Abstract
The biological effect of atrial natriuretic peptide (ANP) in traumatic hemorrhagic shock (THS) is unknown. This study was to evaluate whether ANP therapy can show organ protection in THS. Thirty male Sprague-Dawley rats were divided into three groups: ANP group, sham group, and control group. Pressure-controlled THS was induced in rats in ANP group and control group. ANP at a rate of 0.025 μg/kg/min was infused in ANP group during near-80 min of shock. After that, animals were resuscitated for 60 min and observed until 24 h. Hemodynamic parameters during shock and resuscitation were measured. Serum levels of ANP and lactate dehydrogenase, tissue oxidative stress and inflammatory factors, as well as liver and kidney function were determined. Tissue apoptosis was also assessed. There was no statistically significant difference between ANP group and control group in arterial pressure throughout the 150 min monitoring period. Blood urea nitrogen at 90 min and 24 h in ANP group was significantly lower than control group. Alanine transaminase and aspartate aminotransferase activity at 90 min in control group were significantly higher than that in sham group. However, hepatic enzyme activity at 90 min in ANP group was not significantly different compared with sham or control group. After 24 h, myocardial expression of caspase 3 protein in ANP group was significantly reduced compared with control group. Jejunal and hepatic Malondialdehyde was increased following ANP treatment. ANP therapy during early THS has no significant adverse effect on hemodynamics but can exert oxidative stress and certain protective effect on multiple organs. Our study may shed light on the novel therapy of THS with regard to organ protection. The mechanisms underlying the organ protection require further study.
Introduction
Trauma remains a leading cause of mortality and morbidity among populations of all ages. Damaged cells after injury release large amounts of danger-associated molecular patterns which can be recognized by toll-like receptors on the surface of immune cells, thereby triggering the activation of myeloid differentiation factor 88 dependent inflammatory pathway.1–3 Activation of nuclear transcription factor-Kappa B (NF-κB) consequently stimulates innate immune cells to release inflammatory factors. 2 The inflammatory cascade initiates systemic inflammatory response syndrome (SIRS), leading to the development of multiple organ failure (MOF). 3 Prevention of trauma-induced MOF is filled with many challenges as care strategies for trauma patients recommended in clinical guidelines usually neglect the immune aspect.
Atrial natriuretic peptide (ANP) is a 28-amino acid peptide that is produced in several tissues. This ANP is a biologically active hormone that regulates blood pressure as well as the balance of the salt and water. 4 Recent research has reported that ANP has anti-inflammatory properties and is protected by natriuretic peptide receptor (NPR)-mediated signaling pathways against multiple organ injury.4,5 ANP significantly decreased NF-κB activation and tumor necrosis factor (TNF)-α intrapancreatic levels in cerulein-induced acute pancreatitis in rats. 6 During cardiac surgery, an intravenous ANP infusion demonstrated a renal protective effect caused by the inhibition of the intrarenal renin-angiotensin-aldosterone system. 7 It is not clear if ANP plays a part in the early treatment of traumatic hemorrhagic shock (THS).
In our previous study, serum atrial natriuretic peptide (ANP) level was positively correlated with expression of intestinal Protein kinase B (Akt) protein in THS but negatively correlated with interleukin 10 (IL-10) expression in bronchoalveolar lavage fluids. 8 Although the study did not find a correlation between ANP and early survival, it does imply that ANP may have a potential role in the initial treatment of THS, which needs to be studied further. ANP has a blood pressure lowering effect, but it is not clear whether ANP can aggravate hemodynamic disorders during hemorrhagic shock. In this study, we aimed primarily to evaluate the influence of exogenous ANP therapy on organ protective profiles in THS. We focused on assessing the non-side effects of ANP and blood pressure in the THS model and the impact on oxidative stress.
Materials and methods
Animals
Eight-to-twelve-week-old male Sprague-Dawley rats, weighing 342–484 g, were provided by Zhejiang Academy of Medical Sciences. The protocol for this research was approved by the Intitutional Ethical Committee at the Second Affiliated Hospital of Zhejiang University School of Medicine. Animals were treated per guideline from “Guide for the Care and Use of Laboratory Animals-Eighth Edition” (National Institutes of Health publication, 2011). Rats were maintained on an alternating 12-h light/dark cycle at a room temperature of 22°C. Rats were fasted for 12 h but allowed free access to water before the experiment. The sample size of 10/group was sufficient to detect a relative difference of 80% between treated groups with an α of 0.05 and power (1-β) of 0.80. They were allocated into different groups according to a randomized digital table.
Surgical procedures
Prior to surgical procedure, rats were anesthetized with 2% sodium pentobarbital (50 mg/kg, i.m.). The rectal temperature (Tr) was monitored continuously and maintained at normothermia. Intratracheal intubation and mechanical ventilation were performed with a tidal volume of 0.75 ml and 75 beats/min respiratory rate. For blood screening, bloodletting, and pressure control, a 24-gauge Angiocath (Becton-Dickinson, Sandy, UT) was inserted through the left popular carotid artery. A polygraph (PM-9000; Shenzhen Mindray Bio-Medical Electronics Co, Ltd, Shenzhen, China) was used to monitor hemodynamics. A 22-gauge Angiocath was implanted into the left femoral vein for infusion. A midline laparotomy was conducted to mimic the abdominal trauma, a 2-0 nylon suture was mounted along the edges of the laparotomy.
THS model and intervention
All animals in the experimental design were divided into three stages: phase 1, pressure-fixed THS (0–90 min); phase 2, resuscitation (90–150 min); and phase 3, observation (150 min–24 h). We chose a near 90 min of shock because it induced a strong intestinal bacterial shift compared to that of 30 or 60 min when the target mean arterial pressure (MAP) was reduced to 30–40 mmHg. At 0 min, hemorrhage from the carotid artery was induced by bloodletting at a rate of 2 ml/kg/min until mean arterial pressure (MAP) reduced to 40 mm Hg (usually took 10 min). The target MAP was maintained stable by either bleeding or infusing normal saline (NS) until 90 min.
The rats were divided into three groups at random (n = 10 for each group): group A, ANP group; group B, sham group; group C, control group. At 30 min, ANP (ANF 3-28, A6791, Sigma-Aldric, St Louis, Mo) was infused in group A at a rate of 0.025 μg/kg/min until 90 min through a Pump 11 Elite (Harvard Apparatus, Holliston, MA). Animals in group C were not treated in phase 1 except for maintaining the arterial blood pressure of 40 mmHg. During phase 2, animals in group A and C were resuscitated with shed blood and NS (1:2) to achieve baseline MAP targets. MAP was recorded every 10 min during 0–150 min, urine volume and the amount of infused blood and NS were also recorded. All animals were allowed to eat and drink during phase 3 until 24 h, after which time they were sacrificed by intravenous injection of an overdose of pentobarbital sodium.
Serum ANP detection
Blood samples at 0 min, 90 min, and 24 h were analyzed to test the level of serum ANP using commercialized assays (Rat ANP ELISA kit, ab108797, Abcam, Cambridge, MA) by the enzyme-linked immunosorbent assay (ELISA) method. Blood sampling by cardiac puncture was performed from 10 healthy male SD rats to obtain normal reference range for random serum ANP level.
Liver and renal function
To assess damage to the hepatic and renal parenchyma, serum activity of alanine transaminase (ALT) and aspartate aminotransferase (AST) as well as levels of blood urea nitrogen (BUN) and creatinine were assessed. Blood samples were analyzed with assay kits (Nanjing Jiancheng Bioengineering Institute, Nanjing, China) in a microplate reader (ELX-808, BioTek Instruments, Inc., Winooski, VT) at different interval of 0 min, 90 min, and 24 h.
Necrosis and inflammation biomarkers
ELISA kits of rat lactic dehydrogenase (LDH) (ab102526, Abcam, Cambridge, MA), TNF-α (BMS622, eBioscience, San Diego, CA), and interleukin-10 (IL-10) (BMS629, eBioscience, San Diego, CA) were used to measure serum LDH activity and levels of TNF-α and IL-10. Malondialdehyde (MDA) level and myeloperoxidase (MPO) activity in the liver, jejunum, and heart tissues were measured to assess the oxidative stress status. Wet-to-dry weight ratios (W/D) of the liver and jejunum tissues were measured to assess tissue edema. In the sham group, rats also received the same anatomic procedures such as invasive arterial and venous cannulations and midline laparotomy. These procedures would augment inflammation, which would cause increased TNF-α level.
Western blot analysis for inflammation and apoptosis-related protein
To assess the inflammation and apoptosis-related protein, the isolated tissues of jejunum, myocardium, and liver were minced and washed with PB solution (phosphate-buffered saline) followed by proper homogenization in the Western blot lysis buffer for 30 min. The extracts were cleared by centrifugation at 12,000g for 5 min. Bicinchoninic acid assays were used to quantify the concentration of protein. β-actin was used as the loading control. The resolved proteins were separated by electrophoresis with the help of 10% sodium dodecyl sulfate-polyacrylamide gel and transferred to the membrane of polyvinylidene difluoride. After transferring, this membrane was further blocked with 5% nonfat milk in phosphate buffered saline for 1.5 h at room temperature. These membranes were then incubated with primary antibodies such as anti-HMGB-1 (ab79823, 1:10000; Abcam, Cambridge, MA), anti-NF-κB (ab32360, 1:5000; Abcam, Cambridge, MA), and anti-caspase-3 (sc-7148, 1:1000, Santa Cruz Biotechnology, Santa Cruz, CA) overnight at 4°C. Blots were washed twice over 30 min in tris-buffered saline. Horseradish peroxidase conjugated goat anti-rat immunoglobulins (CW0104, 1:5000; CWbio. Co. Ltd, Beijing, China) were incubated at room temperature for 1 h. All membrane images were acquired using ChemiDocTM MP imaging system (Bio-Rad Laboratories, Hercules, CA). The density of each band was quantified using Quantity One software (Bio-Rad Laboratories, Hercules, CA).
Statistical analysis
All data in this study are presented in the form of the mean ± standard deviation. Two-way repeated measures analysis followed by post hoc Turkey tests were used to analyze the variables that have been tested several times. One-way multivariate analysis was used for only one time recoded data. Differences between two groups were assessed by an independent-samples t-test. All the statistical analyses were done by IBM SPSS version 19.0 software (SPSS, Inc, Chicago, IL). A p-value of <0.05 was considered significant.
Results
All animals survived over 24 h. There was no statistically significant difference between groups with respect to MAP and body weight in the baseline characteristics as shown in Table 1 and Figure 1(a).
Animal characteristics.
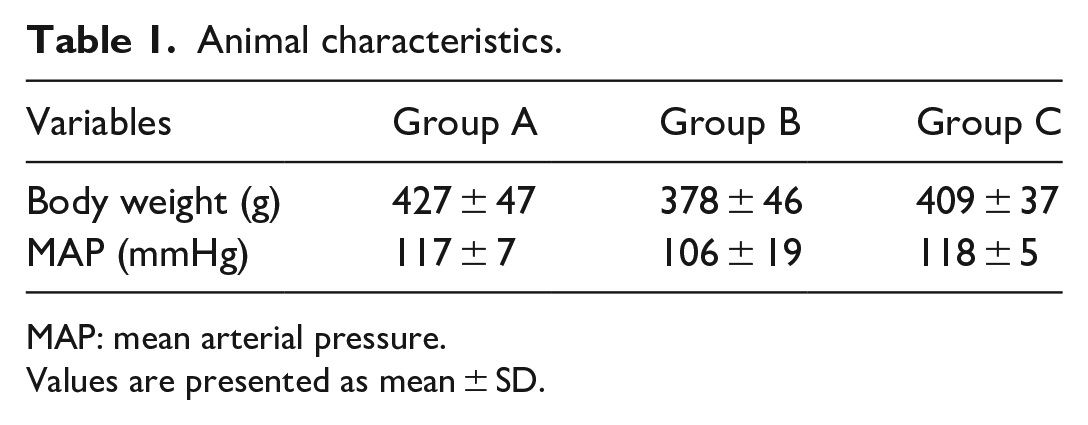
MAP: mean arterial pressure.
Values are presented as mean ± SD.

MAP changes (a) and NS requirement (b) during hemorrhagic shock and resuscitation. Group A, ANP group; Group B, sham group; Group C, control group. No significant difference was found between group A and group C in MAP and NS infused during the shock stage.
Hemodynamics and fluid management
There was no statistically significant difference between group A and group C in MAP throughout the 150 min monitoring period. There was no substantial difference between group A and group C in the amount of NS infused during the hemorrhagic shock and resuscitation time (Figure 1). The output of urine between group A and group C was not substantially different, but was significantly increased in group A or group C compared to group B (p < 0.01) (Table 2).
Fluid requirement and urine output during shock and resuscitation.

NS: normal saline.
Values are presented as mean ± SD.
Serum ANP level
In this study, the normal serum ANP concentration in rats was 0.19 ± 0.11 ng/ml (95% CI: 0.11–0.28). The injury resulted in an average of a 12-fold increase in baseline serum ANP level in group A, B, and C (all p < 0.01 compared with normal value). After 90 min, the level of serum ANP in group B tended to fall. Although animals in group A received 60 min infusion of exogenous ANP, serum ANP concentration only increased slightly but without reaching statistical significance, the level of which was not significantly different compared to that in group B or group C at 90 min. After 24 h, serum ANP in groups B and C decreased to baseline level (both p < 0.01 compared to 90 min). However, serum ANP in group A remained an approximately 11-fold increase as compared with normal value (p < 0.01) (Table 3).
Comparison of serum ANP level (ng/ml) between groups.

ANP: atrial natriuretic peptide.
Values are presented as mean ± SD.
p < 0.01 versus group B or group C. #p < 0.01 versus baseline value.
Liver and renal function
At the end of phase I, serum creatinine and BUN in groups A and C were all significantly increased compared to the baseline level (Table 4). There was no statistical difference in creatinine at 90 min and 24 h between group A and group C, but BUN at 90 min and 24 h in group A were significantly lower than Group C. There was no significant change in serum activity of ALT and AST in groups A, B, and C at the end of phase I. After 24 h, serum activity of ALT and AST were significantly increased in groups A and C compared to that at 90 min or 0 min (both p < 0.01). Moreover, ALT and AST activity at 90 min in group C were significantly higher than that in group B (p < 0.05). However, there was no substantial difference in ALT and AST activities in group A compared to group B or group C.
Hepatorenal function between groups.

BUN: blood urea nitrogen; ALT: alanine transaminase; AST: aspartate aminotransferase.
Values are presented as mean ± SD.
p < 0.05 and #p < 0.01 versus group A. †p < 0.01 and ‡p < 0.05 versus group B.
Assessment of inflammation and inflammatory exudation
The baseline serum IL-10 level did not vary significantly between the groups. At 90 min, the level of IL-10 was significantly increased in groups A and C while level of IL-10 was significantly higher in group C than the group B (p < 0.05) (Figure 2). At 24 h, serum IL-10 returned to baseline in groups A and C. No significant difference was found with regard to serum IL-10 level when comparing group A to group B at 90 min or 24 h. Serum TNF-α levels at 0 min and 24 h could not be detected in most animals. There was no substantial difference between group A and group C in the amount of NS At the end of phase 1, there was a substantial increase in three groups, the amount was significantly higher in group B than in group C (p < 0.01). No substantial difference in serum TNF-α was observed between group A and group B or C. At 24 h, the liver tissue W/D ratio was significantly lower in group A than in group C (p < 0.05). However, the W/D ratio of jejunal tissue was not significantly different between group A and group C.
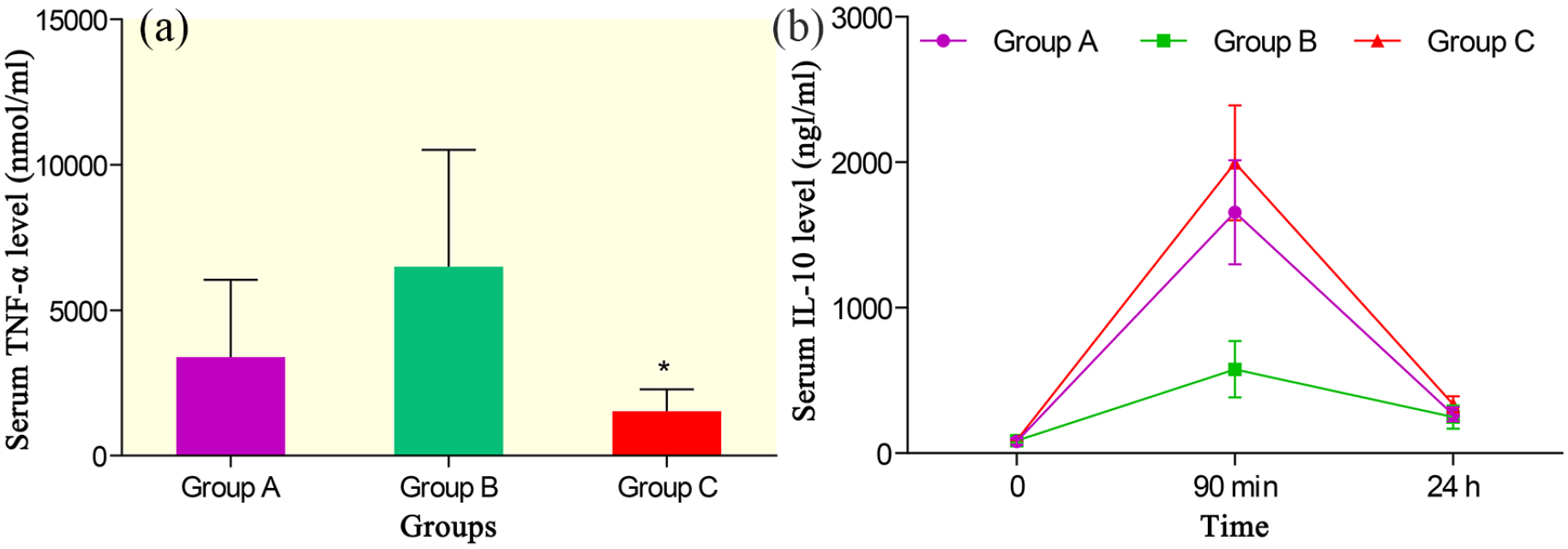
Serum TNF-α (a) and IL-10 levels (b) in THS. Group A, ANP group; Group B, sham group; Group C, control group.
Tissue oxidative stress
The level of cardiac MDA was significantly higher in group A than in group B (p < 0.05), but no substantial difference was observed between group A and group C. The activity of cardiac MPO was significantly higher in group A than in group B and group C (both p < 0.01). Similarly, the level of jejunal MDA was significantly higher in group A than in group B (p < 0.05) and group C (p < 0.01), whereas the behavior of jejunal MPO did not vary significantly between the groups. The amount of MDA in liver tissue was significantly greater in group A than in group B (p < 0.05) and group C (p < 0.01) (Table 5).
Oxidative stress between groups.

MDA: malondialdehyde; MPO: myeloperoxidase.
Values are presented as mean ± SD.
p < 0.01 and †p < 0.05 versus group A. #p < 0.05 versus group B.
Serum LDH
No major difference in baseline serum LDH was observed between the groups. LDH activity in groups A and C was substantially increased at the end of phase 1 relative to baseline, which was further elevated after resuscitation. At 24 h, serum LDH activity appeared to be decreased in three groups. Among them, serum LDH activity decreased to baseline levels in groups A and B, and serum LDH activity was significantly higher in group C than in group B (p < 0.05). When comparing group A with group B at 24 h, no substantial difference was observed in serum LDH activity (Figure 3).
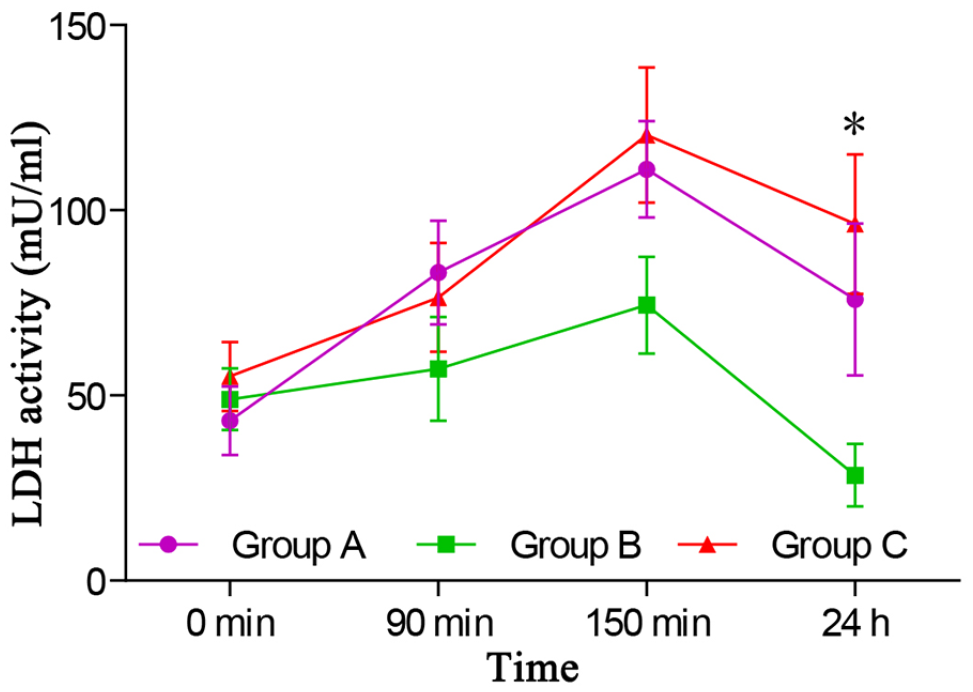
Serum lactic dehydrogenase (LDH) activity changes in THS. Group A, ANP group; Group B, sham group; Group C, control group.
Tissue expression of apoptosis- and inflammation-related protein
At 24 h, the myocardial expression of caspase 3 proteins, including procaspase-3 (32 kDa) and its degradation subunits (11 and 17 kDa) was significantly increased in group C (Figure 4). Compared with group C, myocardial expression of caspase 3 proteins in group A was significantly reduced (all p < 0.01). In addition, relative to group C, myocardial expression of HMGB-1 in group A was significantly decreased (p < 0.01). However, there was no substantial difference between group A and group C in the myocardial expression of NF-κB.

Comparison of myocardial expression of apoptosis- and inflammation-related protein between groups. Group A, ANP group; Group C, control group. (a) representative Western blots of NF-κB, HMGB-1, caspase-3, and β-actin (as loading control) in group A and group C. (b) semiquantitative comparison of band intensities (normalized to β-actin) corresponding to the blots in panel A.
There were no major variations between group A and group C in the expression of the above proteins in tissues of the liver and jejunum. There was a minimal expression of caspase 3, HMGB-1, and NF-κB in different tissues.
Discussion
We demonstrated for the first time that early ANP therapy has no significant adverse effect on hemodynamics during hemorrhagic and resuscitation but exerts a certain protective effect on organs such as the liver, kidney and heart. ANP also increased oxidative stress in THS. Our study may shed light on the novel therapy of THS with regard to organ protection.
ANP is one of the members of the natriuretic peptide family (NPs). Recent studies show that the function of NPs is not limited to regulating blood and fluid homeostasis. NPs have been found to protect multiple organs, including the heart, 9 the lungs, 10 the kidneys, 11 and the liver. 12 ANP exerts its biological effects by activating the signaling pathway for natriuretic peptide receptors (NPR)-A/cGMP or NPR-C/phospholipase C. ANP receptors have been identified in several tissues, including the lungs, the brain, the cardiovascular system and various types of cells. As THS and fluid resuscitation therapy represent a typic process of global IR injury, theoretically in this condition, ANP should have a role in preventing the occurrence of multiple organ injury.
ANP has a short half-life of only 2.4 min after intravenous injection in human, which makes it possible to use ANP clinically via continuous infusion. Theoretically, serum ANP level should have been eliminated to the pre-infusion level in a certain time after a short-term period (i.e. 60 min) of ANP infusion. However, intravenous infusion of ANP at 0.025 μg/kg/min during hemorrhagic shock resulted in persist expression of endogenous ANP in serum. The mechanisms underlying this phenomenon are still unclear and warrant further research. Nevertheless, our experimental data and literature review do provide certain clues to the mechanism analyses, which obviously beyond the scope of the present study.
According to our data, ANP treated animals showed significantly increased oxidative stress level in the tissues of heart, liver, and jejunum. This does not mean that ANP aggravated oxidative injury to multiple tissues, on the contrary, the enhanced oxidative stress may have triggered production of endogenous ANP (and perhaps vice versa). Decreased ANP clearance may also be responsible for the high level of serum ANP, which can be attributed to the changes of the affinity of ANP with its degrading enzyme (neutral endopeptidase) and/or degradation receptor. 13 However, there is little evidence in this study that this was involved. Although the MDA level and MPO activity of jejunum tissue in ANP group were higher than that in the control group, the W/D ratio and inflammation and apoptosis-related protein were not significantly different between groups, suggesting that the increased oxidative stress did not contribute to tissue injury. Early studies have shown that ANP can stimulate macrophages and polymorphonuclear leukocytes to produce superoxide, thereby enhancing innate immunity.14,15 Conversely, in vitro study by Shah et al. showed that intracellular ROS could stimulate atrial secretion of ANP through activating the MAPKerk pathway and/or Na-H exchanger. 16 At this point, the phenomenon of increased oxidative stress coupled with sustained endogenous ANP expression suggests that there may be a positive feedback loop between ANP and oxidative stress proposed by us in a previous study. 17
Among all NPs, ANP has the most potent biological activity and the shortest half-life. 18 This makes it easy to control the underlying adverse clinical effects. ANP can be increased when an injury happens, the level of which would be much higher in bone injury. 19 This may be related to catecholamine-induced stress and release of endothelin after endothelial damage. 20 Hypovolemia can also cause an increase in serum ANP, which can be reduced by appropriate volume replacement and might be elevated if volume overload happens.21–23 In the present study, we confirmed that the serum level of ANP could be increased after trauma and hemorrhagic shock. However, serum ANP levels were not further elevated when treated with exogenous ANP, which may be explained by the short half-life properties, low infusion loading (0.025 μg/kg/min), and/or high post-traumatic ANP levels.
One important finding of this study was that administration of ANP at 0.025 μg/kg/min during 60 min of shock had no evident influence on hemodynamics. There was no difference in blood pressure and the amount of fluid injected at that time between the ANP group and the control group. This is especially important, as one of the main side effects of ANP is lowering blood pressure. However, it is unclear whether higher dosages of ANP therapy will affect the hemodynamics or not. In our study, ANP has shown to promote the recovery of liver and kidney function, reduce liver edema and myocardial apoptosis, and to some extent reduce necrosis. It is worth noting that among all variables of hepatorenal function in the ANP group, only BUN was found to be lower than that in the control group. Our data, however, implies that ANP had a tendency to reduce ALT and AST activity as well as creatinine level. This difference might be better identified by enlarging sample size.
This study does not support the idea that ANP treatment can inhibit the inflammatory response, but instead, it may have a tendency to correct post-traumatic immunosuppression. Accordingly, compared with the sham group, animals in the control group showed an increase in IL-10 and a decrease in TNF-α at the end of hemorrhagic shock, suggesting that inflammatory response was inhibited to a certain extent after shock. At this time, however, IL-10 in the ANP group tended to decrease, and TNF-α tended to increase as compared with the control group, although the difference was not statistically significant. At 24 h, although serum ANP level in ANP group remained high, TNF-α and IL-10 levels in each group fell back to normal levels, this further supports that short-term ANP treatment has no significant effect on the long-term inflammatory response. In addition, whether or not the continuous infusion of ANP controls inflammation needs to be investigated in future studies.
Our study has also limitations. Firstly, the dose-effect analysis of ANP treatment was not performed as we only studied the usual dosage of 0.025 μg/kg/min, whether higher dosages may be superior is unknown. Secondly, it’s not prudent to make a conclusion that ANP has no significant effect on the inflammatory response because our study may be limited by potential biases and variability due to small samples.
Conclusion
In summary, in THS, early ANP therapy has no significant adverse effect on hemodynamics but can exert oxidative stress and certain protective effect on multiple organs. The mechanisms underlying organ protection require further study.
Footnotes
Animal welfare
Animals received humane care in accordance with the Guide for the Care and Use of Laboratory Animals: Eighth Edition (National Institutes of Health publication, 2011).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Zhejiang Provincial Natural Science Foundation of China (LQ17H150001) and Natural Science Foundation of China (81701933).
Ethical approval
Ethical approval for this study was obtained from the Intitutional Ethical Committee at the Second Affiliated Hospital of Zhejiang University School of Medicine (approval no. No 27. 2018).
