Abstract
We previously reported that CD1d, a molecule responsible for the presentation of lipid antigens, is expressed in atherosclerotic lesions and that its expression is restricted to dendritic cells. Recent studies demonstrating that CD1d-restricted natural killer T (NKT) cells are involved in atherogenesis prompted the present study investigating whether NKT cells are present in human atherosclerotic lesions and, if so, whether there is an association between NKT cells and dendritic cells. We found that NKT cells do accumulate in rupture-prone shoulders of atherosclerotic plaques and observed direct contacts of dendritic cells with NKT cells in rupture-prone regions of plaque.
A
Inflammatory infiltrates in atherosclerotic plaques contain activated T cells (Hansson et al. 2002). Antigen-specific T-cell activation is dependent on the interactions of T-cell receptors (TCR) with antigens presented by major histocompatibility complex (MHC) molecules (Hansson et al. 2002). ICAM-1/lymphocyte function-associated antigen-1 and vascular cell adhesion molecule-1 (VCAM-1)/very late antigen-4 interactions are critical in T cell activation (Hansson et al. 2002). In atherosclerotic lesions, DCs display HLA-DR, ICAM-1, VCAM-1, and heat shock protein-70 and locate among and contact T cells (Bobryshev and Lord 1998,2002), suggesting that DCs might activate T cells directly in the injured arterial wall. CD1d, an antigen-presenting molecule responsible for the presentation of lipid antigens (Porcelli 1995), is expressed in atherosclerotic lesions, and its expression is restricted to DCs (Bobryshev and Lord, 2002). We hypothesized that CD1-restricted responses may play a role in atherosclerosis (Bobryshev and Lord 1995,1998; Bobryshev 2000; Bobryshev and Lord 2002) and suggested that some DCs interact with T cells directly within atherosclerotic lesions, whereas others may migrate to regional lymph nodes to activate T cells (Bobryshev and Lord 1998; Bobryshev 2000).
Recent studies revealed that natural killer T (NKT) cells are involved in atherosclerosis (Nakai et al. 2004; Tupin et al. 2004). Earlier studies implicated NKT cells in the regulation of autoimmunity, particularly in diabetes and experimental allergic encephalomyelitis in animal models (van Kaer, 2004). NKT cells comprise a heterogeneous subpopulation of T cells that coexpress a TCR and natural killer (NK) surface antigen CD161 in humans and NK1.1 in mice (van Kaer 2004). NKT cells are CD1d-restricted and express an evolutionarily conserved TCR with invariant α-chain (Vα14-Jα18 in mouse and Vα24-Jα18 in human). NKT cells react to exogenous α-galactosylceramide (αGalCer), which is presented by the monomorphic HLA class-I–like molecule CD1d (van Kaer, 2004). The treatment of apoliprotein E–deficient (apoE−/-) mice with αGalCer resulted in accelerated atherosclerosis (Nakai et al. 2004; Tupin et al. 2004) accompanied by the recruitment of NKT cells to aortic atherosclerotic-like lesions in apoE−/- mice. Similar treatment of CD1d−/- apoE−/- mice had no impact on disease progression, suggesting that the effect of αGalCer on atherosclerosis progression is CD1d-dependent (Nakai et al. 2004; Tupin et al. 2004).
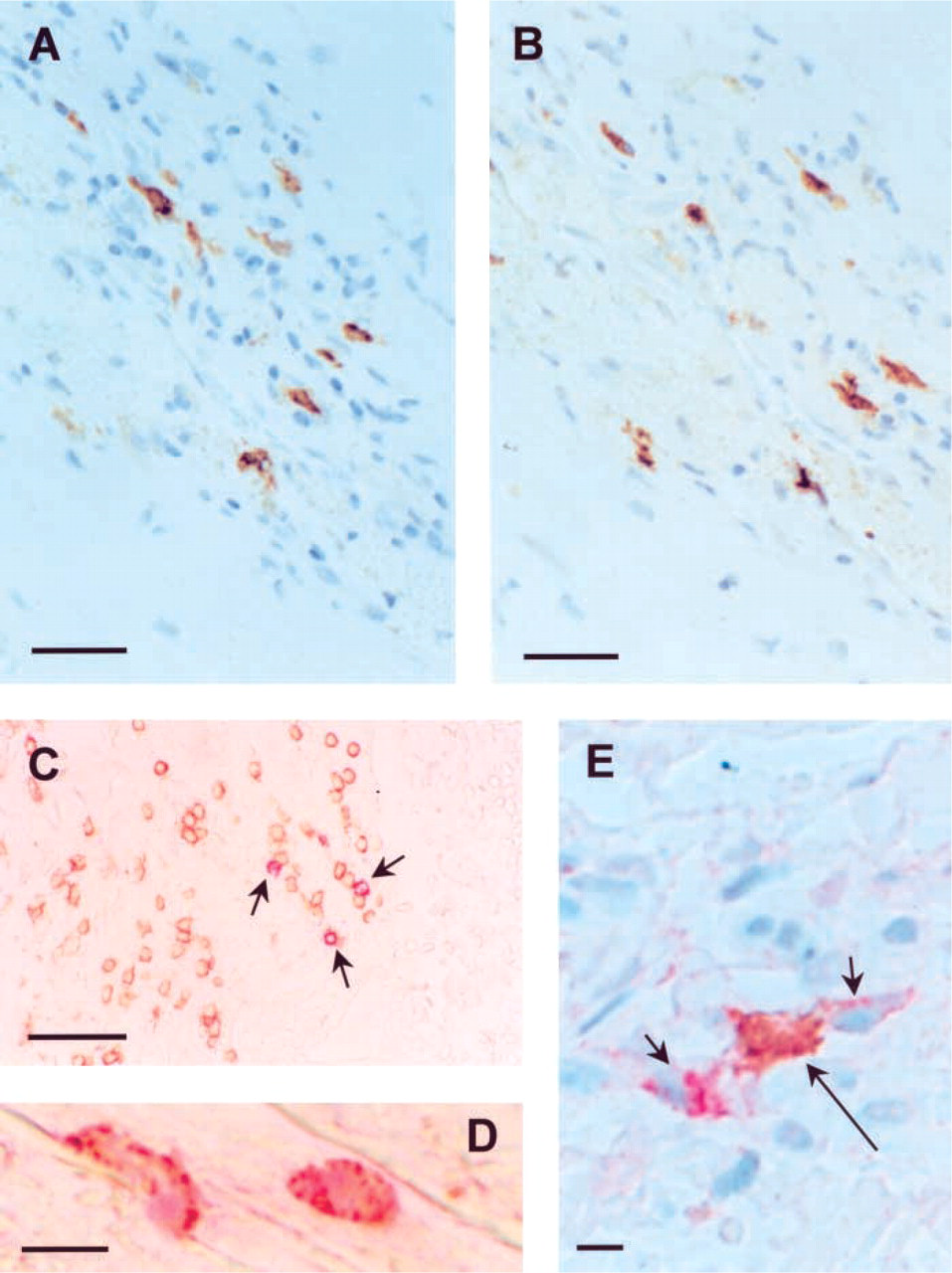
Dendritic cells (DCs) and natural killer T (NKT) cells in atherosclerotic plaques.
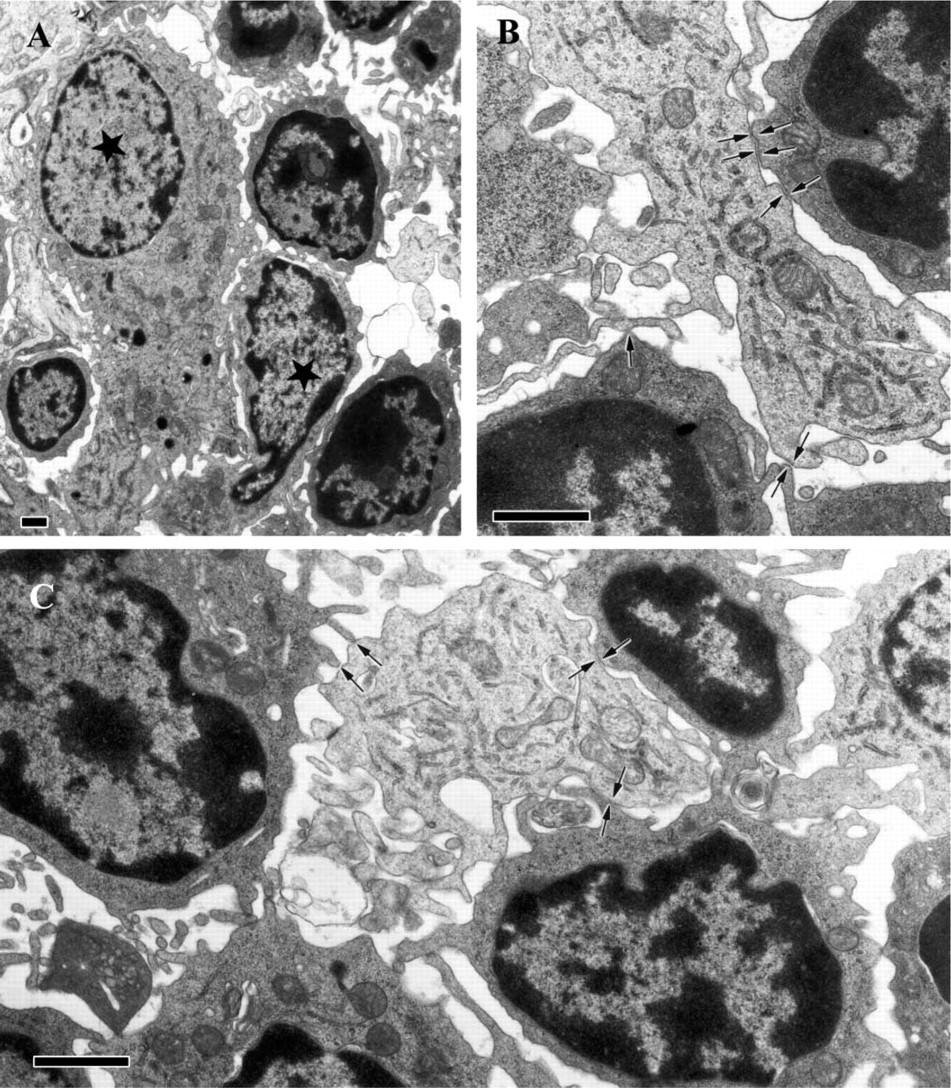
Electron microscopic visualization of contacts between DCs and lymphocytes in a plaque shoulder region. (A) DCs are marked by stars.
No previous studies have investigated whether NKT cells are present in human atherosclerotic lesions and, if so, whether there is an association between NKT cells and DCs. In the present study, using immunohistochemical techniques, we examined human carotid artery specimens collected as previously reported (Bobryshev and Lord 1998). For immunohistochemical examination, unfixed specimens were immediately embedded in OCT compound, rapidly frozen in liquid nitrogen, and stored at −70C until cryostat sectioning. For the present study, frozen sections were obtained from eleven carotid artery specimens containing advanced atherosclerotic lesions and adjacent normal-appearing arterial wall. The material was collected in accordance with the Helsinki Declaration of 1975, and some characteristics of these specimens have been reported previously (Bobryshev and Lord 2002). All atherosclerotic plaques selected for the present analysis were unstable plaques according to the criteria described by Yilmaz et al. (2004). The sections were single and double immunostained. The total population of T cells was identified with anti-CD3 (Dako, Glostrup, Denmark; dilution 1:50). NKT cells were identified using anti-NK surface antigen CD161 (Santa Cruz Biotechnology, Santa Cruz, CA; 1:100). DCs were identified with anti-CD1a (Dako; 1:50) and fascin (Dako; 1:100). CD1d expression was visualized using anti-CD1d (Santa Cruz Biotechnology). Macrophages, smooth muscle cells, and endothelial cells were identified as previously described (Bobryshev and Lord 1998,2002). For single immunostaining, the avidin-biotin immunoperoxidase method was used as previously detailed (Bobryshev and Lord 1995). Double immunostaining included combinations of peroxidase-anti-peroxidase and alkaline phosphatase-antialkaline phosphatase techniques, which were carried out as described previously (Bobryshev and Lord 1998,2002). Negative controls were also carried out as described previously (Bobryshev and Lord 1998).
The distribution patterns of DCs in atherosclerotic lesions were similar to those described previously (Bobryshev and Lord 1998). Consistent with our previous observations, DCs were located predominantly in plaque shoulders containing inflammatory infiltrates consisting of T cells and macrophages. Comparison of consecutive sections stained with anti-CD1d and with different cell-type–specific antibodies confirmed that CD1d expression is restricted to DCs (Figures 1A and 1B). Immunohistochemical examination demonstrated that there were no NKT cells in the non-diseased arterial sites, but atherosclerotic plaques contained NKT cells (Figures 1C and 1D). Although NKT cells represented a minor cell population among T cells (0.3–2%), they were found in all specimens studied. NKT cells were observed most often within inflammatory infiltrates in the deep portions of plaques underlying the necrotic cores and in plaque shoulders. In plaque shoulders, the colocalization of CD1d+ DCs with NKT cells was identified (Figure 1E). Electron microscopic examination of areas corresponding to zones where DCs were colocalized with NKT cells demonstrated direct contacts between DCs and lymphocytes, some of which contained well-developed cytoplasm typical of NKT cells (Figures 2A–2C). In clusters of DCs with lymphocytes, cisterns of the DC tubulovesicular system were hypertrophied (Figures 2A–2C), suggesting that DCs were activated.
Atherosclerotic plaque stability is determined by a variety of factors, of which immune and inflammatory pathways are particularly important (Hansson et al. 2002). Both innate and adaptive immuno-inflammatory mechanisms contribute to plaque instability (Hansson et al. 2002). Unstable plaques contain an infiltrate of T cells, DCs, and macrophages resembling a delayed hypersensitivity reaction. Activated T cells secrete cytokines that regulate the function of arterial cells. T cells may also differentiate into effector cells with tissue-damaging potential (Hansson et al. 2002). The present observations suggest that the interaction between DCs and NKT cells occurring directly within the atherosclerotic arterial wall may indicate plaque instability.
Emerging evidence suggests that DCs are involved in the regulation of the helper T cell Th1/Th2 balance and may interact with effector cells of innate immunity (Creusot and Mitchison 2004). It has been reported that DCs can activate NKT cells by presenting αGalCer in association with CD1d (Creusot and Mitchison, 2004).
DCs may represent a useful tool for the regulation of NKT cell function in atherosclerosis. DCs pulsed with αGalCer are used today for cancer immunotherapy (Nieda et al. 2004). In one such technique, DCs pulsed with αGalCer ex vivo are returned to the bloodstream (Nieda et al. 2004). In atherosclerosis, in contrast to cancer immunotherapy, NKT cells need to be suppressed (Nakai et al. 2004; Tupin et al. 2004). It is known that the presentation of MHC complexes to T cells by DCs in the absence of costimulatory signals does not lead to T cell activation but, quite the opposite, leads to the deletion or inactivation of these T cells (Austyn 1998; Lotze and Thomson 2001). Perhaps, for atherosclerosis immunotherapy, costimulatory molecules on DCs pulsed with αGalCer need to be downregulated/ligated before they are returned to the bloodstream.
Footnotes
Acknowledgements
This research was supported by the St Vincent's Clinic Foundation, Sydney.
