Abstract
Vitamin D receptor (VDR) and 25-hydroxyvitamin D3 1-α-hydroxylase expression have recently been shown to be upregulated in several tumors and thought to represent an important endogenous response to tumor progression. Little is known about the expression of these proteins in thyroid carcinoma, although previous reports have documented evidence of the biological effect of vitamin D in thyroid cells. Using paraffin-embedded and frozen sections of papillary thyroid carcinoma, we utilized real-time quantitative RT-PCR and immunohistochemistry to characterize the expression of VDR and 1-α-hydroxylase in thyroid follicular cells, with special emphasis on papillary thyroid carcinoma (PTC). VDR and 1-α-hydroxylase expression were increased in PTC compared with normal thyroid tissue and especially high in areas of lymphocyte infiltration. Expression of VDR and 1-α-hydroxylase in PTC may be compatible with an overall favorable prognosis for this tumor type and may constitute important prerequisites for using vitamin D and/or vitamin D analogs in the treatment of PTC.
P
Active vitamin D3 and its analogs have recently become potentially therapeutic agents for treatment of patients with various tumors such as colon or breast carcinoma (Thomas et al. 1999; Verlinden et al. 2000). Expression of the vitamin D receptor (VDR) and 25-hydroxyvitamin D3 1-α-hydroxylase (1α-hydroxylase), the rate-limiting enzyme in production of active vitamin D3, has been found in several tissues and tumor types (Zehnder et al. 2001). Active vitamin D is important for regulation of differentiation and cell growth in many different organs, and expression of 1α-hydroxylase/VDR has been regarded as an important response against tumor progression. Active vitamin D3 acts by binding to its nuclear VDR, which may form heterodimers with the retinoic receptors (RAR, RXR). In several tumors, vitamin D has an antiproliferative and differentiating effect (Cross et al. 1992; Miller et al. 1995; Kawa et al. 1996; Okano et al. 1999; Suzuki et al. 1999; Pourgholami et al. 2000; Verlinden et al. 2000; Verstuyf et al. 2000). Several potential mechanisms for these actions have been proposed (Reitsma et al. 1983; Studzinski et al. 1993). In a subgroup of patients with thyroid carcinoma, retinoic acid may increase radioiodine uptake and be used for adjuvant therapy (Simon et al. 1998).
In the present study we have investigated expression of VDR and 1α-hydroxylase in PTC and normal thyroid tissue to clarify a possible endogenous vitamin D regulatory response in tumors and prerequisites for potential use of vitamin D and vitamin D analogs or precursors for treatment of PTC.
Materials and Methods
Patients and Tumors
Thirty five patients (26 females) with PTC, mean age of 44 ± 18 years, were chosen for analysis because they represented different tumor stages. The patients lived in Sweden or Germany and had not been exposed to irradiation from the Chernobyl accident. Paraffin-embedded specimens from primary tumor (n=35) or lymph node metastases (n=9) were collected and processed for immunohistochemistry. Immediately snap-frozen tumors as well as normal thyroid tissue were used for evaluation of mRNA expression. Paraffin-embedded specimens and frozen tissue were partially derived from different patients. All tumors were classified according to the tumor–node–metastasis (TNM) stage, as well as the tumor stage adjusted for age according to the American Joint Commission on Cancer/International Union against Cancer (Rosai et al. 1992; Bearhs 1997). Radioiodine uptake efficiency at the time of therapeutic postoperative treatment was noted for correlation with expression of 1α-hydroxylase and VDR.
Immunohistochemistry
Paraffin-embedded 5-μm sections were dewaxed in xylene and rehydrated through decreased concentrations of ethanol. Immunohistochemistry for 25(OH)-1α-hydroxylase was performed with the use of a polyclonal sheep antiserum (diluted 1:150; kind gift of Dr. M. Hewison, Birmingham, UK) (Zehnder et al. 2001). This antiserum has been thoroughly characterized and is specifically directed against 1α-hydroxylase as described (Bland et al. 1999; Zehnder et al. 2001). Antigen retrieval was performed in a microwave oven for 2.5 min in 10 mM citric acid solution (pH 6.0). Endogenous peroxidase activity was blocked by using 0.3% H2O2 in methanol for 15 min. The 1α-hydroxylase antiserum was then applied to the tissue sections (n=38), which were incubated for 90 min at room temperature. After washing, the slides were exposed for 30 min to biotin-labeled donkey anti-sheep Ig (1:500; Vector Laboratories, Inc., Burlingame, CA), after which avidin and development of visible staining followed (Vectastain ABC kit; Vector Laboratories). All reagents were diluted in 0.1 M Tris-HCl buffer (pH 7.6) containing 5% normal swine serum.
Negative control sections included use of primary 1α-hydroxylase antiserum incubated with an excess of immunizing peptide (RHVELREGEAAMRNQGKDEEDMPS) or substitution of primary antiserum.
For immunostaining of VDR, we utilized a well-characterized rabbit Ig polyclonal antibody (C20; Santa Cruz Biotechnology Inc., Santa Cruz, CA) on 49 sections (7 from lymph node metastases). After blocking of endogenous peroxidase activity the tissue was hydrolyzed in 2 M HCl at 37C for 30 min. Nonspecific antibody binding was blocked with normal goat serum (1:10) for 20 min (Thomas et al. 1999). The primary C20 antiserum was applied (diluted 1:100) for 90 min at room temperature, followed by a biotinylated goat anti-rabbit Ig antibody (1:200; Vector Laboratories) for 30 min and finally avidin for another 30 min. Tris-buffered saline (pH 7.4) was used as a rinsing buffer, and all reagents were diluted in 2% BSA in Tris-buffered saline (pH 7.4).
Negative control sections for VDR staining included use of primary antibody incubated with an excess of specific blocking peptide (sc-1008p; Santa Cruz Biotechnology) or replacement of the primary antibody with a nonspecific antiserum. Slides were developed using 3-amino-9-ethylcarbazol and counterstained with Mayer's hematoxylin.
Kidney tissue is known to express 1α-hydroxylase and VDR and was used as positive control. Immunohistochemical staining was assessed semiquantitatively by two independent observers and in comparison with proximal tubule cells. Staining intensity was divided into 0) absent staining, 1) low expression, 2) medium expression, and 3) high expression.
Real-time Quantitative RT-PCR Analysis
Real-time quantitative RT-PCR (Applied Biosystems; Foster City, CA) was performed on RNA extracted from normal thyroid glands (n=5), primary PTC (n=16), and lymph node metastases of PTC (n=7). All tissues were obtained from patients undergoing neck exploration and were snap frozen in liquid nitrogen and stored at −70C. To avoid gross contamination of non-tumor cells, specimens were microdissected in a cryostat. Tumor pieces subjected to RNA extraction usually comprised more than 75–90% tumor cells as determined by histochemical evaluation. Total RNA was extracted with Trizol Reagent (Gibco BRL; Life Technologies, Gaithersburg, MD) according to the manufacturer's instructions. RNA was treated with RQ1 DNase (Promega Corp.; Madison, WI) and proteinase K. Using a first-strand cDNA synthesis kit (Amersham Biosciences; Uppsala, Sweden), total RNA was transcribed to cDNA using random primers. Expression levels of 1α-hydroxylase and VDR mRNA were compared with that of glyceraldehyde-3-phosphate dehydrogenase (GAPDH) in each sample. Real-time quantitative PCR was performed on an ABI PRISM Sequence Detecting System (Applied Biosystems) as previously described (Segersten et al. 2002). cDNA-specific PCR primers and the fluorescence-labeled oligonucleotide probe for 1α-hydroxylase, VDR, and GAPDH were (5′–3′) 1α-hydroxylase-F: TTG CTA TTG GCG GGA GTG G; 1α-hydroxylase-R: TGC CGG GAG AGC TCA TAC AG; 1α-hydroxylase probe: AGC GTG TCC AAC ACG CTC TCT TGG G; VDR-F: GCA TCC AAA AGG TCA TTG GC; VDR-R: TCA CGT CAC TGA CGC GGT AC; VDR-probe: CCT GGA CCT GTG GCA ACC AAG ACT ACA; GAPDH-F: GAA GGT GAA GGT CGG AGT C; GAPDH-R: GAA GAT GGT GAT GGG ATT TC; GAPDH-probe: CAA GCT TCC CGT TCT CAG CC.
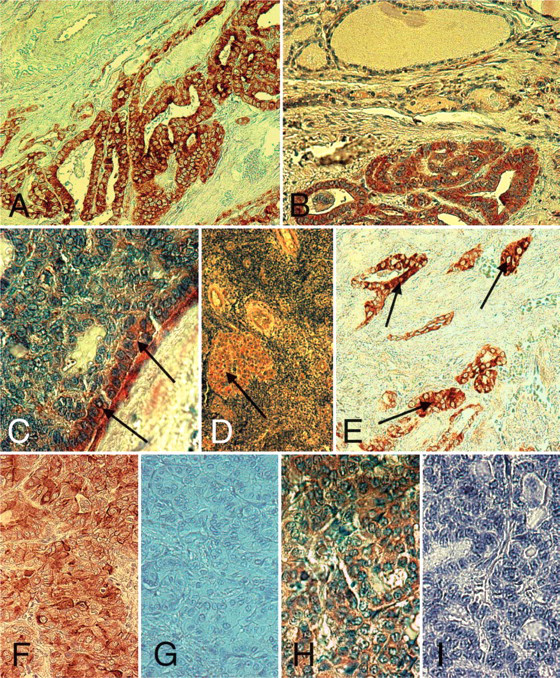
1α-Hydroxylase and vitamin D receptor (VDR) immunoreactivity in papillary thyroid carcinoma (PTC). (
Each cDNA sample was analyzed in triplicate. All reagents were supplied in the TaqMan PCR core Reagent Kit (Applied Biosystems). PCR reactions had a final volume of 50 μl and contained 5 μl cDNA template, TaqMan buffer A1 X, 5.5 mM MgCl2, 200 μM of dATP, dCTP, dGTP, 400 μM dUTP, 100 nM probe, 200 nM of each primer, 0.01 U AmpErase UNG, and 0.05 U AmpliTaq Gold.
Statistical Analyses and Ethics
All values are presented as mean ± SEM. ANOVA followed by Fisher's Protected Least Significant Difference was used to calculate differences among investigated groups; p<0.05 was considered significant. All calculations were done with the StatView software (Stata Corp.; Cary, NC). The work was approved by the local Ethics Committee.
Results
1α-Hydroxylase immunohistochemical staining was not detected in normal thyroid follicular cells and was positive in 33/38 samples with PTC. Follicular thyroid cells without any evidence of malignant changes on the same section but outside the PTC tumor were considered as normal cells. High expression was seen in 10 PTC specimens (26.3%), medium expression in 17 (44.7%), and low expression in 6 (15.8%), whereas 5 (13.2%) were considered negative. The staining was generally diffuse and cytoplasmic, and in nine cases the expression was more intense close to the tumor capsule. In 10 primary PTC specimens, lymphocyte infiltration was evident, and all these specimens were highly positive for 1α-hydroxylase staining (Figure 1). In several sections, the area of the tumor with lymphocyte infiltration demonstrated higher 1α-hydroxylase expression than areas without lymphocytes. No clear correlation between staining intensity and TNM stage was seen. There was no clear correlation between staining intensity for 1α-hydroxylase and uptake levels of radioiodine.
Immunohistochemistry for VDR revealed positive staining in 43/44 PTC specimens. High expression was evident in 26 cases (59.1%), medium in 13 (29.5%), and low in 4 (9.1%), whereas one (2.3%) was considered negative. Immunohistochemical expression of VDR was most obvious in the cytoplasm but in ∼20% of the cells also in the nucleus. In four (9.1%) specimens, an enhanced VDR expression was detected close to the tumor border, in the vicinity of the capsule. Similar to the 1α-hydroxylase staining, PTC cells within areas of lymphocyte infiltration demonstrated more intense VDR expression (n=10; Figure 1). Normal thyroid follicular cells, designated similarly as for 1α-hydroxylase staining, were usually negative, as well as in normal thyroid sections from patients not suffering from PTC. However, in seven sections (15.9%), small areas of normal follicular cells situated outside the tumor capsule and adjacent to a highly positive tumor demonstrated positive, albeit low, staining.
Immunohistochemical staining of 1α-hydroxylase correlated with that of VDR. The tumor that was negative for VDR was also negative for 1α-hydroxylase. This tumor was classified as T2N1M0. Generally, no correlation between VDR immunostaining and TNM stage, sex, or age or radioiodine uptake was present.
Real-time Quantitative RT-PCR
Results from the real-time quantitative RT-PCR analyses using cDNA-specific primers and probes did not show large differences in 1α-hydroxylase expression in PTC and PTC metastasis compared with normal tissue (Figure 2; ratios 6.8 ± 2.0; 6.4 ± 2.3 vs. 10.8 ± 4.1, respectively). A large spread of the sample data was evident (∼10-fold) and was notable in all three sample groups.
Results from the real-time quantitative RT-PCR analysis for VDR showed more evident difference among the three sample groups (Figure 3). VDR/GAPDH ratios varied considerably within the PTC as well as the normal thyroid tissue (mean 17.7 ± 10.0 and 5.9 ± 2.4, respectively). Data for the PTC samples were spread within a larger range than the normal thyroid tissue, and it was possible to divide them among three groups expressing high, medium, and low VDR mRNA levels compared with normal thyroid tissue. VDR/GAPDH ratio was significantly lower in the lymph node PTC metastases (1.5 ± 0.3) compared with both normal thyroid tissue (5.9 ± 2.4) and primary PTC (17.7 ± 10.0; p<0.05). However, primary tumors associated with the investigated lymph node metastases were spread among the groups expressing high, medium, and low VDR mRNA levels. Within the investigated specimens, a group of six primary PTC tumors displayed concomitantly low 1α-hydroxylase/GAPDH and VDR/GAPDH ratios. TNM stage of these tumors did not differ from the rest of the tumors.
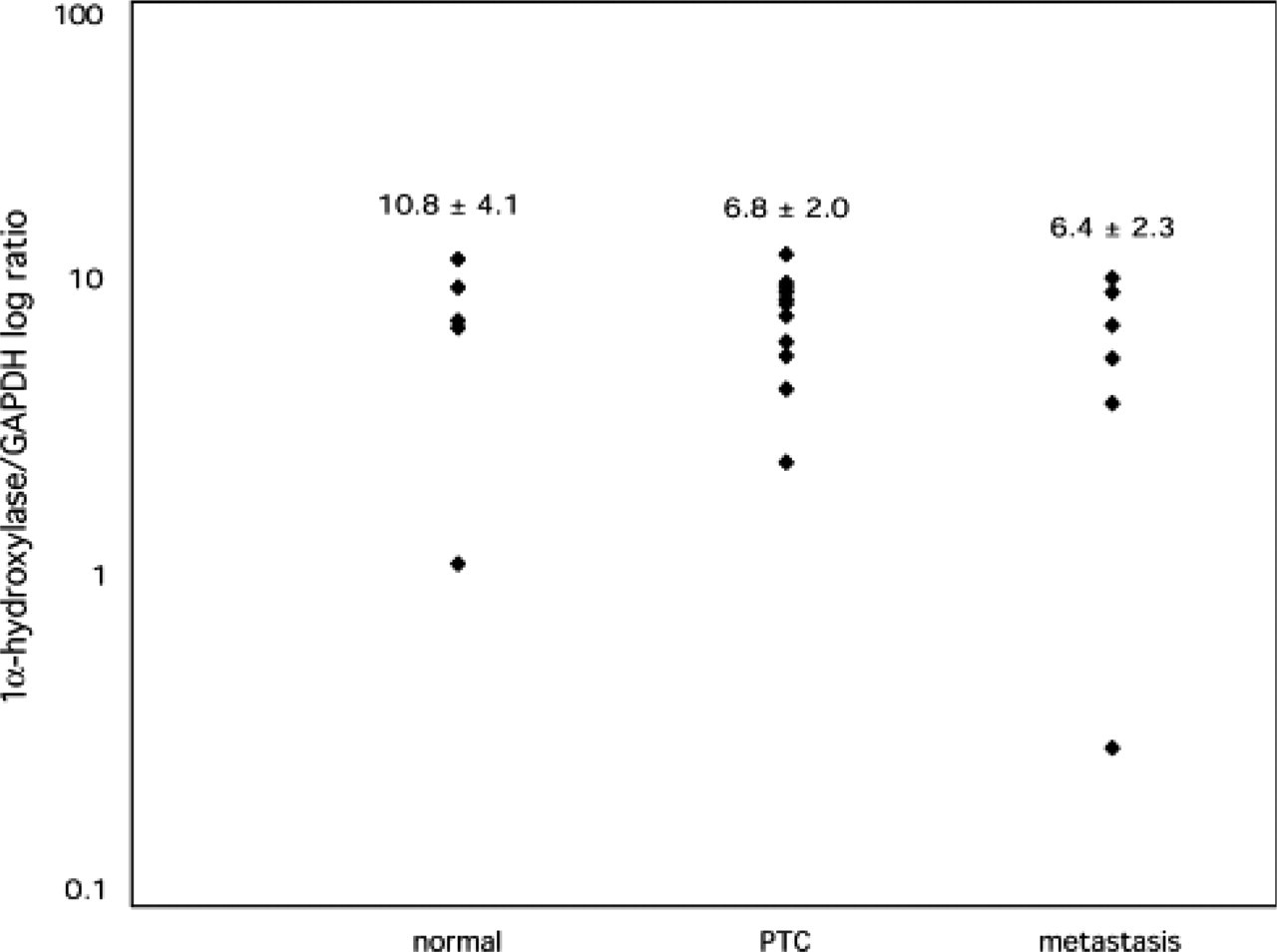
Results of quantitative RT-PCR analyses using cDNA-specific primers for 1α-hydroxylase. Numbers presented as ratio of 1α-hydroxylase and glyceraldehyde-3-phosphate dehydrogenase (GAPDH) cDNA expression, to normalize against mRNA amount and quality in each sample (normal, normal thyroid tissue; PTC, primary papillary tumor in the thyroid; metastasis, tumor tissue from lymph node metastases).
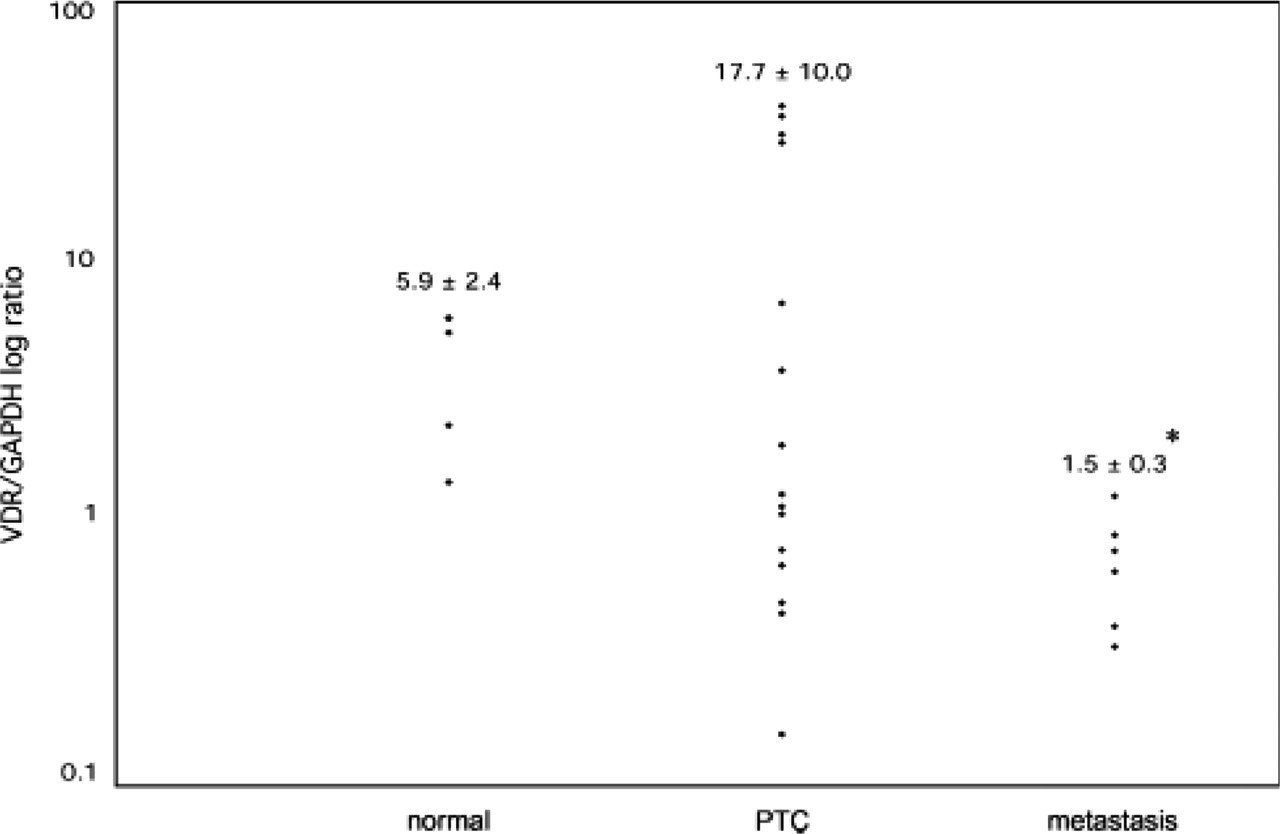
Results of quantitative RT-PCR analyses using cDNA specific primers for VDR. Numbers presented as ratio of VDR and GAPDH cDNA expression, to normalize against mRNA amount and quality in each sample (normal, normal thyroid tissue; PTC, primary papillary tumor in the thyroid; metastasis, tumor tissue from lymph node metastases). ∗p<0.05 toward normal thyroid tissue as well as PTC tissue.
Overall, no correlation between 1α-hydroxylase/GAPDH and VDR/GAPDH ratios and stage of disease, age, or sex were found.
Discussion
The present study demonstrates expression of 1α-hydroxylase and VDR in PTC at both the protein and mRNA levels. Although mRNA expression levels did not differ significantly between primary PTC and normal thyroid tissue, immunohistochemistry revealed evident signs of higher expression of 1α-hydroxylase and VDR proteins in PTC. The results demonstrate the presence of molecular prerequisites for a role of vitamin D in the thyroid, particularly in PTC.
VDR is expressed in normal thyroid follicular cells and PTC cells in culture (Lamberg-Allardt et al. 1991; Suzuki et al. 1999). Among the few reports about vitamin D effects in thyroid cells, the analog 22-oxacalcitriol was shown to inhibit cell proliferation of thyroid carcinoma cells in culture (Okano et al. 1999; Suzuki et al. 1999), indicating a similar differentiating role for vitamin D in thyroid tumors as described for other tissues (Verstuyf et al. 2000). Recent studies have further substantiated an important differentiating and anti-proliferative effect of vitamin D on thyroid carcinoma cell lines derived from papillary as well as follicular cancer, possibly mediated through accumulation and dephosphorylation of nuclear p27 (Liu et al. 2002; Dackiw et al. 2004).
The enzyme 1α-hydroxylase, previously exclusively expressed in renal tubule cells, was recently expressed in a wide variety of tissues, i.e., parathyroid, prostate, cervix, colon, breast, and pancreas (Cross et al. 1997; Bell 1998; Schwartz et al. 1998; Friedrich et al. 2001; Reichrath et al. 2001; Zehnder et al. 2001; Segersten et al. 2002). These findings indicate a potential local production of active vitamin D in the target tissues themselves, as long as the precursor 25-hydroxyvitamin D3 is supplied. The present results from the thyroid clearly demonstrate expression of 1α-hydroxylase in normal thyroid, although more evident on the mRNA level. In PTC the expression is clearly higher than in normal thyroid tissue. The results indicate that thyroid tissue, and especially PTC, may be added to the list where local production of active vitamin D may occur. It is interesting to note that the precursor 25-hydroxyvitamin D3, bound to its transporting serum protein DBP (vitamin D-binding protein), is taken up through the cell surface protein megalin in proximal tubule cells in the kidney (Nykjaer et al. 1999) and presented to the 1α-hydroxylase enzyme expressed in these cells. Megalin is also expressed in thyroid follicular cells and, thus, the special molecular prerequisite for 25-hydroxyvitamin D3 uptake is present in the thyroid (Marino et al. 1999).
Immunohistochemistry results indicated a higher difference between tumor and normal thyroid tissue 1α-hydroxylase protein than mRNA expression. One explanation might be a faster degradation of the protein in normal follicular cells compared with PTC cells. The finding of higher expression of 1α-hydroxylase in a subset of normal follicular cells close to the tumor may indicate that a factor released from tumor cells or infiltrating immunological cells mediate higher expression of the enzyme. It is known from the situation in sarcoid granuloma that interferon-α released from immunocompetent cells increases expression of 1α-hydroxylase (Reichel et al. 1987). Such cells are often present in PTC, appearing as lymphocyte infiltration. It is also noted in our immunostainings that the expression of 1α-hydroxylase is increased in the tumor cells in the immediate vicinity of the lymphocyte accumulations. VDR expression demonstrated a pattern similar to that of 1α-hydroxylase. Thus, expression was high in PTC, both on the protein and mRNA levels, but lower in normal thyroid tissue.
Immunohistochemistry revealed low, scarce, or even absent VDR staining in normal thyroid. However, using the more sensitive technique of quantitative RT-PCR, expression was nevertheless demonstrated. It is interesting to note the higher VDR expression in normal thyroid follicular cells just outside the tumor capsule and the increased VDR expression in PTC cells close to areas of lymphocyte infiltration, in parallel to what was noted for the 1α-hydroxylase staining. The presence of 1α-hydroxylase and the potential local production of active vitamin D3 may induce VDR expression as seen in other tissues. For example, in keratinocytes, vitamin D itself or proteosome inhibitors cause increased VDR protein expression without affecting VDR mRNA levels (Li et al. 1999). However, the mechanisms for transcriptional and translational regulation of 1α-hydroxylase and VDR in the thyroid and in PTC are unclear, and further investigations are needed.
Effects of vitamin D in the thyroid may be important in relation also to known effects of retinoic acids. In a subset of patients, retinoic acid may increase the uptake of radioiodine into tumor cells, possibly by affecting the iodine symporter or increasing the differentiation level. In several systems, retinoic acid and vitamin D act in concert through heterodimerization of their respective receptors. Although no correlation between expression levels of 1α-hydroxylase/VDR and radioiodine uptake in the remaining tissue in these patients was found, the present results continue to support such an effect in thyroid tumors with increased expression of VDR. Indeed, documented radioiodine uptake levels did not reflect the extirpated tissue that we performed expression analyses on.
The higher protein expression of 1α-hydroxylase and VDR in PTC compared with normal thyroid cells may be associated with differentiation, reduced proliferation, and the generally favorable prognosis in PTC. Corroborating this, VDR expression was significantly lower in PTC metastases. The results are similar to those in colon adenocarcinoma, where expression of VDR and 1α-hydroxylase mRNA decrease to subsequently lower levels during advancement from low- to high-grade cancer (Cross et al. 2001). In addition, vitamin D may induce differentiation, at least on a cellular level and in animal experiments (Liu et al. 2002; Dackiw et al. 2004).
In summary, the expression of 1α-hydroxylase/VDR in PTC tumor cells may be utilized for treatment of PTC. Thus, the present results support that vitamin D analogs or the precursor 25-OH-vitamin D may be useful as adjuvant treatment in PTC.
Footnotes
Acknowledgements
The Swedish Institute and the Swedish Cancer Foundation supported this work.
We are greatly indebted to Birgitta Bondeson, Daniel Lindberg, and Peter Lillhager for excellent technical assistance and advice.
