Abstract
This study investigated if formalin fixation duration affects HER2/neu gene amplification detection by fluorescence in situ hybridization (FISH) in breast cancer. Tumor tissues from 35 cases were divided into three groups and subjected to two formalin fixation protocols per group (12 hr, 27 hr in the first; 2 hr, 17.5 hr in the second; 28.5 hr, 541 hr in the third) before FISH analysis. There was no significant difference in FISH signal detection between the two different fixation protocols in the first two groups. In the third, no signal was detected in 4/6 cases fixed for an extended duration.
The HER-2/neu gene (otherwise known as c-erbB2 gene), a proto-oncogene located on the long arm of chromosome 17, encodes a 185-kD transmembrane glycoprotein that belongs to the type I growth factor receptor family (Vang et al. 2000). Amplification and/or overexpression of the HER2/neu gene have been observed in 25% of primary breast carcinomas (Hoang et al. 2000). The determination of HER2/neu status in breast cancers is important for selection of suitable candidates for anti-HER2/neu therapy (Ellis et al. 2000; Slamon et al. 2001). Hence, considerable attention has been given to the evaluation of HER2/neu gene and protein expression because accurate and reliable testing methods are essential in ensuring that patients are appropriately managed (Perez et al. 2002). There is currently no clear standardization for HER/2neu testing, although known laboratory methods include Southern, Northern, and Western blotting techniques, immunohistochemistry, and fluorescence in situ hybridization (FISH) (Vang et al. 2000).
FISH is a powerful molecular cytogenetic method based on the hybridization of specific DNA sequences to the target genome and involving both histochemical and solid matrix hybridization techniques (Pauletti and Slamon 1999). A permeabilization step is required to facilitate penetration of the fluorescence labeled probe and a denaturation step is necessary for converting double-stranded DNA in tissue sections to single-stranded DNA to allow reannealing of the probe and target genome. Many factors can affect the detection of fluorescent signals by FISH analysis. Technical limitations include the type of fixative used, artifacts caused by sectioning of tissue blocks, thickness of sections, storage of cut sections, and probe penetration (Thompson et al. 1994; Ellis et al. 2000; Gozzetti and Le Beau 2000). In our surgical pathology laboratory, specimens are usually fixed in 10% buffered formalin for a duration of approximately 8–32 hr owing to the inherent vagaries of surgical schedules. The aim of this study was to ascertain if variations in fixation duration have an impact on the reliability of HER2/neu amplification results obtained via FISH and how these results compared with protein overexpression by immunohistochemistry.
Paraffin-embedded breast carcinoma tissues from 35 women who underwent surgical resection of their tumors at the Singapore General Hospital were used for this study. Paraffinized sections were routinely stained with hematoxylin-eosin for histopathological diagnosis. Two sets of tumor tissues from each case were divided into three groups. They were fixed in 10% buffered formaldehyde for 12 ± 2.5 hr (range 9–18 hr, median 12 hr) and 27 ± 3.0 hr (range 20–32 hr, median 27 hr) in the first group, 2 hr and 17.5 ± 1.5 hr (range 16–20 hr, median 17.5 hr) in the second group, and 28.5 ± 2 hr (range 26–30 hr, median 28.5 hr) and 541 hr ± 285 hr (range 193–1010 hr, median 541 hr) in the third group. All cases in the first group represented the usual range of fixation durations for breast surgical specimens routinely handled in the laboratory. The second and third groups were used to test the effect of a shortened fixation duration (2 hr), and an extended fixation duration (median 541 hr) on FISH results compared with routinely fixed cases.
Three-μm sections were cut for FISH analysis and mounted on glass slides coated with silane (3-aminopropyltriethoxysilane; Sigma, St Louis MO). The sections were deparaffinized and pretreated to facilitate probe permeability using a Pretreatment Kit (Vysis; Downers Grove, IL) according to manufacturer's instructions with minor modifications. Briefly, slides were heated overnight (56C) and dewaxed in xylene, air-dried, and treated with 0.2 M HCl for 20 min at room temperature (RT). This was followed by incubation in 1 M sodium thiocyanate in 2 X SSC (0.5 M NaCl, 0.015 M sodium citrate) at 80C for 30 min, rinsed, and digested with protease (1.25 mg/ml) for 30 min at 37C. The slides were finally dehydrated in an ethanol series (70–100%) and air-dried.
ISH was performed using the PathVysion HER2 DNA Probe Kit (Vysis) basically according to the manufacturer's recommendations (Hoang et al. 2000). The hybridization solution containing the probe was applied to the treated slides, the area coverslipped, and sealed. The slides were then placed in a slide thermocycler (Hybaid Omnislide) programmed to codenature probe, target DNA, and incubate the slides for hybridization (75C 5 min, 38C 16 hr, or overnight). Post-hybridization washes were performed (0.5 X SSC, 0.1% NP-40 pH 7) at 70C followed by counterstaining in DAPI (0.1 g/ml in 2 X SSC) at RT. The slides were mounted with antifade (Vectashield) and viewed under an epifluorescence microscope (Olympus) and images captured (CytoVision) (Figures 1A and 1B). Sixty nuclei were counted. Signal enumeration was performed with the following conditions: overlapping nuclei were excluded and split signals were counted as one chromosome component (Hoang et al. 2000). Stromal and inflammatory cells were excluded from analysis on the basis of the morphological features of their nuclei. The criteria for gene overamplification were (a) >4 signals per cell and (b) the HER2/neu to CEP 17 ratio >2 (Ellis et al. 2000; Ridolfi et al. 2000).
For HER2/neu IHC, 4-μm sections were mounted on glass slides coated with silane (3-aminopropyltriethoxysilane; Sigma), and dried overnight at 37C. The sections were deparaffinized in xylene and rehydrated via a series of graded alcohols. Endogenous peroxidase activity was blocked by incubating the sections in 12 ml of methanol containing 200 μl of 30% H2O2 for 10 min, followed by washing in running tapwater. Antigen unmasking was carried out by microwaving sections in citrate buffer for 10 min. Nonspecific binding sites were blocked by 10% normal swine serum for 10 min. Sections were then incubated with polyclonal anti Her2/neu antibody (Dako A0485; Carpinteria, CA) at 1:1000 dilution in a humid chamber for 30 min at RT. The primary antibody was rinsed off with Tris-buffered saline (TBS) at pH 7.4 and incubated with linking biotinylated antibody for 20 min, followed by incubation with peroxidase-conjugated streptavidin complex (Dako LSAB2 kit, K0675) for 20 min. Freshly prepared DAB solution (5 mg 3, 3'-diaminobenzidine tetrahydrochloride; Sigma D5637) was applied for 10 min after the tertiary layer was rinsed off with TBS. DAB was removed by rinsing with distilled water and slides were counterstained with hematoxylin, and mounted in depex. A breast tumor known to be reactive with HER2/neu antibody was used as positive control in each staining batch. IHC staining was classified according to the criteria recommended by the DAKO protocol: 0, no staining is observed or membrane staining is observed in <10% of tumor cells; 1+, faint/barely perceptible membrane staining is detected in >10% of tumor cells; 2+, weak to moderate complete membrane staining is observed in >10% of tumor cells; and 3+, strong complete membrane staining is observed in >10% of tumor cells.
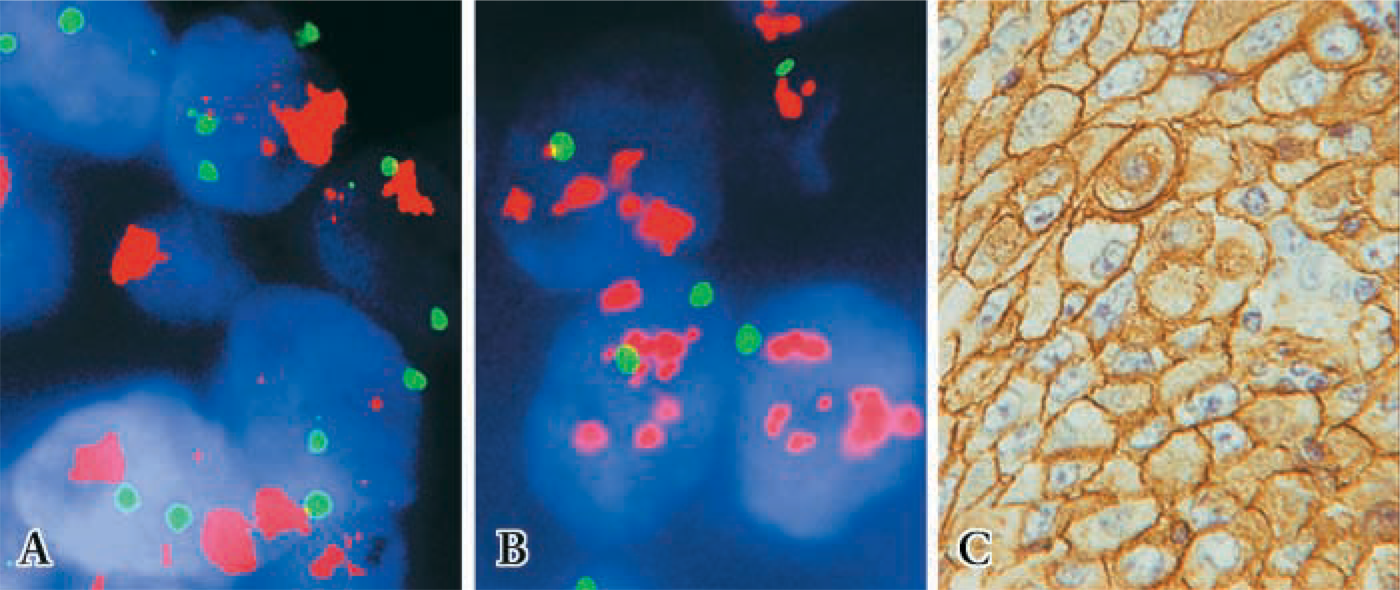
FISH and immunostaining of formalin-fixed breast cancer tissues (case 12) in group 1. (
Statistical analysis was performed using the Graph-Pad Prism statistical Package. The Student's paired t-test was performed to compare means and chi-squared test for comparing proportions. p<0.05 was considered as statistically significant.
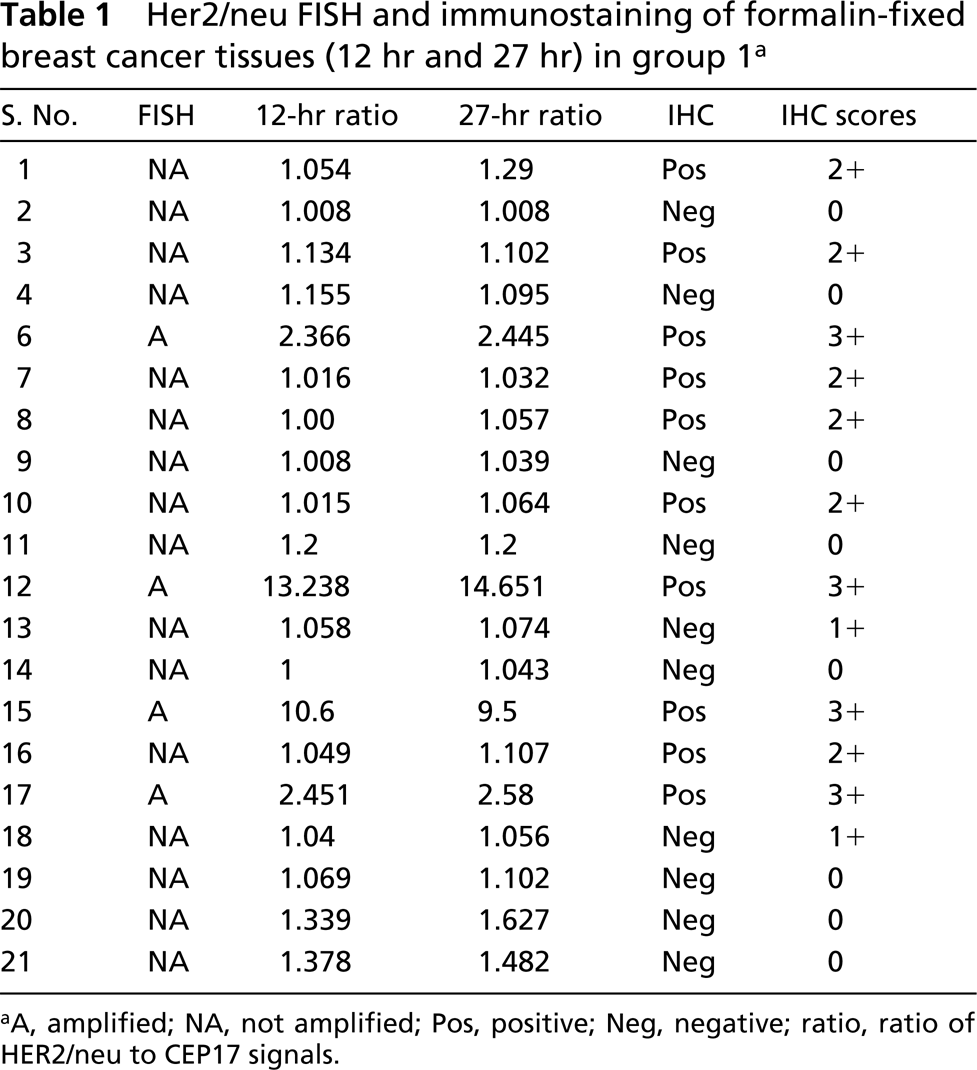
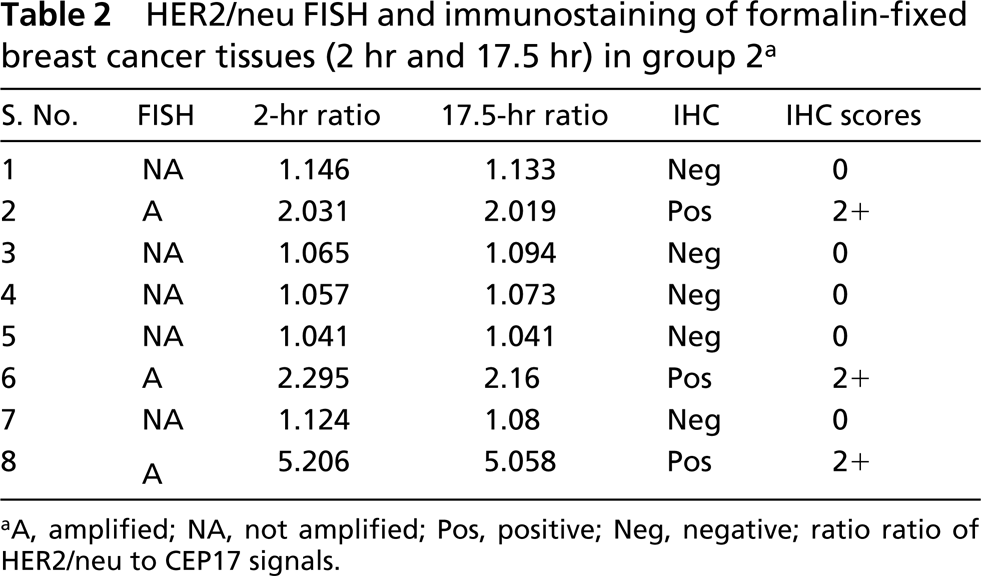
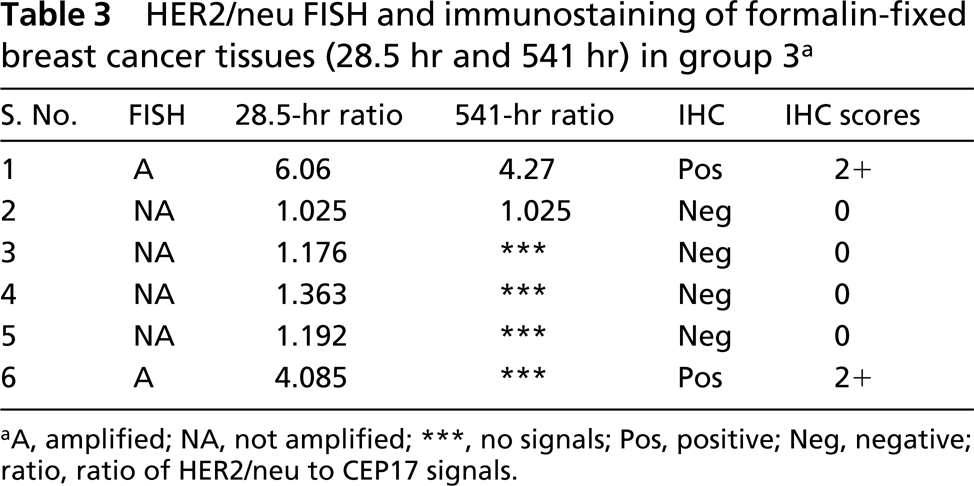
There was no significant difference in detection of FISH signals for the 12-hr or 27-hr fixation protocols in the first group (p=0.476; Table 1; Figures 1A and 1B), indicating that the range of fixation times used routinely in our surgical pathology laboratory did not affect FISH results. Of four amplified cases in this group, two cases revealed very high levels of gene amplification as shown by the HER2/neu/CEP17 ratio of 13.24, 10.6, and 14.65, 9.5 for the 12-hr and 27-hr fixation protocols, respectively. All tissues with 3+ IHC staining disclosed HER2/neu amplification (Figure 1C), whereas those with negative immunostaining (0 and 1+) showed no HER2/neu gene amplification (Table 1). No gene amplification was detected in the five immunopositive cases with 2+ IHC staining. In the second group there was also no significant difference in FISH results between the 2-hr and 17.5-hr fixation protocols (p=0.151; Table 2). In the third group signals were detected in all six cases in the 28.5-hr fixation protocol but only two cases showed signals in the extended-duration fixation protocol (cases 1 and 2; Table 3). In case 2, the fixation duration was approximately a week (the “shortest” period among those with protracted fixation). Case 1 showed high amplification, which likely accounted for the fact that despite prolonged formalin fixation for over 3 weeks, signals were observed, although diminished compared to those of the routinely fixed counterpart. When FISH signals were detected, there was a positive correlation between HER2/neu gene amplification by FISH and protein expression by IHC (p=0.0002) in all the groups.
Her2/neu FISH and immunostaining of formalin-fixed breast cancer tissues (12 hr and 27 hr) in group 1 a
aA, amplified; NA, not amplified; Pos, positive; Neg, negative; ratio, ratio of HER2/neu to CEP17 signals.
HER2/neu FISH and immunostaining of formalin-fixed breast cancer tissues (2 hr and 17.5 hr) in group 2 a
aA, amplified; NA, not amplified; Pos, positive; Neg, negative; ratio ratio of HER2/neu to CEP17 signals.
It appears from our study that a fixation period ranging from 2 hr to 1 week would not affect FISH results. Beyond 1 week however, no signals could be detected, with the exception of the highly amplified case 1 in group 3. Formalin fixation increases the complexity of cell structure and chromatin condensation, making it difficult for the probe to penetrate and interact with the target DNA (Thompson et al. 1994). It has been suggested that the longer the primary fixation, the more aggressive the permeabilization step of pretreatment and enzyme digestion must be (Pauletti and Slamon 1999). However, we obtained similar results for the different fixation protocols in all three groups in this study even though we did not vary the permeabilization step, with the exception of four cases in the extended fixation protocol where no signals were obtained. Our finding is at variance with that of Goelz et al. (1985), who reported previously that DNA isolated from formaldehyde-fixed and embedded tissues was not affected by the length of time in fixation.
HER2/neu FISH and immunostaining of formalin-fixed breast cancer tissues (28.5 hr and 541 hr) in group 3 a
aA, amplified; NA, not amplified; ∗∗∗, no signals; Pos, positive; Neg, negative; ratio, ratio of HER2/neu to CEP17 signals.
The observation of a good concordance between FISH and HER2/neu IHC is in accord with those of other investigators (Couturier et al. 2000; Hoang et al. 2000; Vang et al. 2000). In this study, 2+ IHC staining cases showed no HER2/neu gene amplification whereas in studies with larger samples the HER2/neu gene was reportedly amplified in 12–36% of such cases (Ridolfi et al., 2000; Perez et al., 2002). Possible reasons for the disparity in results obtained by FISH and IHC analyses include transcriptional or posttranscriptional regulation for increased surface receptor expression in the absence of gene amplification (Hoang et al. 2000) and intrinsic variability of the IHC assay in terms of specimen processing and antigen retrieval (Wang et al. 2000).
In conclusion, reliable results for HER2/neu amplification are not compromised by the usual range of routine fixation periods of surgical breast specimens before processing and FISH analysis. Breast specimens fixed for a shortened period are also suitable for FISH analysis. However, a fixation period of more than a week appears to compromise the results obtained by FISH. Further study is necessary to determine if variable Her2/neu FISH results are also obtained on stored paraffin blocks.
Footnotes
Acknowledgements
Supported by a grant from the SingHealth Cluster Research Fund No. BF006/2001.
