Abstract
Because incongruous controversial staining results are a common phenomenon in the placenta, methodical investigations are important to prevent researchers from obtaining misleading results. While investigating dendritic cells (DC) at the human fetomaternal interface, we observed staining of endothelial cells (EC) in chorionic villi for CD83. Given the high specificity of this antigen for DC, this did not seem credible. Previous studies had revealed the same surprising staining pattern with human leukocyte antigen (HLA)-G antibodies. We therefore analyzed human placental EC staining more closely. Both CD83 and HLA-G antibodies were of the same mouse IgG2b isotype. We also observed EC staining with a panel of control antibodies of the IgG2b isotype. This suggests a high affinity of human placental capillaries for mouse IgG2b. Several commonly used techniques for blocking nonspecific binding of antibodies could not prevent this nonspecific EC staining. A new preincubation step with purified human IgG was introduced. This abolished any placental EC staining with CD83, HLA-G, and IgG2b isotype control antibodies, presumably by blocking Fc receptors, whereas specific staining patterns remained unchanged. Mouse antibody of the IgG2b isotype are bound nonspecifically by vascular endothelial cells in human placenta and this can be overcome by blocking with purified human IgG. This blocking procedure could also be appropriate for frozen tissues other than placenta in which Fc receptors are expressed.
Keywords
I
We investigated frozen samples of human first trimester placenta with a mouse monoclonal antibody against CD83 (clone HB15a), which is specific for a subset of the dendritic cell (DC) lineage. As anticipated, decidual DC showed a clear and reproducible staining pattern. However, to our surprise, vessels in the placental villi were also stained. Because CD83 is a highly specific surface marker for DCs at a certain stage of their maturation, its expression on endothelial cells (EC) did not seem plausible (Zhou and Tedder 1995; Banchereau and Steinman 1998).
We obtained similar unexpected staining results with the anti-human leukocyte antigen (HLA)-G antibody clone BFL-1 (Bensussan et al. 1995). HLA-G is an atypical major histocompatibility complex class 1 antigen with limited polymorphism. It has been described to be specifically expressed by extravillous cytotrophoblast (Kovats et al. 1990; McMaster et al. 1995; Hiby et al. 1999), although staining of EC of villous vessels for this antigen has also been described (Blaschitz et al. 1997). Because of the similarity of the two unexpected results, we hypothesized that the staining pattern observed with the two antibody clones on frozen sections of placental tissue was unrelated to CD83 and HLA-G antigen expression. Both the antibody clones were of the IgG2b isotype. Because staining of placental EC was not observed with numerous antibodies of the IgG1 and IgG2a isotypes, the aberrant staining pattern observed seemed to be restricted to IgG2b antibodies. To confirm this hypothesis, we investigated the “specificity” of this binding of antibodies to placental villous EC with several different antibodies. In addition, various established blocking procedures known to prevent nonspecific binding of antibodies in immunohistochemistry and the application of purified human immunoglobulin were tested in an attempt to overcome the problem of nonspecific binding in placental sections.
Materials and Methods
Tissue Specimens
The investigations were approved by the institutional Ethics Committee. Tissue was obtained from 10 healthy women undergoing legal therapeutic abortion of an intact pregnancy at 7-12 weeks of gestation via suction curettage. Chorionic villi and fragments of decidual tissue were identified macroscopically and taken from each specimen to be snap-frozen in liquid nitrogen. Samples were stored at -80C until histological examination and immunohistochemical investigations were performed
Immunohistochemistry
The antibodies applied in the study are listed in Table 1. The specificity of the primary antibodies was confirmed by staining of frozen sections of human lymph nodes and skin. Serial frozen sections were cut at 4 μm and placed onto APES (3-amino-propyltriethoxy-silane; Roth, Karlsruhe, Germanycoated slides. They were air-dried overnight to destroy endogenous peroxidases, then fixed in acetone and rehydrated in TBS (25 mM TRIS/HCl, pH 7.4, 137 mM NaCl, 2.7 mM KCl) for 10 min each.
For standard immunohistochemical staining, the sections were exposed to the primary antibodies at appropriate dilutions in antibody diluent with background-reducing components (Dako; Hamburg, Germany) for 30 min at room temperature. For negative controls, sections were incubated in parallel with the antibody diluent alone. After being rinsed in TBS five times, all sections were incubated with a horseradish peroxidase-labeled goat anti-mouse specific secondary antibody (dilution 1:100; Dako) for 30 min at room temperature. The detection reaction was developed with 3, 3'-diaminobenzidine (Sigma; Deisenhofen, Germany). Sections were counterstained with Meyer's hematoxylin (Sigma), dehydrated through graded ethanols and xylene, and embedded in Vitro-Clud (Langenbrink; Emmendingen, Germany). The sections were examined and photographed with a Leika Othoplan microscope (Leica Microsystems; Bensheim, Germany).
Additional Blocking Procedures and Solutions
Several different blocking procedures were applied to prevent the postulated nonspecific binding: before the application of the primary antibodies, the sections were incubated for 30 min at room temperature in one of the following blocking solutions: (a) 5% bovine serum albumin (Sigma), (b) 1% heat-inactivated (30 min 56C) fetal calf serum (PAA Laboratories; Cölbe, Germany), (c) 2.5% human AB serum (PAA Laboratories), and (d) 20% normal goat serum (Dako). All four blocking reagents were diluted in Dulbecco's PBS (without Ca2+ and Mg2+; PAA Laboratories).
Preincubation with purified IgG was performed with a mixture of human IgG immunoglobulins (stock solution: 30 mg/ml; Beriglobin; Behringwerke, Marburg, Germany) diluted in PBS, with which the sections were incubated for 30 min at room temperature. A series of dilutions (1:10, 1:50, 1:100, 1:500) was tested. After the blocking procedure, excess liquid was removed and the standard immunohistochemical procedure was performed immediately as described previously.
Results
Standard Immunohistochemistry
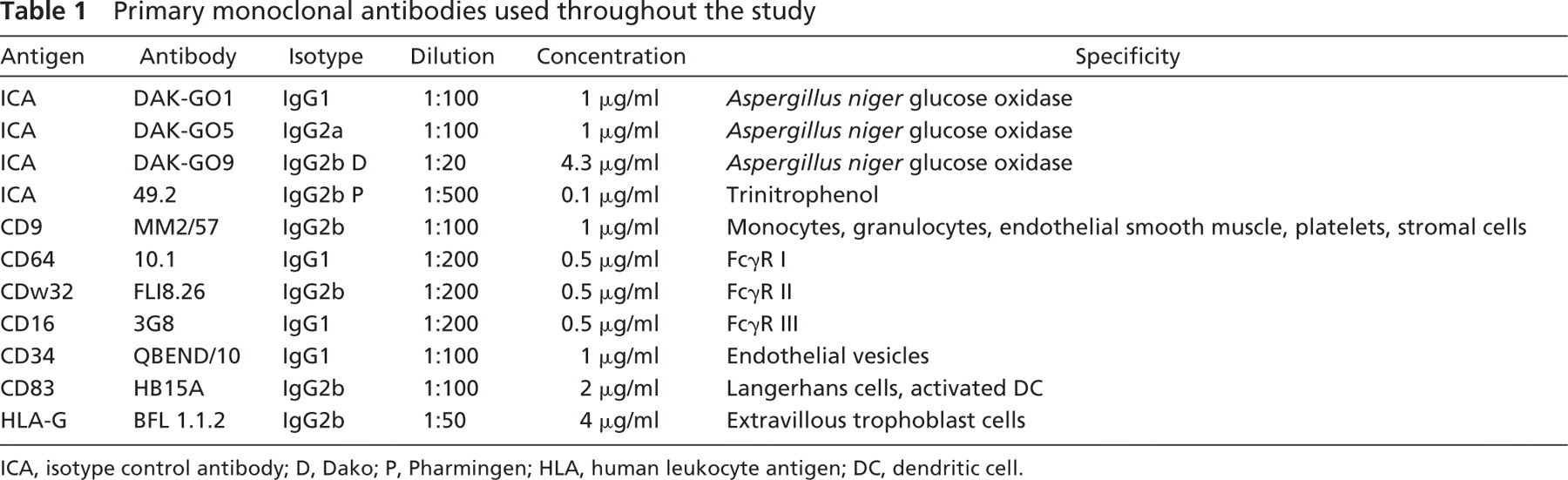
All the antibodies listed in Table 1 were applied to each of the 10 different tissue samples. When the standard immunohistochemical procedure was applied, antibodies against CD83 and HLA-G and two different IgG2b isotype control antibodies produced the staining of EC in chorionic villi under investigation antibody (Figures 1A, 1C, 1E, and 1G). Negative control sections incubated with the substrate solution only showed no background staining, confirming satisfactory elimination of endogenous peroxidases by the airdry step. Control samples stained with the horseradish peroxidase-labeled secondary antibody alone did not exhibit any nonspecific staining. As shown in Figures 1E and 1G, placental villous EC were clearly stained by both IgG2b isotype control antibodies, whereas the IgG1 and IgG2a antibodies did not produce any staining with the standard protocol, even when used at higher concentrations than recommended by the supplier (not shown). One of the two IgG2b isotype control antibodies (Dako) also produced weak staining of Hofbauer cells (Figure 1E). These cells were not stained by the other IgG2b isotype antibodies or the antibodies against HLA-G and CD83. The staining results are summarized in Table 2.
Primary monoclonal antibodies used throughout the study
ICA, isotype control antibody; D, Dako; P, Pharmingen; HLA, human leukocyte antigen; DC, dendritic cell.
Immunohistochemical staining of human early pregnancy placenta with (right) and without (left) blocking with human IgG. The staining results are compared in corresponding sections. Magnification ×250. (
Blocking Procedures
Several different procedures were applied in an attempt to block possible nonspecific binding of IgG2b antibodies to the placental tissue. Incubation of the sections in BSA (5%), fetal calf serum (1%), human AB serum (2.5%), and goat serum (20%), respectively, did not prevent nonspecific binding to villous EC and Hofbauer cells (not shown). Blocking with antibody diluent with background reducing component also failed to prevent nonspecific binding of IgG2b isotype control antibodies. Unfortunately, even when two of these blocking procedures were performed consecutively on one section, the IgG2b isotype control antibodies still bound to villous EC (data not shown).
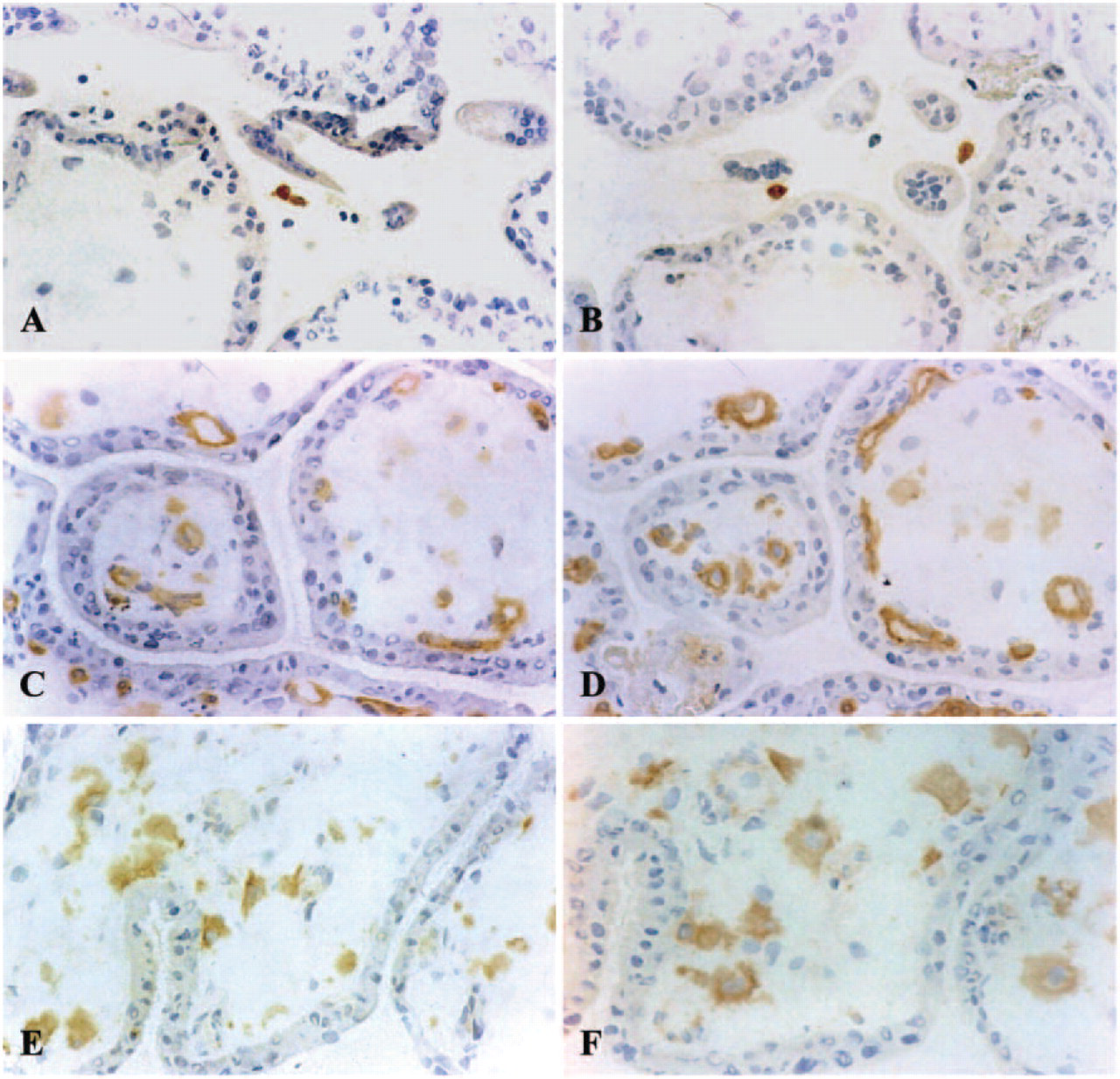
We hypothesized that one or more Fc receptors could be responsible for the nonspecific binding and applied purified and concentrated human IgG immunoglobins diluted in PBS (at 1:10, 1:50, 1:100, 1:500) before the sections were incubated with the primary antibody. After this preincubation step, the nonspecific binding of all antibodies (both isotype control antibodies, anti-CD83 clone HB15a, and anti-HLA-G clone BFL-1) to villous vessels at dilutions of 1:10 and 1:50 totally disappeared (Figures 1B, 1D, 1F, and 1H). There was still nonspecific background staining at a dilution of 1:50, and no blocking effect was seen at a dilution of 1:100 (not shown). To optimize the cost:effect ratio, a dilution of 1:50 (corresponding to 0.6 mg/ml human immunoglobulin) was selected as standard for our blocking protocol. This blocking step did not interfere with the specific detection of CD83-positive dendritic cells with the HB15a clone (Figures 3E and 3F) or HLA-G-positive extravillous trophoblast cells (Figures 3G and 3H) in the decidua basalis.
Expression of Fc Receptors
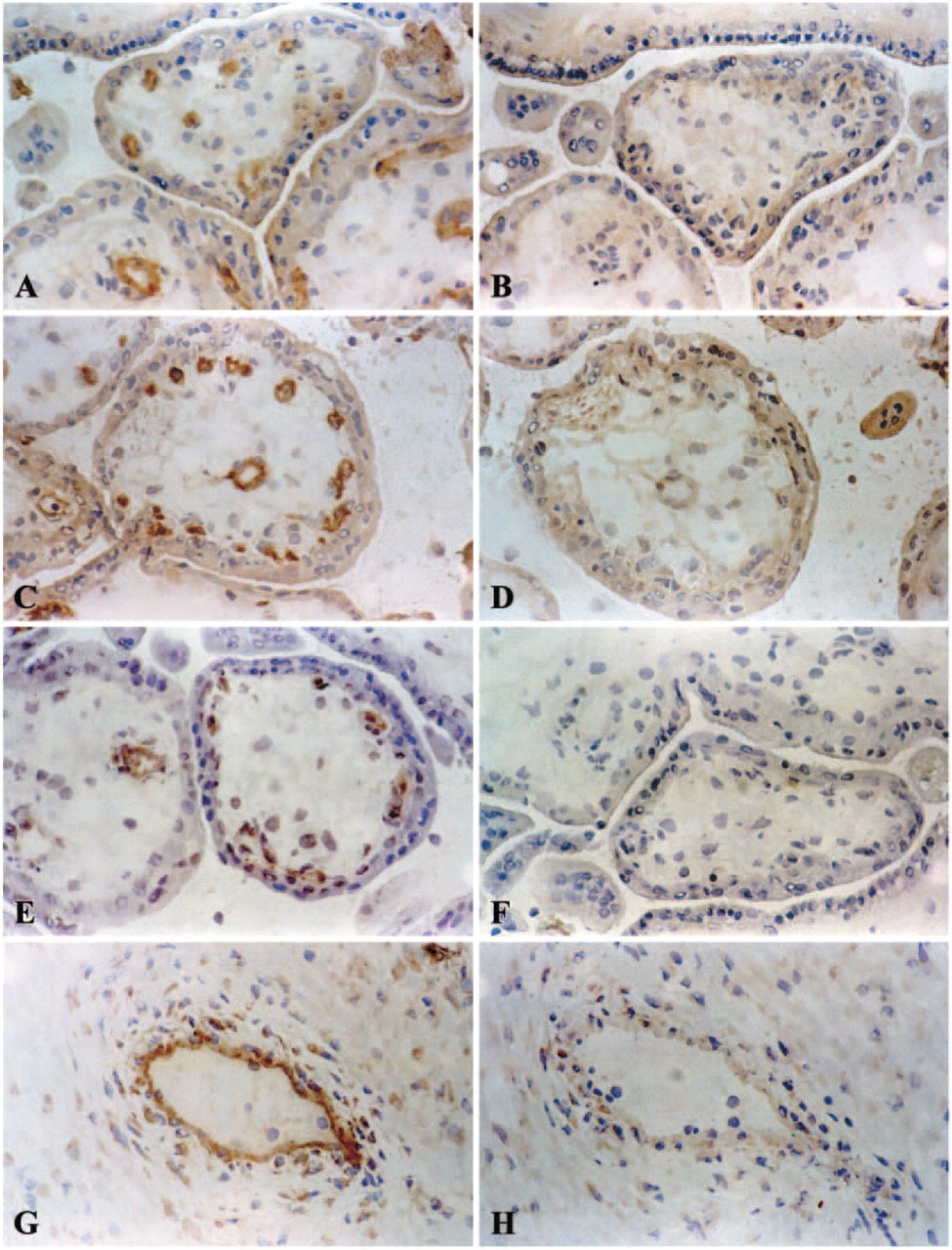
To identify the receptors or antigens responsible for the nonspecific binding, we analyzed Fc receptor expression in human placenta specimens by staining tissue sections with antibodies against CD64 (FcγRI), CDw32 (FcγRII), and CD16 (FcγRIII; Table 1).
No staining for FcγRIII was observed in fetal endothelial cells or placental Hofbauer cells (Figures 2A and 2B). Figures 2C and 2D demonstrate that placental vessel EC are clearly positive for FcγRII (CDw32), even if incubation with the primary antibody is preceded by incubation with human IgG immunoglobulin (Figure 2D). Decidual macrophages showed moderate staining for FcγRII. The antibody against CD64, stained only Hofbauer cells in the placental villous stroma (Figures 2E and 2F).
Does Preincubation with IgG Interfere with Specific Detection?
To determine whether the “preincubation” step with human IgG interferes with specific antibody binding to the endothelial cells of placental vessels, we used anti-CD34 (IgG1). This antibody is directed against endothelial cells and CD34 is also expressed on hematopoietic progenitor cells (Table 2). In addition, we applied a anti-CD9 (IgG2b) antibody clone to specifically stain endothelial cells (Table 1). Preincubation with human IgG did not interfere with specific staining of villous endothelial cells by either antibody, although the staining intensity for CD34 was slightly decreased (Figures 3C and 3D). Fetal endothelial cells stained for CD9 and this was not influenced by the preincubation procedure with human IgG (Figures 3A and 3B).
Cell type-dependent results of immunohistochemistry with and without blocking by human IgG1

Grades of staining intensity: -, negative; +, weak; + +, moderate; + + +, strong. Pre-IgG = Preincubation with human IgG (0.6 mg/ml) as blocking procedure. ICA, isotype control antibody; D, Dako; P, Pharmingen; EVT, extravillous trophoblast; HLA, human leukocyte antigen.
Analysis of placental Fc receptors in early pregnancy placenta. Immunohistochemical detection with (right) and without (left) the blocking procedure. (A-E) Magnification: ×250; (
The antibodies directed against FcγRI (CD64) (Figures 2E and 2F), FcγRIII (CD16) (data not shown), and CD9 are known to detect antigens that are expressed on monocytes. Macrophage staining with these antibodies in decidual areas attached to the placenta was not altered by preincubation with purified IgG (data not shown). Likewise, specific staining of endothelial cells for CD9 was not affected and for CD34 staining intensity decreased only slightly by the IgG-blocking procedure (Figures 3B and 3D). In summary, the specific staining of endothelial cells, macrophages, mature dendritic cells (Figure 3F), and extravillous cytotrophoblast (Figure 3H) in the decidua was not affected by preincubation with IgG, but nonspecific binding of IgG2b isotype antibodies disappeared.
The blocking procedure does not interfere with specific antigen detection. Left: conventional immunostaining; right: immunostaining after preincubation with human IgG at a dilution of 0.6 mg/ml. (
Discussion
In the course of investigations into immunological interactions between trophoblasts and immune cells in early pregnancy placenta and decidua, we observed staining of endothelial cells in placental villi with clone HB15a (anti-CD83) and clone BFL-1 (anti-HLA-G) antibodies. Although this staining seemed to be specific, the expression of CD83 and HLA-G on the same structure did not seem plausible. HLA-G is a frequently investigated antigen in placenta, and there was only one study describing HLA-G expression by endothelial cells in placental chorionic villi (Blaschitz et al. 1997). A search for features common to the two unexpected immunohistochemical reactions revealed that both primary antibodies were mouse monoclonal antibodies of the IgG2b isotype. We formulated the hypothesis that placental villous endothelial cells might possess a specific affinity for mouse monoclonal antibodies of the IgG2b subtype. A finding reported by Johnson et al. (1975) also pointed to the nonspecific nature of the observed staining: they found nonspecific binding of aggregated IgG to fetal villous endothelial structures.
To test this hypothesis in experiments, we applied mouse monoclonal IgG2b isotype control antibodies and found that this indeed led to endothelial cell staining in chorionic villi. That all these staining reactions vanished after purified human IgG was applied as a preincubation step suggested that they were nonspecific.
We used antibodies against CD34 (IgG1) and CD9 (IgG2b), which belong to different IgG subtypes, to investigate whether this preincubation would interfere with specific endothelial staining. In these experiments, specific detection of EC was found to be largely unaffected by preincubation. Further experiments revealed that preincubation with IgG does not interfere with other specific IHC reactions.
We can only speculate as to which structure in human placenta is responsible for the nonspecific binding of mouse IgG2b isotype antibodies observed. The obvious candidates are Fc receptors, which are known to mediate maternofetal IgG transport. Because of the known homology of mouse and human placental transport mechanisms, it is possible that human Fc receptors might have an affinity for mouse antibody (Lyden et al. 2001; Simister 2003). Both species have a hemochorial placenta and exhibit parallels in terms of IgG transport from mother to fetus. It is therefore conceivable that mouse IgG2b could bind to human placental Fc receptors that normally bind human IgG in the course of maternofetal immunoglobulin transport. Such crossreactivity could result in stable Fc binding of these antibodies, which would explain the immunohistochemical results we obtained.
In keeping with the results of Kameda et al. (1991) and Lyden et al. (2001), we found the human placenta to be negative for FcγRIII (CD16). Our findings were also consistent with those of Wainwright and Holmes (1993) and Bright et al. (1994) in that FcγRII (CDw32) was clearly detectable on vascular endothelium in the villi. After the preincubation step, the intensity of staining with anti-CDw32 (IgG2b) decreased. The more intense staining of this antibody without preincubation is most probably the result of additional binding of antibodies via this Fc receptor in the human placenta. Our results with FcγRI (CD64) confirmed the findings of Lyden et al. (2001) with regard to vascular endothelium, which was negative for this receptor.
Conflicting research findings concerning the presence of Fc receptors in the placenta have been published (Simister and Story 1997; Saji et al. 1999; Gafencu et al. 2003). FcγRII detection nicely exemplifies how challenging the immunohistochemical investigation of the placenta can be. Five different investigators used clone IV3, a mouse monoclonal antibody, of the IgG2b subclass to detect FcγRII expression by fetal EC in term placenta. Three studies found expression of FcγRII (Kristoffersen et al. 1990; Micklem et al. 1990; Sedmak et al. 1991), whereas two did not (Stuart et al. 1989; Kameda et al. 1991), which underlies the necessity for improvements in immunohistochemistry techniques in human placenta.
Hofbauer cells are fetal macrophages that are known to express multiple Fc receptors (Saji et al. 1999; Lyden et al. 2001). Although one of the IgG2b isoform control antibodies stained these cells, nonspecific IgG preincubation prevented this staining, suggesting that receptors responsible for nonspecific binding could be expressed on these macrophages.
Whether one of the fetal Fc receptors known to be expressed on placental vessels or other, still to be identified receptors mediate crossreactivity with mouse IgG2b has not yet been fully elucidated. One potential candidate is FcγRIIb2, which was found by Lyden et al. (2001) to be expressed on fetal EC.
In conclusion, our study has shown that the interpretation of immunohistochemical studies performed on frozen sections of human placental tissue is a demanding task. The difficulties arise from one of the organ's functions, maternofetal IgG transport. There is an obvious tendency of this tissue to absorb antibodies in an antigen-independent manner. This nonspecific binding of antibodies is not seen in formalinfixed placental tissue. However, numerous primary antibodies fail to stain fixed (and paraffin-embedded) tissue, so the problem of nonspecific binding associated with the use of frozen tissue needs to be solved. As demonstrated in the present study, this nonspecific staining can be eradicated by the application of purified human IgG before the primary antibody. The blocking procedure with immunoglobulin could also be appropriate for frozen tissues other than placenta, such as the tissue of the immune system and certain epithelia, in which Fc receptors are expressed (Stuart et al. 1989). However, even when this reliable tool is employed to improve the specificity of immunohistochemical staining, suitable controls on serial sections should still always be included. These controls should involve staining with antibodies of the same IgG isotype but irrelevant specificity, which are applied at the same concentrations as the original primary antibodies.
