Abstract
Neurofibromatosis type 1 (NF1) is an inherited disease with an incidence of about 1:3000 worldwide. Approximately half of all patients with NF1 present osseous manifestations, which can vary from mild to severely debilitating changes such as congenital pseudarthrosis. In the present study, fracture healing of mouse tibia was followed and specimens were collected 5, 9, 14, and 22 days postoperatively. Experimental pseudarthrosis of rat was followed up to 15 weeks postoperatively. In situ hybridization and immunohistochemistry were used to demonstrate expression of NF1 tumor suppressor and phosphorylated p44/42 mitogen-activated protein kinase (MAPK), an indicator of the Ras-MAPK pathway. The results showed that ossified callus was formed in mouse fracture 22 days after the operation. The final outcome of rat pseudarthrosis was detected 9 weeks after the operation, presenting abundant cartilaginous callus at the pseudarthrosis. NF1 gene expression was noted in the maturing and in the hypertrophic cartilages during normal mouse fracture healing, and in rat pseudarthrosis. Phosphorylated p44/42 MAPK was detected in a subpopulation of the hypertrophic chondrocytes in both models. Furthermore, positive labeling for NF1 mRNA and protein was detected in endothelium in both the pseudarthrosis and in the fracture. In conclusion, NF1 gene expression and function are needed for normal fracture healing, possibly restraining excessive Ras-MAPK pathway activation.
N
Mutation in the NF1 gene causes neurofibromatosis type 1 syndrome (NF1), which is an autosomal, dominantly inherited disease with an incidence of 1:2600–1:3500 (Huson et al. 1989; Poyhonen et al. 2000; Lammert et al. 2005a). Cafe-au-lait pigment spots, neurofibromas, hamartomas of the iris (Lisch nodules), and selected osseous dysplasias are hallmarks of neurofibromatosis 1 (Stumpf et al. 1988; Riccardi 1999; Evans et al. 2004). Skeletal manifestations in NF1 include macrocephaly, short stature, kyphoscoliosis, sphenoid wing dysplasia, decreased bone mineral density and content in the load-carrying parts of the body, and congenital bowing and pseudarthrosis of the tibia (CPT) (Crawford and Bagamery 1986; Illes et al. 2001; Vitale et al. 2002; Kuorilehto et al. 2005; Lammert et al. 2005b). In addition, decreased bone strength and muscle cross-sectional area have been detected in NF1 patients (Stevenson et al. 2005).
Approximately 50% of NF1 patients present osseous manifestations (Crawford and Bagamery 1986). A recent study has demonstrated an abundant expression of the NF1 gene in normal mature and developing skeletal tissues (Kuorilehto et al. 2004). Specifically, NF1 expression was detected in maturing and hypertrophic cartilages in the growth plate and in osteoblasts, osteocytes, and osteoclasts (Kuorilehto et al. 2004).
The objective of the present study was to characterize the expression of the NF1 mRNA and protein in rodents during bone fracture healing. Fracture repair of long bones offers a fitting way to study bone development in an adult organism because it mirrors the various steps of endochondral ossification that occur during embryonic bone formation. In addition, because 50–90% of congenital pseudarthroses, especially in tibia or fibula, are due to NF1 (Morrissy 1982; Murray and Lovell 1982; Hefti et al. 2000), we also studied NF1 gene expression and protein localization in an experimental rat pseudarthrosis model. We used mouse fractures and rat pseudarthrosis because of the availability of tissue specimens. Previous studies have shown that tibial fracture healing in mouse and rat follow similar timetables, and that the histological findings are similar (Ekholm et al. 2000, 2002). Furthermore, because neurofibromin functions as a negative regulator of Ras, phosphorylated p44/42 MAPK was used as an indicator of activated Ras-MAPK pathway.
Materials and Methods
Animals
Histological specimens for mouse fracture healing and experimental pseudarthrosis of rat were available from two previous studies (Ekholm et al. 1995, 2002). Fracture healing was analyzed in 129sv/tae mice (n=27) and experimental pseudarthrosis in male Wistar rats (n=21). Fracture of tibia in mice was produced bilaterally under anesthesia as described previously (Hiltunen et al. 1993). Unrestricted mobilization of the operated limb was allowed immediately, and weight bearing was achieved within a few days. Callus formation was observed at 5, 9, 14, and 22 days after the operation. Calluses were carefully dissected from surrounding tissues and fixed in buffered formaldehyde, decalcified in 0.5 M EDTA, embedded in paraffin, and sectioned longitudinally for histology.
Experimental pseudarthrosis was generated as described previously (Hietaniemi et al. 1995). In short, fracture of mid diaphysis of right femur was fixed loosely with 0.7 mm steel wire. Periosteum was electrocauterized within 2 mm of the fracture site. Weight bearing was allowed immediately after the operation, and the rats returned to normal activity within a few days. The development of pseudarthrosis was followed at 1, 5, 7, 9, 11, and 15 weeks after the operation. Femurs were carefully dissected from surrounding tissues and prepared for histology following a procedure similar to that used for mice fractures. The experimental procedures were approved by the ethical committees of the University of Turku (mouse fractures) and the University of Helsinki (rat pseudarthrosis).
In Situ Hybridization
In situ hybridization was carried out as previously described in detail (Yla-Outinen et al. 2002). The plasmid, containing a 652-bp cDNA fragment corresponding to bases 145–797 of mouse NF1 cDNA (gi:26102424), was linearized to generate templates for digoxigenin (DIG)-labeled sense- and antisense-oriented RNA probes. Tissue sections embedded in paraffin were deparaffinized and processed further for in situ hybridization. DIG-labeled probe was detected with a sheep anti-digoxigenin antibody coupled to alkaline phosphatase and the color substrate nitrobluetetrazole/5-bromo-4-chloro-3-indolyl phosphate according to the protocol provided by the manufacturer (Roche Diagnostics; Mannheim, Germany). In situ hybridization with sense probe served as a negative control, showing only faint uniform background hybridization.
Immunohistochemistry
Paraffin-embedded tissues were cut into 5-μm-thick sections, plated on silanated glass slides, and deparaffinized. The sections were treated with H2O2 in PBS for 5 min to remove endogenous peroxidase. The sections were subsequently incubated in 1% BSA-PBS for 30 min to prevent nonspecific binding. To detect NF1 protein, two polyclonal rabbit primary antibodies recognizing different sequences were used (NF1GRP(D) (cat. Sc-67; Santa Cruz Biotechnology, Santa Cruz, CA; and cat. NB 300–155; Novus-Biologicals, Littleton, CO). Both antibodies yielded apparently similar labeling patterns. Polyclonal rabbit antibodies were used to detect phosphorylated p44/42 MAPK (Thr202/Tyr204) (cat. #9101; Cell Signaling Technology, Beverly, MA) and type X collagen (cat. RDI-COLL10abr; Research Diagnostics Inc., Flanders, NJ). The Histostain-plus kit (Zymed Laboratories, Inc.; San Francisco, CA) was used to provide secondary antibodies and DAB color reaction. As a negative control, immunoreaction primary antibody was replaced with 1% BSA-PBS. In all negative controls, only a faint uniform background labeling was observed.
Results
General Histology of Mouse Fractures and Experimental Rat Pseudarthrosis
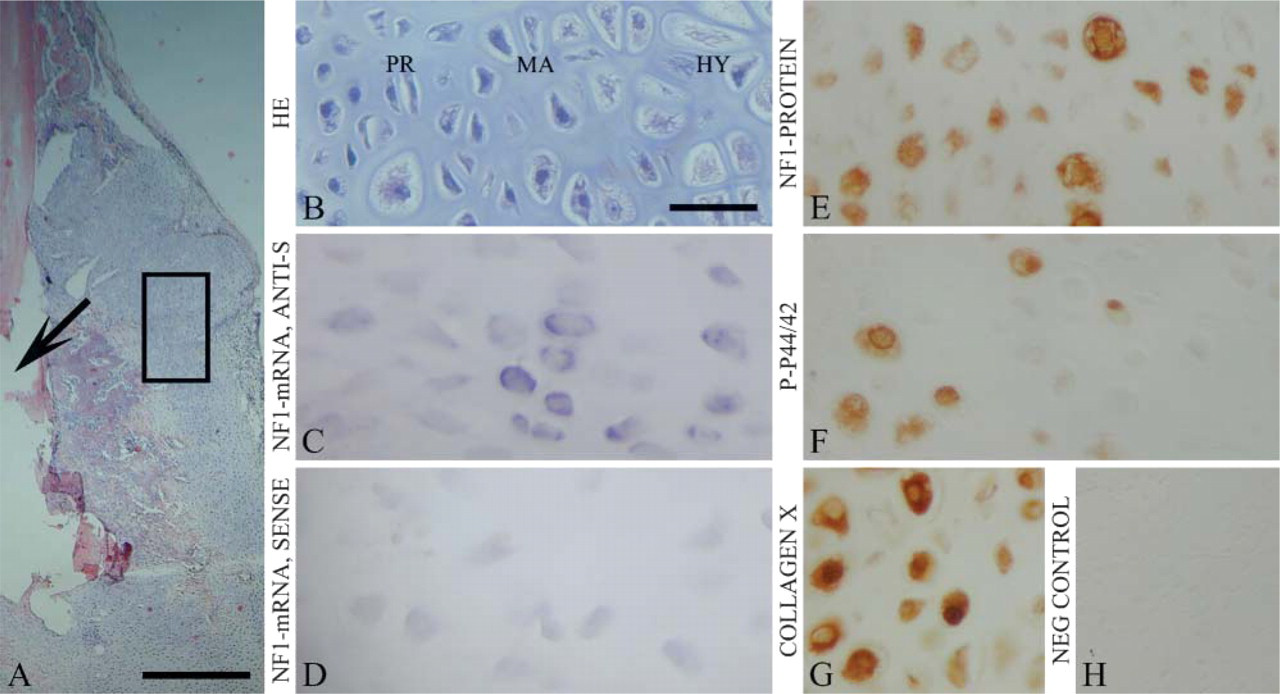
The histological results on mouse fracture were in good agreement with a previous report (Hiltunen et al. 1993). Specifically, mouse fracture healing started with the production of the highly vascular collagenous tissue that forms after the blood clot (granulation tissue; day 5). Granulation tissue was replaced by fibrous tissue and hyaline cartilage and subsequent endochondral ossification (day 9; Figure 1). New trabecular bone in the callus was abundant on the 14th day after the operation and joined the fracture fragments by day 22.
Detailed analyses and methodological aspects of the experimental pseudarthrosis have been presented previously (Ekholm et al. 1995; Hietaniemi et al. 1995). One week after the operation, granulation tissue was observed in the gap between fracture ends (Figure 2). Only a little cartilaginous callus was seen at this point. A distinct hard callus but no bridging with new bone over the fracture gap was detected after 3–15 weeks (Figures 2B and 2C). The final outcome was detectable 9 weeks after the operation, presenting abundant cartilaginous callus at the pseudarthrosis (Figure 2). Cartilage was still detected at both ends of the fracture at 15 weeks (Figure 2).
Throughout the present study, cartilage was defined as avascular tissue with abundant extracellular matrix and basophilic reaction in hematoxylin-eosin (HE) staining. Differentiation of hyaline cartilage was identified by morphology (Figure 1) and expression of the collagen type X (Figure 1G and Figure 2I), which is a marker for hypertrophic cartilage (Cancedda et al. 1995). Differentiation of cartilage proceeds, in brief, as follows: proliferative cells are flattened, in columns, and gradually become rounded in maturing cartilage. Ossification begins as the cells become larger and start to develop into mineralizing hypertrophic cartilage (Cancedda et al. 1995).
NF1 Gene Expression and Activation of Ras-MAPK Pathway in Mouse Fracture and Rat Pseudarthrosis
In situ hybridization and immunohistochemistry detected NF1 gene expression in the resting, maturing, and hypertrophic cartilages of healing mouse fracture callus (Figures 1C and 1E). Proliferating cartilage cells did not express NF1 mRNA or protein. The expression patterns in mouse fracture callus remained constant throughout the surveillance period. In the callus associated with experimental rat pseudarthrosis, NF1 mRNA was detected in the mature and hypertrophic cartilages, most notably 7 weeks after the operation (Figure 2E). Only a faint NF1 mRNA hybridization signal was seen 1 week after the operation (Figure 2E). The most intense NF1 protein labeling was detected in the mature and hypertrophic cartilages in analogy to the findings on cartilaginous callus in mouse fracture healing (Figure 1E and Figure 2G). Only a weak NF1 protein labeling was detected in the soft tissue around the cartilaginous ends of the pseudarthrosis gap (Figure 2G).
Experimental mouse tibial fracture at 9 days after operation. (
Phosphorylated p44/42 MAPK was observed in a subpopulation of hypertrophic chondrocytes in both normally healing mouse fractures (Figure 1F) and in rat pseudarthrosis (Figure 2H). In the rat pseudarthrosis, the p-p44/42 MAPK expression seemed to increase during the surveillance period and reached its maximum at 15 weeks after the operation (Figure 2H). However, the present study includes descriptive data only, because the material was not sufficient for further analyses.
In addition, positive signal for NF1 mRNA and protein was detected in osteoblasts and osteoclasts, in analogy to the results of our previous study (Figures 3B and 3C) (Kuorilehto et al. 2004). Positive signal for NF1 mRNA and protein was seen in the endothelium of blood vessels near cartilaginous tissue (Figures 3B and 3C). Only a few endothelial cells of the blood vessels in the vicinity of the pseudarthrosis or the fracture were positive for p-p44/42 (Figure 3D). The results in osteoblasts, osteoclasts, and endothelium were similar throughout the surveillance period in both mouse fractures and rat pseudarthrosis. The HE staining of the osteoblasts, osteoclasts, and endothelium is shown in Figure 3A to clarify the high-magnification panels and to show the morphological features of the cells.
Negative controls included hybridization with sense probe in in situ hybridization (Figure 1D and Figure 2F), and immunization without primary antibody in immunohistochemistry (Figure 1H and Figure 2I), both showing only faint background labeling. Collagen type X served as a positive control for hypertrophic cartilage (Figure 1G and Figure 2I).
Discussion
Previous studies have demonstrated that the NF1 gene is upregulated during human dermal tissue repair, and that NF1 regulates mouse skin wound healing, fibroblast proliferation, and collagen deposition by fibroblasts (Yla-Outinen et al. 1998; Atit et al. 1999). In this study, NF1 gene expression was evaluated for the first time during mouse fracture healing and in experimentally induced pseudarthrosis in rats. The results showed distinct expression of the NF1 gene in the cartilaginous callus of mouse fracture. Specifically, NF1 mRNA and protein were detected in maturing and hypertrophic cartilages, in osteoblasts and osteoclasts, and in endothelium, but not in proliferating chondrocytes. The NF1 expression profile in the cartilage between fracture ends resembled that in the growth plate of normal mice (Kuorilehto et al. 2004). The similarities observed here between developing and healing bones can be attributed to the fact that most bone growth and fracture healing are attained in part via endochondral ossification.
Pseudarthrosis is the development of a false joint in a long bone. Most frequently, pseudarthrosis develops in a malpositioned or infected fracture and can occur in almost any bone of the body (Hayda et al. 1998). In the present study, experimental pseudarthrosis was induced using inadequate fixation by a loose steel wire and electrocauterization of the reactive periosteum (Hietaniemi et al. 1995). Healing and new bone formation are severely affected in this model, and show altered cartilage-specific gene expression profiles (Ekholm et al. 1995). Normally, cartilage reaches its maximal size during the second week of healing in long-bone fractures of young adult rats and is rapidly and completely replaced by bone within 4 weeks. In the pseudarthrosis model used in the present study, cartilage synthesis reached its maximum at 7 weeks. The stimulus of endochondral ossification becomes, however, exhausted, and the fracture gap develops into an inactive fibrous scar (Ekholm et al. 1995; Hietaniemi et al. 1998). As in stable mouse fracture repair, NF1 mRNA and protein were expressed in resting and hypertrophic cartilages in experimental pseudarthrosis, in particular at week 7, correlating with maximal cartilage-specific gene expression. Furthermore, NF1 mRNA and protein were expressed in osteoblasts, osteoclasts, and endothelium in experimental rat pseudarthrosis. Positive labeling for phosphorylated p44/42 MAPK was detected in a subpopulation of hypertrophic chondrocytes of fracture callus and in experimental rat pseudarthrosis. All maturing and hypertrophic chondrocytes were positively labeled for NF1 protein and collagen type X, and only a subpopulation of chondrocytes was labeled using p-p44/42 MAPK antibody. Whether the expression of NF1 protein and that of p-p44/42 MAPK are mutually exclusive cannot be determined in the present study and using the material that was available to us. This topic should be addressed in an independent study.
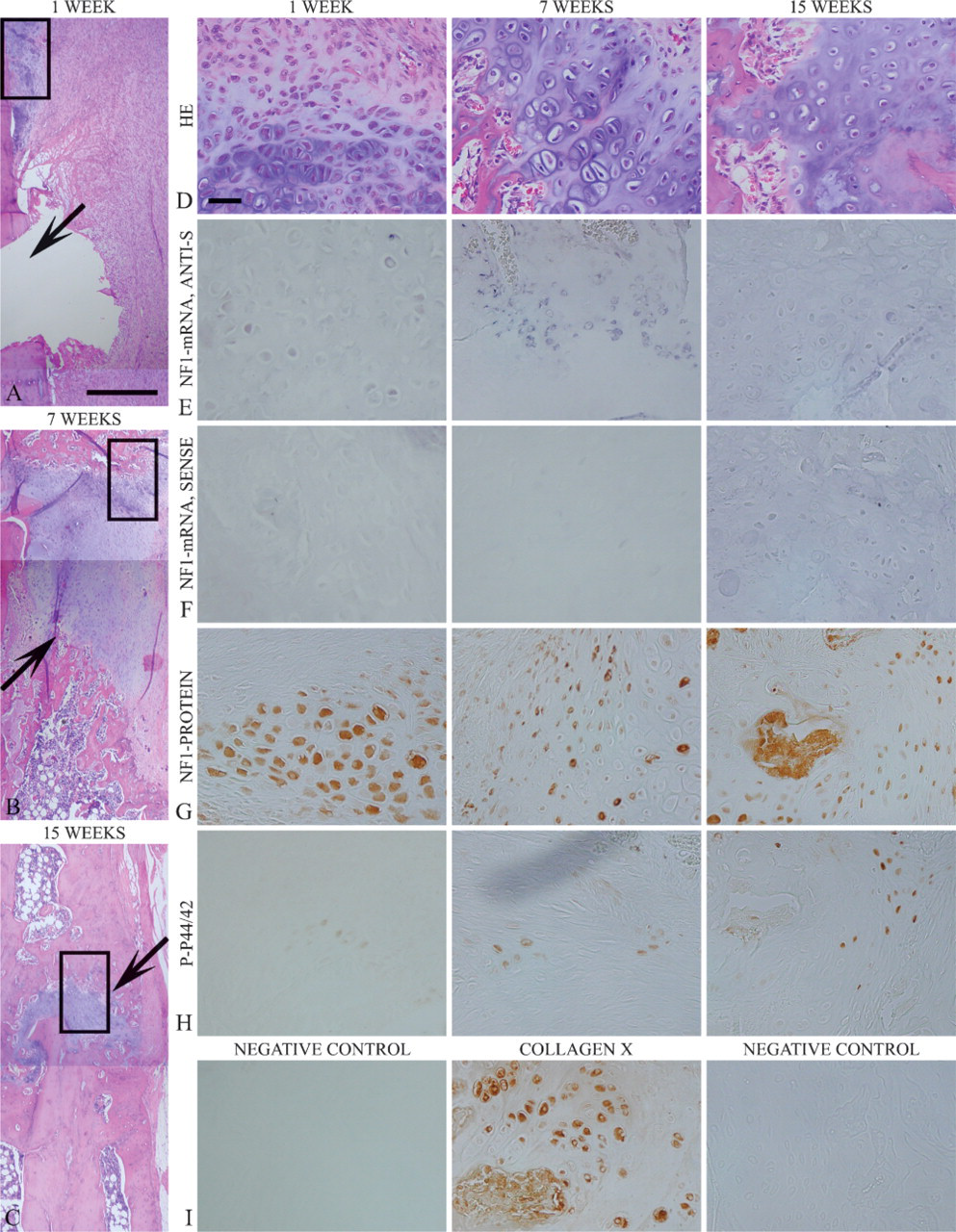
Experimental pseudarthrosis in rat 1, 7, and 15 weeks after operation. Low-magnification HE-staining frames (
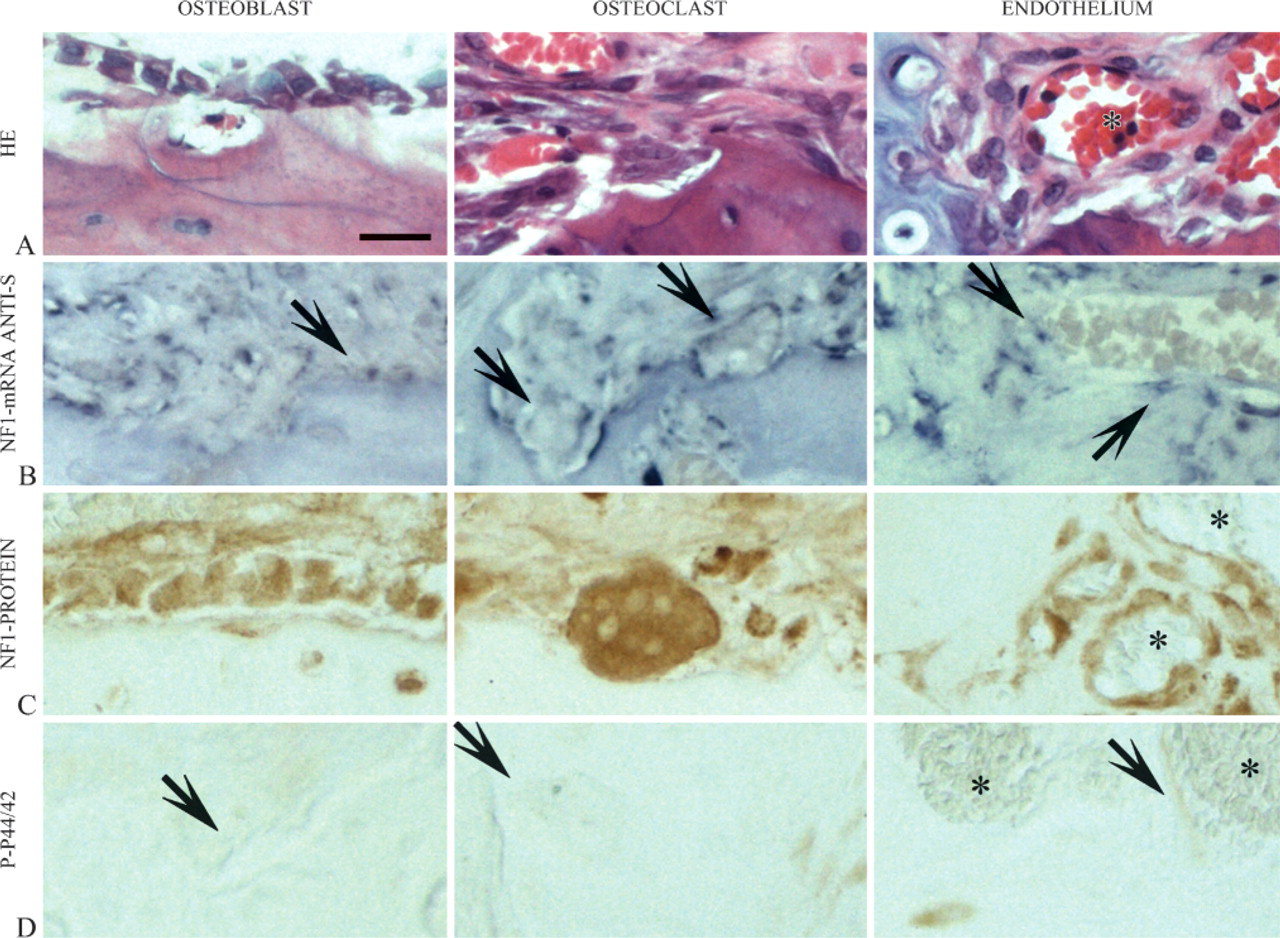
Expression patterns of osteoblasts, osteoclasts, and endothelium in experimental pseudarthrosis of rat. (
A review of the literature reveals only a few reports on aberrant fracture healing in NF1 patients (Hayashi et al. 2005). A general genotype of NF1 patient with one functional NF1 gene allele is thus enough for normal fracture healing. On the other hand, the results presented here may suggest that NF1 gene expression is necessary for normal fracture healing. The role of the NF1 gene in fracture healing may be that of restraining the excessive activation of the Ras-MAPK pathway in hypertrophic chondrocytes. Furthermore, increased Ras-MAPK activation, together with altered osteoblastic differentiation, has been detected in NF1 heterozygous mice (Yu et al. 2005). Some NF1 patients suffer from congenital pseudarthrosis, which represents primary dysplasias rather than consequences of trauma or nearby neurofibroma (Stevenson et al. 1999; Hefti et al. 2000). Congenital pseudarthrosis of tibia often manifests when a child with NF1 starts to stand and walk (Stevenson et al. 1999; Vitale et al. 2002). Histological studies have shown that the tissue of the pseudarthrosis gap consists mainly of fibroblasts, not neurofibroma-like tissue, i.e., Schwann cells, perineurial cells, and axons (Briner and Yunis 1973; Ippolito et al. 2000). Furthermore, in a recent publication, vasculopathy was detected in vessels at the pseudarthrosis in both NF1-associated CPTs and non-NF1 CPTs (Hermanns-Sachweh et al. 2005). The chain of events leading to congenital pseudarthrosis in NF1 remains to be elucidated. One can speculate that impaired endochondral ossification (Kuorilehto et al. 2004) or poor osteoblastic differentiation (Yu et al. 2005) could lead to the growth within the bone of soft tissue prone to developing pseudarthrosis (Briner and Yunis 1973; Ippolito et al. 2000) following increased mechanical strain when a child with NF1 starts to stand and walk.
In conclusion, the NF1 gene is expressed in cartilage, fibrous tissue, and endothelium in normal healing of a long-bone fracture and in all phases of experimental pseudarthrosis. Thus the altered expression of the NF1 gene in NF1 patients could lead to the skeletal aberrations of the disease. The congenital nature, rare occurrence, and constant clinical picture of this disease emphasize the possibility that the loss of neurofibromin function and the resultant increase in Ras activity could lead to poor differentiation and bone formation and eventually to NF1-related pseudarthrosis.
Footnotes
Acknowledgements
This study was supported by grants from the Cancer Society of Finland; Oulu University Hospital, grant # H01139; Academy of Finland; Turku University Foundation; and The Finnish Medical Foundation.
We thank Marja Paloniemi and Pirkko Peronius for expert technical assistance.
