Abstract
Muscle side population (SP) cells have demonstrated hematopoietic and myogenic activities in vivo upon intravenous (IV) injection into lethally irradiated mdx mice. In contrast, muscle main population (MP) cells were unable to rescue the bone marrow of lethally irradiated mice and, consequently, their in vivo myogenic potential could not be assessed using this method. In the current study, muscle SP or MP cells derived from syngeneic wild-type male mice were delivered to sub-lethally irradiated mdx female mice by single or serial IV injections. Recipient mice were euthanized 12 weeks after transplantation at which time the quadriceps and diaphragm muscles were analyzed for the presence of donor-derived cells. Mice injected with 104 muscle SP cells or with 106 MP cells appeared to have similar numbers of dystrophin-positive myofibers containing fused donor nuclei. Analysis of the remaining tissue via real-time quantitative PCR indicated that mice injected with muscle SP cells had a higher percentage of donor-derived Y-DNA in the quadriceps than mice injected with MP cells, suggesting that muscle SP cells may be enriched for progenitors able to engraft dystrophic skeletal muscles from the circulation. Although the overall engraftment did not reach therapeutically significant levels, these results indicate that further optimization of cell delivery techniques may lead to improved efficacy of cell-mediated therapy using muscle SP cells.
D
In both DMD patients and the mdx mouse, dystrophin expression is restored in a small percentage (0.1–1%) of myofibers, termed revertants (Hoffman et al. 1990; Danko et al. 1992; Fanin et al. 1995). The mechanism by which mdx revertant myofibers express dystrophin involves exon skipping surrounding the mutated exon 23, resulting in a truncated but in-frame protein product (Lu et al. 2000). The expression of this shortened dystrophin in revertant myofibers may complicate the analysis of mdx muscle while assessing the efficacy of cell (Skuk and Tremblay 2000; Smythe et al. 2001; Partridge 2002), gene (Hartigan-O'Connor and Chamberlain 2000; Takeda and Miyagoe-Suzuki 2001), or pharmacological-based (Khurana and Davies 2003) therapies in vivo. Truncated dystrophin expressed by revertant myofibers in mdx mice is detectable with antibodies typically used for immunofluorescence and is often indistinguishable from full-length dystrophin by this technique.
Currently no effective treatment is available for DMD, although numerous efforts are being investigated, including stem-cell-based therapy (Ferrari and Mavilio 2002; Jankowski et al. 2002; Sohn and Gussoni 2004). Stem cells are defined as primitive cells that retain the ability to self-renew, but given the proper signals they also generate committed cells. Several types of cells have been tested for optimization of cell-based therapy of muscle diseases. Initial studies have demonstrated that unfractionated bone marrow cells can fuse to myofibers of mdx mice when injected intravenously (IV) (Ferrari et al. 1998; Bittner et al. 1999; Ferrari et al. 2001).
These studies lead to testing the possibility that bone-marrow-derived stem cells may exhibit myogenic potential in vivo. Bone marrow-derived side population (SP) cells are isolated using the fluorescence-activated cell sorter (FACS) based on their ability to efficiently exclude the vital DNA dye Hoechst 33342 (Goodell et al. 1996), which is mediated via the ABC transporter bcrp1 (ABCG2) (Zhou et al. 2001; Zhou et al. 2002). IV injection of bone marrow SP cells into lethally irradiated mice demonstrated that these cells have both myogenic and hematopoietic potential in vivo (Gussoni et al. 1999; Camargo et al. 2003; Corbel et al. 2003). However, the percentage of donor-derived cells that had fused into myofibers from the circulation was low (1–2%) and therapeutically not significant in most skeletal muscles, with the exception of the panniculus carnosus (Brazelton et al. 2003).
Similar to findings using bone-marrow-derived SP cells, individual clones of skeletal muscle-derived stem cells (MDSC) isolated via the preplating technique, expanded in vitro, and injected into lethally irradiated mice demonstrated the ability to reconstitute the bone marrow and engraft into 1–2% of skeletal muscle myofibers (Cao et al. 2003). Muscle SP and main population (MP) cells have also been identified by adapting the technique used for the isolation of bone marrow SP cells (Gussoni et al. 1999; Jackson et al. 1999; Montanaro et al. 2004). Characterization by FACS analysis of cell surface antigens present on muscle SP cells indicated that these cells may be similar to MDSC isolated via the preplating technique, as both cell populations appear to express Sca-1 and CD34, whereas they are negative for the hematopoietic marker CD45 (Gussoni et al. 1999; Cao et al. 2003; Rivier et al. 2004). IV injection of unfractionated, uncultured muscle SP cells from wild-type male mice into lethally irradiated mdx female mice demonstrated that this cell population contains myogenic and hematopoietic precursors (Gussoni et al. 1999). In contrast, parallel injections of muscle MP cells demonstrated the inability of these cells to efficiently engraft into recipient bone marrow, leading to premature death of the hosts (Gussoni et al. 1999). Muscle SP cells are able to adapt a myogenic fate in vitro upon coculture with primary myoblasts, suggesting that specific cell–cell interactions may influence their specification (Asakura et al. 2002). In support of these findings, intramuscular (IM) injections in mice demonstrated that muscle SP cells are able to give rise to satellite cells in vivo (Asakura et al. 2002), as well as to contribute to vascular regeneration after local injections into chemically damaged regenerating muscle (Majka et al. 2003). Muscle MP cells were not able to provide short-term radioprotection of lethally irradiated mdx mice; therefore, it remains unclear whether these cells could fuse to dystrophic myofibers from the circulation and be utilized for optimization of cell-based therapy of muscular dystrophy.
In the current study, the myogenic potential of muscle-derived SP and MP cells was evaluated after single or serial IV injections into mdx mice that received a sub-lethal dose of X-irradiation. Results demonstrate that both SP and MP cells were able to engraft skeletal muscle from the circulation in sub-lethally irradiated or non-irradiated mdx mice. No dose response was observed in animals receiving booster injections compared with animals that received a single cell injection. Real-time quantitative PCR analysis of recipient female quadriceps indicated that mice injected with SP cells had a higher percentage of male-derived DNA than mice injected with MP cells, despite an injection of significantly lower numbers of cells. Although the overall level of engrafted donor cells within the muscles appears to be therapeutically insignificant, muscle SP cells are more likely than MP cells to be enriched for precursors able to target skeletal muscle from the circulation. Further optimization studies using systemic cell delivery may improve the efficacy of muscle SP cell-mediated therapy.
Materials and Methods
Isolation of Muscle MP and SP Cells
Muscle MP and SP cells were isolated as previously described (Gussoni et al. 1999; Montanaro et al. 2004). Briefly, 3–8-week-old C57BL10/SnJ wild-type male mice were euthanized, and 2–4 g of muscle tissue was collected per mouse. Cells were dissociated enzymatically using dispase II and collagenase IV as described (Rando and Blau 1994; Gussoni et al. 1999; Montanaro et al. 2004). Cells were filtered, and red cells were lysed using Puregene (Gentra Systems; Minneapolis, MN) red cell lysis buffer. Remaining cells were counted and resuspended at 1 × 106 cells/ml in PBS with 0.5% (w/v) BSA. Hoechst dye was added at a final concentration of 12.5 μg/ml, and samples were incubated for 90 min at 37C. A parallel control aliquot was also incubated with 50 μM verapamil. Cells were washed and counterstained with 2 μg/ml propidium iodide for dead-cell exclusion and then were analyzed and sorted into MP and SP populations on a FACS sorter (Becton Dickinson; Franklin Lakes, NJ). Cells were collected in sterile PBS and kept on ice until injection.
IV (Tail Vein) Injection of Muscle Cells in mdx Recipient Mice?
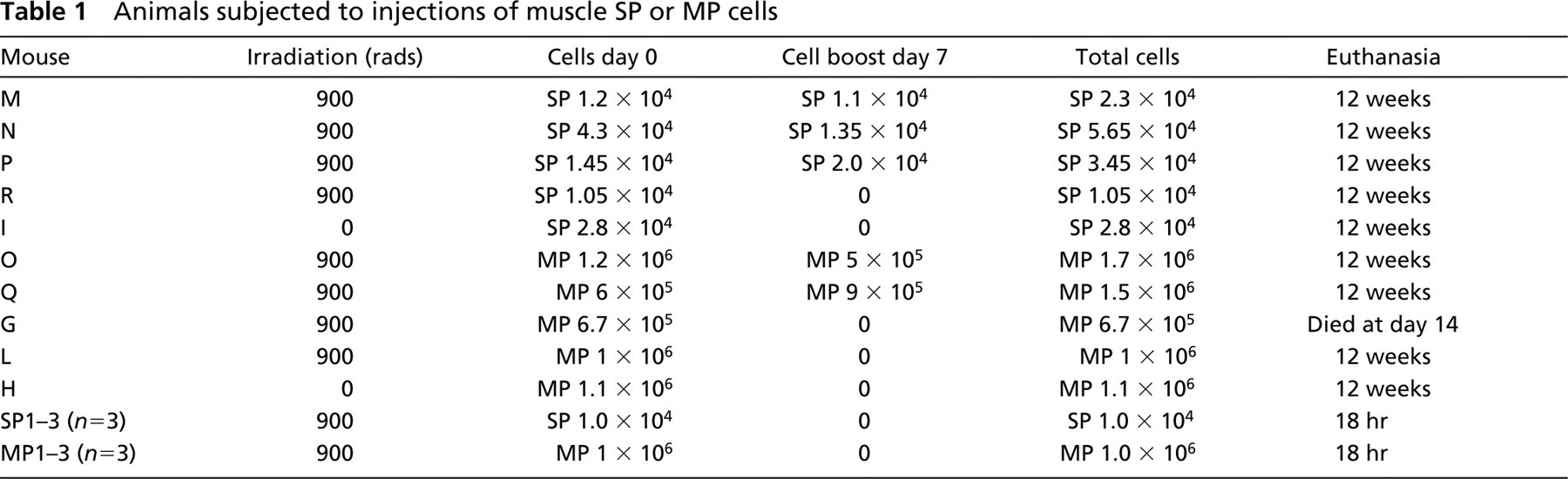
Eight recipient female C57BL10/ScSn mdx mice were sub-lethally irradiated (900 rads) in a split dose and four of these mice were injected with muscle SP while the remaining four mice were injected with muscle MP cells derived from syngeneic male mice (Table 1). Animals H and I did not receive irradiation, serving as cell-injected littermate controls. Recipient mice were injected IV with purified muscle MP or SP cells immediately after the second irradiation dose (when applicable). A booster injection of muscle SP or MP cells was given to five mice 1 week after the initial injection (Table 1). Because muscle SP cells are much less abundant than MP cells, the number of injected SP cells was consistently on the order of 104, whereas MP cells were delivered on the order of 106 cells/mouse (Table 1). All surviving mice were sacrificed 12 weeks after the initial injection, with the exception of mouse G that received 670,000 MP cells and died 14 days after sub-lethal irradiation. Right and left quadriceps and diaphragm skeletal muscles were collected and snap frozen in isopentane.
To determine if injection of cells in the tail vein causes a proportion of muscle MP or SP cells to be sequestered in the lungs, liver, and spleen of recipient mice, six C57BL10/ScSn mdx female mice were sub-lethally irradiated and injected with either 104 C57BL10 male-derived muscle SP cells (SP1–3) or 106 C57BL10 male-derived muscle MP cells (MP1–3, Table 1). Mice were euthanized 18 hr after IV cell injection. The lungs, liver, and spleen were harvested for quantification of male donor-derived DNA by real-time quantitative PCR and fluorescence in-situ hybridization (FISH) analyses.
Animals subjected to injections of muscle SP or MP cells
Immunohistochemistry
Skeletal muscle tissue sections were cut at 10 μm on a cryostat and collected on silanized slides (Polysciences Inc; Warrington, PA). For dystrophin immunostaining, sections were immediately fixed for 3 min in 100% methanol and stored in cold PBS until stained. Sections were blocked for 30 min at room temperature in 10% FBS diluted in PBS. CAP6–10 (anti-dystrophin) polyclonal antibody was used at a dilution of 1:500 in PBS (Byers et al. 1993; Gussoni et al. 1999). Sections were treated with 200 μL of antibody solution and incubated overnight at 4C in a humid chamber. Slides were then washed 3 × 10 min in PBS on a shaker, treated with secondary antibody for 1 hr at room temperature (anti-rabbit FITC diluted 1:100 in PBS; Jackson ImmunoResearch Laboratories, West Grove, PA), and washed again 3 × 10 min in PBS prior to being cover slipped with Vectashield (Vector Labs; Burlingame, CA). A Zeiss Axioplan (Zeiss; Thornwood, NY) microscope was used to examine the sections for presence of dystrophin-positive fibers, and pictures were acquired with a charged-coupled device (CCD) camera (Photometrics; Tucson, AZ/Hamamatsu, Japan). Sections were kept at 4C until used for FISH.
FISH Probe Preparation
The mouse Y-chromosome-specific probe was a kind gift from Dr. Evan Snyder and has been previously described (Harvey et al. 1992). One μg of Y-plasmid DNA was digested via nick translation and labeled with digoxigenin (Roche Applied Science; Indianapolis, IN) via nick translation, by incubating at 16C in the presence of DNase 1 (4 μL/ml stock; Roche) and 20 U DNA polymerase I (New England Biolabs; Beverly, MA). Probe length was confirmed using a 2% agarose gel to be between 200 and 500 bp prior to being precipitated overnight with 5 μg tRNA. Probe was pelleted, washed once in 70% ethanol, air dried, and resuspended in 100 μl of hybridization cocktail (50% formamide, 1 × SSC, 10% dextran sulfate, and 1 × Denhardt's solution), and probe was stored at −20C until used.
FISH
After immunohistochemistry, slides were processed for FISH with all steps carried out in dark- or low-light conditions to prevent fading of the green fluorescent signal (Gussoni et al. 1996). Sections were washed in PBS for 20 min until the coverslips slid off, refixed for 45 min in Histochoice (AMRESCO; Solon, OH), rinsed for 5 min in PBS, and dehydrated through a series of graded alcohols for 5 min each (50%, 70%, 90%, and 100%). Sections were then denatured in 70% formamide (American Bioanalytical; Natick, MA) 2 × SSC at 70C for 12 min and then dehydrated in a series of prechilled (−20C) graded alcohols (70%, 90%, 100%) 5 min each. The Y-chromosome probe was also denatured at 70C for 25 min and then immediately placed on wet ice. Sections were air dried prior to applying 10 μL of denatured Y-chromosome probe (corresponding to 100 ng of labeled Y-plasmid DNA). Slides were covered with an 18 × 18-mm coverslip, sealed with rubber cement, and incubated overnight at 37C in a humidified chamber. Sections were washed in 50% formamide (Sigma; St Louis, MO) 2 × SSC 3 × 5 min at 45C, followed by 0.1 × SSC 3 × 5 min at 60C. Detection of hybridization signals was performed by blocking the slides in blocking solution (150 mM NaCl, 100 mM Tris pH 7.5, 0.5% non-fat dry milk) for 30 min at 37C in a humidified chamber, followed by incubation of 30 min in sheep anti-digoxigenin rhodamine-conjugated antibody (Roche) diluted 1:100 in blocking solution. Slides were then washed three times in 150 mM NaCl, 100 mM Tris, 0.2% Tween-20 at 45C for 5 min, coverslipped in Vectashield (Vector Labs) containing 200 ng/ml DAPI, and analyzed with a Zeiss Axioplan microscope equipped with a CCD camera (Photometrics). Dystrophin-positive myofibers containing a donor male nucleus were counted in at least 16 tissue sections per mouse, 8 sections for each quadricep. For four mice, 25 10-μm serial tissue sections were collected (250-μm total) and analyzed for the presence of dystrophin-positive myofibers. Positive fibers were traced through sections to determine their length, and sections were then hybridized by FISH to detect Y+ nuclei. Dystrophin+ Y + myofibers were counted in each tissue section, and the percentage of these myofibers from the total number of myofibers was calculated in each mouse injected with SP or MP cells.
FISH Analysis of Bone Marrow Nuclei, Spleen Tissue Sections, and Touch Preps from Lungs, Liver, and Spleen
Bone marrow cells, including adherent and non-adherent fractions, were isolated by crushing the femurs and tibias with a mortar and pestle in 5 ml of PBS. Cells were washed, filtered through a 70-μm filter, and red cells were lysed as above. Cells were then incubated with KCl (.075 M) for 10 min and fixed in methanol/acetic acid (3:1) for 30 min at room temperature. Cells were stored at −20C in fixative. Prior to FISH analysis, fixed bone marrow cells were dropped onto uncoated slides using a Pasteur pipette and fixed in a graded (70%, 90%, and 100%) ethanol series. For FISH hybridization, slides were denatured for 2 min in 70% formamide (American Bioanalytical) 2X SSC (prewarmed to 70C in a waterbath) and dehydrated again in prechilled (−20C) ethanol series (70%, 90%, 100%) for 5 min each. Probe denaturation, hybridization, washes, and detection conditions were performed as described for skeletal muscle tissue sections.
Five-μm-thick spleen tissue sections were fixed at room temperature in Histochoice (AMRESCO) for 45 min, washed for 5 min in PBS at room temperature, and dehydrated in ethanol series (50%, 70%, 90%, 100%) for 5 min each before being air dried. Slides were denatured in 70% formamide 2X SSC for 5 min at 70C and hybridized using 100 ng of labeled Y-chromosome probe. Hybridization and washes were performed as described for skeletal muscle tissue sections.
For touch-preps analyses, the liver, lung, and spleen of female mdx mice injected with male muscle SP or MP cells were harvested and the organs were cut in thirds. Pieces were used to make touch preps by gently touching the organ to an uncoated glass slide (VWR International; Bridgeport, NJ). Slides were fixed in Histochoice (AMRESCO) and processed exactly as described for skeletal muscle. The remaining tissue was frozen for DNA extraction and real-time quantitative PCR analysis.
Real-time Quantitative PCR of Y-chromosome DNA
After immunohistochemistry/FISH analysis, genomic DNA was extracted from the remaining tissue of one of the quadriceps and from the diaphragm using the Puregene genomic DNA purification kit, as per manufacturer's instructions (Gentra Systems). Genomic DNA extraction was also performed on lungs, spleen, and liver tissues harvested from the injected female mdx mice SP 1–3 and MP 1–3 (Table 1), euthanized 18 hr after transplantation. Genomic DNA was quantified by spectrophotometer reading and its quality assessed by a ratio 260/280 ≥ 1.8.
For real-time quantitative PCR, control male and female DNA was extracted from the quadriceps of non-injected wild-type C57/BL10 mice as described above (Puregene kit). A series of standards was generated by limited dilutions using control male and female genomic DNA. The prepared standards contained 25%, 10%, 5%, 1%, 0.5%, 0.1%, and 0% male DNA mixed with 75%, 90%, 95%, 99%, 99.5%, 99.9%, and 100% female DNA, respectively. A total of 40 ng of DNA template was used in each PCR reaction for both test samples and standard controls.
Real-time quantitative DNA amplification of Y-target sequences was performed using a modification of a previously described method (Byrne et al. 2002). The mouse Y-chromosome specific primers sense (5′-TGGAGAGCCACAAGCTAACCA-3′) and antisense (5′-TCCCAGCATGAGAAAGATTCTTC-3′) amplified an 81-nucleotide product specific for ZFY2 gene as described (Mardon and Page 1989; Byrne et al. 2002). For each sample, the housekeeping gene glyceraldehyde-3-phosphate-dehydrogenase (GAPDH) was also amplified as a loading control, using sense (5′-GGAGATTGTTGCCATCAACG-3′) and antisense (5′-GTCTCGCTCCTGGAAGATGG-3′) primers, which generate a 140-nucleotide PCR product. ZFY2 primers were used at a final concentration of 300 nM for the sense and 900 nM for the antisense primer as previously described (Byrne et al. 2002), whereas both GAPDH primers were used at a final concentration of 300 nM. Genomic DNA from test samples and standard control curve were amplified in the same reaction using the SYBR Green PCR reagent kit (Applied Biosystems; Foster City, CA) according to the manufacturer's protocol. Real-time amplification was monitored using the ABI 7700 Sequence Detector (Applied Biosystems). All DNA samples were amplified under the following conditions: 95C for 1 min 30 sec, 55C for 2 min for 2 cycles, 95C for 10 min (once), 95C for 15 sec, 58C for 20 sec, and 60C for 35 sec for 45 cycles, as previously described (Byrne et al. 2002). Cycle threshold readings (Ct) from known male/female standard mixtures were used to establish standard curves for each amplification reaction. For each sample, the Ct number was determined using the Sequence Detection Software (Applied Biosystems). Delta Ct (ΔCt) values (ΔCt = Ct for ZFY–Ct for GAPDH amplified from the same sample) were calculated for each standard DNA dilution. A standard curve was generated by plotting the seven ΔCt values (y-axis) corresponding to the known percentage of male DNA in the sample (μ-axis). The percentage of the male DNA for the test samples was then determined by plotting their ΔCt numbers against the standard curve.
Results
Muscle SP and MP Cells Are Able to Engraft into Recipient mdx Skeletal Muscle
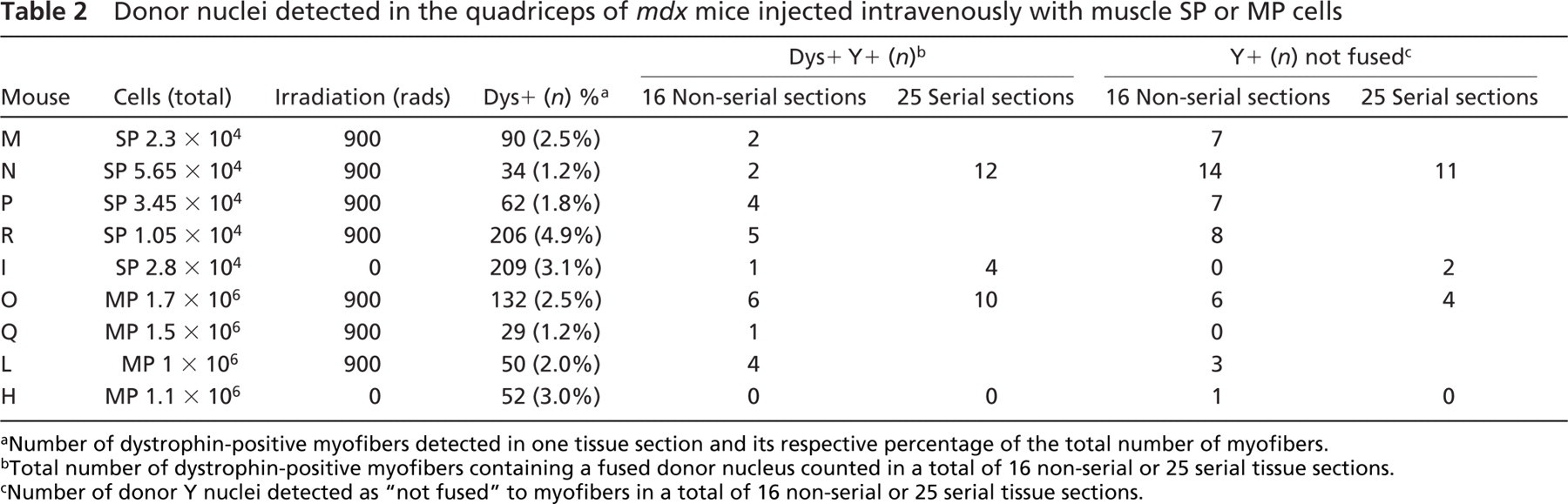
Wild-type male-derived muscle SP and MP cells were injected IV via single or multiple injections into syngeneic C57BL10ScSn mdx female mice that had received either no prior treatment or had been sub-lethally irradiated (Table 1). Animals were euthanized 12 weeks after cell injections, with the exception of mice SP1–3 and MP1–3 that were euthanized 1 day after cell injection. For each animal in the 12-week group, the quadriceps from both legs and the diaphragm were harvested. Frozen tissue was cut in 10-μm sections from both quadriceps (eight consecutive sections per muscle) and stained using an anti-dystrophin antibody (CAP6–10) (Byers et al. 1993). Dystrophin-positive myofibers were detected by immunofluorescence in all samples (Figure 1, green staining), and their percentage in each mdx mouse was calculated for each leg and then averaged (Table 2). In mice injected with SP cells, the percentage of dystrophin-positive myofibers ranged from 1.2 to 4.9%. Similar percentages (1.2–3%) of dystrophin-positive myofibers were detected in the muscles of mice injected with MP cells. FISH analysis was then performed on all 16 sections (8 from each side) using a probe that recognized the donor-specific mouse Y-chromosome. This technique allows detection of male donor nuclei fused within dystrophin-positive myofibers (Figure 1). Donor nuclei were detected within (Figures 1A, 1B, and 1D) or outside (Figure 1C) dystrophin-positive myofibers of the quadriceps, irrespective of whether the animals had received muscle SP or MP cells. From the analysis of both quadriceps, 1–5 donor Y+ nuclei had fused to dystrophin-positive myofibers and 0–14 were detected as interstitial for mice injected with muscle SP cells (Table 2). For mice injected with MP cells, 0–6 donor Y+ nuclei were detected fused to dystrophin-positive myofibers and 1-6 nuclei were found to be interstitial (not fused). Surprisingly, no cell-dose response was observed for animals that received booster injections of SP cells. Mouse R, which was injected with only 10,500 SP cells, had a higher number of dystrophin+ Y+ myofibers than mouse N, which was injected with a total of 56,500 muscle SP cells and had received a booster injection (Table 2). In contrast, for MP-injected mice, mouse O that received the highest cell dose (1.7 × 106) appeared to have the highest number of dystrophin+ Y+ myofibers.
Because previous studies on nuclear domain in myofibers demonstrated that the expression of a protein, such as dystrophin, can extend far away from the nucleus producing it (Hall and Ralston 1989; Pavlath et al. 1989; Gussoni et al. 1997), skeletal muscles from four recipients (N, I, O, and H) were serially sectioned (25 sections of 10 μm) and analyzed by combined immunohistochemistry/FISH analysis. Mice N and O were chosen because they had received sub-lethal irradiation and had been serially injected with the highest number of muscle SP and MP cells, respectively. Mice I and H were chosen because they received a single dose of muscle SP or MP cells without irradiation. The highest number of dystrophin+ Y+ myofibers (n=12) was detected in the mdx mouse N that received SP cells (Table 2), followed by mouse O (n=10) that received MP cells, and by the non-irradiated mdx mouse I that was injected with SP cells (n=4) (Table 2). For the non-irradiated mouse H that received 1.1 × 106 MP cells, only one dystrophin-positive myofiber containing a donor-derived nucleus was detected in the 25-serial-muscle-section analysis (Table 2).
Previous studies have demonstrated that the most affected muscle in mdx mice is the diaphragm (Stedman et al. 1991); therefore, each recipient's diaphragm was analyzed for the presence of dystrophin-positive myofibers associated with fused donor-derived nuclei (Figures 1E and 1F; Table 3). Although all nine animals showed presence of dystrophin-positive myofibers by immunofluorescence, in only four recipients were donor cells found fused to host myofibers producing dystrophin (Table 3). Numerous donor-derived nuclei were again detected as interstitial or not fused to myofibers (Figures 1G and 1H; Table 3).
Quantification of Y-chromosome DNA in the Quadriceps and Diaphragm Muscles
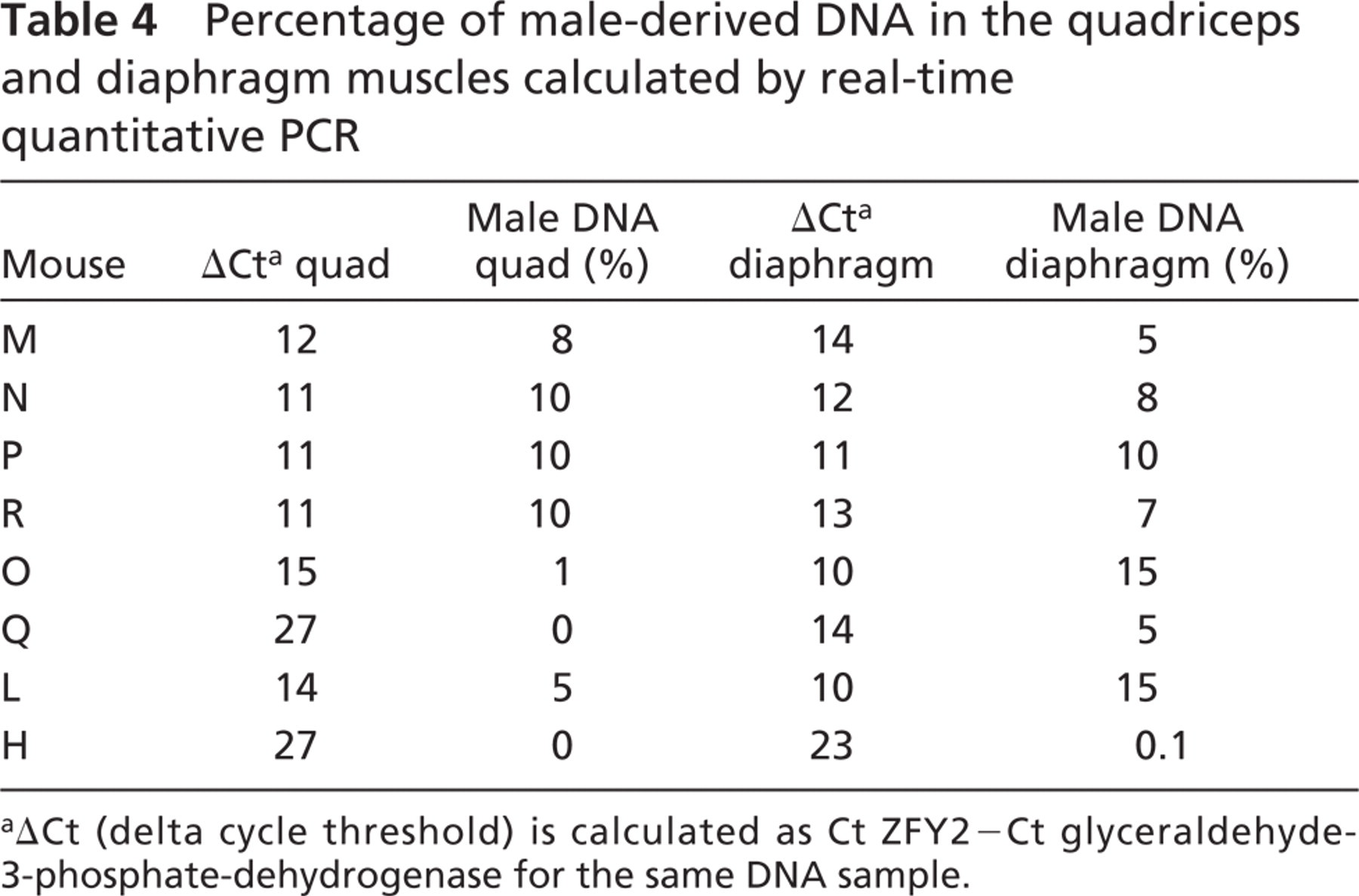
The combined FISH/immunohistochemistry analyses on multiple tissue sections taken from the quadriceps and the diaphragms of recipient mdx mice had suggested that mice injected with either SP or MP cells had a small number of Y+ donor nuclei fused to dystrophin-positive myofibers. However, these assessments were made on a limited number of tissue sections (n=8–25) taken from single regions of the muscle, leaving the possibility that the distribution of engrafted donor cells could differ significantly in other areas. To obtain a more comprehensive assessment of the amount of male-derived cells that were present in the recipient mdx muscles 12 weeks after IV donor cell injection, genomic DNA extractions were performed on the remaining quadriceps and diaphragm samples that had already been subjected to FISH/immunohistochemistry analysis. To calculate the amount of donor-derived DNA present in these muscles, which would include both donor nuclei fused to myofibers and interstitial ones, real-time quantitative PCR was performed via amplification of a gene present on the mouse Y-chromosome, named ZFY2 (Mardon and Page 1989; Byrne et al. 2002) and GAPDH internal control. Real-time quantitation was performed by monitoring the fluorescent emission of SYBR green dye bound to the PCR products, using a modification of a previously described method (Byrne et al. 2002). Real-time quantitative DNA amplification of ZFY2 target sequences was performed for an eight-point control standard curve (Figure 2, circles; Table 4, columns 1 and 2). The known amount of DNA was plotted on the x-axis and the differential Ct numbers (Ct ZFY2–Ct GAPDH) read after PCR amplification on the y-axis. Experimental DNA samples extracted from the quadriceps (Figure 2A) and diaphragm (Figure 2B) muscles were amplified simultaneously with the standard curve. The percentage of male DNA within the test samples was determined by plotting their differential Ct numbers (Ct ZFY–Ct GAPDH) against the standard curve (Figure 2; Table 4). Mice injected with SP cells contained, by this analysis, 8–10% of male-derived DNA in the quadriceps and 5–10% in the diaphragms (Table 4). Mice injected with MP cells had between 0 and 5% of male DNA in the quadriceps and between 0.1 and 15% of donor-derived male DNA in the diaphragm.
Donor nuclei detected in the quadriceps of mdx mice injected intravenously with muscle SP or MP cells
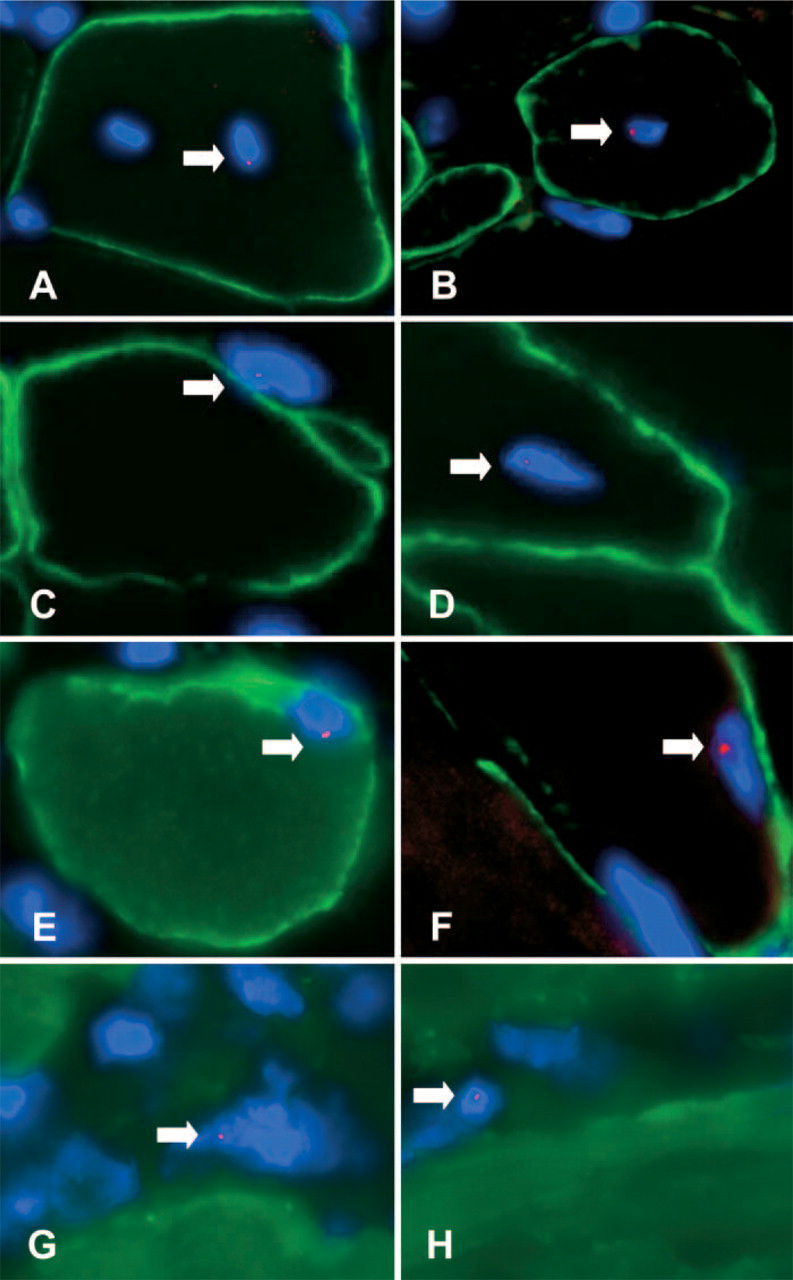
Detection of donor nuclei (arrows) in quadriceps
Detection of Donor Muscle SP and MP Cells in Hematopoietic Tissues
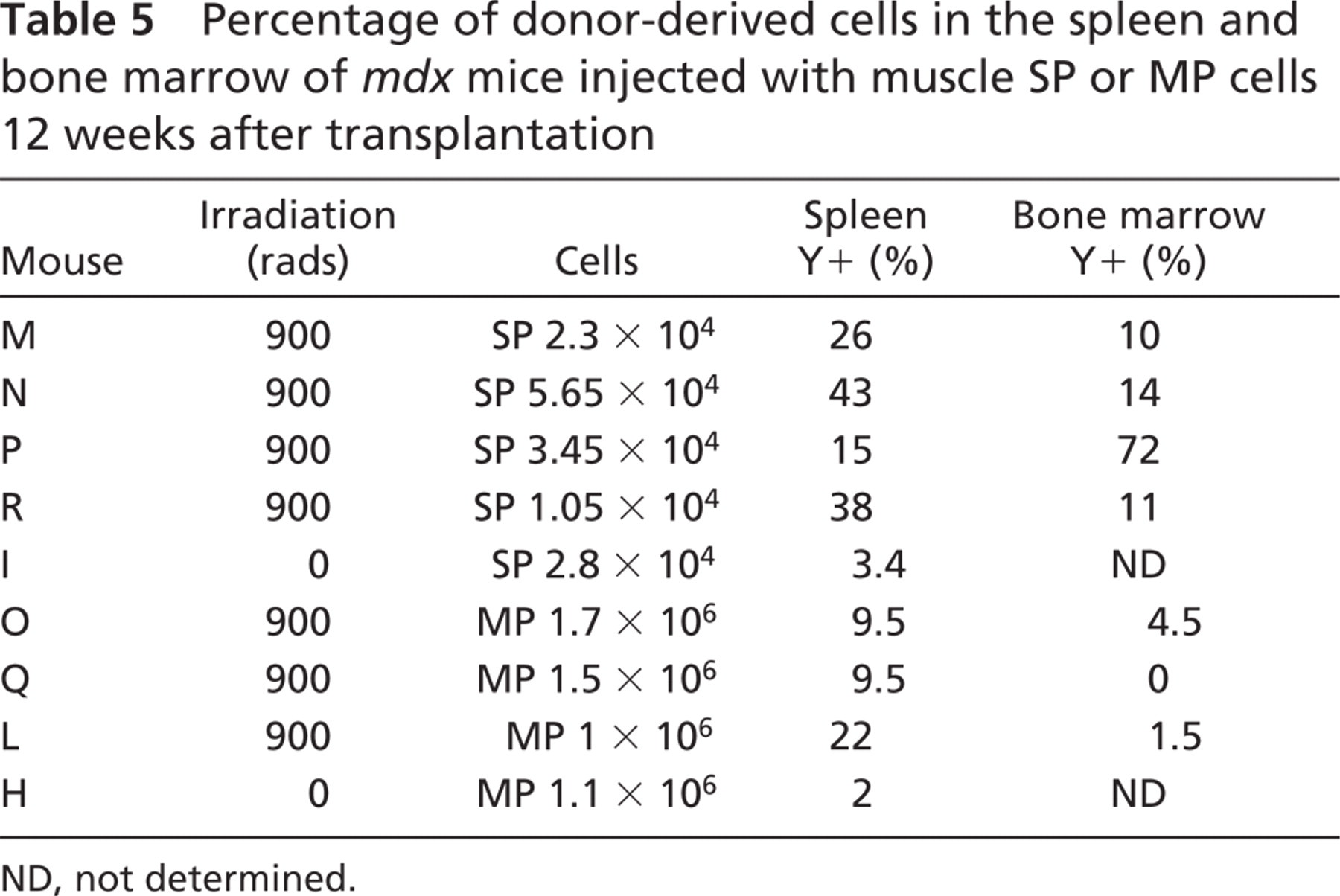
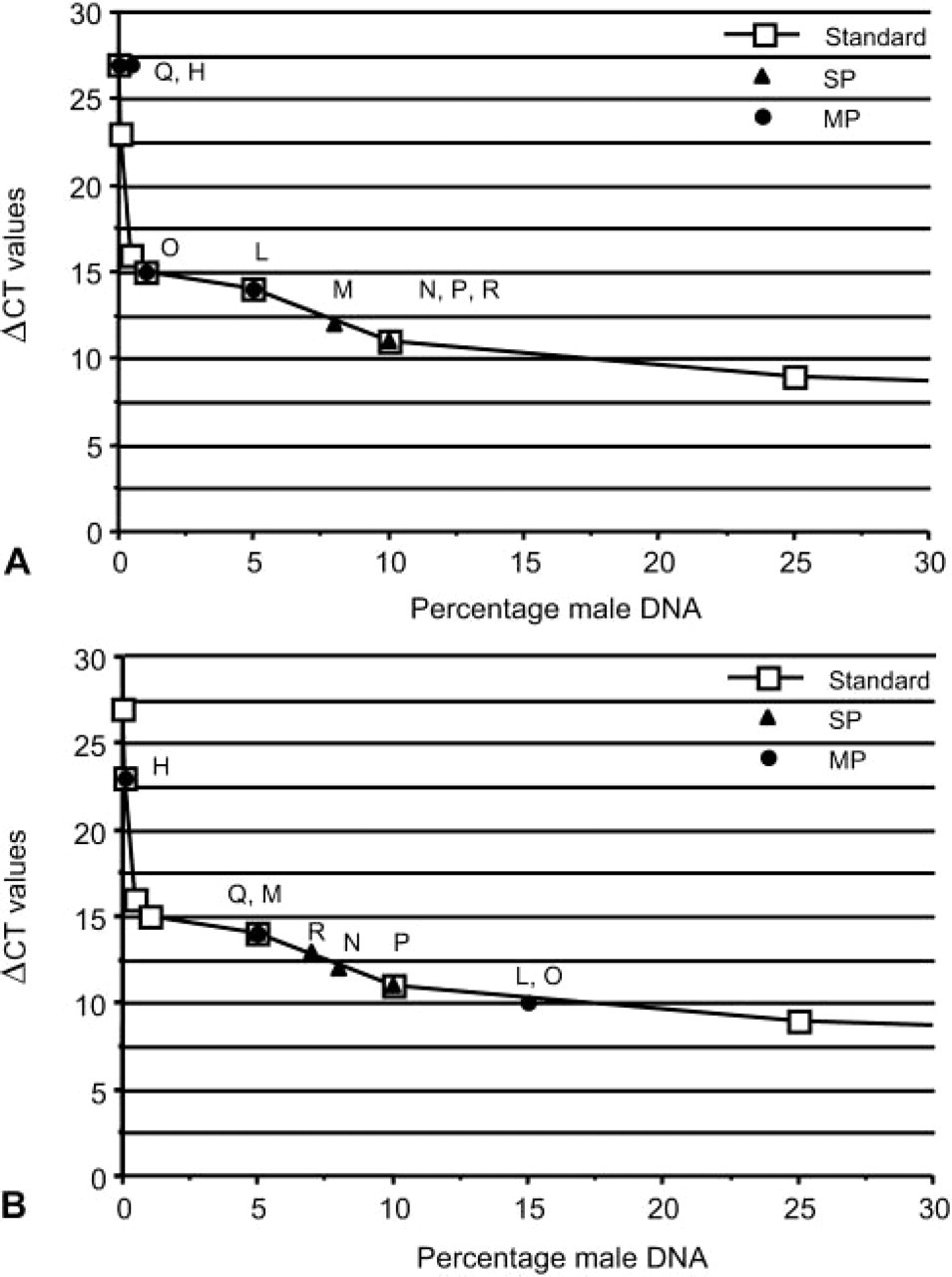
One of the main purposes of this study was to evaluate the myogenic potential of muscle MP cells after IV injection in mdx mice. Recipient mice were subjected to sub-lethal irradiation prior to IV injection of donor muscle SP or MP cells to create a partial ablation of the host bone marrow and to prevent the death of host mdx mice injected with muscle MP cells (Gussoni et al. 1999). Although this experimental design was not aimed at studying the differentiation potential and contribution of muscle SP or MP cells to hematopoietic tissues, presence of donor-derived male nuclei in the bone marrow and spleen was also assessed. To detect the presence of donor-derived cells in the bone marrow, nuclei preparations were hybridized using the Y-chromosome-specific probe. Donor nuclei were detected in the bone marrow of sub-lethally irradiated animals injected with muscle SP cells at a percentage ranging between 10 and 72% (Figures 3A–3D; Table 5), whereas 1.5–4.5% of donor-derived nuclei were detected in two of three sub-lethally irradiated animals injected with muscle MP cells (Table 5). One animal that received sub-lethal irradiation and was injected with only 6.7 × 105 muscle MP cells did not survive (Table 1, animal G). Although these results may suggest that a proportion of muscle SP cells are able to contribute to hematopoietic tissues in vivo as previously reported (Gussoni et al. 1999; Asakura et al. 2002; McKinney-Freeman et al. 2002), additional experiments, such as clonogenic assays or injection of single muscle SP cells in vivo, will be required to obtain a more conclusive assessment of their hematopoietic potential.
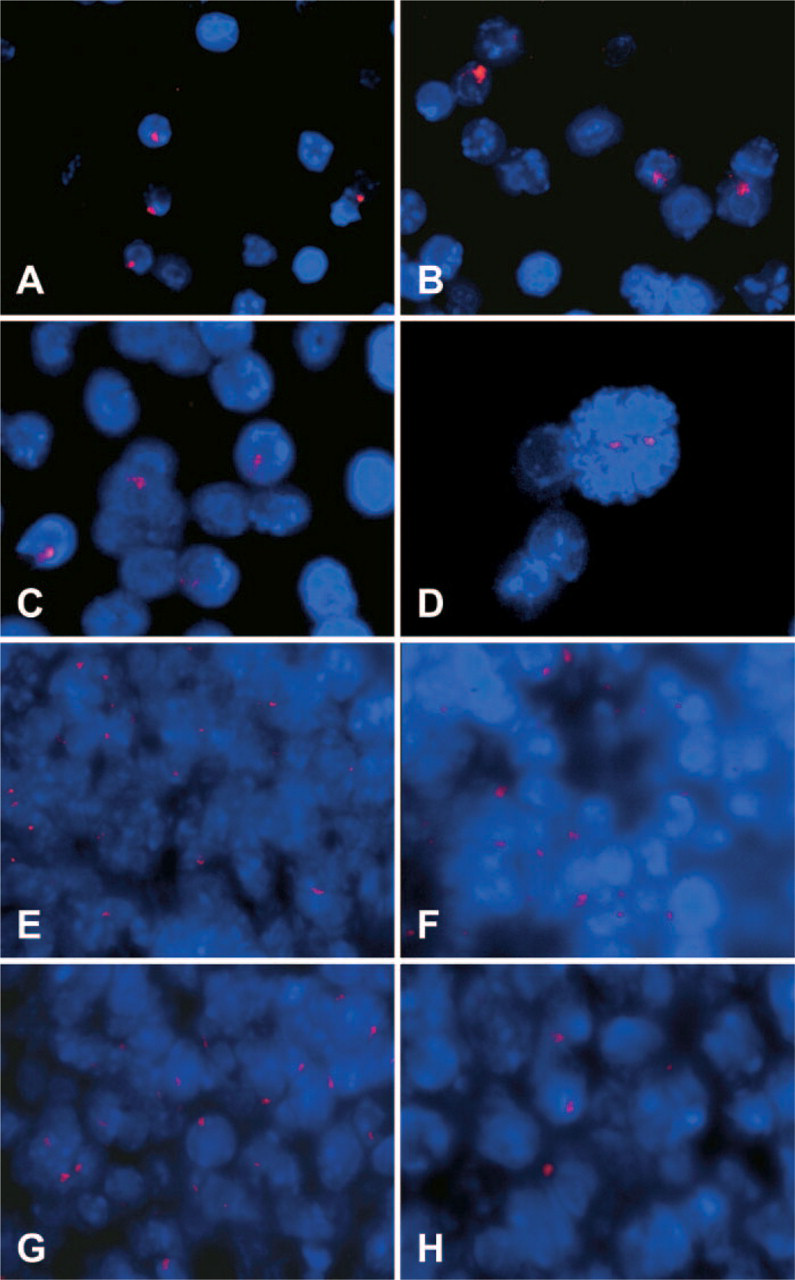
Spleen tissue sections of recipient animals were also analyzed by FISH for detection of donor-derived Y nuclei (Figures 3E–3H). Mice injected with SP cells appeared to have higher percentages of donor cells in the spleen than mice injected with MP cells (Figure 3; Table 5). Interestingly, 2–3% of donor-derived cells were also detected in the spleens of non-irradiated animals I and H, regardless of whether they had been injected with SP or MP cells (Table 5). These results suggest that donor cells delivered in the circulation via tail vein injection may be filtered through other organs, including the spleen, liver, and lungs, thus potentially decreasing the number of cells that could be recruited within the skeletal muscles. To address this possibility, six mdx recipient female mice were sub-lethally irradiated and injected with either 104 male-derived muscle SP cells (SP1–3; Table 1) or 106 male-derived muscle MP cells (MP1–3). One day after cell injection, mice were euthanized and the lungs, liver, and spleen were harvested for analysis. After preparing touch preps for each of these organs for detection of donor-derived nuclei using FISH, the entire organ was digested and genomic DNA was extracted. Genomic DNA from lungs, spleen, and liver of mice SP1–3 and MP1–3 was utilized for PCR amplification and quantitation of the ZFY2 target gene present on the donor Y chromosome. Amplification results demonstrated that 1 day after injection, no male donor-derived muscle SP or MP cell DNA was present in the lungs, liver, or spleen of the recipient mdx female mice (ΔCt >28), whereas the GAPDH control as well as standard control curve could successfully be amplified (Figure 4A). To confirm the results obtained by quantitative real-time PCR, touch-prep slides from the spleen, lung, and liver of each mouse were hybridized by FISH using the Y-chromosome probe (Figures 4B–4D). Results again indicated lack of male-derived cells in these organs, suggesting that if trapping or filtering of SP and MP cells occurs after IV injection, it is not within 1 day.
Donor nuclei detected in the diaphragms of mdx mice injected intravenously with muscle SP or MP cells
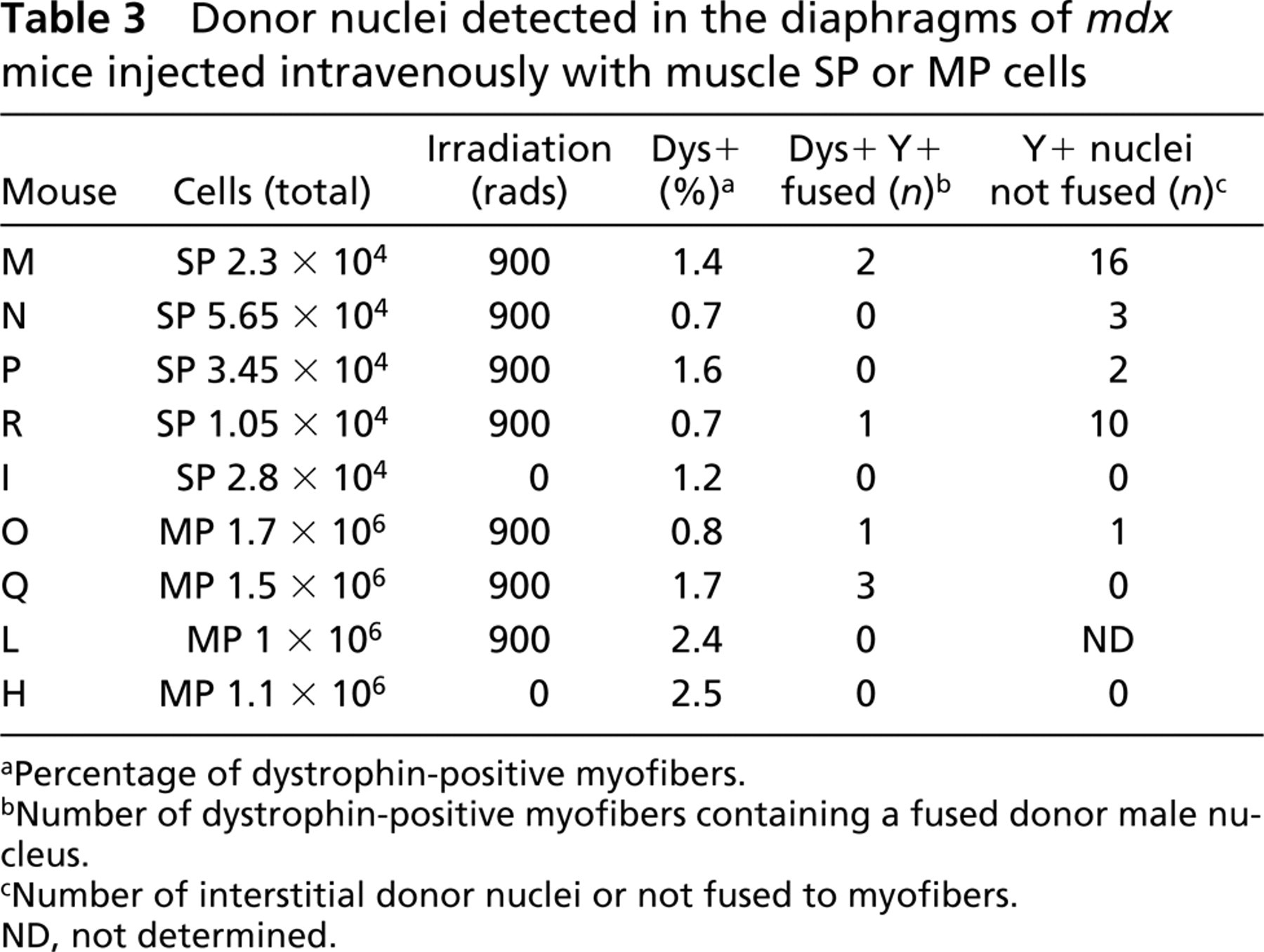
Percentage of male-derived DNA in the quadriceps and diaphragm muscles calculated by real-time quantitative PCR
Real-time quantitative PCR of Y-chromosome DNA amplified from the quadriceps
Discussion
It has previously been shown that 2 to 4 weeks after IV injection of wild-type male muscle-derived SP cells into lethally irradiated mdx females, donor-derived male nuclei can contribute not only to the host bone marrow, but fuse to skeletal muscle myofibers (Gussoni et al. 1999). In contrast, parallel injections of lethally irradiated mdx mice using muscle MP cells resulted in premature death of the hosts, preventing the assessment of the myogenic potential of MP cells by this method of delivery. The goals of the current study were to determine whether muscle MP cells have myogenic potential in vivo, to test whether serial injections of muscle SP or MP cells could increase the yield of donor-derived dystrophin-positive myofibers in mdx mice, and to assess whether preconditioning of the host with X-irradiation would favor donor cell engraftment in the muscle.
To test these parameters, mdx recipient female mice, which received either no irradiation or a sublethal dose, were injected with syngeneic male muscle SP or MP cells. Cells were delivered via either a single IV injection or two serial IV injections given 1 week apart, with the idea that the damage from X-irradiation would generate a niche favoring the engraftment of the donor cell boost. With the exception of one mouse (G), which received 105 MP cells, all sub-lethally irradiated mice survived. Skeletal muscles were analyzed 12 weeks after transplantation, and the presence of dystrophin+ Y+ myofibers was detected both in SP and MP injected mice, indicating that muscle MP cells, like SP cells, contain progenitors able to home and engraft into muscle after IV injections. Preirradiation of the host mice with a sub-lethal dose appeared to positively correlate with increased numbers of dystrophin+ Y+ myofibers in the recipient mdx muscles. These results are in agreement with previous studies reporting that the incorporation of donor cells delivered via IM injections is favored by prior administration of local X-irradiation (Gross et al. 1999).
Percentage of donor-derived cells in the spleen and bone marrow of mdx mice injected with muscle SP or MP cells 12 weeks after transplantation
The current study indicates both by immunohistochemistry/FISH and real-time quantitative PCR analyses that mice injected with muscle SP cells, which received 100-fold less cells than mice injected with MP cells, had similar or slightly higher numbers of fused donor nuclei compared with mice injected with muscle MP cells. These results support the hypothesis that muscle SP cells may be enriched for precursors able to target skeletal muscles from the circulation.
Detection of donor-derived cells in the hematopoietic tissues.
Dissemination of cells via the circulatory system represents an attractive modification over local IM injections for optimization of cell-based therapy of muscular dystrophy.
However, none of the recipient mdx mice in the current study, including the ones that received booster injections, displayed efficacy of IV cell injections improved to levels that could be considered therapeutically significant. In fact, the percentage of engrafted donor cells detected within the muscle after IV injection was at times lower than what was observed with local IM injections. Similar findings were reported after long-term transplantation of whole bone marrow cells injected IV into lethally irradiated mdx4cv mice (Ferrari et al. 2001). Poor efficacy of engraftment of donor-derived cells within the recipient muscle could arise from multiple mechanisms, including poor survival of injected cells within the circulation, inefficient donor cell trafficking to the muscle, and competitive contribution of local muscle satellite cells, which appear to be more effective than circulating donor cells in muscle regeneration and repair (Partridge 2004; Sherwood et al. 2004). When injected into the tail vein, cells must travel through several organs including spleen, heart, and lungs before reaching the hindlimb muscles. In the current study, short-term trapping of muscle SP and MP cells in the lungs, spleen, and liver of recipient mdx mice was evaluated by FISH and quantitative real-time PCR. Results demonstrated that 1 day after IV cell injection, no muscle SP or MP cells are present within these organs. However, FISH analysis demonstrated the presence of donor-derived muscle SP or MP cells within the spleen of recipient mdx mice 12 weeks after IV injection. It is unclear by the current analyses whether a proportion of muscle SP and MP cells injected IV undergoes rapid cell death within the first 72 hr with very few cells surviving, as previously reported for muscle cells delivered via IM injection (Beauchamp et al. 1999; Sammels et al. 2004). Alternatively, it is also possible that freshly isolated, IV-injected muscle SP and MP cells may reside longer than 24 hr in the circulation prior to lodging into other organs. Clearly, additional studies will be necessary to investigate the kinetics and mechanisms of muscle SP/MP cell survival and entrapment in different tissues. These mechanisms are likely to involve modification of the cell surface molecules expressed on muscle SP or MP cells, which can in turn modulate recognition between these cells and different organs.
Detection of donor-derived cells by FISH in the bone marrow of recipient mdx mice 12 weeks after transplantation demonstrated a higher percentage of Y-positive cells in the bone marrow of mice injected with muscle SP cells than muscle MP cells. Although these results may suggest that muscle SP cells contain a higher proportion of hematopoietic progenitors than MP cells, additional studies are necessary to determine whether this is indeed the case. For example, injection of single cells or clones of phenotypically characterized muscle-derived SP cells into lethally irradiated mice will address this question. Such studies have been performed using clones of muscle-derived stem cells (MDSC), which appear phenotypically similar to muscle SP cells (Cao et al. 2003).
One current important caveat with the use of muscle-derived SP cells is lack of methods for in vitro expansion with preservation of their engraftment potential. This limits the number of cells available for injection. Thus far, only numbers in the order of 104 muscle SP cells have been injected in mouse models with muscular dystrophy. The current study shows no positive correlation between the number of dystrophin+ Y+ myofibers and the number of injected SP cells, indicating that factors other than cell number should be considered to improve the efficacy of cell-based therapy of muscular dystrophy. Encouraging findings have been reported using intra-arterial delivery of mesoangioblasts to α-sarcoglycan-deficient mice, leading to phenotypic and functional correction of muscular dystrophy (Sampaolesi et al. 2003). Clearly, more studies will be necessary to optimize parameters for muscle SP cell expansion, survival, trafficking, and robust engraftment within dystrophic muscle.
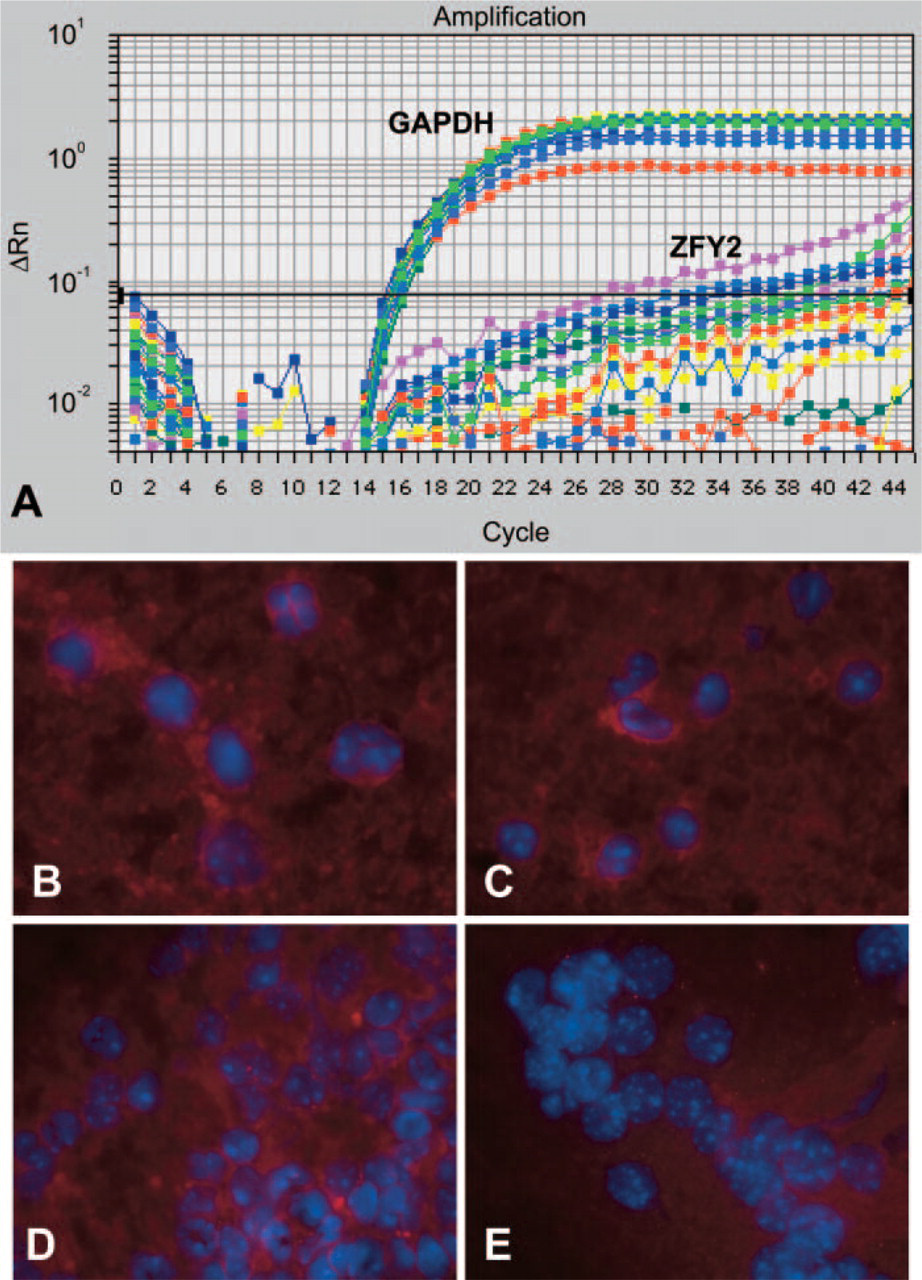
Donor male cells are not detected in the lungs, liver, and spleen of mdx mice injected with muscle SP and MP cells 1 day after intravenous cell injection. (
Footnotes
Acknowledgements
This work was supported by a grant from the Muscular Dystrophy Association, USA (E.G.), and from the Jett Foundation (E.G.).
The authors would like to thank Dr. Louis Kunkel, in whose laboratory this project was initiated, for helpful discussions and support. We also would like to thank Dr. Federica Montanaro, Dr. Kalliopi Liadaki, Jaclyn Schienda, and Michael Molloy for critical review of this manuscript.
