Abstract
Two rat monoclonal antibodies have been generated to plant cell wall (1→4)-β-D-xylans using a penta-1,4-xylanoside-containing neoglycoprotein as an immunogen. The monoclonal antibodies, designated LM10 and LM11, have different specificities to xylans in relation to the substitution of the xylan backbone as indicated by immunodot assays and competitive-inhibition ELISAs. LM10 is specific to unsubstituted or low-substituted xylans, whereas LM11 binds to wheat arabinoxylan in addition to unsubstituted xylans. Immunocytochemical analyses indicated the presence of both epitopes in secondary cell walls of xylem but differences in occurrence in other cell types.
X
To understand the functional roles of cell wall polymers and their various substituted forms, antibodies are required to enable the location of specific structural features of polymers in the context of cells and tissues. Such probes are also useful for the analysis of differences in cell walls of mutants and the characterization of polymers during their isolation (Willats and Knox 2003). Antisera to high- and low-substituted forms of xylans and a monoclonal antibody specific to arabinoxylans have been described (Suzuki et al. 2000; Ordaz-Ortiz et al. 2004; Guillon et al. 2004). Here, we describe the generation and use of two monoclonal antibodies for the analysis of xylans and arabinoxylans in planta.
A neoglycoprotein (X5-BSA) was prepared by coupling xylopentaose (X5, Megazyme, Bray, Ireland) to BSA by reductive amination (Roy et al. 1983). X5 BSA (10 mg) was dissolved in 0.5 ml of 0.2 M sodium borate buffer pH 7.3. This was followed by the addition of 15 mg BSA and then 16 mg of sodium cyanoborohydride, and the tube was maintained in a water bath at 50C for 48 hr with occasional mixing. After 48 hr the pH was adjusted to pH 4.0 by the addition of 5.5 μl of 80% (v/v) acetic acid. The solution was then dialyzed extensively against distilled water with several changes over 72 hr. The coupling efficiency was determined following a phenol-sulphuric acid assay of carbohydrate content and indicated an average of 14 × 5 oligosaccharides per BSA molecule.
Rat immunization, hybridoma preparation, and cloning procedures were performed as described previously (Willats et al. 1998). Two male Wistar rats were challenged with approximately 100 μg X5 BSA in complete Freund's adjuvant, administered subcutaneously on day 0, with the same amount administered with incomplete Freund's adjuvant on days 34 and 64. On day 115, a selected rat was given a prefusion boost of 100 μg X5 BSA in 1 ml PBS by intraperitoneal injection, and the spleen was taken three days later for isolation of lymphocytes and fusion with rat myeloma cell line IR983F (Bazin 1982). Antibodies were selected by ELISA using birch wood xylan and wheat arabinoxylan as antigens. Subsequent characterization involved a combination of immunodot assays (IDAs) and competitive-inhibition ELISAs as described by Willats et al. (1998). Two monoclonal antibodies, designated LM10 (rat immunoglobulin class IgG2c) and LM11 (class IgM), were selected for full characterization.
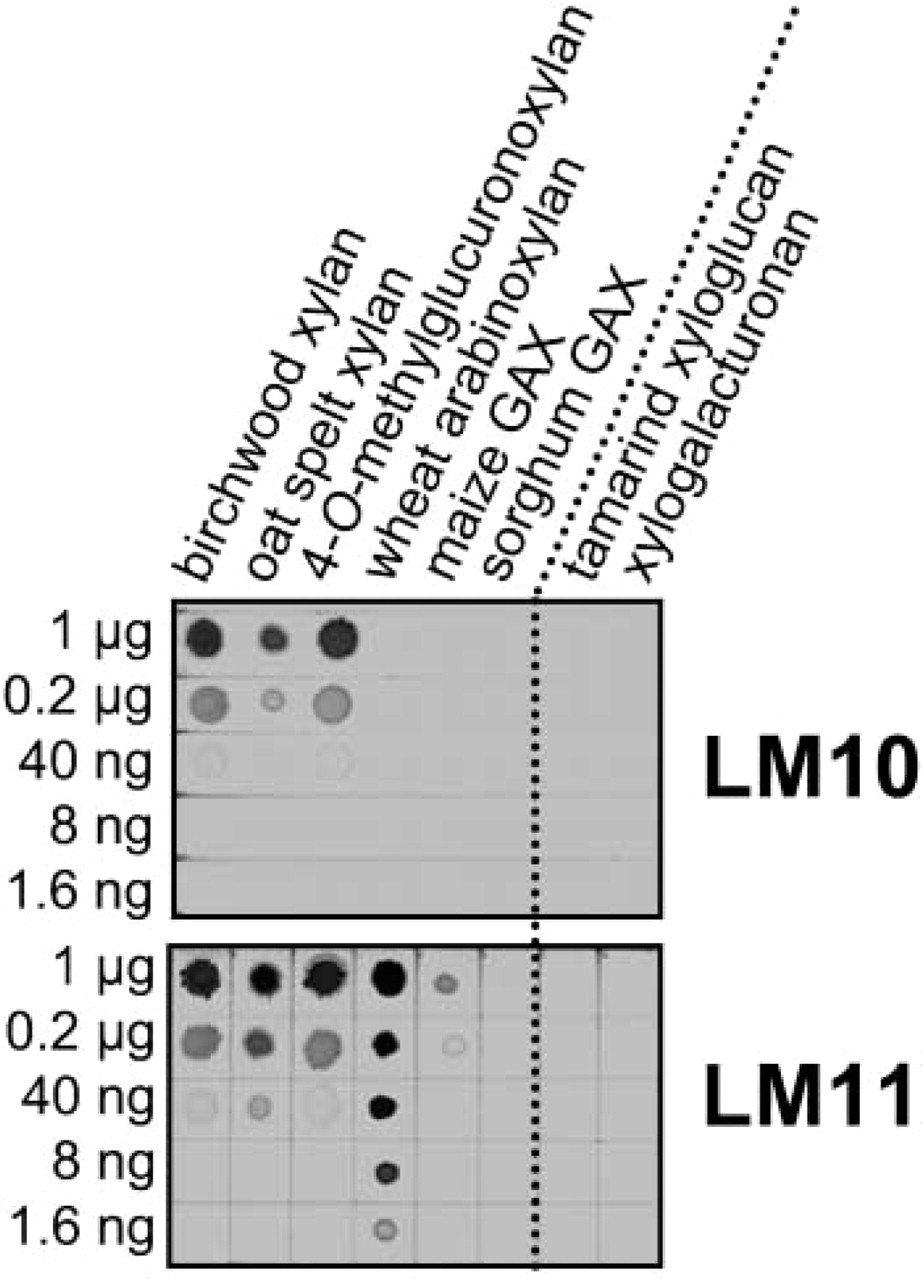
Immunodot assay of the binding of LM10 and LM11 to a series of xylans and other xylose-containing polysaccharides. Samples were applied to nitrocellulose as 1-μl dots from 1 μg to 1.6 ng.
Birch wood xylan (98% Xyl), 4-O-methyl-
For immunolabeling experiments, plant materials were excised and immediately fixed in PEM buffer (50 mM PIPES (piperazine-N-N'-bis[2-ethane-sulfonic acid]), 5 mM EGTA (ethylene glycol bis(β-aminoethylether)-N,N,N'N'-tetraacetic acid), 5 mM MgSO4, pH 6.9) containing 4% paraformaldehyde. Wheat grains were imbibed overnight and then were cut into small cubes of approximately 8 mm3 to include the aleurone layer and were immediately fixed as described previously. Procedures for embedding of plant material in Steedman's wax, sectioning, immunocytochemistry, and use of cellulose-binding Calcofluor White M2R fluorochrome (Sigma-Aldrich; Poole, Dorset, UK) are described elsewhere (McCartney et al. 2003).
Monoclonal antibodies LM10 and LM11 were selected because of their binding to series of soluble polysaccharides. The specificity of LM10 and LM11 is clearly demonstrated by immunodot assay, as shown in Figure 1. They detected approximately 50 ng xylan/dot. LM10 bound only to no- or low-substituted xylans such as birch wood and oat spelt xylans and did not bind to any other samples, including arabinoxylan or GAX. LM11, in contrast, bound strongly to wheat arabinoxylan and, to a lesser extent, to maize GAX in addition to the low-substitution xylans (Figure 1). Neither monoclonal antibody bound to the most substituted sorghum GAX nor to any other of a range of cell wall polysaccharides including glucans and pectins when applied at up to 1 μg/dot.
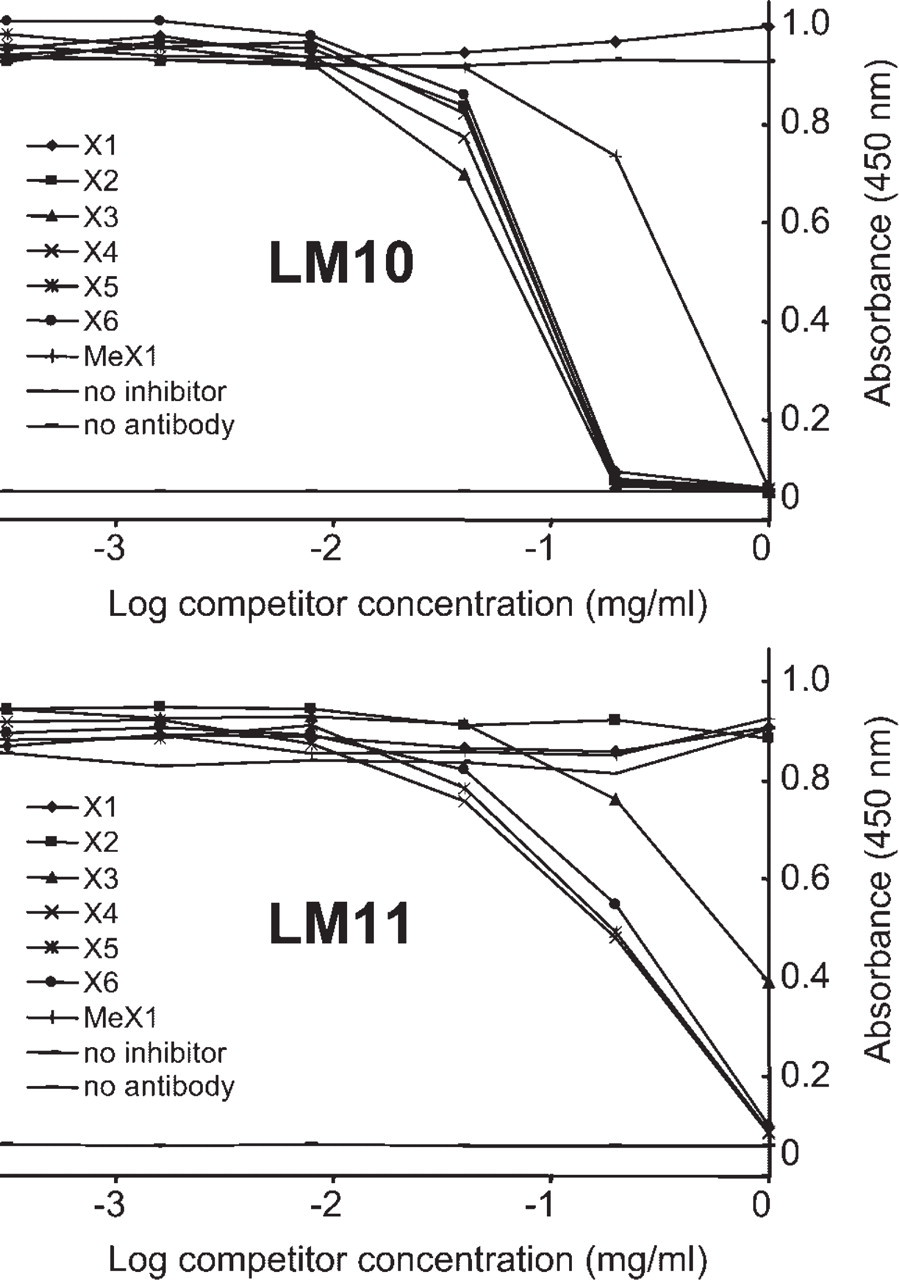
Competitive-inhibition ELISAs of LM10 and LM11 binding to birch wood xylan with xylose (X1), methyl-β-xylanoside (MeX1), and (1→4)-β-xylooligosaccharides (X2 to X6) as potential haptens.
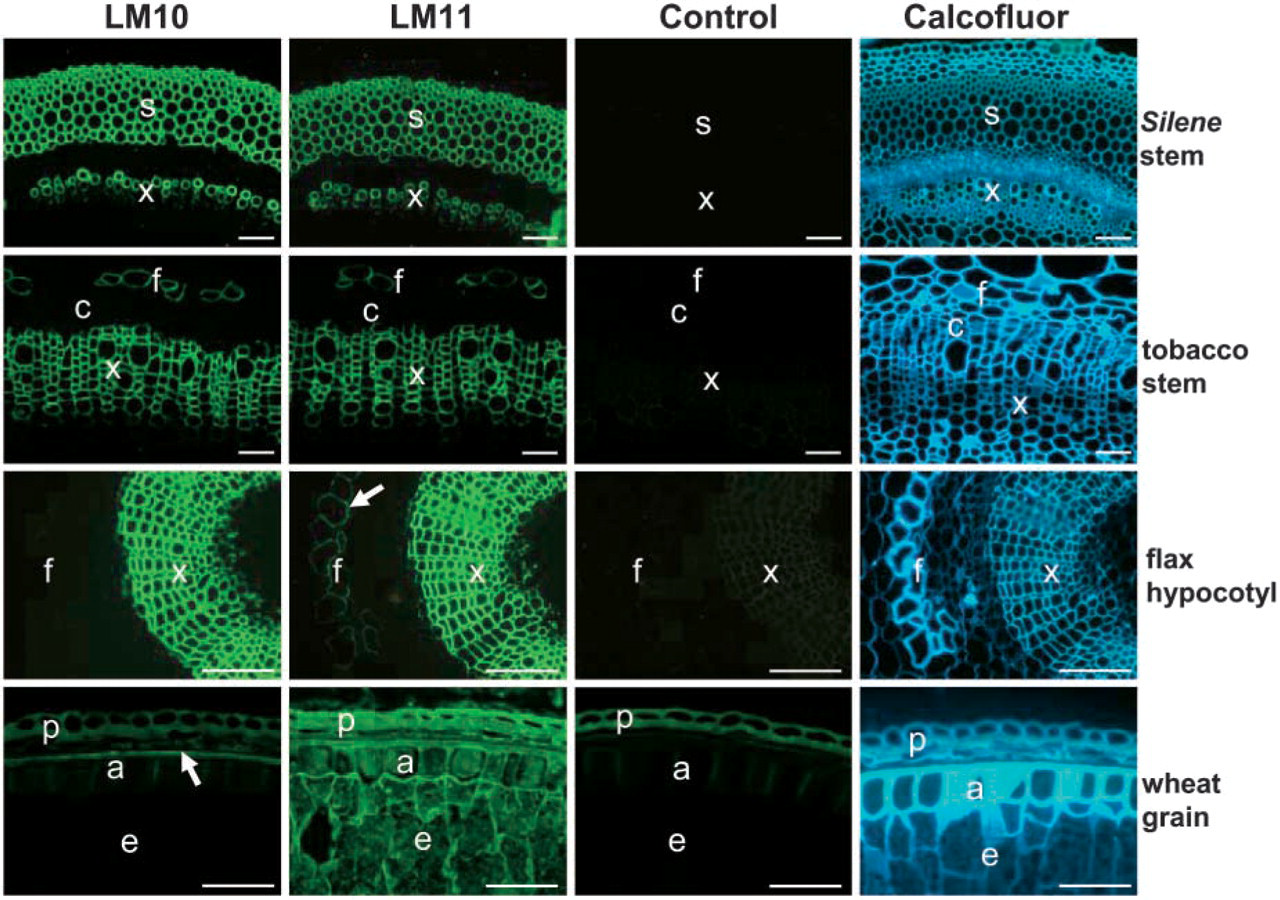
Indirect immunofluorescence micrographs of LM10 and LM11 binding to transverse sections of Silene and tobacco stems, flax hypocotyls, and wheat grain. For flax section labeled with LM11, the arrow indicates phloem fibers. For the wheat grain section labeled with LM10, the arrow indicates the outer cell wall of aleurone cells. Primary monoclonal antibody was omitted for the control micrographs. Calcofluor White staining of cellulose indicated all cell walls. a, aleurone layer; c, cambium; e, endosperm; f, phloem fiber; p, pericarp; s, sclerified parenchyma; X, xylem. Scale bars = 100 μm.
Additional characterization of LM10 and LM11 specificity was carried out by competitive-inhibition ELISA using birch wood xylan as the immobilized antigen, as shown in Figure 2. The potential hapten inhibitors were xylose (X1) through xylohexaose (X6) and methyl β-
Immunocytochemistry on a range of plant materials indicated that the LM10 and LM11 epitopes were restricted in dicotyledons to secondary cell walls, as shown in Figure 3. In Silene, both epitopes occurred exclusively in secondary cell walls of both xylem and sclerified parenchyma. This was the same in pea and Arabidopsis stems (data not shown). In tobacco stems, the LM10 and LM11 epitopes were abundant in the cell walls of xylem vessels and phloem fibers (Figure 3). In flax hypocotyls, both antibodies bound to the secondary cell walls of xylem, but the LM11 epitope also was observed in developing phloem fibers, whereas the LM10 was not, possibly indicating increased substitution of xylan in these cell walls. In a section of wheat grain, the LM10 epitope occurred in the outer cell wall of the aleurone layer/inner seed coat layer only, whereas LM11 labeled all aleurone cell walls in addition to the starchy endosperm cell walls, reflecting its capacity to bind to wheat arabinoxylan. These two rat monoclonal antibodies will be useful probes for the detection and analysis of xylans and arabinoxylans in plant materials.
Footnotes
Acknowledgments
We acknowledge financial support from the UK Biotechnology and Biological Sciences Research Council.
We thank Dr. Christine Andème-Onzighi for the preparation of flax hypocotyls and Dr. Henk Schols and Professor Harry Gilbert for xylans.
