Abstract
We have used a new family of zinc-specific-responsive fluorescent dyes (ZPs) to study the sequestration and secretion of zinc from Paneth cells, which are located in the bases of the crypts of Lieberkühn within the rat small intestine. Vivid ZP fluorescence zinc staining of Paneth cell secretory granules is seen in both cryostat sections and isolated crypts, providing firm evidence for a pool of labile (rapidly exchangeable) zinc within these cells. We further demonstrate that this ionic zinc pool is secreted under physiological conditions. In vivo stimulation of the small intestine by IP injection of the secretagogue pilocarpine results in discrete zinc staining within the lumens of subsequently isolated crypts, concomitant with a decrease in the zinc staining of Paneth cell granules located within the same crypts. In contrast, the secretion of zinc into the lumens of isolated crypts stimulated in vitro with either carbachol or LPS (lipopolysaccharide) is not observed. However, a distinct change in Paneth cell morphology, suggesting attempted secretion, is seen in response to the direct application of cholinergics but not LPS. These findings suggest that zinc is coreleased with other Paneth cell anti-microbials, and that the intact intestine is necessary for secretion into the crypt lumen.
P
The antimicrobial properties of cryptidins have been well studied, and zinc has been shown to play an important role in cryptidin activation by matrilysin, a zinc-dependent metalloprotease (Soler et al. 1994; Wilson et al. 1999). In addition, a depletion of both Paneth cell granules and Paneth cell numbers has been observed in zinc-deficient states, whether induced experimentally, e.g., by zinc chelation, or due to dietary insufficiency (Kelly et al. 2004). Furthermore, zinc itself has antimicrobial properties. Studies in developing countries have demonstrated that zinc supplementation prevents diarrhea and pneumonia (Bhutta et al. 1999; Umeta et al. 2000), leading the World Health Organization and the Indian Academy of Pediatrics to recommend zinc for the treatment of diarrhea. When Sweden banned the use of antibiotic growth promoters in livestock, it was discovered that the addition of zinc oxide to pig feed prevented weaning diarrhea as effectively as the antibiotic growth promoter olaquindox (Holmgren 1994). The antimicrobial action of the zinc ion indicates that labile Zn2+ contributes to the antimicrobial properties of Paneth cell secretions.
To test whether labile (rapidly exchangeable), secretable Zn2+ is present in Paneth cell secretory granules we used the new Zinpyr (ZP) family of zinc-specific-responsive fluorescent dyes to stain cryostat sections and live, isolated crypts from rat small intestine. The non-toxic, fluorescein-based ZP dyes are ideal for studying zinc in biological samples. ZP1 and ZP3 are membrane-permeant probes, which give highly Zn2+-specific responses as demonstrated by metal ion selectivity studies in vitro (Burdette et al. 2001; Chang et al. 2004). Among other advantages, these probes are up to 50 times brighter than the quinoline-based reagents (TSQ, Zinquin, TFLZn) traditionally used for staining ionic Zn2+ in biological samples (Burdette et al. 2001; Frederickson 2003; Chang et al. 2004; Woodroofe et al. 2004). In addition, the ZP dyes fluoresce green when excited with blue light, thus preventing UV-induced cell damage that can occur from the near-ultraviolet excitation of the quinoline-based probes. Paneth cell zinc secretion was examined in both intact animals and isolated crypt preparations using the bacterial antigen lipopolysaccharide (LPS) and cholinergic secretogogues as stimuli, followed by ZP staining to visualize Zn2+.
Materials and Methods
All of the experiments described herein were performed in compliance with Institutional Animal Care and Use Committee guidelines according to approved protocols.
Preparation of Isolated Rat Intestinal Crypts
Adult male and female Sprague Dawley rats were fasted overnight and then deeply anesthetized with isoflurane. After decapitation, the small intestine was removed, cut into 1–2-cm lengths, and either frozen on a liquid-CO2 zinc-free stage or placed in ice-cold physiological medium: (mM) 147 NaCl, 4.0 KCl, 3 CaCl2, 0.9 MgCl2, 11 HEPES, 10 glucose, pH to 7.4. Isolated intestinal crypts were obtained as described below, following literature methods (Ayabe et al. 2000). Briefly, the 1–2-cm lengths of intestine were everted and crypts were “shaken” free by gentle agitation in the physiological medium from which the Ca and Mg salts were omitted and to which 30 mM EDTA was added, for 5 to 10 min. The shaking was done either by hand or by briefly touching the container to a vortexer. The supernatant was then decanted, spun at 700 X g for 2 min, and the resulting pellet was resuspended in the physiological medium without any further exposure to EDTA. Repeated further agitation of the remaining sample, followed by decanting and centrifuging, generated a series of crypt preparations. This procedure was repeated until a sufficient number of crypts were eluted from the sections. The eluted crypts were then stored at 4C until use.
In Vitro Stimulation of Paneth Cells
Isolated crypts were stimulated with either 10 μM carbachol (CCh) or LPS in physiological medium (mM: 147 NaCl, 4.0 KCl, 3 CaCl2, 0.9 MgCl2, 11 HEPES, 10 glucose, pH 7.4) for durations ranging from 10 min to 2 hr and then stained for zinc (see below). Alternatively, crypts were first stained and then stimulated while monitoring for Paneth cell degranulation with confocal microscopy. In addition, lumens of isolated crypts were loaded with a zinc-specific fluorescent dye (ZP1, ZP3, Newport Green) via cannulation with a micropipette, followed by stimulation with either CCh or LPS, while degranulation was monitored under confocal microscopy.
In Vivo Stimulation of Paneth Cells
Rats were fasted overnight and given an IP injection of pilocarpine (200 mg/kg). Isolated crypts were then harvested as described above at times ranging from 2 to 5 min postinjection.
Fluorescence Staining of Paneth Cells by ZP Dyes
Isolated crypts and cryostat sections (20 μm) were stained with 20 μM ZP1 or ZP3 (Burdette et al. 2001; Chang et al. 2004) in physiological medium (mM: 147 NaCl, 4.0 KCl, 3 CaCl2, 0.9 MgCl2, 11 HEPES, 10 glucose, pH 7.4) for 1–2 min and visualized with either a conventional epifluorescence research microscope (Zeiss USA; Thornwood, NY) or in a chamber mounted on an inverted, confocal microscope (Nikon; Melville, NY).
Results
Fluorescence microscopic examination of cryostat sections (20 μm) from rat small intestine revealed a vivid and selective staining pattern in the bases of the crypts of Lieberkühn (Figures 1A and 1B). In favorably aligned sections, the base of each and every crypt was stained brightly. Sections that failed to intercept the base of each crypt showed staining only in the cross-sectioned bases. No other feature of crypts or villi was consistently stained. Preincubation of slices in 100 mM tetrakis(2-pyridylmethyl)ethylenediamine in HEPES buffered saline completely blocked all of the ZP staining.
For closer examination of the zinc staining, freshly harvested intestines were prepared, and individual crypts were separated from their matrix by gentle shaking in 30 mM EDTA. These preparations showed in greater detail that the staining in the bases of the crypts seen in the cryostat sections was restricted to individual Paneth cells, of which two to four typically exhibited zinc ZP staining (Figure 2). In the isolated crypt preparation, however, there were individual crypts that sometimes had no staining. With favorable conditions, individual secretory granules could be resolved (Figure 2). Except for the apical Paneth cell granules, no other feature was consistently stained.
The foregoing histochemical studies establish that Paneth cells of the small intestine contain significant amounts of labile zinc within their secretory granules. Because Paneth cells appear to function as sentinels for the small intestine by releasing peptide defensins from their secretory granules in response to microbes, we sought to test whether the ionic zinc pool within these same granules is available for release under physiological conditions.
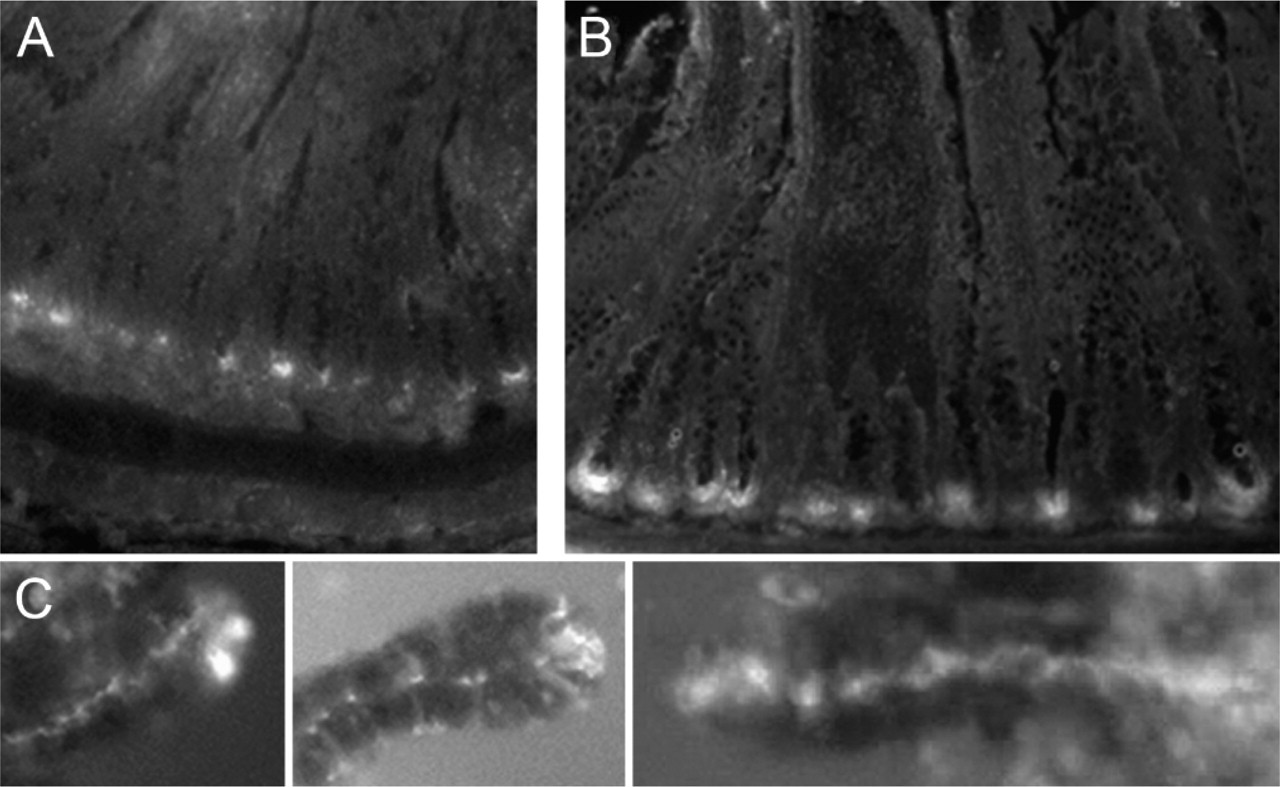
Intestinal zinc sequestration and secretion visualized with Zn:ZP staining. (
When the secretagogue pilocarpine was administered in vivo by IP injection, many of the crypts harvested immediately thereafter (rats sacrificed 2 to 5 min post-injection) consistently demonstrated a striking display of zinc secretion. In these samples, zinc ZP staining was observed throughout the crypt lumen, along with a concomitant decrease in zinc staining within the Paneth cells of the corresponding crypts (Figure 1).
In contrast to the results seen following in vivo, cholinergic stimulation of the intestine, repeated attempts to demonstrate exocytosis of the zinc-rich Paneth cell granules into the lumens of isolated crypts by direct application of pilocarpine or CCh in physiological medium (mM: 147 NaCl, 4.0 KCl, 3 CaCl2, 0.9 MgCl2, 11 HEPES, 10 glucose, pH 7.4) failed. However, direct application of CCh and pilocarpine produced an unexpected change in cell morphology specific to Paneth cells, which suggests attempted secretion. The individual Paneth cells appeared to lose their basal membrane integrity and expand outside the crypt structure, either to break entirely free or to remain poised like the cells shown (Figures 2B and 2C). These cells often lysed and released their contents outside the crypt structure. This cholinergic effect was observed regardless of whether the isolated crypts were stained pre- or poststimulation. No other crypt cells exhibited this response, indicating that the cholinergic effect on crypts is specific to Paneth cells. We also tried to observe secretion by loading the lumens of isolated crypts with fluorescent zinc probe (ZP1, ZP3, or Newport Green) via cannulation with a micropipette, followed by stimulation with either CCh or LPS. This method also failed to detect any Paneth cell zinc secretion into the crypt lumens (not shown).
The stimulation of isolated crypts with LPS in physiological medium failed to produce any evidence of either a specific or secretory effect on Paneth cells (Figure 2D). Rather, all of the cells within the crypt structure began to exhibit cytoplasmic zinc ZP staining, indicating LPS-induced cellular damage and a loss of membrane integrity (Figure 2D) (Courtois et al. 2003; Zhang et al. 2003; Frederickson et al. 2004).
Discussion
We have identified a pool of labile, secretable zinc in Paneth cells of the rat small intestine. Using ZP fluorescence, we have established that these cells, located at the bases of crypts lining the intestinal lumen, store significant amounts of “free” or rapidly exchangeable zinc in their secretory granules. Moreover, this labile pool of zinc is secreted when intact animals are stimulated with the secretagogue pilocarpine, via IP injection. In this regard, Paneth cells resemble certain cerebrocortical neurons, as well as over a dozen somatic secretory cell types—all of these cells store and secrete large amounts of “free” zinc ion by regulated exocytosis (Frederickson et al. 2005). The results of this study indicate that Paneth cells are useful as a model for the study of zinc secretion.
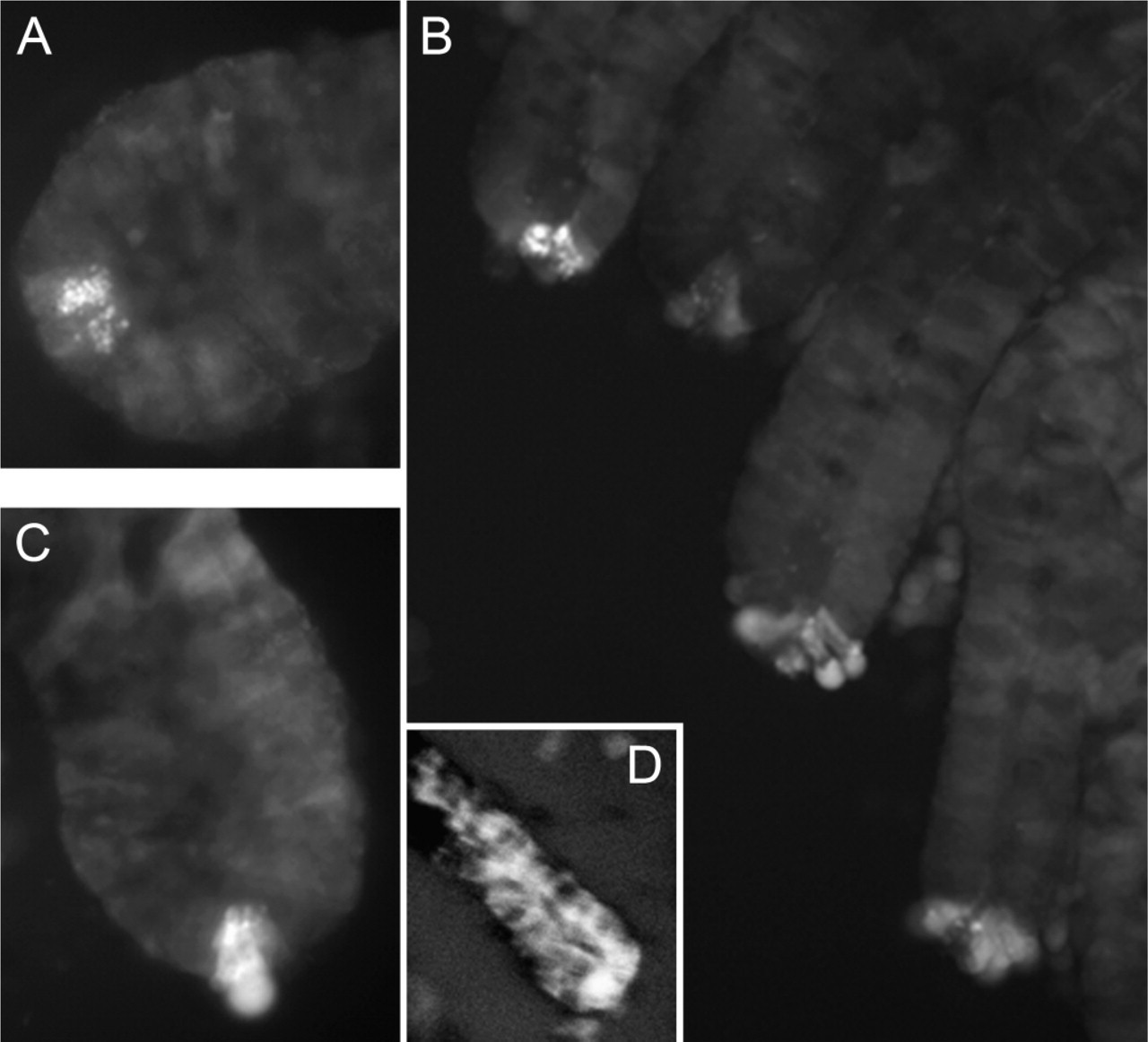
Stimulation of isolated crypts with carbachol (CCh) and lipopolysaccharide (LPS). Crypt diameters are ∼60 m. (
Because a proposed physiological function of Paneth cells is to release microbicidal defensin peptides at effective concentrations, our findings suggest a supporting role for secretable zinc in mucosal immunity (Kelly et al. 2004). The zinc-containing material secreted from Paneth cells, as observed in this study, may represent zinc aggregated with other Paneth cell secretions. This indicates that zinc may be important for the microbicidal activity of the cryptidins secreted by Paneth cells in addition to its role in cryptidin activation by matrilysin, a zinc-dependent metalloprotease (Soler et al. 1994; Wilson et al. 1999; Ayabe et al. 2004). Results of this report also contribute to the emerging notion of free zinc as an effective antimicrobial and suggest that further efforts should be directed toward elucidating the relationship between ionic zinc secretion and antimicrobial defenses.
We have also demonstrated that, upon application of secretagogues, the basal membranes of zinc-containing Paneth cells in isolated crypt preparations began to swell and exhibit diffuse zinc ZP staining (Figures 2B and 2C). This often progressed until these cells lysed and released their constituents outside the crypt structure or broke entirely free from the crypt structure. This result indicates that Paneth cells do respond to direct cholinergic stimulation in the medium used (mM: 147 NaCl, 4.0 KCl, 3 CaCl2, 0.9 MgCl2, 11 HEPES, 10 glucose, pH 7.4). However, in the isolated crypt preparation, the ability of Paneth cells to secrete into the crypt lumen was impaired. In contrast, we did not find evidence that Paneth cells respond to the direct application of LPS. Rather, all of the cells in the isolated crypts began to demonstrate cytoplasmic zinc ZP staining, indicating cell damage (Frederickson et al. 2004, 2005). Courtois et al. (2003) found that LPS-induced lipid peroxidation has an adverse effect on enterocytes, and LPS has also been reported to injure pancreatic acinar cells (Zhang et al. 2003).
In addition to the results discussed above, this study indicates that the structure of the intact intestine plays a necessary role in the movement of Paneth cell secretions into the crypt lumen. Whereas striking evidence of Paneth cell zinc secretion was consistently obtained following cholinergic stimulation in intact animals, zinc secretion into the crypt lumen was never observed upon stimulation of isolated crypts, despite numerous attempts with two different methods. The lack of secretion into the lumens of isolated crypts may be due to the lack of structural support required for the intensive movement of the large apical secretory granules from the Paneth cells into the crypt lumen. In the intact intestine, the muscularis mucosae lie directly beneath the base of the crypts, providing structural support to the immediately adjacent Paneth cells. The difference in structural support can be seen by comparing the cryostat sections with the isolated crypts (Figure 1 and Figure 2). In addition, the contraction of intestinal smooth muscle, which is also stimulated by cholinergics, may play a role in providing a motivating force for the movement of Paneth cell secretions into the crypt lumen.
Our finding that an intact intestine is required for Paneth cell secretion into the crypt lumen has important implications for the study of Paneth cell secretions when they are acquired by the stimulation of isolated crypts. These results suggest that the secretory constituents thus obtained either represent a small portion of the total and/or were released due to alterations in the integrity of Paneth cell membranes. Therefore, investigators should be cautious when interpreting data obtained by stimulating Paneth cell secretions in isolated crypt preparations.
Footnotes
Acknowledgements
This study was supported by grants from the National Institutes of Health (GM-65519 to SJL, NS-42882 and CA-96354 to CJF). C.J.C. thanks the Jane Coffin Childs Foundation for a postdoctoral fellowship.
