Abstract
Serum amyloid A (SAA) is an acute phase reactant, whose level in the blood is elevated in response to trauma, infection, inflammation, and neoplasia. Elevated levels of SAA in the serum of cancer patients were suggested to be of liver origin rather than a tumor cell product. The role of SAA in human malignancies has not been elucidated. We investigated the expression of SAA at various stages of human colon carcinoma progression. Nonradioactive in situ hybridization applied on paraffin tissue sections from 26 colon cancer patients revealed barely detected SAA mRNA expression in normal looking colonic epithelium. Expression was increased gradually as epithelial cells progressed through dysplasia to neoplasia. Deeply invading colon carcinoma cells showed the highest levels of SAA. Expression was also found in colon carcinoma metastases. Cells of lymphoid follicles of the intestinal wall, inflammatory cells, ganglion cells, and endothelial cells, also expressed SAA mRNA. Immunohistochemical staining revealed SAA protein expression that colocalized with SAA mRNA expression. RT-PCR analysis confirmed the expression of the SAA1 and SAA4 genes in colon carcinomas, expression that was barely detectable in normal colon tissues. These findings indicate local and differential expression of SAA in human colon cancer tissues and suggest its role in colonic tumorigenesis.
S
SAA levels were found to be elevated in the serum of patients with a wide range of malignancies, being highest in those with metastatic carcinoma of unknown primary sites (Rosenthal and Sullivan 1979). SAA levels are significantly higher in patients with distant metastases as compared with those with localized disease. The levels are inversely correlated with patient's survival (Weinstein et al. 1984; Biran et al. 1986) and are decreased in response to therapy (Kaneti et al. 1984). Based on these findings SAA was suggested as a marker for neoplastic activity. More recently, using modern mass spectrometry and proteomic technologies, SAA was identified as the most significant protein differentiating the serum of patients with lung cancer from the serum of healthy individuals (Howard et al. 2003) and as a protein whose serum level strongly correlates with the clinical relapse status in nasopharyngeal cancer patients (Cho et al. 2004). In colon cancer patients, SAA levels, when compared with levels of other acute phase proteins including C-reactive protein, showed the best specificity and sensitivity to disease activity (Glojnaric et al. 2001). The SAA protein in the serum of cancer patients, in both the old and the more recent studies, was suggested to be of liver origin, rather than being produced locally in the tumor tissue.
Despite the four decades of research since its first recognition as the precursor protein in inflammation-associated AA amyloidosis (Benditt et al. 1962; Levin et al. 1973; Linke et al. 1975), the biological importance of SAA in health and disease is not well understood. Today SAA is viewed as a multifunctional protein playing a role mainly as a modulator of inflammatory processes and being involved in cholesterol metabolism and transport (Urieli-Shoval et al. 2000). The mode of involvement of SAA in human malignancies has not been elucidated. However, several functions of the SAA protein, described in the context of inflammation, are compatible with the mechanism of tumor cell invasion and metastasis. These include: induction of cell adhesion and migration (Badolato et al. 1994; Xu et al. 1995; Preciado-Patt et al. 1996; Hershkoviz et al. 1997), induction of enzymes degrading the extracellular matrix (ECM) (Migita et al. 1998; O'Hara et al. 2004), and inhibition of cell attachment to ECM proteins by SAA-derived peptides (Preciado-Patt et al. 1994). Furthermore, SAA contains binding sites for the ECM constituents laminin (Ancsin and Kisilevsky 1999a) and heparin/heparan sulfate (Ancsin and Kisilevsky 1999b), it complexes with native ECM and ECM proteins (Preciado-Patt et al. 1996; Hershkoviz et al. 1997) and has functional arginine-glycine-aspartic acid (RGD)-like and tyrosine-isoleucine-glycine-serine-arginine (YIGSR)-like adhesion motifs (Linke et al. 1991; Preciado-Patt et al. 1994; Urieli-Shoval et al. 2002). Cumulatively, these properties place SAA as an ECM-associated adhesion protein, with a potential role in tumor pathogenesis.
Because of the elevation of serum SAA levels in cancer patients and because of the adhesion functions of SAA protein, we felt it was important to directly evaluate the expression of SAA in human cancerous tissues. The main focus of this study was to document, by in situ hybridization, SAA gene expression in colon tissues from normal-looking colonic mucosa through adenoma with different grades of dysplasia to invasive carcinoma and metastases.
Immunohistochemistry and RT-PCR analyses were used on selected tissues to verify that the expression demonstrated by in situ hybridization accurately portrays SAA gene expression and SAA protein synthesis. We demonstrate that the SAA mRNA and protein are locally expressed in human colon carcinoma tissues, predominantly by the tumor cells. Furthermore, we found that SAA mRNA expression in epithelial cells increases gradually as they progress through different stages of dysplasia to overt carcinoma.
Materials and Methods
Tissues
Conventional serial sections of routinely processed formalin-fixed and paraffin-embedded archival tissues from 26 patients with colonic neoplasia operated between 1999 and 2004 were obtained from the Department of Pathology, Hadassah-Hebrew University Medical Center, Mount Scopus. The demographics and histological grading and staging of the colon cancer patients; the type of sections included in the study are detailed in Table 1. The sections included: 25 sections of colon adenocarcinomas, 10 sections of metastases; 8 to regional lymph nodes, 1 to liver, and 1 to omentum; and 11 sections of adenomatous polyps and 18 sections of normal looking colonic epithelium taken from surgical margins. Sections of a liver were used as positive controls. Freshly frozen biopsies of colon adenocarcinomas, as well as normal colons, from 3 patients (patients 2, 8, and 9 in Table 1), were obtained from the Department of Pathology. Studies were approved by the Human Subjects Research Committee of the Hadassah-Hebrew University Medical Center, Jerusalem, Israel.
Colon cancer patients: demographics and histologic staging and grading
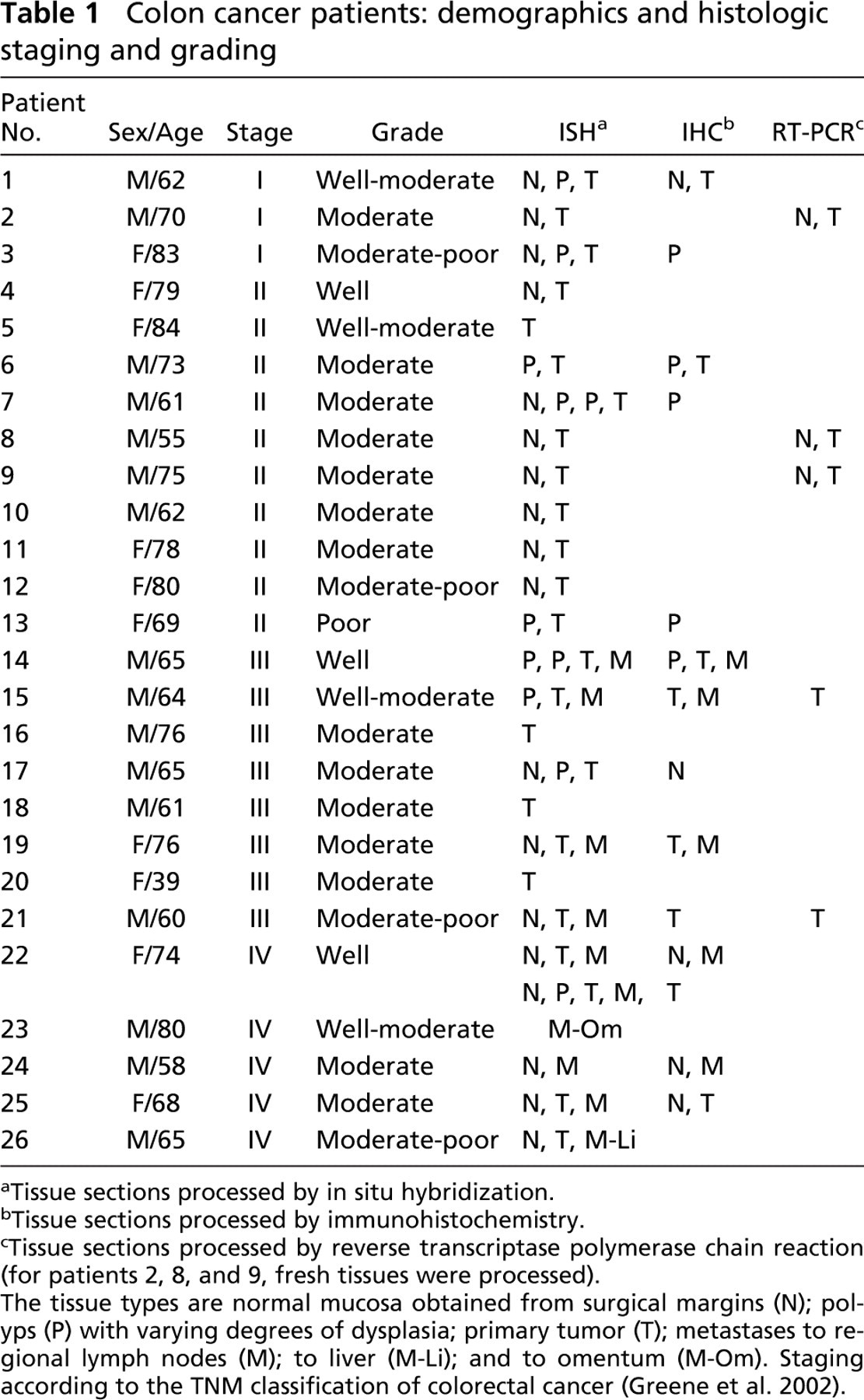
Tissue sections processed by in situ hybridization.
Tissue sections processed by immunohistochemistry.
Tissue sections processed by reverse transcriptase polymerase chain reaction (for patients 2, 8, and 9, fresh tissues were processed).
The tissue types are normal mucosa obtained from surgical margins (N); polyps (P) with varying degrees of dysplasia; primary tumor (T); metastases to regional lymph nodes (M); to liver (M-Li); and to omentum (M-Om). Staging according to the TNM classification of colorectal cancer (Greene et al. 2002).
Probes
The SAA probe was prepared from pGEM transcription vector that contained a 110-bp sequence of mouse SAA1 cDNA (p125) (Meek and Benditt 1989). This nucleotide sequence encompasses a domain coding for amino acid residues 30–66 that is highly conserved among species and is highly homologous with human SAA genes, as described previously (Meek et al. 1994). To generate digoxigenin labeled probe, p125 was linearized with HindIII (antisense) or EcoRI (sense) and incubated in a transcription reaction containing 500 μM each of GTP, ATP, and CTP (Promega Biotec; Madison, WI) and 500 μM digoxigenin-labeled UTP (Boehringer-Mannheim; Indianapolis, IN) as described (Urieli-Shoval et al. 1992; 1998). In addition to the sense control probes, nonlabeled antisense probes were generated by including UTP instead of digoxigenin-labeled UTP, in the transcription reaction.
In Situ Hybridization
Nonradioactive in situ hybridization was performed as previously described (Urieli-Shoval et al. 1992;1998) with slight modifications. Deparaffinized and rehydrated sections were digested with proteinase K (5 μg/ml, 40 min, 37C) and prehybridized in 100 μl hybridization buffer (50% deionized formamide/0.3 M NaCl/20 mM Tris-HCl, pH 8.0/5 mM EDTA/1 × Denhardt's/10% dextran sulfate) for 2 hr at 50C. Hybridization was initiated by addition of 50 μl hybridization buffer containing the labeled antisense probe and was allowed to proceed for 15–18 hr at 50C. Parallel sections were incubated with either the sense probe or the antisense probe mixed with a 20-fold excess of nonlabeled antisense probe, to be used as negative controls. Liver sections were used as positive controls. After hybridization the sections were treated with RNase A (20 μg/ml, 30 min, room temperature), followed by 0.1 × SSC/0.5% Tween 20 (3 × 40 min, 37C), and proceeded with the immunodetection, which was accomplished with the Genius Nonradioactive Nucleic Acid Detection Kit (Boehringer-Mannheim) with modifications. Slides were incubated in 100 mM Tris HCl, pH 8.0/150 mM NaCl (buffer 1) containing 2% normal sheep serum and 0.3% Triton-X-100 at room temperature for 30 min. One hundred ml of anti-digoxigenin antibody conjugated to alkaline phosphatase (1:1000 dilution in buffer 1 containing 1% normal sheep serum and 0.3% Triton X–100) was applied onto the slides, which were incubated in a humid chamber overnight at 4C. After washing in buffer 1, a color solution (alkaline phosphate substrate kit III; Vector Laboratories, Burlingame, CA) was applied onto the slides and incubation was carried out at room temperature in a dark chamber until a satisfying intensity of color developed (usually 60 min). Sections were counterstained with acridine orange/safranin O (Urieli-Shoval et al. 1992;1998). The intensity of staining was graded by a trained pathologist (DP) using a three-point scale: negative (0), weak (1), and moderate to strong (2 to 3).
Immunohistochemistry
Immunohistochemistry was performed using the Histostain Plus SP kit which offers superior sensitivity (Zymed Laboratories Inc.; San Francisco, CA). Briefly, deparaffinized and rehydrated sections were digested with protease (0.1% in PBS, 15 min, room temperature) and then treated according to the manufacturer instructions using the ready-to-use kit components. Two anti-SAA monoclonal antibodies were used: clone mcl (DAKO Corporation; Carpinteria, CA), was directed against AA-amyloid fibril protein and detecting also SAA and clone mc29, directed against the highly conserved (invariant) region of SAA. The preparation and specificity of these antibodies were previously described and demonstrated (Linke 1984; Linke et al. 1991). The specificity of clone mc29, as assessed by Western blotting, as well as its neutralizing activity were demonstrated recently again (Urieli-Shoval et al. 2002). Antibodies were diluted 1:20 (mc1) and 1:600 (mc29) in 0.1 M Tris-HCl pH 7.6 and incubation lasted 2 hr at room temperature. Color was developed using AEC substrate (Zymed Laboratories Inc.) for 10 min followed by counterstaining with hematoxylin. Negative controls included replacement of the primary antibodies by PBS and by normal mouse isotype-matched IgG (IgG2a, kappa; DAKO Corporation). Liver sections were used as positive controls.
RT-PCR
Total RNA was extracted from tissue sections cut from formalin-fixed paraffin-embedded blocks using Paraffin Block RNA Isolation Kit (Ambion; Austin, TX). RNA was also extracted from freshly frozen tissue biopsies using Tri Reagent (Molecular Research Center; Cincinnati, OH). Synthesis of cDNA was performed using random hexamer primer and the SuperScript II RNase H− reverse transcriptase (Invitrogen Life Technologies; Paisley, UK). Amplification of cDNA was done using Supertherm DNA polymerase (JMR Holdings; London, UK) and primers specific for each of the four known human SAA genes; SAA1, SAA2, SAA3, and SAA4. The sequence of the primers and their specificity were previously described (Urieli-Shoval et al. 1994). Amplification of a fragment of the housekeeping gene β-actin (220 base pairs) was used as a positive control for successful amplification of the cDNA, using primers previously described (Kaneko-Ishino et al. 1995). Negative controls included replacement of the cDNA mixture with H2O in the PCR reaction. Mixtures were incubated in a thermocycler (MJ Research Inc.; Watertown, MA) under the following conditions: 1 cycle of 2 min at 95C followed by 40 cycles each consisting of 30 sec at 95°C, 30 sec at 59°C, and 30 sec at 72C and at the end followed by 1 cycle of 2 min at 72C. The identity of the PCR amplification products was confirmed by their sequencing using the SAA primers and the BigDye Terminator cycle sequencing chemistry from Applied Biosystems (Foster City, CA). Sequencing was carried out on an automated DNA sequencer (Model 3700 DNA Analyzer; Applied Biosystems), in the center for genomic technologies, the Hebrew University, Jerusalem, Israel.
Results
In Situ Hybridization and Immunohistochemistry of Human Colon Tissues
We applied the nonradioactive in situ hybridization technique, on human colon tissue sections from 26 colon cancer patients (as detailed in Table 1), to determine whether human colon tissues express SAA mRNA, what cell types are responsible for such an expression, and whether SAA is differentially expressed in the progressive stages of human colon carcinoma. Two controls were used for nonspecific hybridization: the sense probe and the antisense probe mixed with a 20-fold excess of nonlabeled antisense probe, both resulted with diminished signal. Liver sections used as positive controls and showed positive staining of hepatocytes, as described (Urieli-Shoval et al. 1998). Our findings are described in the following section.
We applied immunohistochemistry on selected sections, as detailed in Table 1, to document that the presence of SAA mRNA is accompanied by SAA protein synthesis. Two different anti-SAA monoclonal antibodies were used, resulting with a similar pattern of staining. Negative controls included replacement of the primary antibody by either PBS or normal mouse isotype-matched IgG, both resulting in diminished signal. Liver sections used as positive controls and hepatocytes were positively stained with anti-SAA antibody but not with control IgG (Figures 2G and 2H).
The following is a description of our findings.
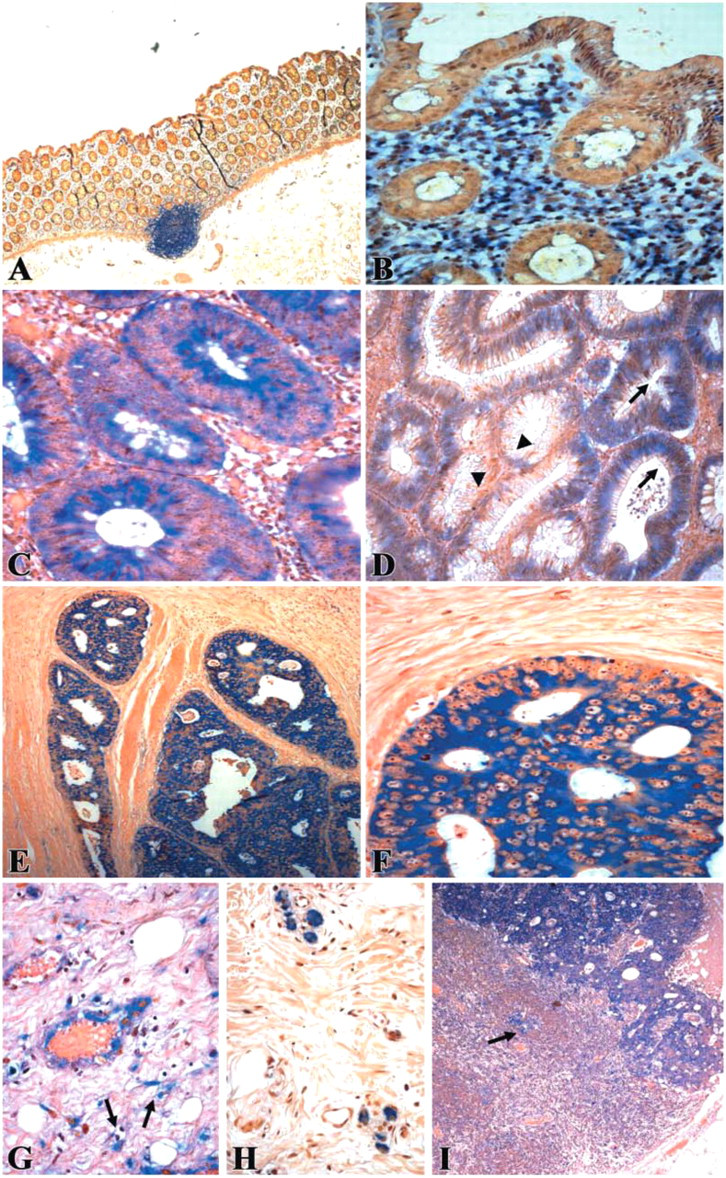
In situ hybridization demonstrating serum amyloid A (SAA) mRNA expression in neoplastic colon. The blue cytoplasmic staining represents positive SAA mRNA signal, counterstaining is pink-brown.
RT-PCR Analysis of Human Colon Tissues
We carried out RT-PCR analysis on RNA extracted from sections cut from paraffin blocks participating in this study to further support the in situ hybridization findings and to determine which of the four known human SAA genes are expressed. Oligonucleotide primers specific for each of the four known human SAA genes—SAA1, SAA2, SAA3, and SAA4—were used. In Figure 3A, results obtained from tumor sections of patient 21 in Table 1 are shown. Fragments of the predicted sizes were amplified when the SAA1- and SAA4-specific primers were used and no PCR products were obtained for the SAA2 and SAA3 genes. The identity of the PCR products was confirmed by their sequencing and comparison to SAA sequences reported in the GenBank/EMBL Databases. The colon-derived SAA1 sequence had 99% nucleotide homology with clones with the accession numbers NM 000331 and NM 199161. The colon-derived SAA4 sequences had 99% nucleotide homology with clone with the accession number M81349. Similar results were obtained applying this analysis on tumor sections of patient 15 in Table 1 (not shown).
We also performed RT-PCR on RNA extracted from freshly frozen tumor and normal colon biopsies, from three patients (patients 2, 8, and 9 in Table 1). In Figure 3B, the SAA1 and SAA4 genes are strongly expressed in the tumor tissues, whereas their expression is weak to barely detectable in the normal tissues obtained from the same patients. The control gene β-actin is expressed at equal levels in both the tumor and the normal samples.
Discussion
High levels of SAA in the serum of cancer patients, including colon cancer patients, were suggested to be of liver origin. Expression of SAA in tumor tissues has not been investigated and its role in human malignancies has not been elucidated. In the present study, using nonradioactive in situ hybridization, immunohistochemistry, and RT-PCR analyses, we demonstrate local expression of SAA mRNA and protein in human colon adenocarcinomas. We found that the tumor cells are the predominant SAA expressing cells. Moreover, the SAA mRNA expression in epithelial cells is gradually increased as they progress through different stages of dysplasia to overt carcinoma. Local and differential expression of SAA in human colon cancer tissues suggests its role in colonic tumorigenesis and may have both prognostic and therapeutic applications.
In most of the normal-looking colonic epithelium, no SAA mRNA was detected and in the few sections in which some staining was observed, it was weak and confined to the surface epithelium. In only two sections, was there a diffuse strong staining of the normal epithelium. In adenomatous polyps, weak to moderate staining for SAA mRNA was found in the dysplastic epithelium and more so in the severely dysplastic cells. In all sections of colon adenocarcinomas, there was strong diffuse cytoplasmic staining for SAA mRNA in the tumor cells. In about half of the sections, stronger and more diffuse staining was found in deeply invading tumor areas as compared with the more superficial tumor glands. In the metastases studied, there was expression of SAA mRNA in almost all the sections studied; however, staining was somewhat weaker compared with the primary tumors. These results, which are summarized in Figure 4, indicate positive correlation between epithelial SAA mRNA expression and the progressive stages of colonic neoplasia. Post hoc comparison of the four groups (Tukey) showed that normal epithelium stained significantly different from staining of dysplastic polyp epithelium, primary tumor, and metastatic epithelium. Staining of dysplastic polyp epithelium was significantly different from staining of primary tumor and metastatic epithelium. There was no significant difference in staining between the primary tumor epithelium and the metastatic epithelium.
Immunohistochemistry experiments revealed SAA protein expression that colocalized with mRNA expression. Cytoplasmic staining in dysplastic, neoplastic, and metastatic epithelial cells was observed. Colocalization of SAA mRNA and protein was found also in stromal elements including inflammatory cells, endothelial cells, and fibroblasts, predominantly in desmoplastic areas. In addition to cell-associated protein staining, there was also extracellular staining mainly in the primary tumor sections. This staining presumably represents SAA bound to the extracellular matrix, because SAA binds to extracellular matrix proteins, as revealed by in vitro studies (Preciado-Patt et al. 1996; Hershkoviz et al. 1997). Extracellular immunostaining could be derived not only from locally synthesized protein but could also represent liver-derived SAA arriving from the blood and diffused into the tissue.
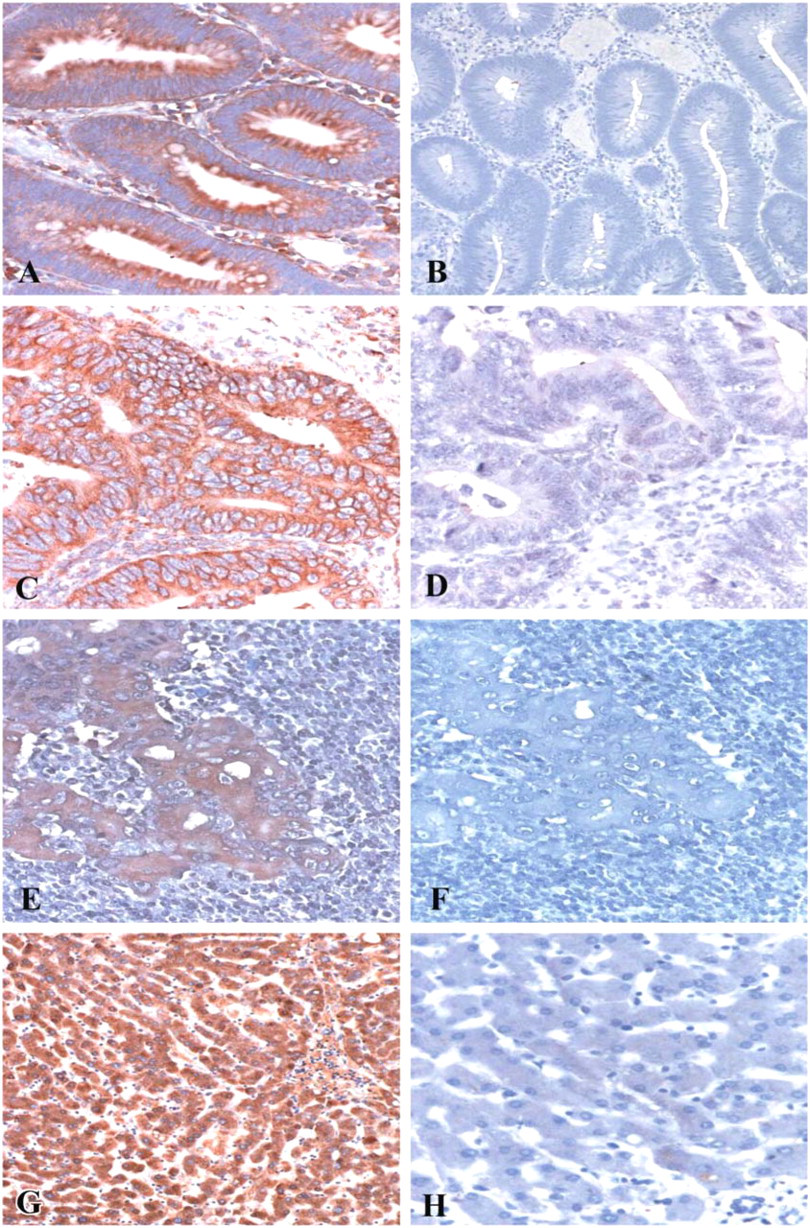
Immunohistochemistry demonstrating serum amyloid A (SAA) protein expression in neoplastic colon. Tissue sections were immunostained with monoclonal anti-SAA antibodies
The human SAA gene family comprises four discrete loci containing two highly homologous genes, SAA1 and SAA2, and two less related genes, SAA3 and SAA4. SAA1 and SAA2 corresponding proteins are the predominant circulating SAA proteins during the acute phase response (Betts et al. 1991; Uhlar et al. 1994). The SAA3 gene was considered a nonexpressed gene (Kluve-Beckerman et al. 1991), but recently its expression was demonstrated in mammary epithelial cells (Larson et al. 2003). The gene for SAA4 is constitutively expressed and its protein product is a constituent of normal, nonacute phase, high-density lipoprotein (Whitehead et al. 1992). Expression of the genes SAA1, SAA2, and SAA4 can be induced in human cultured smooth muscle cells (Meek et al. 1994) and in human monocyte/macrophage cell lines (Urieli-Shoval et al. 1994). Constitutive expression of SAA4 and inducible expression of SAA1 and SAA2 genes in some human carcinoma cell lines was demonstrated (Steel et al. 1996). Here we show that human colon carcinomas express the SAA1 and SAA4 genes, as revealed by RT-PCR analysis of paraffin sections and freshly frozen tissues. RT-PCR also revealed that these genes are preferentially expressed in carcinomas as compared with normal colons, consistent with the preferential staining observed by in situ hybridization.
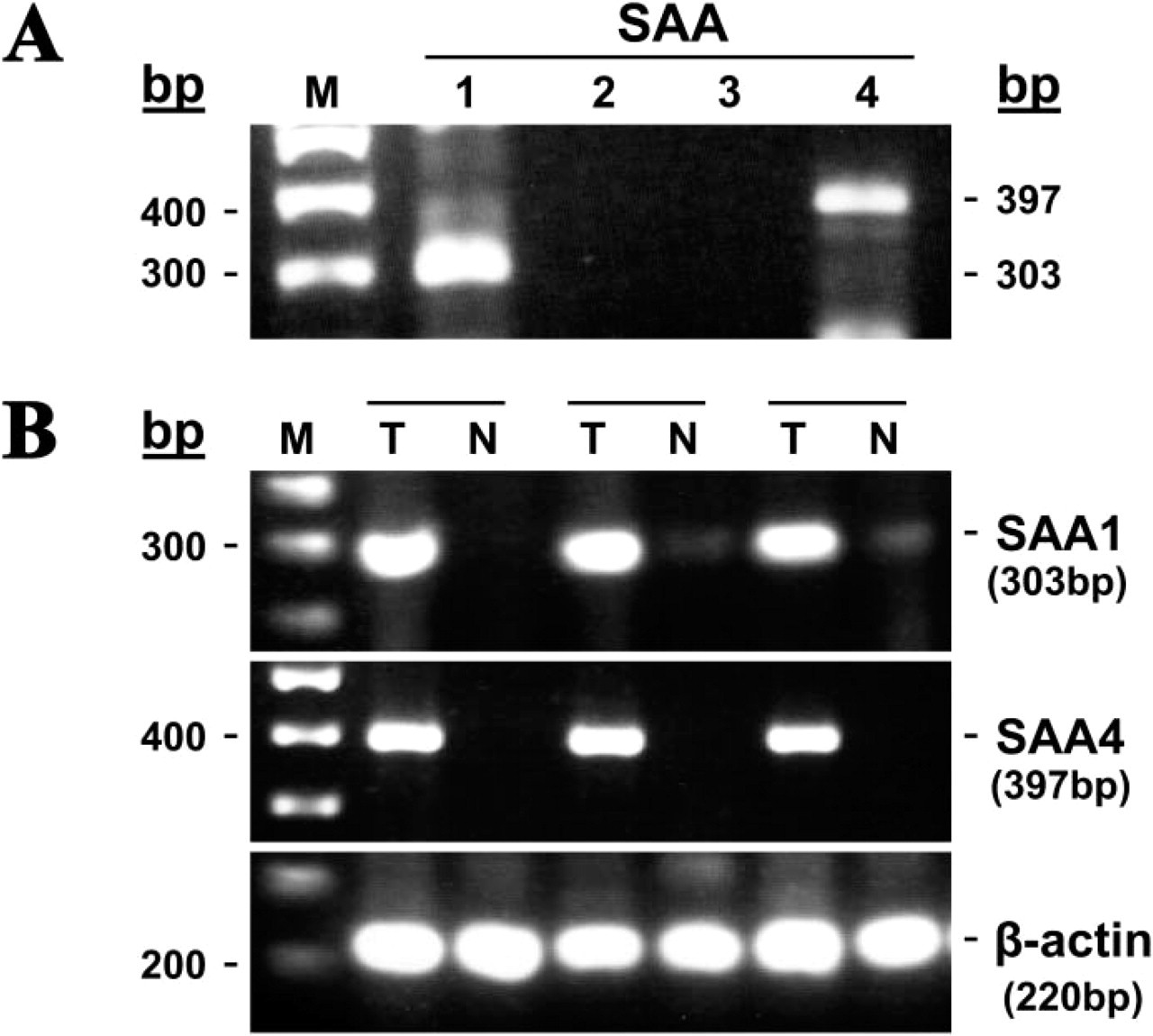
RT-PCR demonstrating expression of SAA1 and SAA4 genes in neoplastic colon. RNA was extracted from paraffin sections of colon adenocarcinoma
Was the SAA transcript identified in molecular studies looking for differentially expressed genes in human cancer? Using serial analysis of gene expression (SAGE), SAA was found among 548 differentially expressed genes in human neoplastic vs normal gastrointestinal tissues (Zhang et al. 1997). In another SAGE analysis, SAA was found among three genes whose expression is reduced in metastatic as compared with primary colon tumor cell lines (Parle-McDermott et al. 2000). This reduced expression is in line with the weaker staining of the metastasizing colon adenocarcinoma cells compared with the primary tumor, as found in our study. SAGE analysis also identified SAA among 13 genes, all oxidative stress-related genes, whose expression is markedly increased in p53-infected colorectal cancer cell line (Polyak et al. 1997). Finally, using suppression subtractive hybridization, SAA was identified among nine genes whose expression is increased in renal cell carcinoma compared with surrounding renal tissue (Nishie et al. 2001). The significance of the differential expression of SAA, as found in these studies, to human cancer is not yet known.
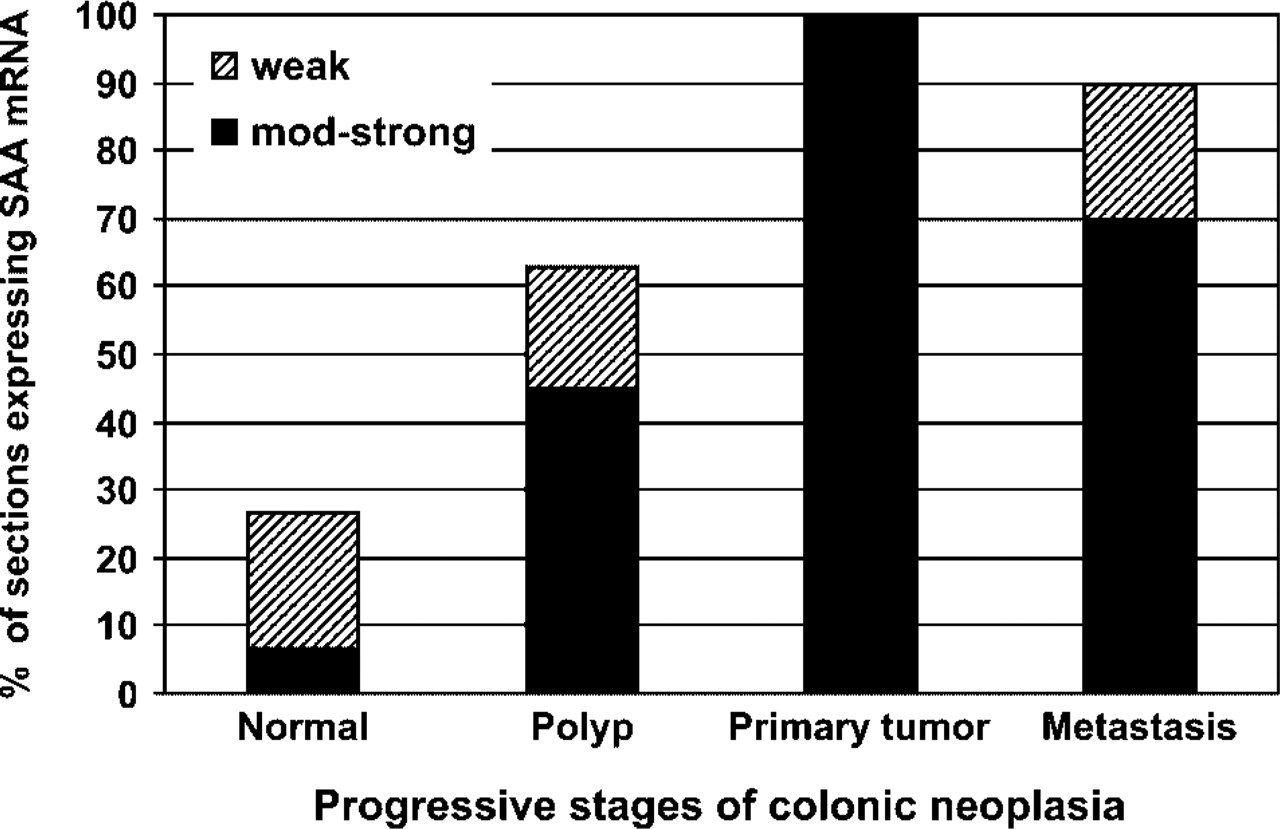
Summary of in situ hybridization staining of colonic epithelium. Presented is the percentage of sections expressing SAA mRNA in the epithelium in each category: normal epithelium, polyp, primary tumor, and metastasis. The total number of sections examined in each category was (see Results) normal,
Although the role of SAA expressed in human colon cancer tissues is not known at present, several proposed functions for the SAA protein could be relevant to the mechanism of tumor cell invasion and metastasis: (1) SAA may serve as an adhesive ligand for tumor cells as has been shown for human lymphocytes (Badolato et al. 1994; Xu et al. 1995; Preciado-Patt et al. 1996), mast cells (Hershkoviz et al. 1997), platelets, and certain melanoma cells (Urieli-Shoval et al. 2002); (2) SAA may induce the migration of tumor cells to the perivascular tissue toward metastases sites, because it induces the migration and tissue infiltration of lymphocytes (Badolato et al. 1994; Xu et al. 1995) and vascular muscle cells (Kumon et al. 2002); (3) SAA may influence tumor cell invasion through the extracellular matrix because it induces matrix metallo-proteinases (Migita et al. 1998; O'Hara et al. 2004); (4) SAA-derived peptides may inhibit tumor cell attachment to proteins of the extracellular matrix, as has been shown for T-lymphocytes and platelets (Preciado-Patt et al. 1994; Urieli-Shoval et al. 2002); (5) SAA may play a role in p53-induced apoptosis, as was suggested based on its identification as an induced gene in this system (Polyak et al. 1997), the mechanism by which is not yet known; and (6) in light of recent studies revisiting the concept of inflammation-associated tumorigenesis (Balkwill and Mantovani 2001; Pikarsky et al. 2004; Clevers 2004), SAA may play a role in the local inflammation in the microenvironment of the malignant tissue. SAA may not only induce the migration and tissue infiltration of inflammatory cells (Badolato et al. 1994; Xu et al. 1995) in the tumor mass, but may also induce the production of pro-inflammatory cytokines; tumor necrosis factor-α, interleukin-1β, and interleukin-8 (Patel et al. 1998; Furlaneto and Campa 2000). Whether any of these possibilities have relevance in the development of human colon cancer, or other cancers, requires further investigation.
Finally, to our knowledge, this is the first systematic study describing expression of SAA in human colon cancer tissues or any malignant tissue. Also we have shown preferential expression of the SAA mRNA and protein in colon tumors as compared with the normal-looking colonic epithelium. It is not known whether the SAA produced in the cancerous tissues is secreted and contributes to the elevated SAA levels in the serum of cancer patients. Alternatively, the tumor may secrete mediators that stimulate SAA synthesis in the liver resulting in high SAA serum levels, as has been suggested (Ghezzi et al. 1993). Future prospective study investigating the correlation of SAA serum levels with expression in tissues from the same patients may help clear this issue.
Footnotes
Acknowledgements
Supported by the Israel Cancer Research Fund and the Israel Science Foundation (no. 686/00-1) (to SU-S), the Deutsche Forschungsgemeinschaft, Bonn, Germany (Li 247/12-3), and the courtesy of Prof. R. Huber, Max-Planck-Institute of Biochemistry, Martinsried, Germany (to RPL).
We wish to dedicate this article to the memory of Prof. Yaacov Matzner, the Head of the Hematology Unit, who died tragically in a plane crash on November 24, 2001. His wisdom, encouragement, and scientific legacy continue to inspire us in this study and the research projects in general. We thank R.L. Meek (the Heart Institute of Spokane) for the p125 clone.
