Abstract
Gata-3 has been shown to specifically alter its expression patterns in different types of cancers. Recent evidence suggests that an interference of Gata-3 exists in the TGF-β signaling pathway. To determine the role of Gata-3 in pancreatic cancer, pancreatic cancer samples were analyzed in comparison to normal pancreatic tissues. Furthermore, four different pancreatic cancer cell lines with different alterations of the TGF-β pathway were studied. To evaluate if a potential relationship with TGF-β signaling pathway exists, we correlated mRNA expression levels with the expression of TGF-βs, TGF-β receptors, and Smad-3. Finally, we analyzed the influence of TGF-β on Gata-3 expression in vitro. All pancreatic cancer samples demonstrated a marked overexpression of Gata-3 mRNA and protein. Immuno-histochemical staining revealed strong and persistent cytoplasmic Gata-3 immunoreactivity in cancer cells. In an electrophoretic mobility shift assay, a disturbed nuclear translocation was confirmed. The expression of Gata-3 showed a significant correlation with the expression of TGF-βs, TGF-β receptors, and Smad-3. TGF-β responsive cell lines showed a down-regulation of Gata-3 mRNA upon TGF-β exposure, whereas in TGF-β-unresponsive cell lines, Gata-3 mRNA expression persisted at high levels. Furthermore, strong specific upregulation of Gata-3 impaired nuclear translocation and its cooperative action with the TGF-β pathway, suggesting that Gata-3 plays a central role in human pancreatic cancer.
P
The development and progression of pancreatic cancer is very rapid and leads to early localized invasion and metastasis. Recent studies established that certain molecular alterations may modify the pathological behavior of this tumor. These include gene mutations, chromosomal and microsatellite instability, alterations in growth factor pathways, and alterations in the activity of transcription factors (Fernandez-Zapico et al. 2003). Transcription factors are central in regulating gene expression through the alteration of gene transcription and by integrating different exogenous and endogenous signals.
Gata-3 is in fact a well-known, crucial transcription factor in T-cell development and differentiation. It plays a significant role in the early development of thymocytes (Ting et al. 1996) and the differentiation of adult T-cells in a subtype of T helper cells (Th2), producing a specific set of cytokines and promoting humoral immunity.
Recent reports have also illustrated altered expression patterns of Gata-3 in other human cancers, such as in breast (Hoch et al. 1999), cervical (Steenbergen et al. 2002), and esophageal carcinoma (Shiga et al. 1993). In breast cancer, for example, Gata-3 expression correlates strongly with estrogen receptor expression, which suggests that Gata-3 may be involved in establishing the hormone-responsive phenotype (Hoch et al. 1999). In cervical cancer, a progressive downregulation of Gata-3 expression was observed during carcinogenesis. Furthermore, human papillomavirus-mediated immortalization of cell lines led to a loss of Gata-3 expression (Steenbergen et al. 2002). In contrast, a clear upregulation of Gata-3 expression was observed in human esophageal cancer cell lines, in comparison to normal esophageal mucosal cells and primary carcinoma cells (where no expression of Gata-3 was observed) (Shiga et al. 1993).
However, the precise role of Gata-3 in the pathogenesis of different cancers still remains unclear. Recent studies suggest a noted interference of Gata-3 in the TGF-β pathway (Blokzijl et al. 2002), which is known to play a crucial role in pancreatic carcinogenesis (Friess et al. 1993).
In the present study we investigated the expression and localization of Gata-3 in human pancreatic cancer. We then went on to analyze the potential function of this transcription factor in the pathogenesis of pancreatic cancer.
Materials and Methods
Patients and Tissue Collection
Pancreatic carcinoma tissues were obtained from 27 patients (13 women and 14 men) undergoing a partial duodenopancreatectomy (Whipple resection) for pancreatic carcinoma. The mean age of the pancreatic carcinoma patients was 66.2 years (range, 36–81 years). According to the TNM classification and histopathologic grading system of the International Union Against Cancer, there were four Stage I, 4 Stage II, 14 Stage III, and 5 Stage IV tumors.
Normal pancreatic tissue samples were obtained, through an organ donor program, from 20 individuals (7 women and 13 men) who were free of pancreatic disease. The median age of the organ donors was 46.2 years (range, 20–74 years). All normal tissue samples were obtained from the head of the organ donor's pancreas to ensure comparability with the tumor samples. In all experiments, tissue sections of normal and cancerous pancreas samples were processed simultaneously to ensure comparability in the results. Freshly removed tissue samples were immediately fixed in formaldehyde solution for 12–24 hr and embedded in paraffin for immunohistochemistry and confocal microscopy. Simultaneously, tissues for RNA extraction were placed in RNALater (Ambion; Huntingdon, UK), whereas tissues for protein extraction were snap frozen in liquid nitrogen in the operating room upon surgical removal and maintained at −80C until use. The Ethics Committee at the University of Heidelberg approved the study, and informed consent was obtained from each patient.
Cell Culture
Four human pancreatic cancer, cell lines (Panc-1, MiaPaCa-2, BxPC-3, and Colo 357) and an acute lymphoblastic leukemia cell line (MOLT-4) as positive control for Gata-3 transcriptional activity were purchased from the American Tissue Type Culture Collection (ATCC; Rockville, MD). The pancreatic cancer cell lines were chosen because of their different degrees of cellular differentiation. Cell lines were cultured either in DMEM (Panc-1 and MiaPaCa-2) or in RPMI (BxPC-3 and Colo 357) supplemented with 10% heat-inactivated fetal bovine serum, with penicillin G (100 U/ml) and streptomycin (100 μg/ml) (Life Technology; Rockville, MD). All cell lines were grown through fewer than 10 passages. Cells were grown as a monolayer at 37C in humidified air with 5.7% CO2. For TGF-β1 experiments, subconfluent cells were incubated overnight in serum-free medium and subsequently incubated with 1 nM of TGF-β1 for the indicated time. Cells were then harvested for RNA extraction.
RNA Extraction and Quantitative PCR
Tissue samples were manually ground into a fine powder in liquid nitrogen with mortar and pestle. Total RNA was extracted from tissue samples using the single-step guanidinium isothiocyanate method. The integrity of all mRNA preparations was confirmed by gel electrophoresis.
At subconfluence, pancreatic cancer cell lines were rinsed twice in ice-cold PBS and solubilized in MagNA Pure lysis buffer. mRNA was prepared by automated isolation using a MagNA Pure LC instrument and an isolation kit I (Roche Applied Science; Mannheim, Germany).
cDNA was prepared using 1st Strand cDNA Synthesis Kit for RT-PCR (AMV) according to the manufacturer's instructions (Roche Applied Science). Reverse transcriptase products were amplified by real-time polymerase chain reaction (PCR) with a LightCycler FastStart DNA SYBR Green kit, as described previously (Roche Applied Science) (Hornung et al. 2002). The number of specific transcripts (Gata-1, −2, −3, TGFβ-1, TGFβ-2, TGFβ-I, TGFβR-II, and Smad-3) was normalized to housekeeping genes (cyclophilin B and HPRT) before graph blotting (all primers obtained from Search-LC; Heidelberg, Germany).
Western Blot Analysis
Proteins from pancreatic cancer cell lines and human pancreatic tissue were homogenized in RIPA lysis buffer (Tris-HCl 50 mM, pH 6.8, NaCl 150 mM, 0.1% SDS, 1% NP-40, 0.5% Na-deoxycholate) and supplemented with 500 μM PMSF and protease inhibitor (Sigma; Deisenhofen, Germany). The lysate was collected and centrifuged at 4C for 5 min at 14,000 rpm to remove the insoluble material. The protein concentration of the supernatant was measured by spectrophotometry using the BCA protein assay (Pierce Biotechnology; Rockford, IL). Equal amounts of protein extracts were fractionated on a 10% Bis Tris gel (Invitrogen; Karlsruhe, Germany). After electrophoresis, proteins were transferred to polyvinylidene difluoride membranes. Ponceau S (Sigma) staining of the membrane was used to ensure equal loading. For immunoblotting, unspecific binding was blocked with 5% non-fat milk, and the membranes were then incubated with the primary rabbit polyclonal anti-GATA-3 antibody (Santa Cruz Biotechnology; Santa Cruz, CA) in a dilution of 1:200 in 5% non-fat milk TBS-Tween at 4C overnight. Subsequently, the membranes were washed with TBS-Tween buffer (5% nonfat milk in 20 mM Tris-HCL, 150 mM NaCl, 0.1% Tween 20) and incubated with horseradish peroxidase-conjugated goat anti-rabbit IgG (Bio-Rad Laboratories; Hercules, CA) at room temperature (RT) for 1 hr. After washing with TBS-Tween buffer, the signal detection was performed with an enhanced chemiluminescence reaction (ECL Western blotting detection; Amersham Life Science, Amersham, UK).
Immunohistochemistry
Immunohistochemical staining for GATA-3 on paraffin sections was performed by an alkaline phosphatase anti-alkaline phosphatase (APAAP) procedure using polyclonal rabbit-anti human GATA-3 antibodies as primary reagent (dilution 1:20; Santa Cruz Biotechnology). Paraffin sections of 1-μm thickness were cut and dewaxed, followed by microwave pretreatment in 0.01 M citrate-buffer, pH 8.0, for 15 min at 600 W. Primary antibodies were diluted in Tris-buffered-saline (TBS) with an additional 2.5 mg/ml of normal human immunoglobulins (γ-venin; Behring, Marburg, Germany) and 0.2% BSA (Sigma) and then incubated overnight at RT. Parallel incubations with normal rabbit immunoglobulins (DAKO; Hamburg, Germany) instead of the GATA-3 antibodies served as negative controls. After washing, polyclonal mouse anti-rabbit antibodies (1:50; DAKO) were incubated for 30 min in TBS/BSA, followed by polyclonal rabbit anti-mouse-bridging antibodies (1:25; DAKO) and the APAAP-complex (1:50; DAKO), which were added in two different incubation cycles of, respectively, 30 and 15 min. Naphthol-AS-biphosphate (Sigma) and new fuchsin (Merck; Darmstadt, Germany) were used as the substrate for alkaline phosphatase. Finally, the sections were counter-stained with hematoxylin (Merck) and mounted.
Confocal Microscopy
Pancreatic adenocarcinoma cells were grown for 24 hr on Teflon-covered slides (Menzel-Glaeser; Braunschweig, Germany) at a confluency of 5000 cells/well. The acute lymphoblastic leukemia cell line MOLT-4 was used as positive control for Gata-3 nuclear localization. After rinsing in PBS, cells were fixed in 4% formalin in PBS for 1 hr at 4C, followed by ice-cold methanol for 5 min, and acetone for 2 min. Thereafter, slides were permeabilized by 0.05% Saponin for 20 min at RT. After blocking the slides in normal goat serum (KPL; Gaithersburg, MD) for 1 hr at RT, the primary polyclonal rabbit anti-GATA-3 antibody (Santa Cruz Biotechnology) was added in a dilution of 1:50. Incubation was performed overnight at 4C. After thorough rinsing in PBS containing 0.1% BSA-C (Aurion; Wageningen, Netherlands) for 3 × 5 min, slides were incubated with goat anti-rabbit secondary antibody, conjugated with fluorescein dye Cy3 (Biomedica Corporation; Foster City, CA). Thereafter, for nuclear visualization, slides were incubated with TOTO-3 (Molecular Probes; Eugene, OR) at a dilution of 1:1000 for 10 min. Slides were mounted and visualization was performed by scanning the slides with confocal microscopy (Leica GmbH; Wetzlar, Germany).
Electrophoretic Mobility Shift Assay (EMSA) for Gata-3 in Pancreatic Cancer Cell Lines
Nuclear proteins were isolated from pancreatic cancer cell lines (Panc-1, Capan-1, MiaPaca-2) and from an acute lymphoblastic leukemia cell line (MOLT-4) by placing 107 cells in cold hypotonic buffer (10 mM Tris-HCl, pH 7.5, 10 mM NaCl, 3 mM MgCl2, 1 mM PMSF, 0.05% NP-40). The samples were then immediately centrifuged at 700 rpm for 5 min at 4C in an Eppendorf centrifuge. Supernatants were decanted, the nuclear pellets after a single wash with hypotonic buffer without NP-40 and PMSF were suspended in cold hypertonic buffer (20 mM HEPES, pH 7.9, 0.4 M NaCl, 1 mM EDTA, 1 mM EGTA, 1 μM DTT, 1 μM PMSF; protease inhibitors), incubated on ice for 15 min, and centrifuged at 15,000 rpm for 15 min at 4C. The supernatants were collected as nuclear extracts and stored at −80C. Concentration of total proteins in the samples was determined using the BCA Protein Assay Kit (Pierce Biotechnology).
Gata-3 consensus oligonucleotides for EMSA were synthesized by Invitrogen (Carlsbad, CA) and annealed to generate double-stranded DNA probes. Double-stranded Gata-3 consensus probe (5′-CGC AAC TAT TGT CTT TCA CTA TTG AGA-3′) was 5′ end-labeled with γ-[32P]ATP and T4 polynucleotide kinase. Nuclear extracts and control cytoplasmic protein extract (10 mg) were incubated with 100,000 cpm of labeled probe in a 20-μl volume containing 5X binding buffer (10 mM Hepes, 25 mM KCl, 2.5 μM DTT, 0.5 μM EDTA, 70% (v/v) glycerol), and 2 mg of nonspecific DNA (salmon sperm DNA). The binding reaction mixture was incubated at RT for 20 min and analyzed by electrophoresis on 5% polyacrylamide gels (37.5:1) in 0.5X TBE buffer for 2 hr at 160 V RT. After electrophoresis, gels were dried by Gel-Drier (Bio-Rad Laboratories) and exposed to Kodak X-ray films at −70C. The acute lymphoblastic leukemia cell line MOLT-4 served as positive control for Gata-3 transcriptional activity.
Statistical Analysis
Because the hypothesis of “normal distribution of data” was rejected by the Shapiro-Wilks test, non-parametric statistical tests were used. The Mann-Whitney test was used for comparison of mRNA expression levels between groups, and the Spearman's rho coefficient test for evaluation of correlation between variables. Significance was defined as
Results
Gata-3 mRNA and Protein Are Significantly Upregulated in Pancreatic Cancer
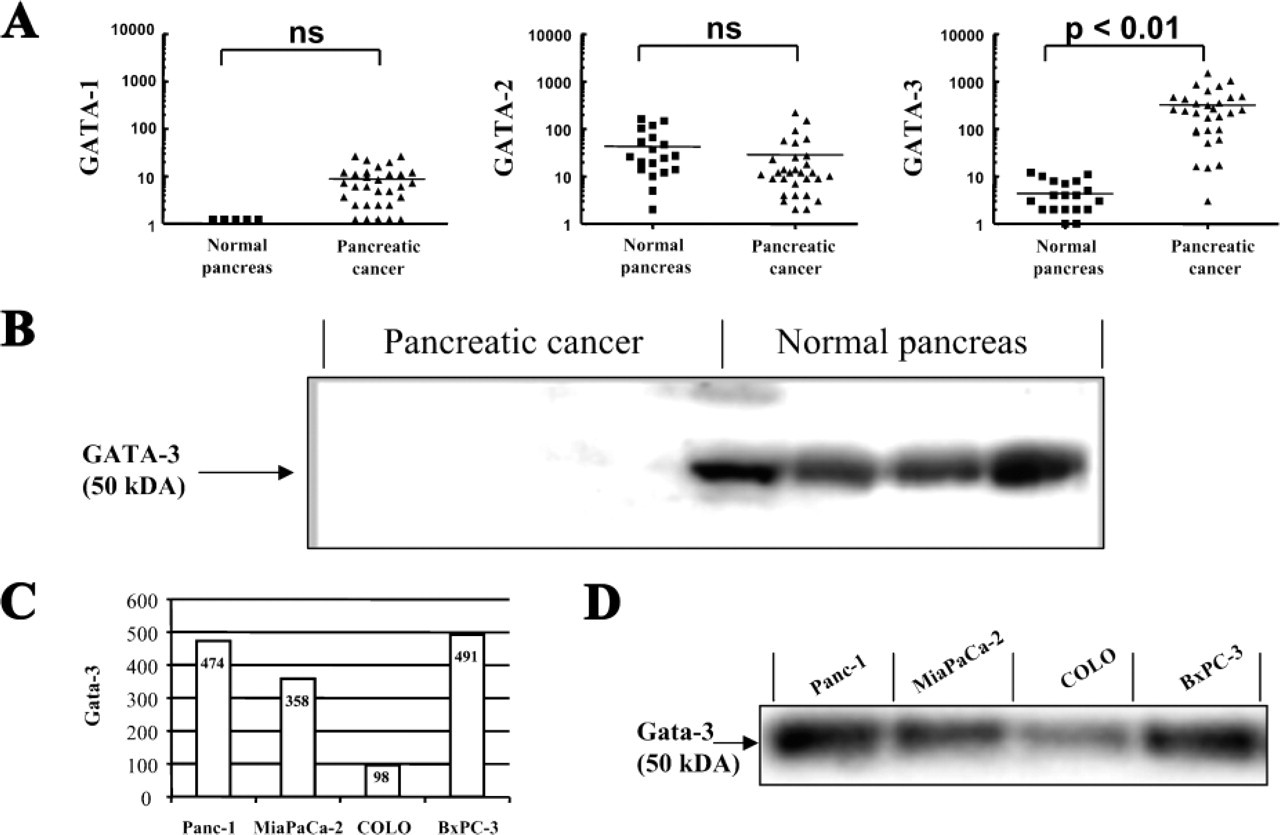
Twenty-seven human pancreatic cancer samples compared with 20 normal pancreatic tissues were analyzed by quantitative PCR. All pancreatic cancer samples demonstrated a significant overexpression of Gata-3 mRNA, whereas normal pancreas showed only very few or no copies of Gata-3 mRNA. On average, a 69-fold upregulation of Gata-3 mRNA (
(
In contrast, other members of the Gata family such as Gata-1 and −2 revealed no alteration in the expression patterns between normal pancreas and pancreatic cancer (Figure 1). Patients with different tumor stages and grading did not reveal significant differences in the expression of the three Gata mRNA isoforms.
Analysis of the protein content of the same tissue using Western blot analysis revealed similar results: in normal pancreatic tissue samples Gata-3 protein was below detection level, whereas the pancreatic cancer tissues showed clear upregulation of Gata-3 protein levels, which was independent of the tumor stage (Figure 1).
Through quantitative PCR, as well as in Western blot analysis, an abundant expression of Gata-3 mRNA and protein was observed in all four human pancreatic carcinoma cell lines (BxPC-3, Colo 357, MiaPaCa-2, Panc-1) (Figures 1C and 1D).
Cytoplasmic Localization of Gata-3 in Pancreatic Cancer Cells
To further determine whether pancreatic cancer cells are the only origin of Gata-3 production, pancreatic cancer tissues were analyzed by immunohistochemistry. No or only a faint staining in some acinar cells was detected in the normal pancreas (Figure 2). In contrast, strong and persistent Gata-3 immunoreactivity was found in the cancer cells (Figures 2B and 2C). Furthermore, some tubular complexes in the surrounding cancerous environment and infiltrating mononuclear cells also showed positive immunostaining (Figures 2D and 2E). Surprisingly, all cancer cells showed cytoplasmic staining of Gata-3 (Figures 2B and 2C), whereas in tubular complexes and mononuclear immune cells nuclear staining was observed (Figures 2D and 2E). All stained pancreatic samples showed a similar staining pattern. This is in contrast to the nuclear Gata-3 immunostaining pattern in human breast carcinomas, which has been reported recently. In our study we also confirmed nuclear staining in cancer cells of human breast cancer samples (Figure 2F).
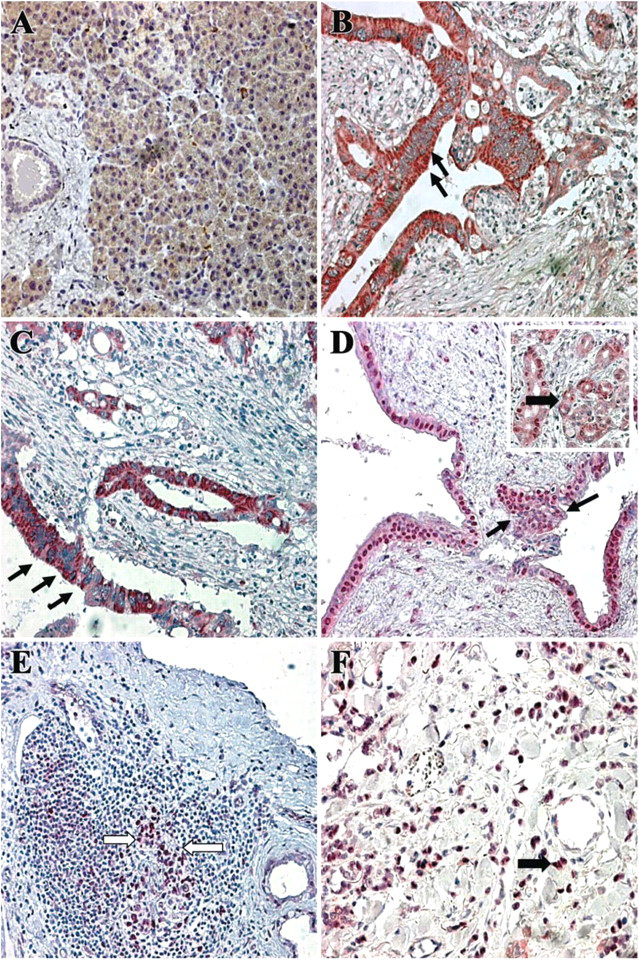
Unique cytoplasmic localization of GATA-3 in pancreatic cancer cells. Immunohistochemical staining did not show GATA-3 immunoreactivity in the normal pancreas (
To determine the exact localization of Gata-3 in pancreatic cancer cells, the four human pancreatic carcinoma cell lines (BxPC-3, Colo 357, MiaPaCa-2, Panc-1) were analyzed by confocal microscopy using double staining with the nuclear stain TOTO-3. All cell lines demonstrated a strong cytoplasmic immunofluorescence activity of Gata-3 with an intense perinuclear zone (Figures 33-A3), whereas no Gata-3 immunoreactivity could be detected in the nucleus.
Gata-3 Is Transcriptionally Inactive in Pancreatic Cancer Cells
To test whether Gata-3 has any transcriptional activity in pancreatic cancer cells, we measured the DNA-binding activity of nuclear extracts in EMSA. Gata-3 nuclear translocation and DNA binding in pancreatic cancer cells was assessed in comparison to MOLT-4, an acute lymphoblastic leukemia cell line which is known to have transcriptionally active Gata-3 (certified positive control for Gata-3 from Santa Cruz Biotechnology). None of the four tested pancreatic cancer cell lines revealed Gata-3 DNA binding (Figure 3E), suggesting that in pancreatic cancer cells Gata-3 is transcriptionally inactive.
Gata-3 Expression in Pancreatic Cancer Correlates with TGF-β Signaling
To evaluate a potential relationship, a correlation analysis of Gata-3 mRNA expression levels and several TGF-β signaling pathway-related molecules (TGF-β1 and −β2, TGFβR-I and -II, and Smad3) was performed. The correlation analysis revealed a statistically significant relationship between the expression of Gata-3, the ligands TGF-β1 and −β2, and their receptors TGFbR-I and -II (Spearman's rho 0.6, 0.4, 0.6, 0.5, respectively;
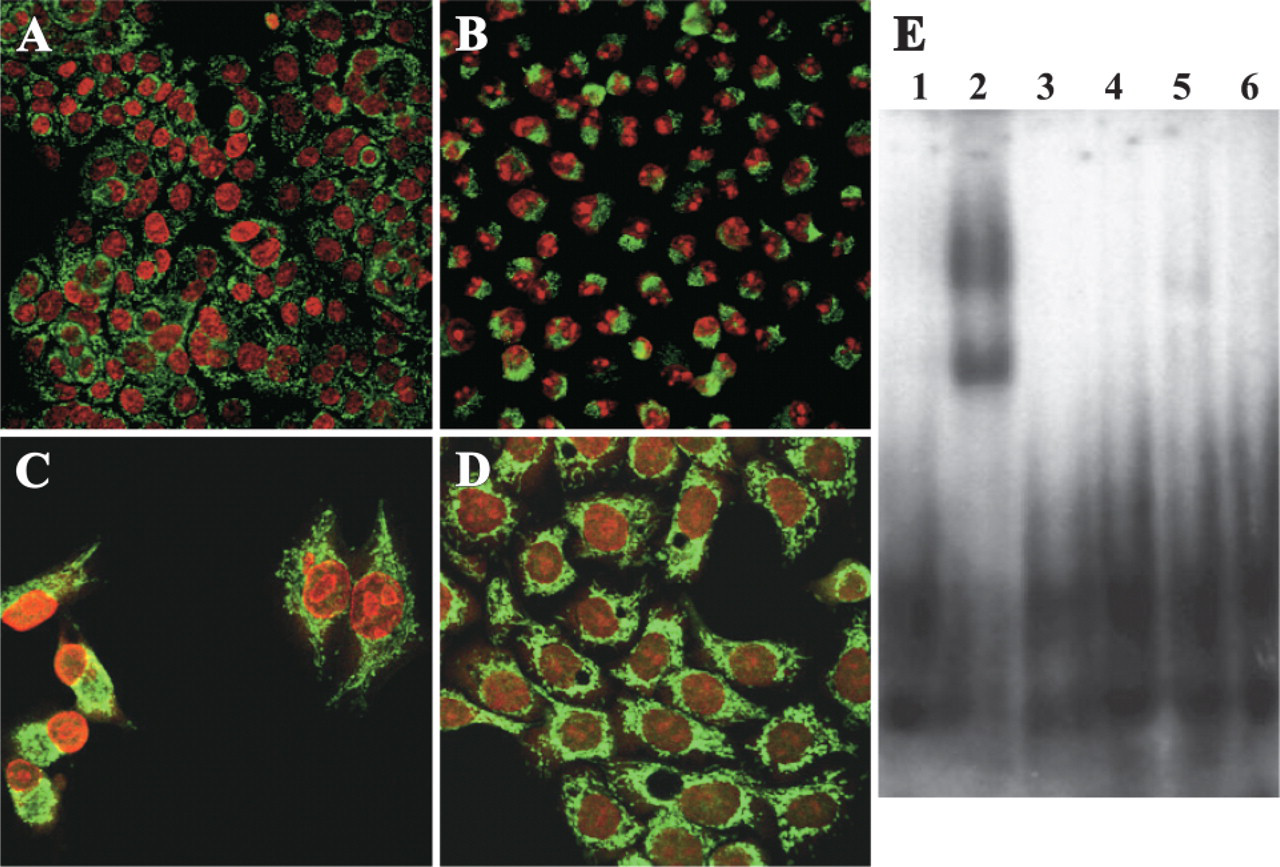
Localization of GATA-3 in pancreatic cancer cell lines. Confocal microscopy exposed GATA-3 immunoreactivity in the cytoplasm of all four pancreatic cancer cells lines BxPC-3 (
Exogenous TGF-β1 Transiently Inhibits Gata-3 Expression in TGF-β-responsive Cell Lines
To determine whether the association between Gata-3 and TGF-βs in human cancerous tissues is based on the regulation of Gata-3 by TGF-βs, we exposed the previously described TGF-β-responsive (Colo 357, Panc-1) and -unresponsive pancreatic cancer cell lines (Mia-PaCa-2 and BxPC-3) to exogenous TGF-β1. The TGF-β-responsive cell lines (Colo 357, Panc-1) showed transient but marked growth inhibition in addition to an upregulation of p21 and an auto-induction of TGF-β1. In contrast, Gata-3 expression was significantly downregulated (
Discussion
Gata-3, a crucial transcription factor in T-cell development and differentiation, has recently shown variable expression patterns in different human cancers (Shiga et al. 1993; Hoch et al. 1999; Steenbergen et al. 2002). The presumed role of Gata-3 in the pathogenesis of cancer, however, still remains unclear. This study investigated, for the first time, the role of Gata-3 in human pancreatic cancer.
Our study results demonstrated a clear overexpression of Gata-3 at the mRNA and protein level in human pancreatic cancer, compared with the normal human pancreas. Conversely, other members of the GATA family, such as Gata-1 and Gata-2, revealed no altered expression between the cancerous and normal pancreas. This suggests that Gata-3 plays a specific role in pancreatic cancer. Abundant expressions of Gata-3 were found in four different human pancreatic carcinoma cell lines. In most of the cancer cells, immuno-histochemical stainings revealed a strong Gata-3 signal that, surprisingly, was located in the cytoplasm, whereas no staining was observed in the nucleus. In contrast, some tubular complexes and mononuclear immunocytes showed, in fact, a Gata-3 signal in the nucleus. Double staining with a nuclear stain and Gata-3 by confocal microscopy demonstrated the same cytoplasmic staining pattern in all four pancreatic cancer cell lines. This finding contrasts the nuclear Gata-3 staining pattern in human breast carcinomas recently reported by Hoch et al. (1999), suggesting that the cytoplasmic pattern is a specific feature in pancreatic cancer cells. In the electrophoretic mobility shift assay, Gata-3 did not bind to the specific Gata-3 consensus-binding site, neither in the nuclear nor in the cytoplasmic fraction. These two observations may suggest that Gata-3 is somehow blocked in the cytoplasm and not able to translocate into the nucleus. Impaired phosphorylation, mutations, or inhibiting signals may be responsible. Further studies looking at cancerous precursor lesions using non-transformed pancreatic ductal cancer cells will be needed to demonstrate the significance of blocked nuclear translocation of Gata-3 in the pathogenesis of pancreatic cancer.
While searching for a functional association between Gata-3 upregulation and known gene alterations in pancreatic cancer, we found a moderate to strong correlation between Gata-3 expression, the ligands TGF-β1 and −β2 and their receptors TGFbR-I and -II, as well as between Gata-3 and Smad-3, in human pancreatic cancer samples. Pancreatic cancer is known to have redundant barriers to TGF-β signaling, which allows cancer cells to escape TGF-β-induced growth inhibition (Ellenrieder et al. 2002). Thus far, it is believed that the growth-inhibitory effects of TGF-β are altered in most pancreatic cancers either by an imbalance in the expression of the signaling receptors (Friess et al. 1993; Wagner et al. 1998), mutations in intracellular signaling genes (Smad4) (Hahn et al. 1996), or by upregulation of TGF-β pathway signaling inhibitors (Smad6 and Smad7) (Kleeff et al. 1999a, b). Nevertheless, the exact molecular mechanism of the resistance of pancreatic cancers to the inhibitory growth action of TGF-βs is still not fully understood (Korc 1998). Recently it has been shown that TGF-β inhibits the differentiation of Th2 helper cells and the production of cytokines by impeding Gata-3 expression. This is suggested to represent an inhibitory feedback mechanism that limits interaction between Gata-3 and TGF-β signaling (Gorelik et al. 2000; Blokzijl et al. 2002). Correspondingly, this study also revealed that Gata-3 expression is regulated by TGF-β in pancreatic cancer cells. Cell lines that are still partly TGF-β responsive, such as Colo 357 and Panc-1, showed significant downregulation of Gata-3 expression, in contrast to marked growth inhibition, p21 upregulation, and TGF-β1 auto-induction. On the other hand, TGF-β1-resistant pancreatic cell lines exhibited no downregulation of Gata-3 expression, leading to persistently high levels of Gata-3 in the cancer cells. This suggests that the barriers to TGF-β signaling in pancreatic cancer may be responsible for the persistent upregulation of Gata-3, and that this uninhibited Gata-3 expression supports persistent growth phenotype of pancreatic cancer cells.
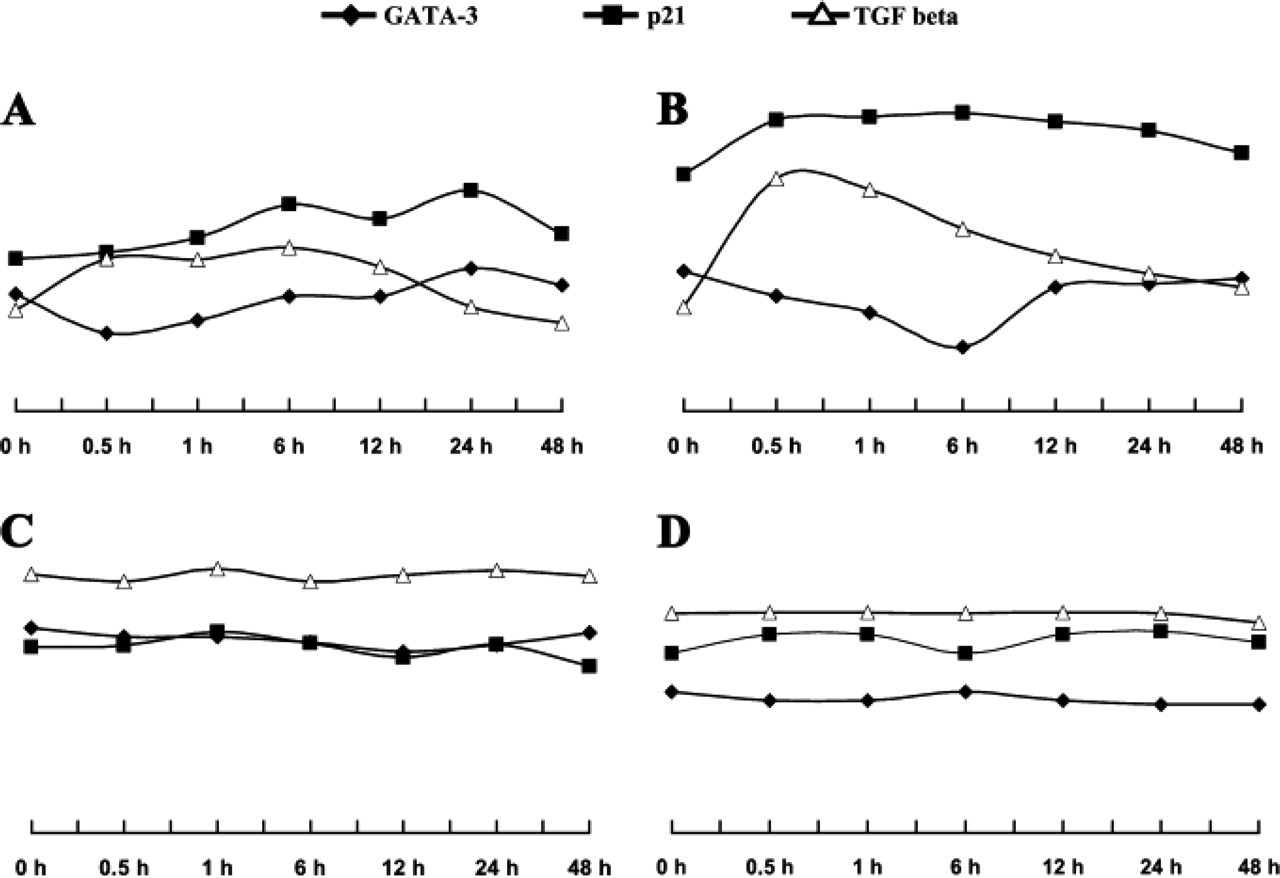
TGF-β transiently inhibits GATA-3 expression. Pancreatic cancer cell lines were exposed to exogenous TGF-β. GATA-3, TGF-β1, and p21 mRNA expression was quantified by quantitative PCR. TGF-β-responsive cell lines Panc-1 (
Furthermore, a recent study in lymphoid cells revealed that a direct physical interaction between Smad3 and Gata-3 is needed to control TGF-β-dependent regulation of cell-specific target genes (Blokzijl et al. 2002). Gata-3 overexpression interferes not only with the differentiation of T-cells but also several other cell systems such as neurons and adipocytes (Lim et al. 2000; Tong et al. 2000). In both cell types, dysregulation of Gata-3 expression leads to a severe disturbance in differentiation processes. Also, in neurons and adipocytes, Gata-3 seems to interfere with the TGF-β signaling pathway. The authors concluded that through this interaction the cell might be able to integrate its genetic differentiation program with an extracellular signal. Dedifferentiation processes are suspected to be of great importance, especially in the initial phase of pancreatic carcinogenesis (Real 2003). If the interaction of TGF-β and Gata-3 signaling is involved in de-differentiation, processes in pancreatic cancer cells need to be evaluated in further studies.
In conclusion, this study suggests that Gata-3 transcriptional function is disturbed in pancreatic cancer cells, whereas tubular complexes and immunocytes show pronounced upregulation; however, with normal nuclear translocation. Furthermore, Gata-3 seems to have a connection to the frequently altered TGF-β pathway in pancreatic cancer. The exact role of the high levels of Gata-3 in pancreatic cancer cells remains to be explained in additional studies. However, we speculate here that disturbed Gata-3 transcriptional function in concert with TGF-β signaling gives pancreatic cancer cells a distinct growth advantage and might be associated with dedifferentiation mechanisms, which may finally lead to malignant transformation of pancreatic cells.
