Abstract
The Ca2+/calmodulin-activated protein phosphatase, calcineurin, is believed to regulate the development and function of skeletal and cardiac muscle. Striated muscle contains many calcineurin substrates, a few of which have been colocalized or found in molecular complexes with calcineurin. We examined the subcellular distribution of calcineurin in developing rat skeletal muscle cells and adult mouse skeletal muscle fibers by immunofluorescence microscopy. We found low levels of calcineurin immunoreactivity in the cytoplasm of myoblasts and higher levels in cytoplasmic vesicles of myotubes. Most of these vesicles were not immunoreactive for ryanodine receptors and, those that were, represented a small fraction of nascent triad junctions. In adult myofibers, calcineurin was largely associated with triads. Weaker calcineurin immunoreactivity occurred in the sarcoplasmic reticulum at the level of the M line. Unexpectedly, we found tiny clusters of calcineurin associated with nucleoli of developing myofiber nuclei. There were one to three clusters per nucleolus, either within or at the edges of fibrillar centers where ribosomal genes are transcribed. This suggests a role for calcineurin in regulating ribosome synthesis. Our findings suggest a variety of potential new targets and pathways through which calcineurin could regulate skeletal muscle development and plasticity and underscore the importance of spatial specificity in this regulation.
C
Calcineurin consists of two subunits: a 58- to 69-kDa catalytic and calmodulin-binding subunit, calcineurin A, and a regulatory 16- to 19-kDa Ca2+-binding subunit, calcineurin B. Mammalian calcineurin A has three isoforms, α, β, and γ, which are the products of three different genes. Calcineurin B has four EF-hand sites that bind four molecules of Ca2+, one with high affinity (Kd < 10−8 M) and three with affinities in the micromolar range (Aramburu et al. 2000).
Activation of calcineurin depends both on the amplitude and oscillation frequency of [Ca2+]i (Timmerman et al. 1996; Dolmetsch et al. 1997). This ability to function as a calcium flux decoder makes calcineurin an ideal candidate to decipher and transmit specific signals in tissues such as muscle, which routinely experience a variety of calcium fluxes. To date, calcineurin has been well documented to play key regulatory roles in many aspects of both cardiac and skeletal muscle growth and plasticity (Olson and Williams 2000; Wilkins and Molkentin 2004). We previously found that calcineurin has an important role in determining skeletal muscle phenotype (Torgan and Daniels 2001). We found that calcineurin's effects were mediated via both NFAT-dependent and -independent routes, suggesting a variety of targets are involved in calcineurin's ability to regulate muscle phenotype.
Phosphatases are typically positioned near their target substrates, and thus their localization can help shed light on potential substrates (Alto et al. 2002). Therefore, the goal of this study was to examine more closely the intracellular distribution of calcineurin in skeletal muscle with a view toward elucidating potential targets through which it can act. We found calcineurin at nucleolar and cytoplasmic sites that have not previously been shown. Our findings indicate that calcineurin may have more targets and play more roles as a signaling molecule than previously thought.
Materials and Methods
Cell Culture
Skeletal muscle cell cultures were obtained as previously detailed (Dutton et al. 1995) but without the replating step to enrich for myoblasts. Briefly, muscles were stripped from the hindlimbs of 21-day-old fetal rats, trypsinized to dissociate, and plated for 1 hr to remove fibroblasts. Non-adherent cells were plated in 35-mm dishes on 0.5% carbon- and gelatin-coated 13-mm round coverslips (Clay Adams Gold Seal; Thomas Scientific, Swedesboro, NJ) at a density of 0.5 × 106 cells per dish. Cells were fed Dulbecco's modified Eagle's Medium (DMEM) supplemented with 10% horse serum (Gibco BRL; Gaithersburg, MD) and 10% fetal calf serum (Intergen; Purchase, NY) (growth medium). Forty eight hr after cell plating, cultures were refed with 90% DMEM and 10% horse serum to discourage fibroblast growth. Three days later, and every 48 hr thereafter, cultures were refed with 95% DMEM and 5% horse serum. All media also contained penicillin/streptomycin (P/S 100 U/100 mg/ml) and fungizone (2.5 μg/ml). Cells were maintained at 37C, 100% humidity, and 8% CO2 for up to 9 days.
COS-7 cells were plated on coverslips coated with 0.1 mg/ml poly-L-lysine (Sigma-Aldrich; St Louis, MO), in 35-mm dishes at a density of 0.1 × 106 cells per dish. Cells were fed DMEM supplemented with 10% fetal calf serum and maintained at 8% CO2, 37C, and 100% humidity.
Transfection
A GFP-RPA194 construct used to visualize the fibrillar centers of nucleoli was created by inserting a full-length 4125 bp clone of mouse RPA194 as a PseI-ApaI fragment into modified pEGFP-C2 (Dundr et al. 2002) and was a gift from Dr. T. Misteli (National Cancer Institute; Bethesda, MD). Myoblasts were transfected with GFP-RPA194 or GFP alone 5–6 hr after final cell plating using 2 μg of DNA per 35-mm dish with a DNA:transfectant ratio (μg DNA:μl reagent) of 1:3 (FuGENE 6; Roche Molecular Biochemicals, Indianapolis, IN).
Immunostaining
Primary muscle cells or COS-7 cells were rinsed with Dulbecco's phosphate-buffered saline without calcium and magnesium (DPBS) and then fixed with prewarmed (37C) 4% paraformaldehyde in 0.1 M sodium phosphate buffer (pH 7.2–7.4) containing 4% sucrose at room temperature for 30 min. Following a series of washes in DPBS, cells were permeabilized with 0.2% Triton X-100 in DPBS for 30 min and then blocked with 1% BSA, 0.05% saponin, and 10% normal goat serum in DPBS (perm/block solution) to reduce nonspecific antibody binding. Cells were incubated with primary antibodies diluted in perm/block solution (see Table 1 for sources and dilutions) for 1 hr at 37C, washed in perm/block solution for 30 min, and then incubated in secondary antibodies diluted in perm/block solution (see Table 1 for sources and dilutions) for 1 hr at 37C.
In the case of double labeling with two different mouse IgG1 antibodies, cells were first incubated with the calcineurin antibody followed by a Cy3-labeled secondary antibody, refixed with the paraformaldehyde solution for 45 min, reblocked overnight, and then incubated with either the myogenin or nucleophosmin antibodies followed by Alexa Fluor 488- or FITC-labeled secondary antibodies, respectively. Some cells were additionally incubated for 5 min with the nuclear stain Hoechst 33342 (0.5 μg/ml), rewashed, and mounted with Vectashield (Vector Laboratories; Burlingame, CA) to prevent fading.
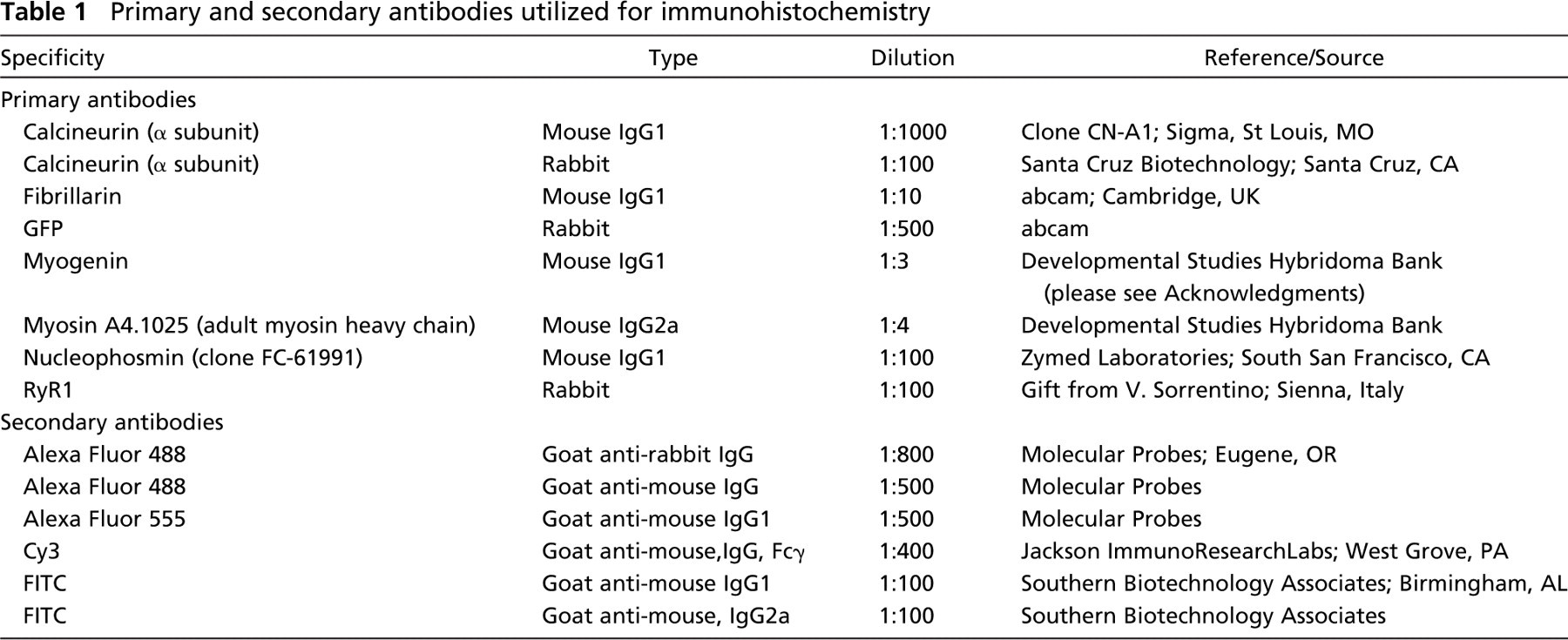
Primary and secondary antibodies utilized for immunohistochemistry
Single Adult Muscle Fibers
Extensor digitorum longus (EDL) and soleus muscles of adult mice were removed, rinsed in saline, pulled taut, and pinned down in a stretched position (EDL) or in a shortened position (soleus) in Petri dishes coated with Sylgard 184 (Dow Corning; Midland, MI). Muscles were washed briefly with DPBS and then fixed for 3 hr in 4% paraformaldehyde and 4% sucrose warmed to 37C. Following extensive rinsing in DPBS, individual fibers were teased apart from the fixed muscles with fine forceps under a dissecting microscope. Fibers were permeabilized with 1% Igepal CA-630 (Sigma-Aldrich) in DPBS for 1 hr and then blocked in 1% BSA, 10% normal goat serum, and 0.1% Igepal in DPBS (fiber perm/block solution) overnight. Fibers were then incubated in primary antibodies diluted in fiber perm/block solution for 2 hr, washed extensively, incubated for 2 hr with secondary antibodies, washed three times, and mounted with Vectashield (Vector Laboratories) onto glass slides for viewing.
Specificity of the monoclonal calcineurin antibody (Table 1) was verified by performing a Western blot. Adult rat tibialis anterior muscle and brain cerebral cortex were homogenized in 1:30 (w/v) HES buffer (20 mM HEPES, 1 mM EDTA, and 250 mM sucrose, pH 7.4) and stored at −80C until analysis. Protein analysis and Western blotting were performed as previously described (Torgan and Daniels 2001) on a 4–12% gel. Membranes were blotted with the monoclonal calcineurin antibody at a dilution of 1:5000, and immunoreactivity was visualized with the ECL system (Amersham Biosciences; Piscataway, NJ). Due to the low abundance of calcineurin in skeletal muscle relative to other proteins, 50 μg of total protein was loaded per lane. Although this large amount of protein resulted in some background bands, a discrete band at the expected size (57–59 kDa in mammals) was clearly visible in the muscle samples and presented as an extremely strong band in brain tissue (Figure 1). Controls for immunostaining included omitting the permeabilization step, as well as replacing the primary antibodies with perm/block solution, both of which eliminated staining.
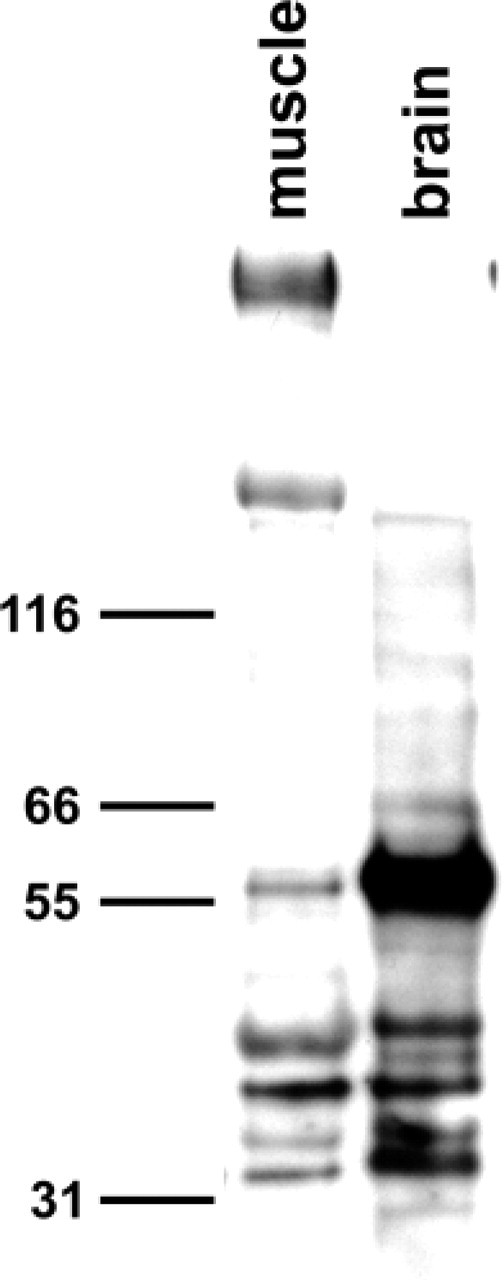
Western blot of homogenates (50 μg of protein) from rat skeletal muscle and brain. The monoclonal calcineurin antibody used in this study labels a band at ∼59 kDa in both muscle and brain, but the band in brain is much stronger due to the higher relative concentration of calcine urin in that tissue. The antibody labels several other bands nonspecifically because of the high concentration of other proteins relative to calcineurin in muscle. Position of molecular weight markers (kDa) is shown on the left.
Photomicroscopy
Cultured cells and adult muscle fibers were viewed with a Zeiss Axioplan IIM microscope and images were recorded with a Zeiss LSM 510 confocal microscope (Carl Zeiss; Thornwood, NY) with a 63× Plan-Apochromat (numerical aperture 1.4) or a 63× Plan-neofluor (numerical aperture 1.3) objective. Images were prepared for publication using Adobe Photoshop (Adobe Systems; San Jose, CA). Brightness and contrast of individual images were adjusted only as necessary to accurately depict details of immunolocalization and colocalization in color overlays.
Results
Calcineurin Expression in Developing Muscle Cells
Calcineurin is believed to play a key role in several aspects of muscle cell development and function (Olson and Williams 2000; Wilkins and Molkentin 2004). Therefore, the initial goal of this study was to assess systematically the distribution of calcineurin in developing muscle cells. Cells isolated from embryonic day 21 hindlimb rat muscle and cultured for 2 days were confirmed to be myoblasts by positive immunostaining for myogenin, one of the myogenic regulatory factors (Figures 2A and 2B). The myoblasts, which demonstrated a classic bipolar shape, exhibited calcineurin throughout much of the cytoplasm in a weak, relatively diffuse pattern, as detected by immunostaining (Figure 2B). Cells other than myoblasts in these cultures showed very little cytoplasmic immunoreactivity for calcineurin. Myoblasts cultured for 5 days fused into multinucleated myotubes (Figure 2C), which expressed calcineurin more robustly than myoblasts throughout the cytoplasm in a punctate pattern (Figure 2D). Thus, expression of calcineurin increases after myoblast fusion.
Localization of Calcineurin Along the Z and M Lines and Colocalization with Ryanodine Receptors
Cells in 9-day-old cultures exhibited organized cross-striations and peripheral nuclei, indicating progressive development from myoblast to myotube to immature myofiber (Figure 2E). Calcineurin staining at this stage revealed a punctate pattern in the cytoplasm that included rows of puncta along the length of the myofiber running parallel to the myofibrils (Figure 2F). This organization was reminiscent of a pattern we previously reported for the ryanodine receptor (RyR) in developing myotubes, where these puncta were also immuno-reactive for the dihydropyridine receptor and hence were thought to be nascent triad junctions (Flucher et al. 1994). To further clarify the location of calcineurin at this stage, we colabeled cells with an antibody against the type-1 RyR. Although both calcineurin and RyR exhibited a pattern consisting mainly of longitudinal rows of puncta that ran the length of the myofiber, only a fraction of puncta that were immunoreactive for either RyR or calcineurin were immunoreactive for both (Figures 2F-2H). Therefore, at this stage of muscle fiber development, most of the cytoplasmic vesicles bearing calcineurin are not likely to be nascent triads.
Localization of calcineurin at or surrounding the Z line in cardiomyocytes has been reported previously (Frey et al. 2000; Heineke et al. 2005). We further examined the sarcomeric and possible triad location of calcineurin in adult mouse single muscle fibers that had been fixed in a lengthened position (sarcomere length of 3 μm). Both calcineurin and RyR appeared as discrete rows of puncta largely colocalized on either side of the Z line in a pattern consistent with that of triads (Figures 2I-2K). In addition, calcineurin immunoreactivity was present at a lower concentration along the M line of the sarcomere (Figures 2I and 2L). The relative position of calcineurin in the sarcomere was confirmed by comparison to myosin heavy chain immunoreactivity in myofibers from soleus muscles fixed in a shortened position. Myosin immunoreactivity occurred in bands alternating with the major bands of calcineurin immunoreactivity. Moreover, calcineurin immunoreactivity was sharply focused in different areas of the optical sections from myosin, indicating that calcineurin surrounds the contractile elements of the sarcomeres (Figures 2L-2N). These results point to a progressive concentration of calcineurin at triads relative to other membrane compartments with myofiber development and suggest the presence of calcineurin in the sarcoplasmic reticulum surrounding the M line.
Nuclear Localization of Calcineurin in Muscle Cells and COS-7 Cells
In addition to calcineurin immunostaining throughout the cytoplasm, we also detected the presence of very small clusters of calcineurin inside a majority of the myotube nuclei (Figure 3A). To confirm the staining pattern, nuclear immunoreactivity in the same pattern was verified with a rabbit antiserum to the calcineurin α subunit (not shown).
Comparison of calcineurin immunoreactivity with phase contrast images suggested a nucleolar localization (not shown). To confirm the nucleolar localization of calcineurin, primary skeletal muscle cells cultured for 6 days were immunostained with antibodies against the abundant nucleolar phosphoprotein, nucleophosmin (Figure 3B). Colocalization revealed that the clusters of calcineurin were associated with nucleoli, often positioned at the edges of these organelles (Figure 3C).
To examine the localization of calcineurin with respect to nucleolar substructure, myoblasts were transfected with a construct that contained GFP-RPA194. RPA194 is a subunit of the RNA polymerase I complex that accumulates in the nucleoli in multiple foci indicative of fibrillar centers (Dundr et al. 2002). Staining of muscle cells cultured for 8 days showed calcineurin clusters in nucleoli among the RPA194 foci (Figures 3D-3F). To distinguish further the location of calcineurin, we double labeled cells with antibodies to calcineurin and fibrillarin, which localizes in the fibrillar centers and dense fibrillar component of the nucleolus (Ochs et al. 1985). The fibrillar centers contain rRNA genes, and the dense fibrillar component surrounding the fibrillar centers is the site of transcription and processing of rRNA. A Z series of 0.58-μm sections showed two to three discrete calcineurin clusters associated with the larger fibrillarin clusters both near the edges and in the centers of nucleoli (Figures 3G and 3H). Due to the small size of these structures, it was not possible to determine whether calcineurin was inside, on the edge of, or in between fibrillarin clusters.
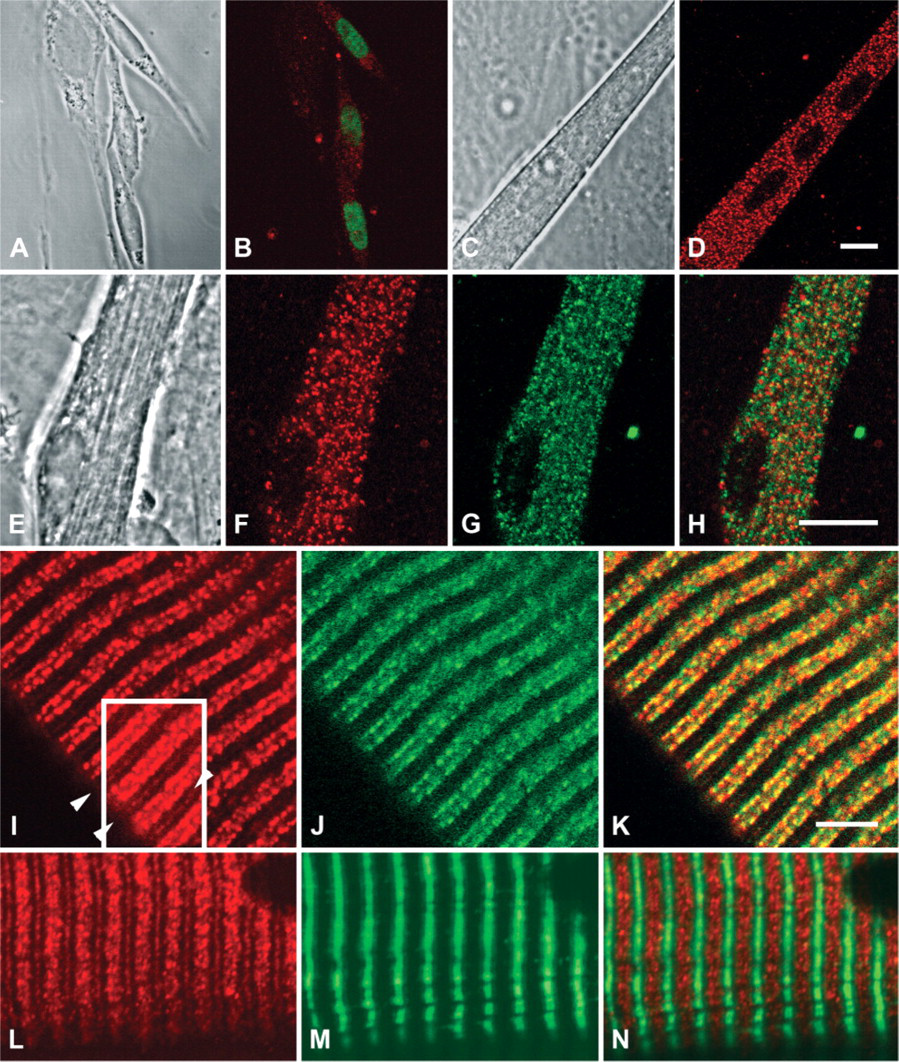
Calcineurin localization in developing primary cultures of rat muscle cells and adult mouse muscle fibers. Top row: Skeletal muscle cells isolated from e21 rats and cultured for 2 days shown in phase contrast (
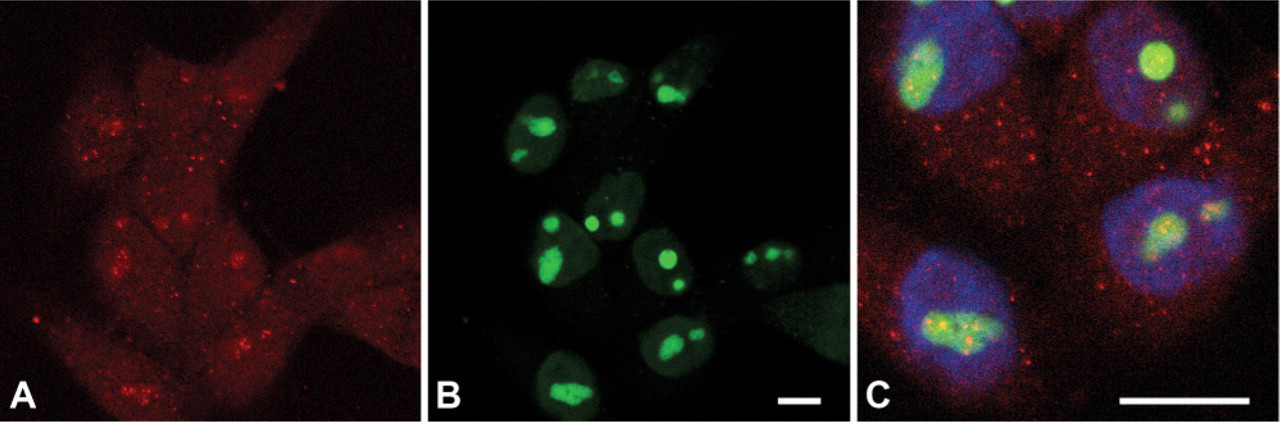
Calcineurin localization in COS-7 cells. A group of COS-7 cells cultured for 3 days express calcineurin (red,
Finally, to determine whether calcineurin is localized to nucleoli in cell types other than muscle, we immunostained COS-7 cells for calcineurin and nucleophosmin. As shown in Figure 4A, these fibroblast-like cells cultured for 3 days expressed calcineurin throughout the cytoplasm. In a minority (<10%) of COS-7 cells, costaining also showed the presence of discrete clusters of calcineurin localized to the nucleoli (Figures 4B and 4C). Interestingly, nucleolar localization of calcineurin most often occurred in small isolated groups of contiguous cells. This may suggest a relationship of nucleolar calcineurin localization to the cell cycle.
Discussion
A recent analysis of the mouse genome identified over 500 candidate protein kinases and over 150 candidate protein phosphatases (Forrest et al. 2003). Mounting evidence suggests that control of these numerous players occurs not only through substrate specificity but via spatial specificity in which key regulators are compartmentalized in the vicinity of their targets (Alto et al. 2002). Because our previous research suggested that calcineurin plays an important role in determining skeletal muscle phenotype through a variety of pathways, we sought to examine the cellular distribution of this phosphatase to elucidate potential targets through which it could be acting.
Calcineurin Distribution in the Cytoplasm during Muscle Development and Localization to the Triad
We detected calcineurin throughout the cytoplasm in myoblasts that also expressed the myogenic transcription factor, myogenin. Our findings support mounting evidence that calcineurin plays key roles in several aspects of muscle cell development. In particular, calcineurin has been shown to participate in muscle differentiation (Delling et al. 2000), in part through transcriptional activation of myogenin (Friday et al. 2000). Calcineurin levels further increased throughout the cytoplasm of myotubes following fusion, consistent with calcineurin's numerous roles in regulating skeletal muscle development that include control of Myf5 gene expression (Friday and Pavlath 2001), myofiber growth (Mitchell and Pavlath 2002), and specification of fiber type (Delling et al. 2000).
As the myotubes matured, calcineurin staining became more prominent and began to exhibit a cytoplasmic pattern that included rows of puncta parallel to the developing myofibrils. Most of these punctae did not appear to be nascent triads. Adult mouse single muscle fibers that had been fixed in a lengthened position exhibited both calcineurin and RyR staining as discrete rows of puncta, largely colocalized on either side of the Z line in a pattern consistent with that of triads. These results extend the work of Sacchetto et al. (2002) who noted the presence of the calcineurin B subunit in a punctate pattern spanning the
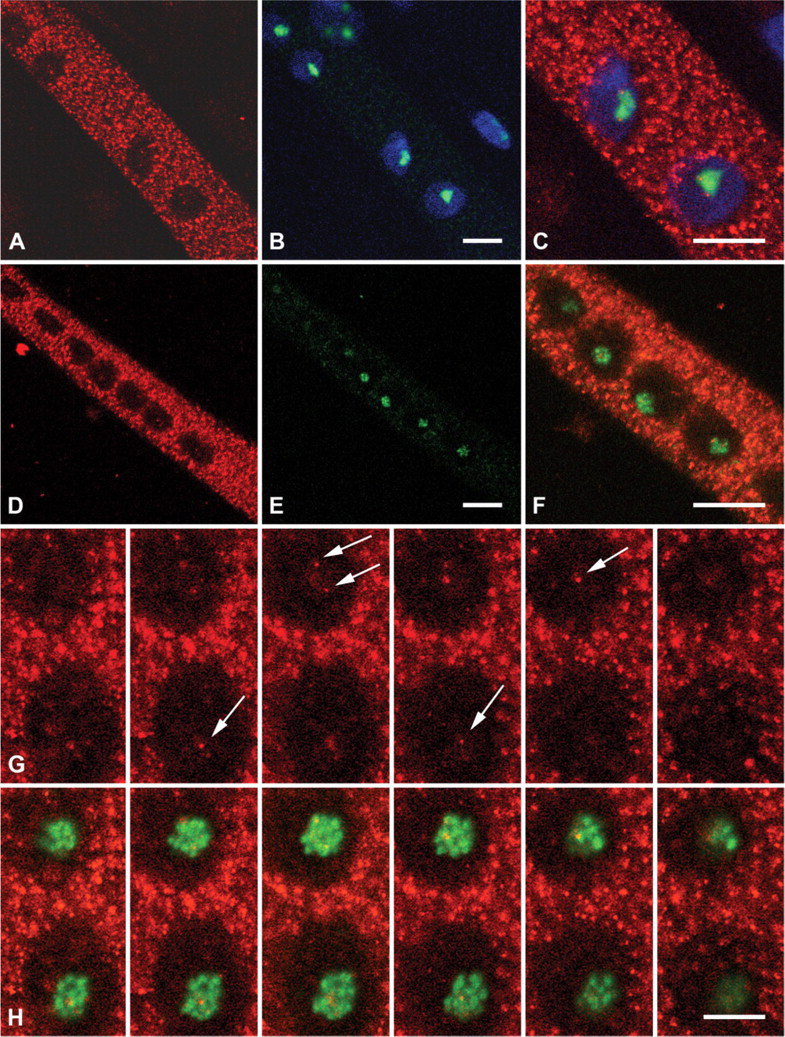
Nucleolar localization of calcineurin in muscle cells. Top row: Rat skeletal muscle cells cultured for 6 days stained for calcineurin (red,
The finding that calcineurin is localized at the triad at first appears to be in contrast to reports that calcineurin is localized to the
Functional Significance of Calcineurin Localization at the Triad
A triad is formed by the juxtaposition of a transverse tubule (T-tubule) with a terminal cistern of the sarco-plasmic reticulum (
In addition to calcineurin localization at the triad, we show for the first time that it is also localized at the M line. The staining occurred in the same focal plane as the triad staining, which suggests calcineurin is localized in a narrow band of the sarcoplasmic reticulum lattice rather than complexed with the myofilament network at the M line. These findings suggest calcineurin may interact with SR proteins in this region of the lattice.
Calcineurin Is Localized in the Nucleoli of Muscle Cells
The most novel and intriguing finding of the present study is the discovery that calcineurin was localized in the nucleoli of muscle cells. The nucleolus is the site of ribosomal RNA (rRNA) synthesis, processing, and the assembly of ribosomes (Spector 2001). This compartment consists of a large, dynamic aggregate of macromolecules that includes the ribosomal genes, precursor rRNAs, mature rRNAs, and rRNA processing enzymes. The nucleolus can be separated into three distinct regions. The ∼30 fibrillar centers in mammalian nucleoli each contain about four rRNA genes, the dense fibrillar component surrounding the fibrillar centers is the site of transcription and processing of rRNA, and the granular region consists of preribosomal particles at various stages of maturation as well as ribosomal subunits (Dundr and Misteli 2001). We attempted to further elucidate the localization of calcineurin within the nucleolus by comparing the distribution of markers for the fibrillar centers and dense fibrillar component. The fibrillar centers were identified by transfecting myoblasts with
This is the first report to demonstrate localization of calcineurin in nucleoli. Previous reports indicate calcineurin in the nuclei in a variety of cell types. In particular, calcineurin was shown to translocate into the nucleus following isoproterenol (Zou et al. 2001) or angiotensin II (Burkard et al. 2005) treatment in cardiomyocytes and to reversibly translocate from the cytoplasm to the nucleus in a complex with NFAT4 in response to calcium signaling in U2OS cells (Shibasaki et al. 1996).
Potential Roles for Calcineurin in the Nucleolus
Calcineurin was found at or on the edges of the fibrillar centers and dense fibrillar component. Given that this is the locale of rRNA transcription and processing, and that there is extensive literature linking calcineurin to hypertrophy in cardiac muscle (Wilkins and Molkentin 2004 and references therein) as well as in skeletal muscle (Musaro et al. 1999; Semsarian et al. 1999), it is tempting to hypothesize a role for this phosphatase in regulation of global ribosome biogenesis. Indeed, had we found that calcineurin extensively colocalized with RPA194 throughout nucleoli, this case could be argued. Calcineurin, however, was not associated with a large number of multiple rRNA synthesis and processing sites but rather presented as only a very few discrete clusters both in myotubes and COS-7 cells. Thus, calcineurin appears to be associated with a subset of ribosomal synthetic sites, although it is possible that there are many sites within each nucleolus that can bind calcineurin, but only a few at a time are in the appropriate state to do so. Given this spatial specificity, it will be intriguing to discern whether calcineurin represents one partner in a domain that may function as a ‘transcription factory’ similar to previously described discrete regional domains (Pombo et al. 1998), and whether the discrete clusters include transcription factors or specific chromosomal loci.
In further support of a role for calcineurin in the nucleus, all the key players involved in calcineurin activation and inhibition are also present. In particular, the required activator of calcineurin, calmodulin, is present in the nucleolus (Portoles et al. 1994), whereas cyclophilin A, the target of the potent calcineurin inhibitor cyclosporine, has recently been shown to reside in the nuclei of yeast cells (Arevalo-Rodriguez and Heitman 2005). Intriguingly, certain calcium waves that occur in the nuclei of primary muscle cells are distinct from those that occur in the cytoplasm (Jaimovich et al. 2000). These nuclear calcium waves, as well as some waves that pass through both cytoplasm and nucleus, have patterns that correspond with those required for calcineurin activation of downstream events (Timmerman et al. 1996; Dolmetsch et al. 1997). In addition, intranuclear extensions of the nuclear membrane may form a “nucleoplasmic reticulum” that allow for the release of calcium in discrete regions within the nucleus (Echevarria et al. 2003) that are associated with nucleoli (Lui et al. 2003). This reticulum is rich in inositol 1,4,5-trisphosphate receptors (IP3R) (Echevarria et al. 2003), which are associated with the slow calcium waves in muscle cells (Jaimovich et al. 2000). IP3R are a target of calcineurin at both the channel and gene level (Bultynck et al. 2003), thus suggesting coordination of complex signaling events through a number of feedback loops.
In conclusion, calcineurin plays a variety of key roles in regulation of muscle development and plasticity through a number of diverse substrates and pathways. As phosphatases are often positioned near their target substrates, our findings regarding the subcellular distribution of calcineurin in skeletal muscle suggest a variety of potential new targets and pathways through which this phosphatase could function. The findings underscore calcineurin's ability to modulate signaling pathways not only through substrate specificity and calcium concentration specificity, but also through spatial specificity.
Footnotes
Acknowledgements
The assistance of Yvonne Chak (cell culture) and Dr. Xufeng Wu (confocal microscopy) is gratefully acknowledged. Monoclonal antibodies against myogenin (F5D, developed by Dr. W.E. Wright) and myosin (A4.1025, developed by Dr. H. Blau) were obtained from the Developmental Studies Hybridoma Bank developed under the auspices of the National Institute of Child Health and Human Development and maintained by the University of Iowa, Department of Biological Sciences, Iowa City, Iowa. We thank Dr. Vincenzo Sorrentino (Department of Neuroscience, Sienna, Italy) for the kind gift of the ryanodine receptor RyR1 antibody. We thank Dr. Tom Misteli (National Cancer Institute, Bethesda, MD) for the gift of the GFP194 construct and for many insightful conversations. We also thank Dr. Kathleen McCormick for thoughtful reading of the manuscript. We regret the omission of many outstanding references due to space constraints.
