Abstract
We recently found that four peptide hormones synthesized by the same gene completely inhibit the growth of human pancreatic adenocarcinomas in athymic mice. The present immunocytochemical investigation was designed to determine where in the adenocarcinomas these peptide hormones localize. Atrial natriuretic peptide, vessel dilator, long-acting natriuretic peptide, and kaliuretic peptide localized to the cytoplasm and nucleus of the human pancreatic adenocarcinomas, which is consistent with their ability to decrease DNA synthesis in the nucleus of this cancer. In this first investigation of where these peptide hormones with anticancer effects localize in any cancer, these peptide hormones also localized to the endothelium of capillaries and fibroblasts within these cancers. This is the first demonstration of growth-inhibiting peptide hormones localizing to the nucleus, where they inhibit DNA synthesis and may interact with growth-promoting hormones that localize there as the etiology of their ability to inhibit the growth of adenocarcinomas both in vitro and in vivo.
A
We have found that vessel dilator, LANP, kaliuretic peptide, and ANP decrease the number of human pancreatic adenocarcinoma cells in culture by 65%, 47%, 37%, and 34%, respectively, within 24 hr (Vesely et al. 2003). This decrease was sustained without any proliferation of the adenocarcinoma cells occurring in the 3 days after this decrease (Vesely et al. 2003). The mechanism of these peptide hormones’ decrease in cancer cell number and antiproliferative effects was a 83% or greater inhibition of DNA synthesis, but not owing to enhanced apoptosis (i.e., programmed cell death) (Vesely et al. 2003). One of the known mediators of these peptide hormones’ mechanisms of action (i.e., cGMP) inhibited DNA synthesis in these adenocarcinoma cells by 51% (Vesely et al. 2003).
In vivo, vessel dilator (139 ng·min−1·kg−1 of body weight) infused for 14 d completely stopped the growth of the human pancreatic adenocarcinomas in athymic mice (
The present investigation was designed to determine where these peptide hormones localize within human pancreatic cancers. Where these peptide hormones localize within any cancer has never been investigated.
Materials and Methods
Tissue Samples
The human pancreatic adenocarcinomas growing in athymic mice were removed after separate four-week infusions via Alzet Model 2004 osmotic pumps (Duret Corporation; Cupertino, CA) of vessel dilator, LANP, kaliuretic peptide, and ANP with each peptide at a concentration of 1.4 μg·min−1·kg−1 body weight) (Vesely et al. 2004). The Alzet Model 2004 osmotic pump for mice delivers all of its contents (200 μl) over 28 days at a rate of 0.25 μl·h−1 and then stops pumping. Untreated human pancreatic adenocarcinomas were also examined 4 weeks after they had became palpable, at which time their volume had increased 299-fold from when they were first palpated. These treated and untreated pancreatic adenocarcinomas were fixed in zinc-formalin for 24 hr, paraffin-embedded, and subsequently processed with routine techniques and immunohistochemistry analysis described previously from our laboratory (Ramirez et al. 1992; Saba et al. 1993). Several tumors of animals treated with vessel dilator, LANP, kaliuretic peptide, and ANP, respectively, and several untreated human pancreatic adenocarcinomas were evaluated.
Immunohistochemistry
After deparaffinization and rehydration with PBS, pH 7.2, 3-μm sections were stained for vessel dilator, LANP, kaliuretic peptide, and ANP by the avidin-biotin peroxidase complex technique (Hsu et al. 1981) using a Vectastain kit (Vector Laboratories; Burlingame, CA). Sections were incubated for 1 hr at 24C in a humidity chamber with the respective primary antibody diluted (1:200–1:1000) with PBS, and then, after rinsing, incubated again in the humidity chamber for 30 min with biotinylated antibody. The illustrations in this manuscript are all at the 1:800 dilution of the respective primary antibodies. The slides were then rinsed followed by a final incubation for 30 min in avidin-biotin peroxidase complex in a humidity chamber. Controls for the immunoperoxidase staining included: (a) substitution of the primary antibody with normal rabbit serum and (b) preincubation of the primary antibody with excess vessel dilator, LANP, kaliuretic peptide, or ANP in their respective immunoperoxidase assays for 24 hr at 37C. These respective human peptide hormone antisera for immunohistochemistry were from Peninsula Laboratories (Belmont, CA).
Cross-reactivity of Vessel Dilator, LANP, Kaliuretic Peptide, and ANP
Cross-reactivities of the various human antisera raised in rabbits to determine their specificity were determined by comparative radioimmunoassays using the human form of pure synthetic peptides vessel dilator, LANP, kaliuretic peptide, and ANP synthesized and assayed by Peninsula Laboratories, Inc. LANP antisera cross-reactivity with pure synthetic LANP was 100%, whereas its cross-reactivity with ANP and vessel dilator was 0%. Vessel dilator antisera had 100% cross-reactivity with pure synthetic vessel dilator, but 0% cross-reactivity with LANP or ANP. The antisera to ANP had 100% cross-reactivity with ANP, and 0% cross-reactivity with either LANP or vessel dilator. Kaliuretic peptide antisera had 100% cross-reactivity with human kaliuretic peptide, but 0% cross-reactivity with LANP, vessel dilator, or ANP.
Results
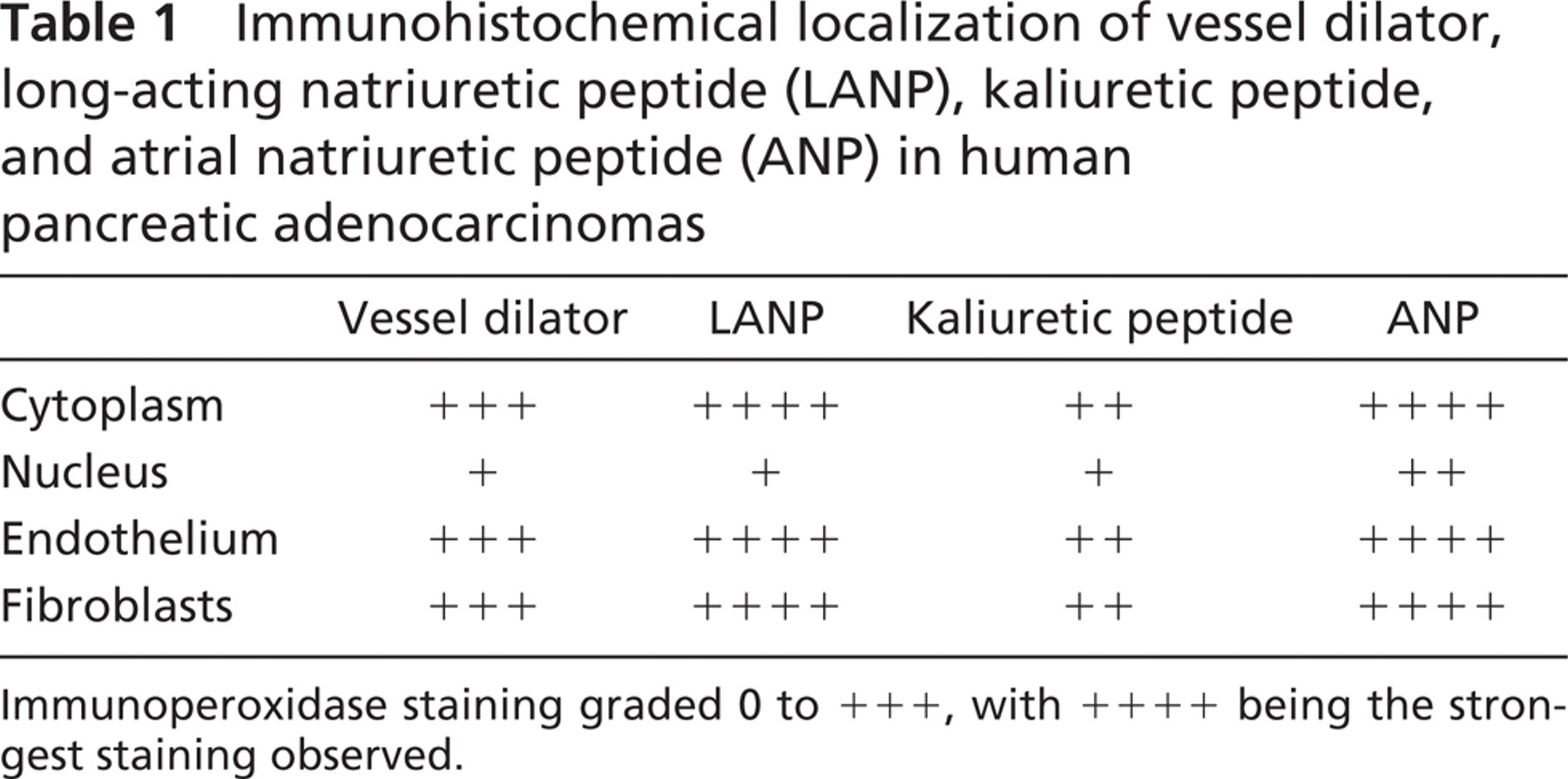
Immunoperoxidase staining of vessel dilator was very strong (+ + +) in the cytoplasm of the human adenocarcinoma cells (Figure 1A) compared with control adenocarcinoma cells (insert, Figure 1D), which did not receive a vessel dilator infusion in vivo. Essentially all of the cytoplasm and perinuclear areas of the pancreatic adenocarcinoma cells had strong staining with vessel dilator (Figure 1A). The nucleus of the adenocarcinoma cells had weaker (+) but discernible vessel dilator immunoperoxidase staining (insert, Figure 1A). Vessel dilator also localized (+ + +) to the endothelium of the small capillaries invading the human pancreatic adenocarcinoma. On histological hematoxylin and eosin (H & E) staining, there were not any large blood vessels (e.g., arteries) within these tumors, but numerous small capillaries invading this tumor could be visualized (Figure 2). In the H & E evaluation, one can observe mitosis occurring within the cancer cells with their nuclei clearly discernible (Figure 2). Each of the tumors had necrotic centers in both the treated and untreated adenocarcinomas. Vessel dilator also localized to the fibroblasts within the adenocarcinomas where the cytoplasm, but not the nuclei, of the fibroblasts stained positive for vessel dilator (Figure 1A).
LANP had a slightly stronger (+ + + +) immunoperoxidase staining of the cytoplasm, but similar intensity (+ + +) of immunoperoxidase staining of the perinuclear area and nucleus (+) of the human pancreatic adenocarcinomas compared with vessel dilator (Figure 1B). Strong (+ + + +) LANP immunoperoxidase staining was noted in the endothelium in the small capillaries in the adenocarcinomas (Figure 1B). LANP staining of the fibroblasts within this tumor was more intense (+ + + +) than with vessel dilator (Figure 1B).
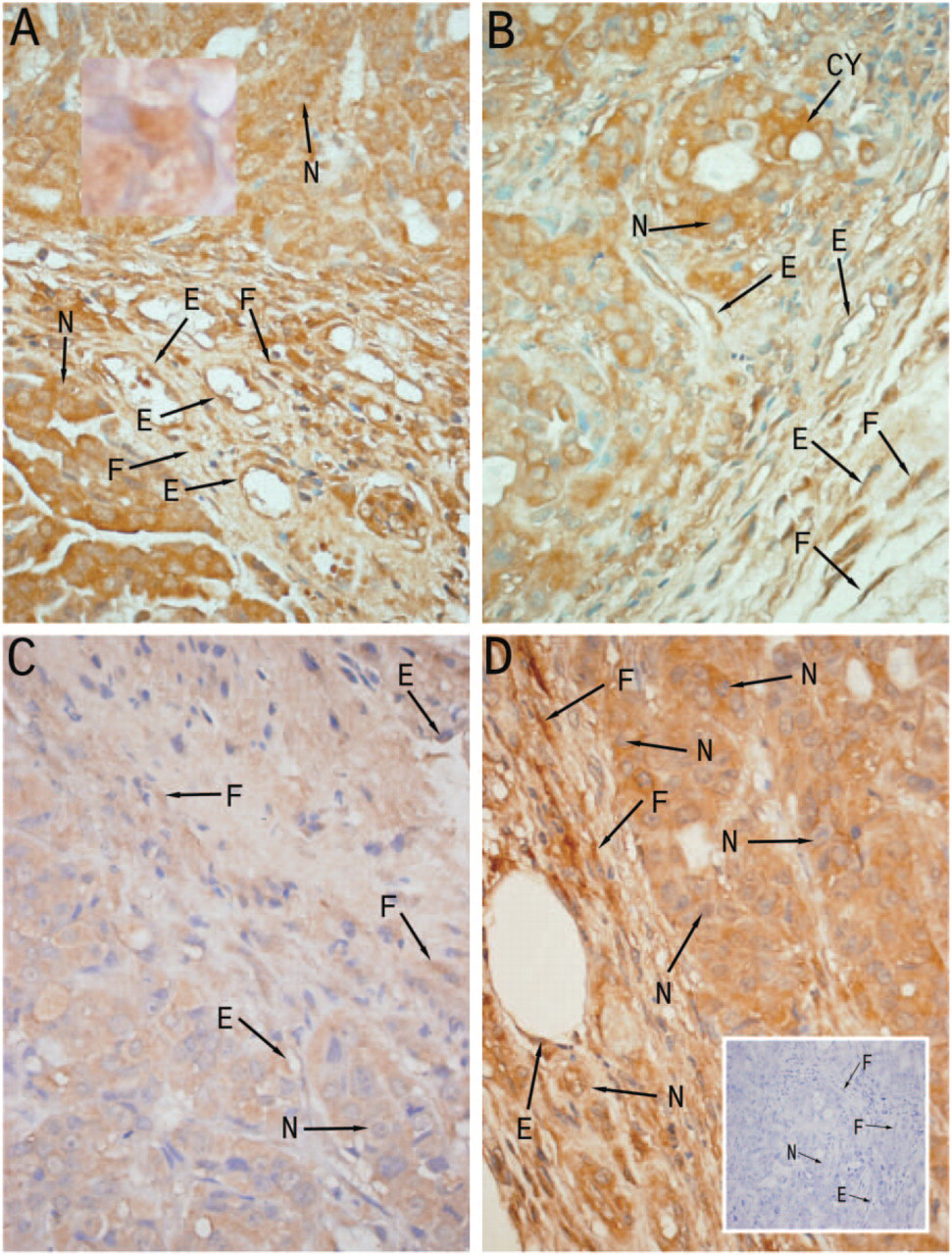
Immunoperoxidase localization of vessel dilator, long-acting natriuretic peptide (LANP), kaliuretic peptide, and atrial natriuretic peptide (ANP) within human pancreatic adenocarcinomas with each of these peptide hormones strongly localizing to cytoplasm (Cy), nucleus (N), endothelium (E), and fibroblasts (F). The light blue stain in the fibroblasts is the nuclei of the fibroblasts. (
Kaliuretic peptide immunoperoxidase staining in the adenocarcinomas infused with kaliuretic peptide had a decreased intensity compared with the other peptide hormones (Table 1 and Figure 1C). Kaliuretic peptide, however, localized to the same structures within the human pancreatic adenocarcinomas (Figure 1C). Thus kaliuretic peptide immunoperoxidase staining (+) localized to the nucleus of the cancer cells similar to that observed with vessel dilator and LANP (Figure 1C). Staining of the cytoplasm (+ +) of the human pancreatic adenocarcinoma cells with kaliuretic peptide was less than that observed with vessel dilator, but there was definite localization of kaliuretic peptide to the cytoplasm. Kaliuretic peptide also localized to the endothelium of the small capillaries and to fibroblasts (Figure 1C), with an intensity similar to that of its localization to cytoplasm.
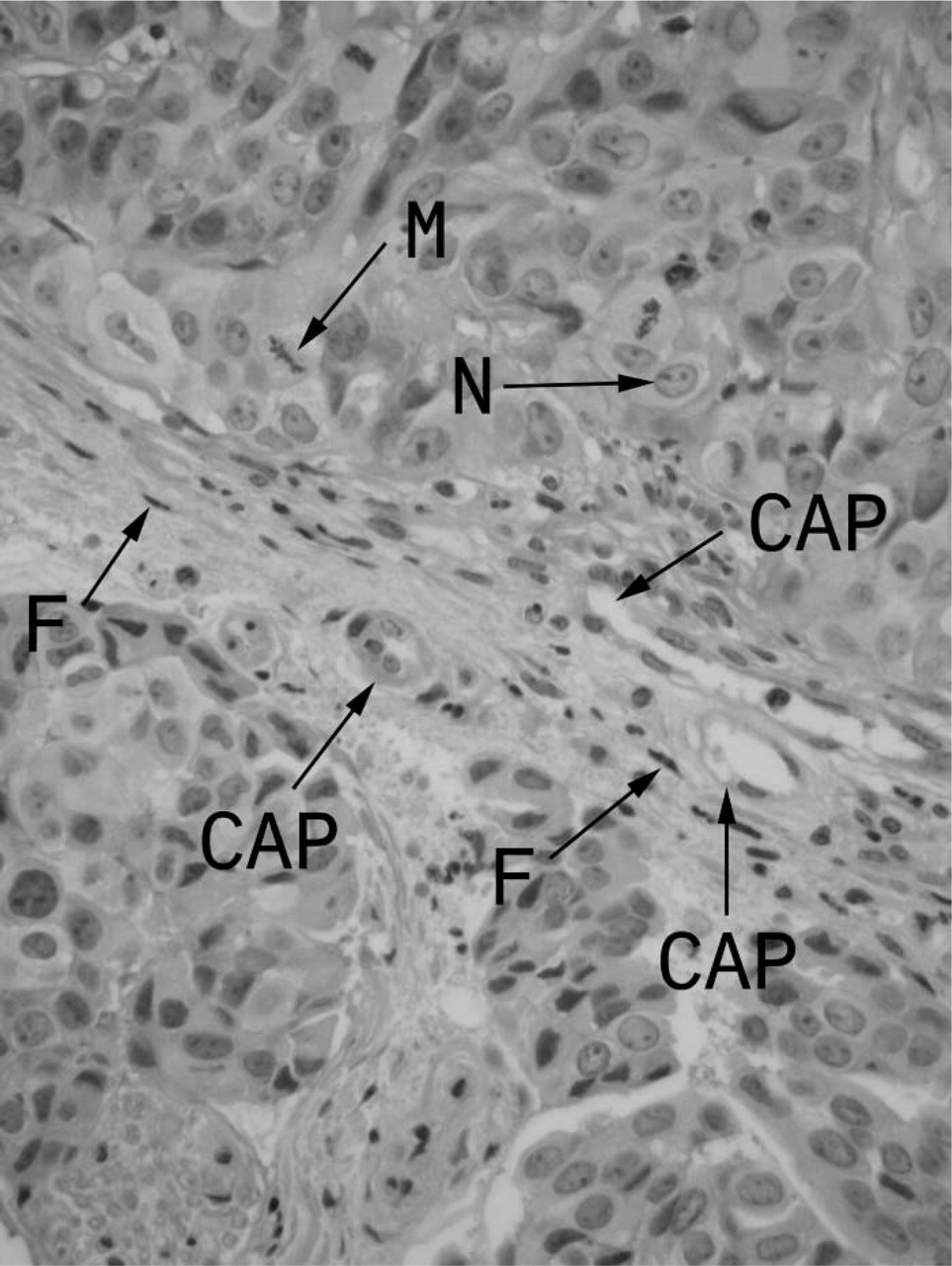
Histology (hematoxylin and eosin) of human pancreatic adenocarcinoma illustrating capillaries (but not large blood vessels) growing into adenocarcinoma and numerous mitosis occurring. CAP = capillaries, n = nucleus of human pancreatic adenocarcinoma cell, M = mitosis within adenocarcinoma cell, and F = fibroblasts with human pancreatic adenocarcinoma. It is important to note that there are several cancer cells undergoing mitosis in this very rapidly growing cancer. Original magnification x60.
ANP had very strong immunoperoxidase (+ + + +) localization to the cytoplasm of the human pancreatic cells (Figure 1D). ANP had slightly stronger immunoperoxidase staining of the nucleus (+ +) than each of the other peptide hormones (Table 1). ANP also had a very strong (+ + +) localization to the endothelium of the capillaries invading this cancer. Strong (+ + + +) ANP immunoperoxidase staining was also present in the fibroblasts in this tumor (Figure 1D). The immunohistochemical data are summarized in Table 1.
Immunohistochemical localization of vessel dilator, long-acting natriuretic peptide (LANP), kaliuretic peptide, and atrial natriuretic peptide (ANP) in human pancreatic adenocarcinomas
There was not any immunoperoxidase staining with vessel dilator, LANP, kaliuretic peptide, or ANP in the human adenocarcinomas when their respective primary antisera were either substituted with normal rabbit serum (Figure 1D) or when the primary antibody was preincubated with an excess of vessel dilator, LANP, kaliuretic peptide, or ANP in their respective immunoperoxidase assays for 24 hr at 37C.
Discussion
This is the first demonstration of the localization of four peptide hormones synthesized by the ANP gene in any cancer. Human pancreatic adenocarcinomas were chosen as the first cancer to be investigated because people with pancreatic adenocarcinoma have the lowest 5-year survival rate (1%; with a median survival of only 4 months) of all common human malignancies (Pitchumoni 1998; Wolff et al. 2000). Surgery and chemotherapy extend survival by a few months, but the 4-month median survival and 5-year survival rate includes those people treated with surgery and chemotherapy (Pitchumoni 1998; Wolff et al. 2000). The human pancreatic carcinomas of the present investigation had their growth completely stopped by each of the four peptide hormones of this investigation, and vessel dilator decreased the volume of these adenocarcinomas 49% after each were infused for a week SC (Vesely et al. 2004). Each of the four peptide hormones in the present investigation had localization in the nucleus of the cancer cells. This information correlates with the knowledge that each of these peptide hormones strongly inhibit DNA synthesis (83% to 91% decrease) within the human pancreatic adenocarcinomas of the present investigation (Vesely et al. 2003) as the nucleus is the site of DNA synthesis. Further correlation of the findings of the localization of these peptide hormones in the nucleus of the human pancreatic adenocarcinomas in the present investigation are flow cytometry studies demonstrating that each of these peptide hormones, except ANP, decrease the number of adenocarcinoma cells in synthetic phase of the cell cycle (i.e., where DNA synthesis occurs) (Vesely et al. 2005). ANP decreases the number of adenocarcinoma cells in the mitotic phase of the cell cycle (Vesely et al. 2005).
Vessel dilator, LANP, kaliuretic peptide, and ANP had a very strong localization to the cytoplasm of these cancer cells. We had hypothesized that if these peptide hormones were reaching the nucleus to inhibit DNA synthesis, they should also be abundantly present in the cytoplasm after binding to the cell surface receptors on plasma membranes of the cancer cells. These peptides are known to bind to specific receptors in the plasma membranes of normal and cancer cells (Vesely et al. 1987,1990,1992). After ANP binds to its receptor, the receptor internalizes and ANP was then thought to be degraded, with the receptors recycling to the plasma membrane (Hirata et al. 1985; Napier et al. 1986; Hughes et al. 1987; Morel and Heisler 1988). Part of the cytoplasmic demonstration of these peptide hormones within the cancer cells may be the ANPs attached to their receptors that are being internalized. However, the intense cytochemical localization throughout the cytoplasm and the new knowledge of the present investigation that these peptides also localize strongly to the perinuclear area and are in the nucleus suggests that these peptides are not all being degraded in the cytoplasm as previously thought, but rather are traveling through the cytoplasm to reach the nucleus to directly inhibit DNA synthesis.
There is evidence that other peptides are transported from the external plasma membrane to the nucleus (Burack and Shaw 2000). One example of this is extracellular receptor kinase (ERK), which is transported to the nucleus to cause proliferation (i.e., a growth-promoting protein) (Burack and Shaw 2000). Proteins such as ERK are thought to move from the plasma membrane to the nucleus via attaching to a scaffolding protein, which moves them to the nucleus (Burack and Shaw 2000). One could envision a similar type of scaffolding protein to transport the four growth-inhibiting proteins of the present investigation, but this protein(s) has not been defined.
These peptide hormones do have an intracellular mediator within the cytoplasm (i.e., cGMP) (Brenner et al. 1990; Vesely 1997). cGMP inhibits DNA synthesis in these cancer cells, but only to about half of the extent of vessel dilator (Vesely et al. 2003). The new knowledge of the present investigation that the peptide hormones are reaching the nucleus via the cytoplasm suggest that these peptide hormones may be directly inhibiting DNA synthesis and decreasing cell-cycle progression (with 62% less cells in the synthetic phase) and indirectly inhibiting DNA synthesis via the intra-cellular messenger cGMP.
It is important to note that all of the previously reported peptide hormones that localize to the nucleus (i.e., insulin, epidermal growth factor, nerve growth factor, platelet-derived growth factor, luteinizing hormone releasing hormone, and human chronic gonadotrophin) (Burwen and Jones 1987) and ERK (Burack and Shaw 2000) have been growth-promoting hormones. The present investigation is the first demonstration we are aware of that growth-inhibiting peptide hormones localize to the nucleus, where they can directly interact to inhibit the effects of the growth-stimulating hormones and act via their demonstrated direct decrease of DNA synthesis in the nucleus (Vesely et al. 2003).
In the present investigation, these peptide hormones also localized to the endothelium of the blood vessels growing into this cancer. This finding is similar to previous finding (Figure 3) that these peptide hormones localized to the endothelium of blood vessels of normal kidneys (Ramirez et al. 1992; Saba et al. 1993). In normal tissues, these peptides localize to the endothelium of larger arteries and veins (Ramirez et al. 1992; Saba et al. 1993), but in the present investigation, when examined by H & E staining (Figure 2), there were not any large blood vessels growing into these tumors, but rather only capillaries that were invading this cancer. Within the capillaries, it was the endothelium that had immunoperoxidase staining of these peptide hormones (Figure 1). That no large blood vessels had grown into these adenocarcinomas helps explain the observation on H & E examination that the centers of each of these tumors were necrotic. This finding is apparently the result of these tumors outgrowing their blood supply (i.e., no large blood vessels were present) rather than from an effect of these peptide hormones per se, because the untreated adenocarcinomas had equally as much necrosis in the center of their tumors as the vessel dilator-, LANP-, kaliuretic peptide-, and ANP-treated tumors.
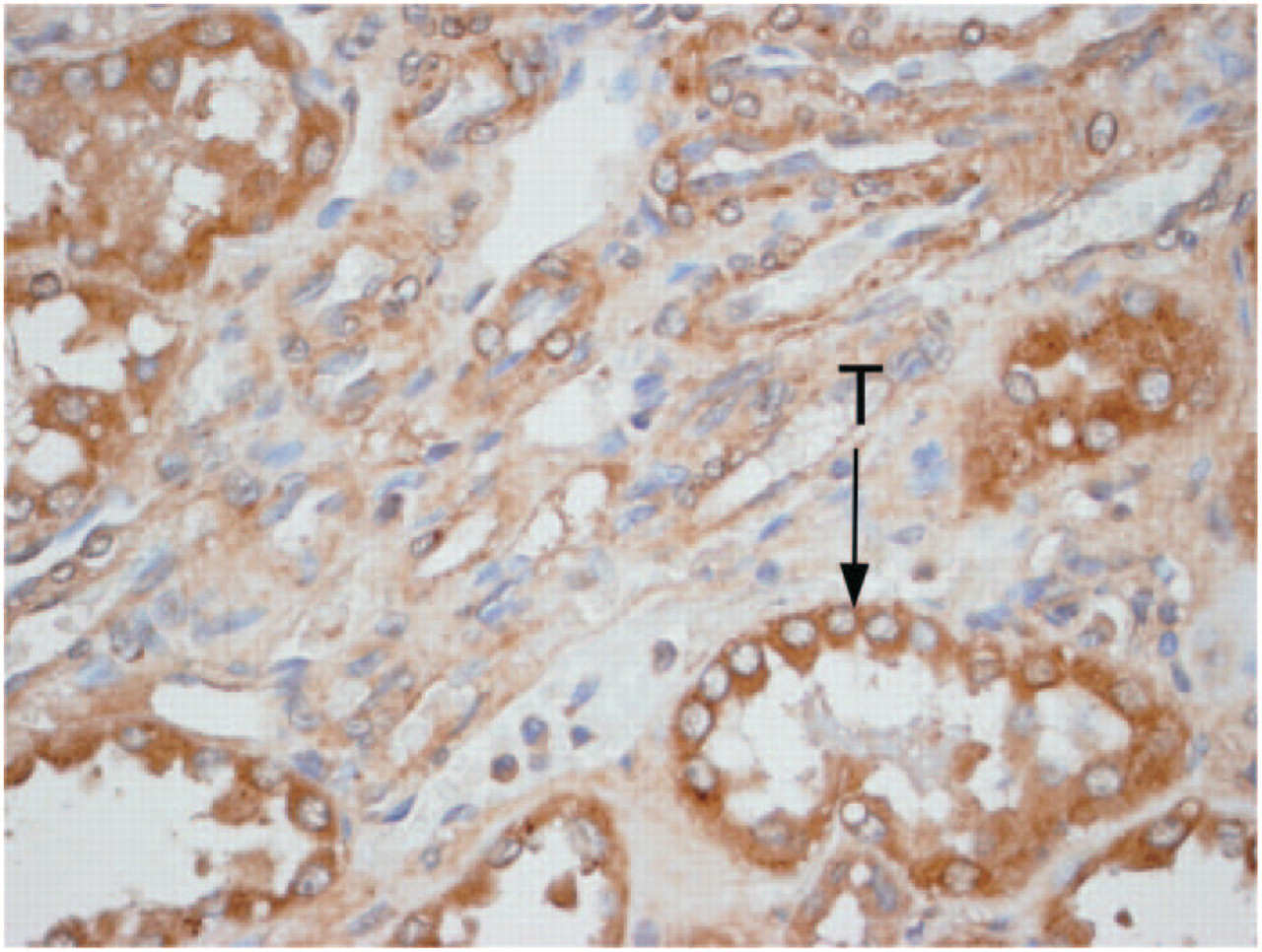
Immunoperoxidase localization of vessel dilator within the kidney demonstrating staining throughout the kidney but of increased intensity of staining in the proximal tubules (T) of this normal rat kidney. Original magnification x60. Primary antibody diluted 1:800.
LANP, vessel dilator, ANP, and kaliuretic peptide also localized to the fibroblasts within the adenocarcinomas in the present investigation. It could not be determined with certainty whether these peptide hormones were being synthesized by the fibroblasts or whether they localized to the fibroblasts after their infusion. ANP prohormone mRNA is present in fibroblasts, which indicates that vessel dilator, LANP, kaliuretic peptide, and ANP, which are derived from the ANP prohormone, are synthesized by fibroblasts (Kawano et al. 2000). It is of interest that these peptide hormones did not localize to the nucleus of the fibroblasts, but only to the nucleus of the cancer cells.
Footnotes
Acknowledgements
We thank Charlene Pennington for excellent secretarial assistance and Quentin W. McAfee for culturing the human pancreatic cells before their injection into athymic mice to form tumors. This study was funded in part by a grant from the Darren Manelski Foundation, New York, NY.
