Abstract
Objective
To investigate the effects of vitamin D (VitD) on GATA-3 and T-bet expression in the nasal mucosa of allergic rhinitis (AR) rats and on regulation of the Th1/Th2 immune balance.
Methods
24 rats were randomly categorized into Control, Model, and VitD groups (n = 8 per group). The Model and VitD groups were sensitized with ovalbumin and aluminum hydroxide to establish an AR rat model. The VitD group received intranasal VitD treatment; the Model group received the equivalent volume of saline. Behavioral scores were recorded before and after modeling and following the final intervention. Nasal mucosal tissues were stained with hematoxylin-eosin to assess histology and eosinophil infiltration. Serum levels of interferon-γ (IFN-γ) and interleukin-4 (IL-4) were measured using enzyme-linked immunosorbent assays. Western blot was used to detect GATA-3 and T-bet expression levels.
Results
Compared with the control, the Model group exhibited typical nasal symptoms with increased behavioral scores; marked inflammatory cell infiltration, tissue edema, and vascular dilation in the nasal mucosa; elevated serum IL-4 levels; reduced IFN-γ levels; downregulated T-bet expression; and upregulated GATA-3 expression. Compared to the Model group, the VitD group showed controlled nasal symptoms, reduced behavioral scores, alleviated inflammatory cell infiltration, decreased serum IL-4 levels, increased IFN-γ levels, upregulated T-bet expression, and downregulated GATA-3 expression.
Conclusion
Intranasal VitD significantly downregulated GATA-3, upregulated T-bet, reduced serum IL-4, and elevated IFN-γ levels. This may be linked to modulation of upstream transcription factors T-bet and GATA-3, potentially altering the differentiation tendency of Th0 cells toward Th1/Th2 subsets.
Introduction
Allergic rhinitis (AR) is a common noninfectious chronic inflammatory disorder of the nasal mucosa, mediated primarily by immunoglobulin E (IgE) in sensitized individuals upon exposure to allergens. It is characterized by paroxysmal sneezing, watery rhinorrhea, nasal itching, and congestion.1,2 AR severely affects patients’ quality of life and work efficiency, and its incidence has increased annually due to urbanization and industrialization. Current treatment approaches include pharmacotherapy, environmental control, immunotherapy, and surgical intervention. While existing therapies demonstrate some efficacy, they are limited by significant variability in individual responses, high recurrence rates after discontinuation, difficulty in effective allergen avoidance, long-term medication requirements, adverse effects, and high costs. Therefore, identifying more effective therapeutic strategies to alleviate clinical symptoms and improve patients’ quality of life remains critical.
The pathogenesis of AR involves genetic predisposition, environmental factors, and immune dysregulation. Current evidence highlights Th1/Th2 immune imbalance as a central mechanism in AR development. 3 Th0 cells, precursors of T-helper cells, differentiate into Th1 or Th2 subsets under the regulation of specific transcription factors. Under physiological conditions, Th1 and Th2 cells maintain dynamic equilibrium. However, in patients with AR, excessive Th2 cell activation skews the immune response toward a Th2-dominant phenotype. 4 Enhanced Th2 immunity elevates levels of IL-4, IL-5, and IL-13. Notably, IL-4 plays a pivotal role in AR progression by promoting IgE synthesis and eosinophil (EOS) activation/recruitment. Th1 cells primarily produce interferon-γ (IFN-γ), suppressing IgE production, inhibiting EOS and mast cell infiltration into nasal mucosa, and dampening local immune responses. 5 IFN-γ further reinforces Th1 differentiation and function through positive feedback. Consequently, reduced Th1 activity and diminished IFN-γ levels exacerbate Th2 dominance.
Th2 cell differentiation is regulated by the upstream transcription factor GATA-3, which induces Th2 cytokine gene expression and biases the Th1/Th2 balance. Activated by STAT6, GATA-3 promotes IL-4, IL-5, and IL-13 expression while suppressing T-bet to inhibit Th1 polarization. 6 Conversely, Th1 differentiation is governed by T-bet, which induces IFN-γ production. 7 Studies suggest that overexpression of GATA-3 and underexpression of T-bet disrupt the Th1/Th2 equilibrium, leading to dysregulated IFN-γ/IL-4 secretion, a hallmark of AR pathogenesis. Restoring the T-bet/GATA-3 ratio may thus represent a therapeutic target for AR management. 8
Emerging evidence indicates that vitamin D (VitD) exerts therapeutic effects on AR by modulating Th1/Th2 imbalance and influencing serum IL-4/IFN-γ levels.9–11 Our preliminary clinical studies demonstrated that VitD supplementation partially corrected IFN-γ/IL-4 dysregulation in patients with AR, 12 which suggests VitD may inhibit Th0-to-Th2 differentiation while promoting Th1 polarization. However, the effect of VitD on the upstream transcription factors T-bet and GATA-3 remains unexplored. This study aimed to investigate the effects of VitD on T-bet and GATA-3 expression in an AR rat model, elucidating its immunomodulatory mechanisms and providing molecular-level evidence for VitD-based AR therapy.
Methods
Experimental animals and grouping
Twenty-four specific-pathogen–free male Sprague-Dawley rats (6–8 weeks old, 200 ± 20 g) were obtained from the Experimental Animal Center of Zunyi Medical University (License No.: SCXK (Qian) 2021-0004). The rats were housed under controlled conditions (20–23°C, 40%–60% humidity) at the Experimental Animal Center of Zunyi Medical University. After 1 week of acclimatization, the rats were numbered from 1 to 24 and randomly categorized into three groups (n = 8 per group): Control, Model, and VitD.
This study was approved by the Animal Ethics Committee of Zunyi Medical University (Approval No.: ZMU21-2304-007; April 17, 2023). Furthermore, all procedures strictly followed both institutional ethical standards and the ARRIVE 2.0 guidelines. 13
Modeling protocol
An AR rat model was established by sensitization with ovalbumin (OVA) and aluminum hydroxide (Al [OH]3) following methods established in the literature.14,15 This sensitization was conducted in two phases: 1) Primary sensitization phase: A suspension containing 0.3 mg OVA, 30 mg Al (OH)3, and 1 mL normal saline (NS) was prepared. From May 1 to May 13, 2024, rats in the two experimental groups received an intraperitoneal injection of this suspension every other day for a total of seven injections, while the Control group received equivalent NS injections. 2) Challenge phase: Starting the day after sensitization (May 14, 2024), 50 μL of 5% OVA-NS suspension was administered intranasally (25 μL per nostril) for eight consecutive days (until May 21, 2024). The Control group received intranasal instillation of an equivalent volume of NS.
Intervention protocol
Beginning the day after modeling completion: 1) Control group: no intervention, standard feeding; 2) VitD group: received an intranasal intervention of active vitamin D3 dissolved in normal saline. The dosage for rats was set at 5.1 × 10-3 mg/kg/day. This dose was calculated based on the common clinical dose for humans (approximately 2000 IU/day for a standard 70 kg adult, equivalent to 8.3 × 10-4 mg/kg/day) and converted according to the body surface area conversion coefficient (typically a ratio of approximately 6.3 for rats to humans) as described in Pharmacological Experimental Methodology. 16 The determination of this dose was based on preliminary experiments and literature reports, falling within the clinically effective and safe supplementation range 17 ; 3) Model group: intranasal NS (equivalent volume once daily for 2 weeks).
Reagents and instruments
Key reagents and instruments: OVA (A5503, Sigma-Aldrich, USA), Al (OH)3 (CAS: 21645-51-2, Chengdu Boruiya Biotechnology Co., Ltd), and vitamin D3 (A146813, Ambeed, purity ≥98%). Enzyme-linked immunosorbent assay (ELISA) kits: Rat IL-4 ELISA kit (JL20894-48T, Jianglai Biological), rat IFN-γ ELISA kit (JL13241-48T, Jianglai Biological). Antibodies: anti-actin primary antibody (1:3,000, HA722023, Huaan Biotechnology), horseradish peroxidase (HRP)-conjugated goat anti-rabbit secondary antibody (1:5,000, RGAR001, Wuhan Sanying Biotechnology), anti-GATA-3 primary antibody (1:1,000, ER1901-20, Huaan Biotechnology), anti-T-bet primary antibody (1:1,000, 13700-1-AP, Wuhan Sanying Biotechnology). Instruments: microplate reader (Thermo Scientific, Model 3020), upright fluorescence microscope imaging system (Leica BM3000), image analysis software (Adobe Photoshop, Adobe Inc.)
Experimental procedures
Behavioral observation and scoring
Symptom scoring criteria for animal models of allergic rhinitis.
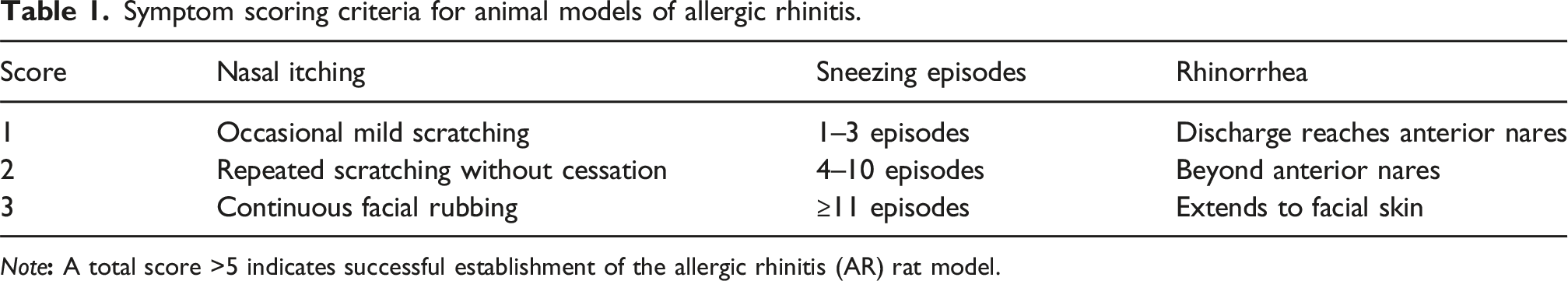
Note
Histopathological examination of nasal mucosa
Thirty min after the final intervention, rats were placed in an induction chamber, and anesthesia was induced with 3% to 5% isoflurane in air. After the rats lost consciousness, the isoflurane concentration was reduced to 1.5% to 3% for maintenance, with delivery controlled by an animal anesthesia machine. Surgical anesthesia was confirmed by the absence of a withdrawal reflex to hind-paw pinching, then blood was collected via the retro-orbital plexus. .After centrifugation (3,000 rpm, 15 min), serum was separated and stored at −80°C. Following cervical dislocation, nasal mucosa was dissected: one portion fixed in 10% neutral buffered formalin (24 h), the remainder stored at −80°C. Fixed tissues underwent gradient ethanol dehydration, xylene clearing, paraffin embedding, and sectioning (4 μm). Hematoxylin-eosin (H&E) staining was performed for light microscopy. To quantify eosinophilic infiltration, EOS were counted in five randomly selected, non-overlapping high-power fields (HPF) in each rat’s nasal mucosa section. The mean value of these five counts was defined as the final EOS count for the individual animal (i.e., n = 1 represents the mean EOS count per rat).
ELISA for serum IFN-γ and IL-4 levels
Thawed serum samples were analyzed using a double-antibody sandwich ELISA kit. Procedures included plate coating, washing, blocking, sample addition (50 μL/well), chromogenic substrate reaction, and termination. Optical density at 450 nm was measured with a microplate reader. Cytokine concentrations (pg/mL) were calculated from standard curves.
Western blot analysis of T-bet and GATA-3 proteins
Nasal mucosal proteins were extracted using a radioimmunoprecipitation assay buffer. After sodium dodecyl sulfate–polyacrylamide gel electrophoresis (10% gel, 80 V stacking/120 V resolving) and polyvinylidene difluoride membrane transfer, blots were blocked with 5% nonfat milk. Primary antibodies (GATA-3 [1:1000], T-bet [1:1000], β-actin [1:3000]) were incubated overnight at 4°C. HRP-conjugated secondary antibodies (goat anti-rabbit IgG, 1:5000) were applied for 1 h at room temperature. Protein bands were visualized by enhanced chemiluminescence. To normalize for protein loading, β-actin was used as an internal control. Band intensities of the target proteins (GATA-3 and T-bet) and β-actin were quantified using ImageJ software. The normalized protein expression level for each sample was calculated as the ratio of the target protein intensity to that of β-actin, and these values were used for subsequent statistical comparison between groups.
Statistical analysis
Data were analyzed in SPSS 29.0 and plotted using GraphPad Prism 9.0, expressed as mean ± standard deviation. Normality (Shapiro–Wilk) and homogeneity of variance (Levene’s test) were verified. Intergroup differences were assessed by one-way analysis of variance and least significant difference post hoc tests. p < 0.05 indicated statistical significance.
Results
Nasal symptom scores
Comparison of behavioral scores across experimental groups (x̄ ± s, n = 8).

Note
Histopathological observations of nasal mucosa
Microscopic examination revealed: 1) Control group: intact mucosal architecture with organized epithelial stratification, absent inflammatory infiltration. 2) Model group: disrupted mucosal barrier accompanied by vasodilatory edema, with significant EOS and neutrophil infiltration (arrows, Figure 1(b)). 3) VitD Group: significant reduction in inflammatory cell density compared to the Model group, accompanied by mucosal structural remodeling and vascular normalization (Figure 1). Pathological changes in nasal mucosa across experimental groups (H&E staining, 40×).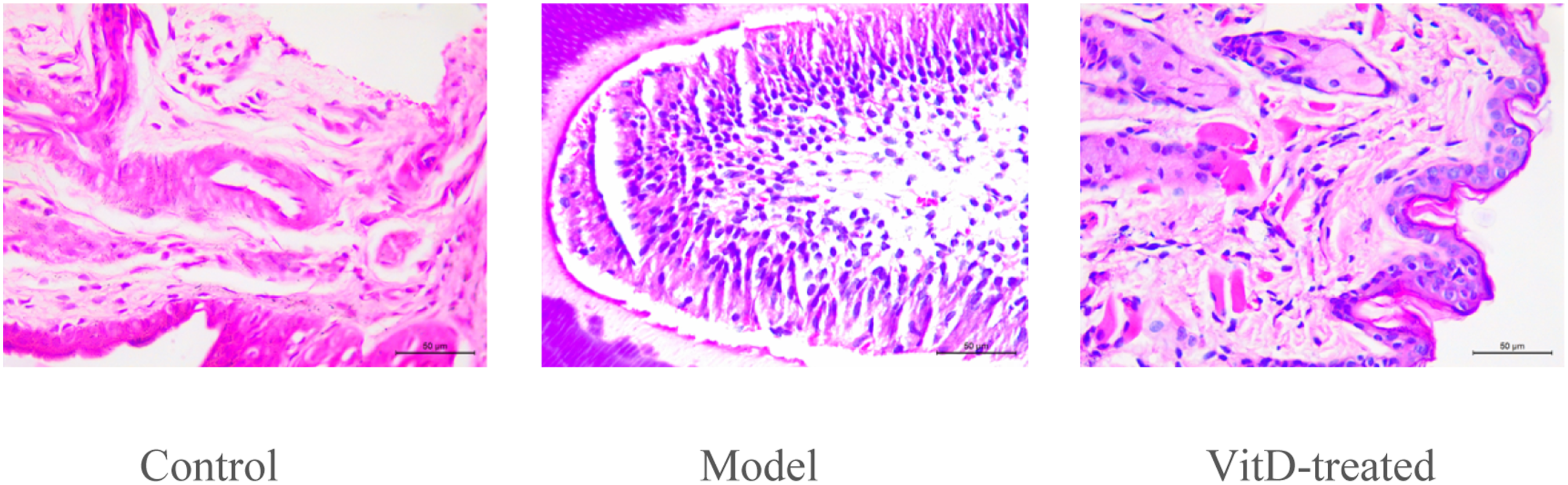
EOS counts in nasal mucosal smears
Comparison of eosinophil counts across experimental groups (x̄ ± s, n = 8).

Note
Serum IFN-γ and IL-4 levels
Comparison of serum IFN-γ and IL-4 levels across experimental groups (x̄ ± s, n = 8).

Note: p< 0.05 versus Control group at the same time point; △p < 0.05 versus Model group at the same time point.
Protein expression of specific transcription factors T-bet and GATA-3 in nasal mucosa
Compared to the Control group, the Model group exhibited significantly elevated GATA-3 expression (p < 0.001) and markedly reduced T-bet expression (p < 0.001) in the nasal mucosa. Following VitD intervention, the VitD group demonstrated restored T-bet expression and suppressed GATA-3 expression compared to the Model group, with statistically significant differences (p < 0.01) (Figure 2). Expression of T-bet and GATA-3 proteins in nasal mucosa of rats **Note: ***p < 0.001 compared with the control group at the same time point; p < 0.05 compared with the model group at the same time point; Note. A, Control group; B, Model group; C, VitD group.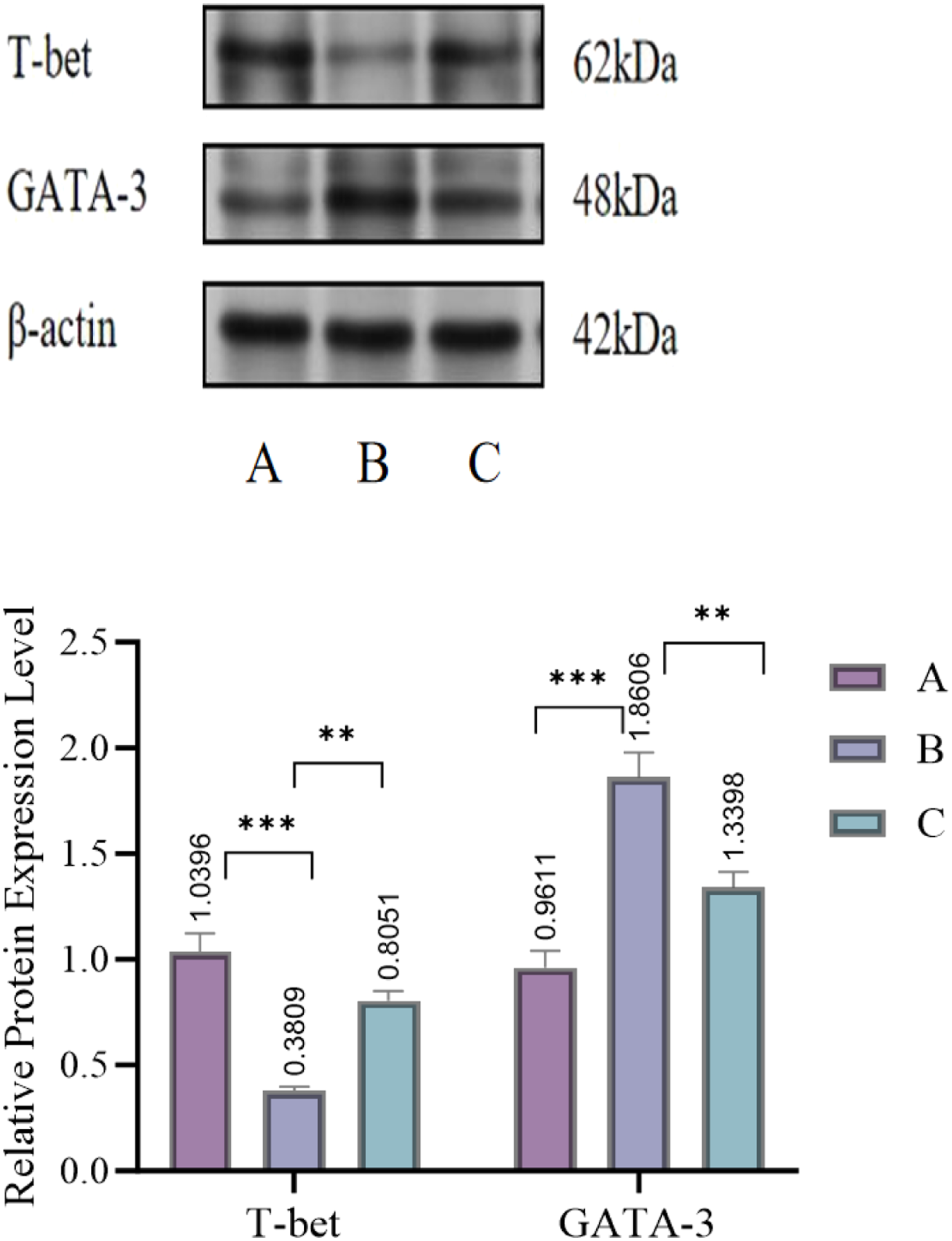
Discussion
Current evidence suggests that the pathogenesis of AR is closely associated with Th1/Th2 immune imbalance. Th1 cells primarily mediate immune responses through IL-2, IL-12, IFN-γ, and TNF-β cytokines. IFN-γ activates macrophages to enhance phagocytic and pathogen-killing capabilities, playing a pivotal role in cellular and antiviral immunity. In contrast, Th2 cells release mediators including IL-4, IL-5, IL-13, and IgE. IL-4 promotes B-cell proliferation, differentiation, and antibody production, particularly inducing IgE synthesis, which is critical in AR pathogenesis. 19 IL-5 activates EOS, amplifying their phagocytic and degranulation functions, while IL-13 modulates airway epithelial function, inducing mucus hypersecretion and airway hyperresponsiveness. 20 Under physiological conditions, Th1 and Th2 cells maintain immune homeostasis through dynamic equilibrium, coordinating responses to external antigens and ensuring stable immune function. However, Th2-dominant polarization disrupts this balance, leading to the overproduction of Th2 cytokines and IgE, which exacerbate mast cell degranulation and inflammatory cascades, thereby intensifying AR symptoms. 21
T-bet, the master transcription factor for Th1 cells, drives Th1 differentiation and upregulates IFN-γ expression. By suppressing Th2 cytokine production, T-bet reinforces Th1 polarization and functional stability. Conversely, GATA-3, the central regulator of Th2 responses, promotes IL-4 secretion while inhibiting IFN-γ, thereby sustaining Th2 dominance.22,23 The reciprocal inhibition between T-bet and GATA-3 establishes a dynamic equilibrium critical for Th1/Th2 homeostasis. In AR, allergen exposure disrupts this balance, manifesting as GATA-3 overexpression and T-bet suppression. This molecular dysregulation triggers aberrant Th2 proliferation, impaired Th1 responses, and perpetuated Th1/Th2 immune deviation, fueling mucosal inflammation and hypersensitivity.22,23
VitD, a lipophilic steroid hormone, exerts immunomodulatory effects via its active metabolite, 1,25(OH)2D3, binding to the vitamin D receptor on immune cells and regulating both innate and adaptive immunity.24–26 In innate immunity, VitD enhances antimicrobial activity and antigen presentation in macrophages and dendritic cells (DCs) while upregulating tight junction proteins (e.g., occludin, claudin) to fortify mucosal integrity, thereby reducing allergen penetration. 27 In adaptive immunity, VitD suppresses Th2 cell activation, inhibits B-cell proliferation/plasma cell differentiation, and reduces IgE production.28,29 Additionally, VitD drives DCs toward a tolerogenic phenotype, curtailing T-cell hyperactivation and disrupting AR-related inflammatory cascades. 30
Clinical studies consistently report lower serum VitD levels in patients with AR compared to healthy controls.31,32 While some evidence suggests VitD supplementation alleviates nasal symptoms in pediatric AR, 33 the correlation between VitD deficiency and AR severity remains controversial,34–36 necessitating further mechanistic and clinical validation. Notably, intranasal VitD administration has emerged as a promising AR therapy. 37 First, it acts directly on the nasal cavity, allowing the drug to rapidly reach the nasal mucosa and exert effects on local immune and epithelial cells, thereby modulating regional immune and inflammatory responses. Furthermore, the required local dosage is significantly lower than that for systemic administration (e.g., 300,000 IU of VitD3 once weekly for 4 weeks in clinical studies), thereby avoiding adverse effects such as hypercalcemia associated with high-dose systemic exposure. 38 Second, VitD has a low molecular weight and demonstrates considerable permeability through the nasal mucosa. As an oily preparation, it can form a protective layer on the mucosa, potentially acting as a barrier against allergens. While studies indicate that oily vehicles alone, despite reducing allergen exposure, cannot suppress mucosal inflammation, 38 we posit that VitD itself contributes positively to the therapeutic effect on AR. 39 Finally, local intranasal delivery minimizes systemic side effects, enhances drug bioavailability at the target site, and avoids first-pass metabolism in the gastrointestinal tract and liver. 40
In AR pathophysiology, allergen exposure triggers inflammatory mediator release (e.g., histamine, leukotrienes). Histamine stimulates nasal nerve endings, inducing pruritus and nasal scratching, while vascular hyperpermeability causes rhinorrhea and congestion—all reflected in behavioral scores. Our study demonstrates that intranasal VitD significantly reduces AR-related behavioral scores, likely by suppressing inflammatory mediators and Th2-driven EOS recruitment (via IL-5 inhibition). Concurrently, VitD acts to downregulate serum IL-4 (Th2 marker) and upregulate IFN-γ (Th1 marker), an effect that restores Th1/Th2 balance. Western blot analysis further revealed VitD-mediated correction of GATA-3/T-bet imbalance, suggesting dual mechanisms: 1) suppressing GATA-3 to attenuate Th2 differentiation, and 2) enhancing T-bet to promote Th1 responses.
Study limitations
This study has several limitations. First, only male Sprague-Dawley rats were used. This decision was primarily made to control for the potential confounding effects of hormonal fluctuations associated with the estrous cycle in female rats, thereby reducing variability in this initial mechanistic investigation. However, we acknowledge that this design limits the generalizability of our findings across sexes, given the known influence of sex differences in allergic diseases. Second, the absence of a pre-experimental sample size calculation is another limitation; the sample size was determined based on historical data from similar models, and while statistically significant changes were observed, the relatively small sample size and the lack of a power calculation may affect the statistical power and reliability of the findings, potentially limiting the detection of subtle biological effects. Future studies should include both sexes and utilize larger sample sizes determined by formal power calculations to comprehensively evaluate the sexual dimorphism and robust efficacy of intranasal VitD therapy, providing a more solid foundation for clinical translation.
Clinical translation and future perspectives
The findings of this preclinical study offer promising translational implications for AR management. Our data suggest that intranasal VitD could serve as a novel adjunctive therapy, particularly for patients suboptimally responsive to intranasal corticosteroids. To bridge the gap between our rodent model and human application, several key issues must be considered. Regarding dosage, the effective dose in rats (5.1 × 10-3 mg/kg/day) requires careful conversion based on body surface area, yielding an estimated human equivalent dose of approximately 2000 IU, which aligns with existing clinical pilot studies using high-dose intermittent regimens (300,000 IU weekly). 17 Concerning safety, while intranasal delivery minimizes systemic exposure and theoretically reduces the risk of hypercalcemia compared to oral supplementation, rigorous human trials must first confirm local tolerability (e.g., avoiding mucosal irritation) and establish a safe dosing window through phase I clinical studies. Future research should focus on randomized controlled trials evaluating the efficacy and safety of intranasal VitD, both as a monotherapy and in combination with standard care.
Conclusion
This study shows that VitD ameliorates AR symptoms by rectifying Th1/Th2 imbalance through transcriptional regulation of T-bet and GATA-3. Our findings highlight the therapeutic potential of targeting the GATA-3/T-bet axis, providing an experimental rationale for VitD supplementation as an adjunctive AR therapy.
Footnotes
Acknowledgment
This research was completed under the meticulous guidance of Professor Li Qiang. From the determination of the research direction, the formulation of the experimental plan to the writing and revision of the thesis, he has devoted a great deal of effort and provided insightful guidance and meticulous help. Here, we would like to express our highest respect and heartfelt gratitude to Professor Li Qiang! Thanks to Lai Rui and Cai Yueyue for their full support and beneficial discussions during the experiment and data collection. This research was funded by the Health Commission of Guizhou Province (Project No.: gzwkj2023 - 198), and we would like to express our special thanks. Finally, I would like to deeply thank my parents and family members for their years of parenting, selfless dedication, and the love and support they have silently given behind the scenes, which have enabled me to fully immerse myself in my studies and research. Thanks to all my friends who have cared for and helped me. Sincere thanks are extended to all the teachers, classmates, friends, and institutions that have provided support and help for this research! The authors sincerely thank the Experimental Animal Center of Zunyi Medical University for their professional support in animal breeding and management. We also gratefully acknowledge the technical staff for their valuable assistance in sample processing and data collection during this research.
Ethical considerations
All experimental procedures involving animals were approved by the Animal Welfare Ethics Committee of Zunyi Medical University (Approval No. ZMU21-2304-007; April 17, 2023).
Author contributions
Tingting Zhu: Conceptualization, Methodology, Writing - Original Draft.Rui Lai: Investigation, Formal Analysis, Visualization. Yueyue Cai: Investigation, Formal Analysis, Visualization. Qiang Li: Supervision, Project Administration, Funding Acquisition. All authors have read and agreed to the final manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Health Commission of Guizhou Province (Grant Number gzwkj2023-198).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
All data generated or analyzed during this study are included in this published article (see Figure 1, ![]() , and Tables).
, and Tables).
Animal welfare
All procedures performed in this study were in accordance with the NIH Guide for the Care and Use of Laboratory Animals. And every effort was made to minimize suffering.
