Abstract
Neurofibromatosis type 1 (NF1) is a common, cancer-predisposing disease caused by mutations in the NF1 tumor gene. Patients with NF1 have an increased risk for benign and malignant tumors of the nervous system (e.g., neurofibromas, malignant peripheral nerve sheath tumors, gliomas) and other tissues (e.g., leukemias, rhabdomyosarcoma, etc.) as well as increased susceptibility to learning disabilities, chronic pain/migraines, hypertension, pigmentary changes, and developmental lesions (e.g., tibial pseudoarthrosis). Pigs are an attractive and upcoming animal model for future NF1 studies, but a potential limitation to porcine model research has been the lack of validated reagents for direct translational study to humans. To address that issue, we used formalin-fixed tissues (human and pigs) to evaluate select immunohistochemical markers (activated caspase-3, allograft inflammatory factor-1, beta-tubulin III, calbindin D, CD13, CD20, desmin, epithelial membrane antigen, glial fibrillary acidic protein, glucose transporter-1, laminin, myelin basic protein, myoglobin, proliferating cell nuclear antigen, S100, vimentin, and von Willebrand factor). The markers were validated by comparing known expression and localization in human and pig tissues. Validation of these markers on fixed tissues will facilitate prospective immunohistochemical studies of NF1 pigs, as well as other pig models, in a more efficient, reproducible, and translationally relevant manner.
Keywords
Introduction
Neurofibromatosis 1 (NF1) is an autosomal dominant disease caused by mutations in the gene encoding neurofibromin, a tumor suppressor protein that negatively regulates multiple proliferative cellular pathways.1–3 The RAS oncogene is the primary target of neurofibromin, a Ras-GAP (GTPase-activating protein) that inhibits Ras signaling by promoting its conversion to the GDP-bound inactive form. 4 As a consequence, loss of neurofibromin increases the activity of Ras and its numerous downstream effectors, including the Raf/MEK/ERK and PI3K/Akt/mTOR pathways.2,3,5–13 Hyperactivated Ras drives tumorigenesis through increased cell proliferation and survival. 14 In fact, nearly all NF1 patients develop benign neurofibromas while a smaller percentage (roughly 15–20%) develops cancers, including optic gliomas, rhabdomyosarcoma, leukemias, and malignant peripheral nerve sheath tumors (MPNST).15–18 In addition to tumors, NF1 patients are also prone to a spectrum of clinical features including pigmentary changes, skeletal deformities, and cognitive and behavioral disorders, to name a few.16,19
Numerous genetically engineered mouse models targeting the NF1 gene have been generated. Unlike humans, however, mice expressing a single mutant allele of Nf1 fail to develop the classic features of NF1 and are considered inaccurate models of the disease. The Nf1+/− mice do exhibit learning and memory deficits that are common in NF1 patients, but they fail to develop the hallmarks of NF1 including neurofibromas, pigmentation defects, enhanced pain perception, and various malignancies such as MPNSTs.20–22 To successfully model typical NF1 tumor types, conditional inactivation of both Nf1 alleles or combined alteration of other cancer genes, such as p53 or INK4a/ARF, with Nf1+/− mutation is required.23–28 Several of these more recent mouse models have served as valuable preclinical tools for testing novel therapeutics for NF1 tumors. Unfortunately, the animals only replicate select aspects of NF1 and are, thus, limited by their inability to mimic the full spectrum of NF1 lesions. As such, there is an ongoing effort within the NF1 research community to develop new animal models that better recapitulate the many NF1 phenotypes seen in patients.
Over the past few years, genetically modified pig models of human diseases have been increasingly and successfully used to study a broad range of diseases including cystic fibrosis,29–32 muscular dystrophy, 33 cancer,34,35 cardiovascular disease,36,37 and ataxia telangiectasia, 38 to name a few. There are several reasons for using pig models, including the similarities to humans such as comparable anatomy, physiology, metabolism, and pathology.39,40 However, compared with rodent models, pig models have limited access to validated reagents and techniques for translational research. 41 In this article, we validate IHC markers for pig and human tissues that will have translational relevance in the study of NF1 in novel pig models.
Materials and Methods
Tissues
Archival tissues (non-NF1 tissues, i.e., “wild type”) from pigs and humans were acquired from the Comparative Pathology Laboratory (University of Iowa). All tissues had been fixed in 10 percent neutral buffered formalin and processed as previously described. 42 Pig tissues were acquired from paraffin-embedded tissue blocks previously used in studies that had received University of Iowa Institutional Animal Care and Use Committee approval. These tissues samples were taken from pigs (Sus scrofa domestica) and included multiple breeds (e.g., Large White, Yucatan, etc.) from commercial farm and/or research sources because (to date) we have not observed variations in immunohistochemical staining between various breeds of pig. Tissues were from pigs less than one year of age and for each marker, a total of three pigs (at least one of each sex) were evaluated. No overt sex-related differences in immunostaining were noted. Human tissues were acquired from de-identified autopsy tissues previously used as IHC control tissues, or through the Cell Culture Core Repository (University of Iowa) that has institutional approval from the University of Iowa Institutional Review Board (IRB #:199507432) for collection of human tissues. Unless otherwise specified, tissues came from individuals that were generally healthy and lacked overt clinical disease. Importantly, we used tissues that had been placed into fixative in a timely manner following harvest to mitigate autolysis, a cellular process that can confound IHC studies. 42
IHC
Markers for optimization and validation were selected based on three criteria: (1) prospective relevance to NF1 studies—for example, through identification of normal tissue structure, diagnostic utility, or study of NF1-related pathogenesis; (2) availability of organs/tissues (human and pig) with known marker expression; and (3) distinct microanatomical localization of the marker for morphological corroboration of human and pig expression. We preferentially evaluated select antibodies/techniques from our lab that were previously optimized/validated for human tissues. Markers and their respective techniques that were successfully optimized and validated are shown in Table 1. For these studies, 3,3′-diaminobenzidine (DAB, brown staining) was used as the chromogen, and Harris hematoxylin (basophilic staining) was used as the counterstain.
IHC Protocols for Validated Cellular Markers in Pig and Human Tissues.
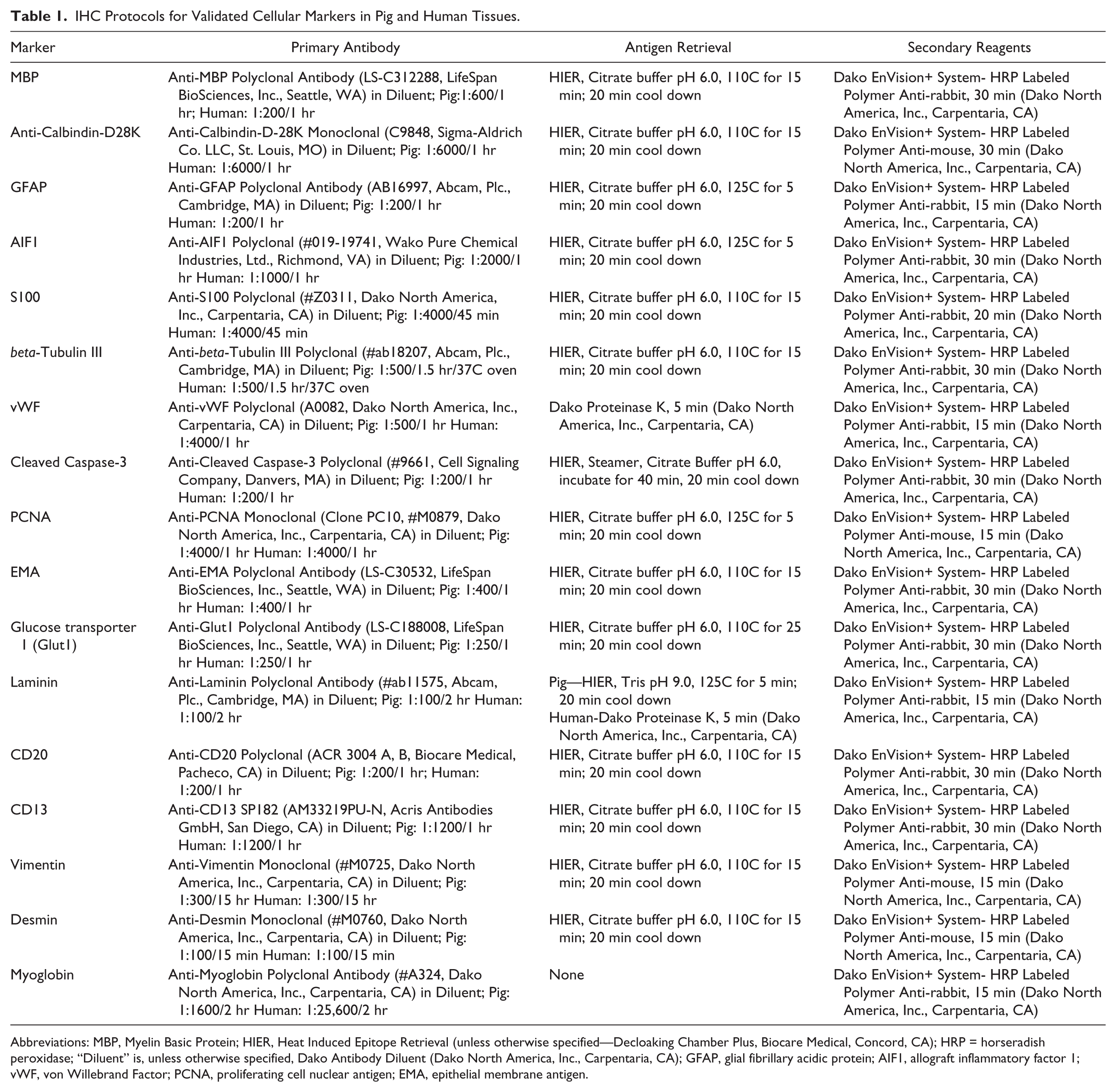
Abbreviations: MBP, Myelin Basic Protein; HIER, Heat Induced Epitope Retrieval (unless otherwise specified—Decloaking Chamber Plus, Biocare Medical, Concord, CA); HRP = horseradish peroxidase; “Diluent” is, unless otherwise specified, Dako Antibody Diluent (Dako North America, Inc., Carpentaria, CA); GFAP, glial fibrillary acidic protein; AIF1, allograft inflammatory factor 1; vWF, von Willebrand Factor; PCNA, proliferating cell nuclear antigen; EMA, epithelial membrane antigen.
Results
Translational markers of the nervous system would be useful as it is commonly affected in NF1. 16 Specifically, markers that allow identification of normal central or peripheral neural structure/organization, or markers that increase the diagnostic sensitivity or specificity for NF1-specific lesions (e.g., neurofibromas) would be useful (see marker selection criteria in section “Materials and Methods”). Myelin basic protein (MBP) was found in myelinated (e.g., white matter) tracts of the brain (Fig. 1A) and can serve as a landmark of brain organization. 43 Calbindin-D28K is a marker of Purkinje cells, 38 specialized neurons of the cerebellum and serve as a marker of cerebellum organization. We observed Purkinje cell immunostaining in the cerebellum (Fig. 1B). Glial fibrillary antigen protein (GFAP) is a marker of glial cells (e.g., astrocytes) and can be useful to detect astrocyte activation (i.e., astrogliosis) in NF1.44,45 GFAP immunostaining was seen in astrocytes of the brain (Fig. 1C). Microglia are resident macrophages in the brain and may play a role in pathogenesis of optic gliomas (an NF1-related tumor) and the ensuing sexual dimorphism of optic glioma-associated retinal injury.46,47 Allograft inflammatory factor 1 (AIF1, also known as ionized calcium-binding adapter molecule-1) is a marker for microglia and can label alveolar macrophages of the lung of pigs and humans. 42 We optimized the AIF1 technique so it immunostained microglia in the brain (Fig. 1D). S100 can be a useful marker in the clinical diagnosis of neurofibromas, intraocular gliomas, and normal peripheral nerves.48–52 We localized S100 in peripheral nerves of muscle tissue (Fig. 1E). Neuronal markers, such as beta-tubulin III, 53 can highlight axonal structure within peripheral nerves. We localized beta-tubulin III in axons of peripheral nerves (Fig. 1F).
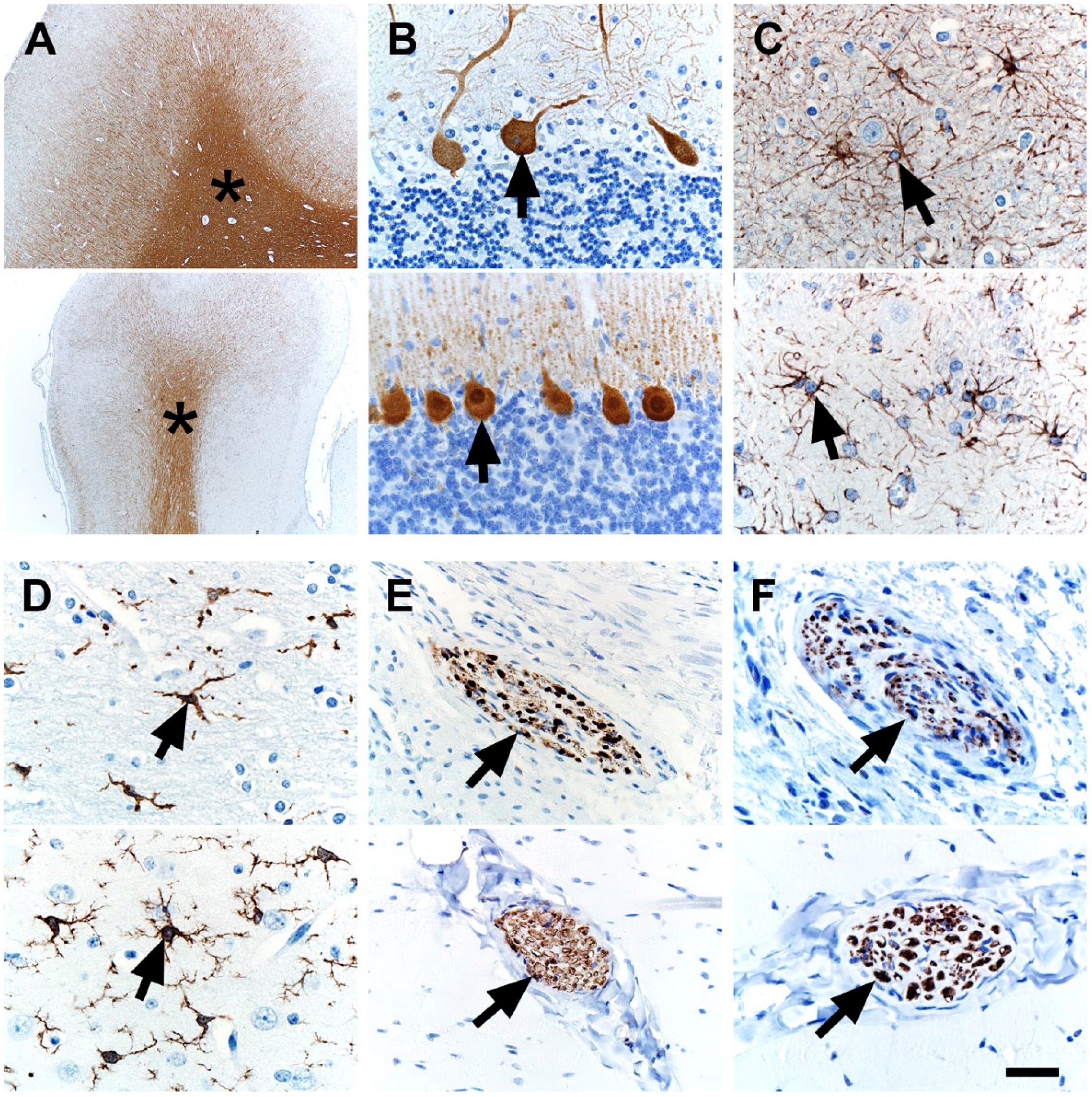
IHC of human (top panel) and pig (bottom panel) tissues. (A) MBP immunostaining was localized to white matter (asterisks) in the cerebrum. (B) Calbindin-D28K immunostaining was localized to Purkinje cells (arrows) in the cerebellum. (C) GFAP immunostaining was localized to glial cells (arrows) in the cerebrum. (D) AIF1 immunostaining was localized to microglial cells (arrows) in the cerebrum. (E) S100 immunostaining was localized to peripheral nerves (arrows) in skeletal muscle. (F) beta-tubulin III immunostaining was localized to axons within peripheral nerves (arrows) of skeletal muscle. Scale bar = 26 (C, D, F), 40 (B, E), and 800 (A) µm. Abbreviations: MBP, Myelin basic protein; GFAP, glial fibrillary acidic protein; AIF1, allograft inflammatory factor 1.
The vasculature is emerging as an important tissue in NF1 pathogenesis. For example, aneurysms and stenoses are vascular anomalies that have been reported in people with NF1. 54 Furthermore, Schwann cells from NF1 neurofibromas have been reported to produce midkine, an angiogenic and mitogenic factor. 55 We evaluated the endothelial marker CD34 using a rabbit polyclonal antibody (#250591, Abbiotec LLC, San Diego, CA), but the immunostaining in both human and pig tissues did not meet our standards for sensitivity and specificity (data not shown) and, thus, did not meet our threshold for inclusion in this study (see section “Materials and Methods”). So, instead, we used von Willebrand Factor (vWF) as a vascular marker, 56 and it distinctly labeled vessels within lymphoid tissues of both species (Fig. 2A). Apoptosis and proliferation are common cellular markers that can be useful in evaluating tumor grade and effects of tumor therapy.57,58 We examined markers of apoptosis (cleaved caspase-3) and proliferation (proliferating cell nuclear antigen, PCNA), and both markers had expected immunostaining that was similar between human and pig tissues (Fig. 2B and C). Epithelial membrane antigen (EMA), 10 glucose transporter 1 (GLUT1), and laminin IHC have been used to rule out other tumors (e.g., perineurioma) and help classify soft tissue tumors.59–61 EMA was localized in the glands (e.g., sebaceus) of normal skin (Fig. 2D). 62 GLUT1 is known to be highly expressed in the lower (more basal) epidermis in humans and was localized there in both species (Fig. 2E). 63 Laminin is expressed in basement membranes and in the perineural tissue, so it can be useful in diagnostics of soft tissue tumors such as neurofibromas and malignant peripheral nerve sheath tumors. 60 Laminin was localized in basement membranes of renal tubules (Fig. 2F).
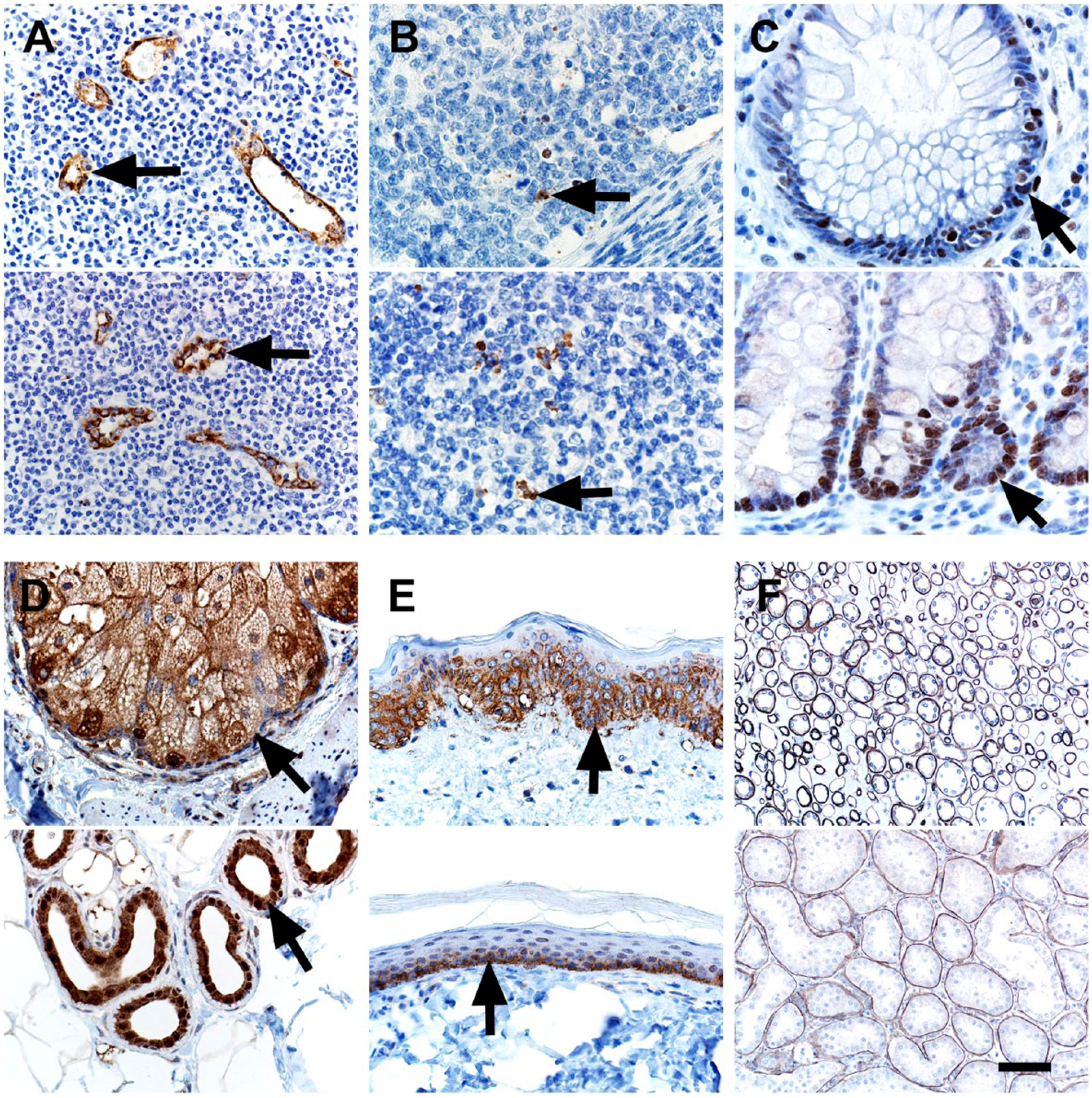
IHC of human (top panel) and pig (bottom panel) tissues. (A) vWF immunostaining was localized to endothelial cells (arrows) of vessels. (B) Cleaved Caspase-3 immunostaining was localized to apoptotic cells (arrows) and debris in lymphoid tissues. (C) PCNA immunostaining was localized to nuclei of proliferating epithelial cells (arrows) in the crypts of the colon. (D) EMA 10 immunostaining was localized to adnexal glands (arrows) of the skin. (E) Glut1 immunostaining was localized to the lower, basal-oriented cells (arrows) of the epidermis. (F) Laminin immunostaining was localized to the tubular basement membranes in the kidney. Scale bar = 26 (B, C), 40 (A, D, E), and 80 (F) µm. Abbreviations: vWF, von Willebrand Factor; PCNA, proliferating cell nuclear antigen; EMA, epithelial membrane antigen; Glut1, glucose transporter 1.
Inflammation (macrophages and lesser numbers of lymphocytes) has been associated with NF1 mutations in glioblastomas. 64 Recently, panels of immune cell markers were evaluated in pig and human tissues 42 ; however, the B cell immunostaining in pigs (i.e., CD79a) was not as robust as in humans. We tested another B-cell marker CD20, and it had the expected B-cell distribution in lymphoid tissues (Fig. 3A), but in similar fashion to CD79a, it had less robust staining in pigs than humans. Children with NF1 may be predisposed to myelogenous leukemias, 65 and CD13 is a myeloid lineage marker that can aid in its diagnosis. 66 CD13 is highly expressed and localized in the renal tubules, and this localization was seen in our tissues (Fig. 3B). Rhabdomyosarcoma is a malignancy of muscle that has increased risks in the NF1 population.15–17 Myoglobin, desmin, and vimentin are potential markers useful for diagnosing skeletal muscle tumors.67,68 In skeletal muscle, these markers exhibited sarcoplasmic-specific staining except for vimentin, which had more sarcoplasmic staining in pigs compared with the scant immunostaining in humans (Fig. 3C–E). In general, vimentin specificity was low as it was seen in other interstitial and mesenchymal tissues (e.g., vascular walls, adipocytes, etc.). The potential for broad or even false positive artifactual immunostaining patterns for vimentin have been recognized69,70 when studying in tissues. Together, these features might limit vimentin usage in situations to corroborate more specific immunostaining (e.g., myoglobin, desmin).
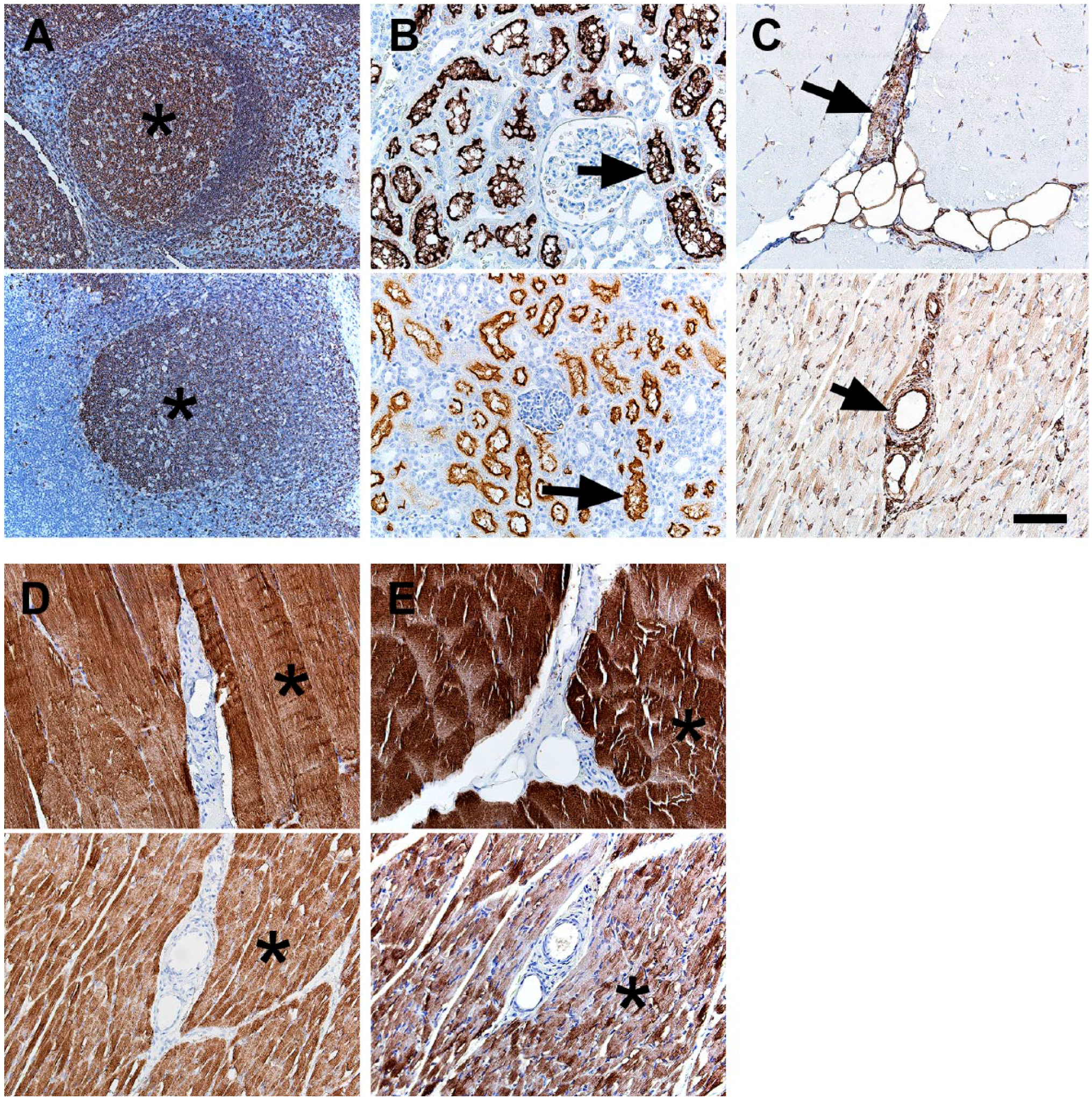
IHC of human (top panel) and pig (bottom panel) tissues. (A) CD20 immunostaining was localized to B-cell rich regions (e.g., germinal centers, asterisks) in lymphoid tissue. (B) CD13 immunostaining was localized to the tubular brush borders of the kidney. (C) Vimentin immunostaining of muscle tissue was preferentially localized to vascular walls (arrows) and interstitial connective tissue with less intense immunostaining of muscle sarcoplasm (pig > human) and adipocytes. (D) Desmin immunostaining of muscle tissue was localized to muscle sarcoplasm (asterisks). (E) Myoglobin immunostaining of muscle tissue was localized to muscle sarcoplasm (asterisks). Scale bar = 80 µm.
Discussion
In this study, we were able to validate several IHC tissue markers in pig and human formalin-fixed paraffin-embedded tissues. Importantly, we did this using the same reagents (i.e., primary antibodies) with infrequent, minor species variation in incubation/concentration (Table 1). We specifically targeted markers that were applicable for prospective NF1 pig studies and, through this, were able to expand the scope and fill in the gaps of validated markers that are available for translational pig studies.
Study of tissues in the nervous system is vital for NF1 research. 16 Tumors of nervous system origin (e.g., neurofibromas) are common in NF1 patients as are cognitive/social deficits, migraines, and chronic pain.71,72 In this study, we were able to validate several markers that would allow for examination of structural organization (e.g., beta-tubulin III, S100) and remodeling (e.g., GFAP, AIF1) of the nervous system in tissues. Markers to study proliferation (e.g., PCNA) and apoptosis (e.g., activated caspase-3), which may be relevant in interrogating the cellular biology of NF, 73 were also validated. These tools allow for detection of abnormal neural structure/organization and tissue remodeling that can help to better detect and clarify the progression of lesions in early NF1 pathogenesis.
People with NF1 are prone to various benign and malignant tumors originating from the nervous system as well as from other tissues. Classification of the tumors in terms of grade (e.g., benign to malignant), cell lineage, and exclusion of other tumors with similar morphology can be critical components in understanding NF1 pig model phenotype and comparing with that of NF1 disease in humans. For instance, S100+ immunostaining is a useful marker to corroborate neurofibroma diagnosis, whereas immunostaining for other markers (e.g., EMA, GLUT1) may point toward different diagnoses (e.g., perineurioma) and, thus, should generally be negative in neurofibromas.50,61 We also were able to validate select markers that could be useful for rhabdomyosarcoma (e.g., myoglobin, desmin) and myelogenous leukemia (CD13)—tumors that are preferentially seen in NF.15–17,65,66
Validated IHC markers, such as these demonstrated in this article, offer investigators a resource for simplicity, consistency, and repeatability in studies of pig models of NF1 as well as other human diseases. With the prospective development of NF1 mutant pigs (unpublished), we expect to further validate and expand the scope of markers that can be used to investigate these models. These markers are part of an emerging “toolbox” that are essential for characterization, diagnostics, and phenotyping of current and future pig models.
Footnotes
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
All authors contributed to the experimental design, writing, and editing of the manuscript. DKM, GKO-A, MRL, JAG performed tissue handling, immunohistochemistry, tissue analysis, and figure preparation. All authors read and approved the manuscript before submission.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the Children’s Tumor Foundation (NF1 Synodos), National Institutes of Health (P01HL051670, P01 HL091842, P30 DK054759), and the Cystic Fibrosis Foundation.
