasize (Ghosh et al. 2004). In the current study, it is noteworthy that the lowest expression of hK6 occurred in PA—a benign tumor—and in the cribriform pattern of ACC. This possibly signifies a lower potential ability for invasion (in the case of PA) and metastasis (in the case of ACC).
The kallikrein gene locus on chromosome 19q13.4 has been well characterized (Diamandis and Yousef 2001; Borgono and Diamandis 2004). The family consists of 15 genes encoding for secreted serine proteases. Among the proteins encoded, prostate-specific antigen (PSA or hK3) is a valuable marker for prostate cancer (Stamey et al. 1987). A number of other kallikreins have also been associated with various forms of malignancy (Diamandis et al. 2001; Yousef and Diamandis 2001; Diamandis and Yousef 2002; Borgono et al. 2004; Borgono and Diamandis 2004). For example, the KLK8 gene is upregulated in ovarian cancer, and its higher expression is associated with a favorable outcome (Magklara et al. 2001; Kishi et al. 2003). In this paper we report for the first time the expression of hK6 in many salivary gland tumors. This is not unexpected, as hK6 is a secreted protein expressed in glandular tissues (Yousef et al. 1999), but further studies are required to determine which way KLK6 is regulated (up or down) in salivary gland tumors and to determine correlation with clinical outcome. Measurement of serum levels of hK6 is required to assess whether it can be used as a marker to monitor salivary gland tumors. Examination of other kallikreins in salivary gland tumors merits investigation because almost all kallikreins are expressed in this tissue (Diamandis and Yousef 2001; Yousef and Diamandis 2001; Borgono et al. 2004; Borgono and Diamandis 2004). hK6, together with the aforementioned kallikreins, may be part of an enzymatic cascade pathway operating in many tissues, including the salivary glands, skin, ovary, etc. (Yousef and Diamandis 2002; Borgono and Diamandis 2004). From these results it is apparent that hK6 cannot be considered a specific marker for any of the salivary gland tumors studied but has the potential to be utilized with other markers. For most if not all cancers, panels of biomarkers are most useful for diagnosis, monitoring, and prognosis. The pathobiological role of other kallikreins in salivary gland tissues also warrants further investigation.
Average a hK6 staining in normal glands and tumors
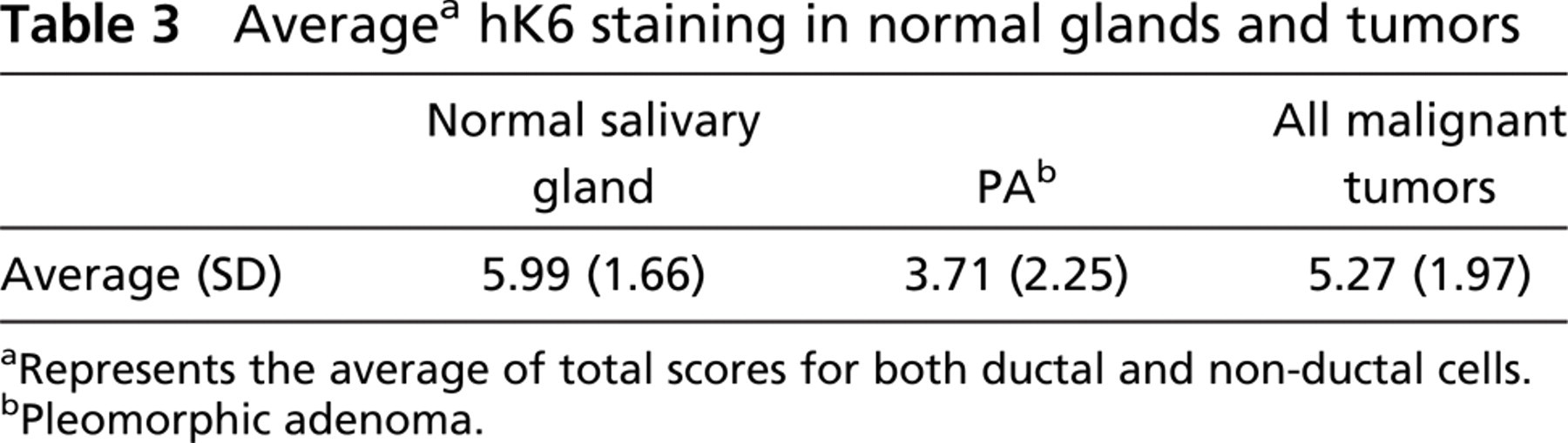
aRepresents the average of total scores for both ductal and non-ductal cells.
bPleomorphic adenoma.
In conclusion, we demonstrate the expression of hK6 in several salivary gland tumors, apparently lower than in normal salivary gland tissue. This and other kallikreins have the potential as useful biomarkers for salivary gland neoplasms.
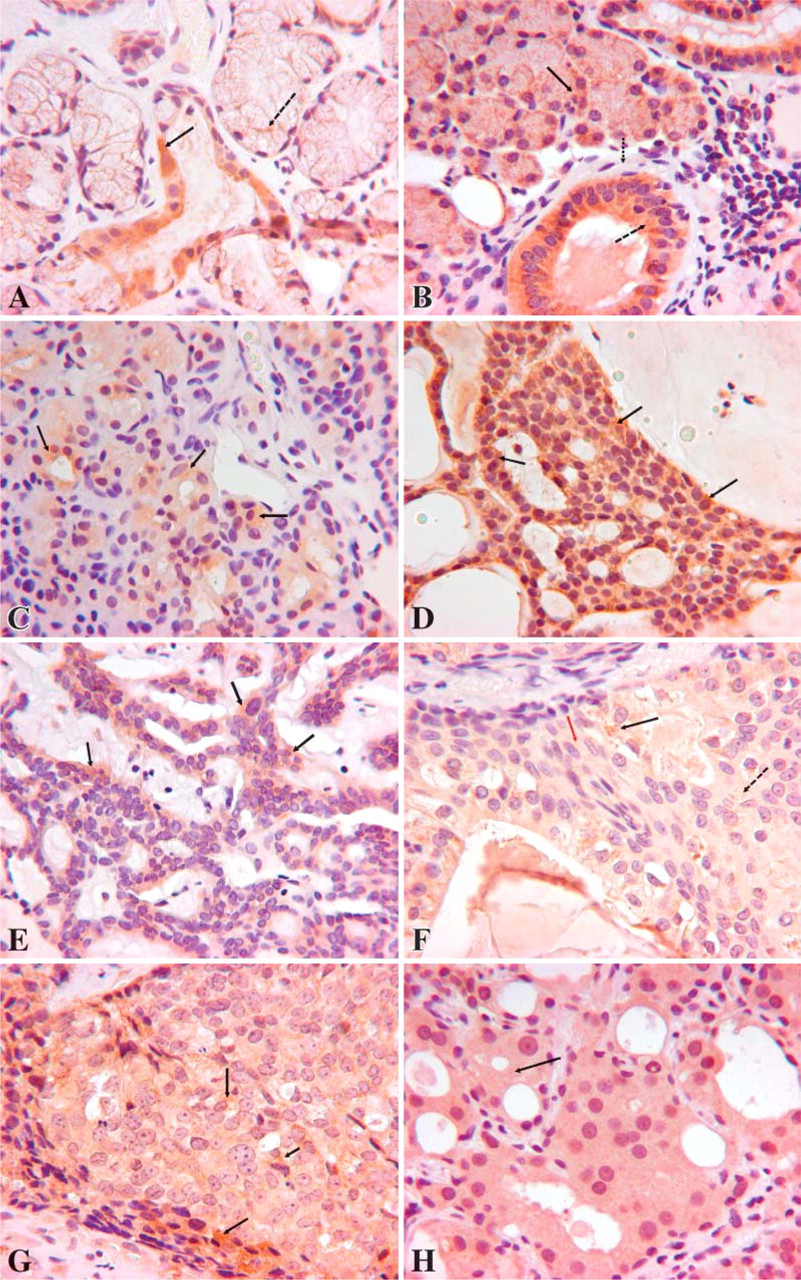
hK6 staining (shown as brown color) in normal salivary glands and salivary gland tumors. (
Footnotes
Acknowledgements
Funding for this study was provided by the National Organization for Rare Disorders.
The authors are grateful to Linda Grass for technical advice.
