Abstract
Introduction
The mechanisms underlying the protective effects of atrial natriuretic peptide (ANP) on the gut barrier during traumatic hemorrhagic shock (THS) remain elusive. This study aimed to explore the potential role of ANP in safeguarding against gut barrier dysfunction after THS, focusing on the PLC-γ1/ROS feedback loop.
Methods
In our THS rat model, we randomly allocated male Sprague-Dawley rats to receive intravenous ANP with or without a concurrent NADPH oxidase/p38 MAPK inhibitor during the shock phase. After 24 h, we assessed circulatory and jejunal ANP, ROS, intestinal tight junction proteins, and apoptosis to evaluate the effects of ANP on the gut barrier and its interplay with intestinal ANP and ROS. Rat small intestinal epithelial cells (IECs) were also treated with ANP and subjected to hypoxia/re-oxygenation injury, with or without PI3K/PLC inhibition, to elucidate the relationship between ANP/ROS signaling and PLC-γ1. Furthermore, we modulated PLC-γ1 expression in these IECs to examine its impact on ROS and ANP production.
Results
Intravenous ANP administration at 0.025 μg/kg/min during THS significantly increased intestinal ANP and ROS levels at 24 h. ANP treatment enhanced the expression of intestinal tight junction proteins and reduced IEC apoptosis. Inhibition of circulatory ROS diminished intestinal ANP levels, while suppression of circulatory ANP led to a reduction in intestinal ROS. Decreasing PLC-γ1 expression in hypoxia/re-oxygenation-treated IECs resulted in lower ROS and ANP levels, whereas augmenting PLC-γ1 expression did not alter these levels. Additionally, PI3K inhibition markedly decreased PLC-γ1 expression in these cells.
Conclusion
ANP-induced protection of the intestinal barrier in THS is mediated by an intrinsic PLC-γ1/ROS positive feedback loop. ANP preserves gut barrier integrity and reduces IEC apoptosis through this mechanism. Further studies are warranted to investigate the interaction between IECs and other cellular components within the PLC-γ1/ROS loop.
Keywords
Introduction
Post-traumatic massive hemorrhage continues to be a predominant cause of potentially preventable multiple organ dysfunction syndrome (MODS) and mortality when not promptly and adequately addressed in an appropriate and timely manner. 1 The incidence of MODS following injury can escalate to 56%, placing an intensive and disproportionate strain on healthcare resources. 2 Despite significant advancements in trauma care, severely injured individuals may still succumb early, even with sufficient blood volume and signs of physiological recovery. 3 There is an urgent need for the innovation of novel therapeutic strategies aimed at combating MODS.
Hemodynamic instability in polytrauma patients, compounded by hemorrhagic shock, results in heightened permeability of the gut-blood barrier. 4 It is widely recognized that intestinal barrier dysfunction post-trauma serves as a ‘second hit’ that exacerbates remote organ damage, thereby sparking substantial interest among researchers to develop preventive and therapeutic strategies against MODS. 5 The primary drivers of intestinal barrier compromise leading to MODS are known to include disruptions in the gut microbiota, apoptosis of intestinal epithelial cells, increased intestinal epithelial permeability, and gut-lymph pathway-mediated distant organ injury. 6 Preliminary research from various groups has suggested that interventions targeting the integrity of the gut barrier may help alleviate subsequent organ damage.7,8 Nevertheless, these approaches have yet to achieve a sufficient level of success for robust translation into clinical practice with a high degree of evidence.
Recent studies have shown that the well-known cardiac hormone, atrial natriuretic peptide (ANP) with a half-life of 2-4 min, can protect against multi-organ injury and function as an immune modulator. 9 It does so through signaling pathways mediated by the natriuretic peptide receptor (NPR). ANP and its receptors are also found in non-cardiac organs such as the kidneys, articular cartilage, spiral ganglion neurons, and intestines, among others.10–12 In our laboratory, ANP has proven to be protective in cases of traumatic hemorrhagic shock (THS). 13 Our previous research revealed that the expression of endogenous serum ANP in rats exposed to THS significantly increased within 30 min post-trauma and then returned to normal levels after 24 h. 14 In contrast, a one-hour intravenous infusion of recombinant ANP at a dose of 0.025 μg/kg/min during hemorrhagic shock led to a sustained high level of circulating ANP. Furthermore, this treatment improved liver and kidney function abnormalities following THS. It also inhibited myocardial apoptosis, while reactive oxygen species (ROS) levels in multiple organs were significantly enhanced. 14
The discovery that a short-term infusion of recombinant ANP triggers sustained expression of endogenous ANP led us to investigate the potential positive feedback regulation mechanisms that protect the gut barrier. 15 Reactive oxygen species (ROS) may serve as a key biomarker within this feedback loop, as their activity was increased in multiple organs. Contrary to inducing oxidative stress, ROS produced by non-phagocytic cell oxidases are known to mediate crucial signaling pathways for physiological functions. 16 It is established that bradykinin can stimulate ROS production in renal tubular cells following ATP depletion via the phospholipase C (PLC)-calcium signaling pathway. 17 Additionally, H2O2 treatment has been shown to enhance the phosphorylation of PLC-γ1. 18 This PLC-γ1/ROS feedback loop has been validated in primary human CD4 + T cells. 19 Notably, ROS can facilitate the translocation of nuclear factor-kappa B (NF-κB) to the nucleus and promote ANP gene expression through the p38 MAPK pathway. 20 Despite these insights, it remains unclear whether this positive feedback loop regulates ANP expression. Therefore, this study aimed to investigate the role of the PLC-γ1/ROS feedback loop in the regulation of ANP expression and its association with gut barrier protection.
Materials and methods
Animals and cells
This study was approved by the ethics committee of the Second Affiliated Hospital of Zhejiang University School of Medicine. We acquired 72 male Sprague-Dawley (SD) rats, 8-12 weeks old and weighing 250-300 g, from Shanghai SIPPR-BK LAB Animal Ltd. The animals were cared for according to the US National Research Council’s 1996 Guidelines for the Care and Use of Laboratory Animals. They were kept in a 22 °C room with a 12-h light/dark cycle and fasted for 12 h with water available before experimentation. Rat small intestinal crypt epithelial cells (IECs) were sourced from iCell Bioscience Inc, Shanghai, China, and maintained in DMEM medium supplemented with 10% fetal bovine serum, 0.01 mg/mL insulin, and 1% penicillin-streptomycin at 37 °C in a 5% CO2 humidified incubator.
Animal THS and cell hypoxia/re-oxygenation (HR) model
Rats were anesthetized intraperitoneally with 2% pentobarbital sodium at 50 mg/kg. THS was simulated by midline laparotomy and bleeding from the left common carotid artery based on a previous model. 21 Invasive arterial blood pressure was monitored on a polygraph (PM-9000; Shenzhen Mindray Bio-Medical Electronics Co, Ltd, Shenzhen, China). Mean arterial pressure was reduced to 40 mmHg within 30 min, maintained for 60 min, followed by resuscitation with a 1:2 mixture of pre-stored blood/normal saline intravenously for 1 h. The rats were euthanized 24 h later with an overdose of pentobarbital sodium intravenously (100 mg/kg). Death was confirmed by no electrical activity and blood pressure readings on monitor. Similar to a previous model, 22 to simulate cell H/R injury, the cell culture medium was replaced with 6 mmol/L of Na2S2O4 for 2 h. Following induction of hypoxia, IECs were re-oxygenated for 1 h.
Study of ideal concentrations of recombinant ANP in rats
To determine the optimal recombinant ANP concentrations for further study, 40 rats were randomly divided into five groups (n = 8): sham, control, and groups A, B, and C. Groups A, B, and C received recombinant ANP (1912, Tocris Bioscience, Ellisville, Missouri) infusions at 0.0125, 0.025, 0.05 μg/kg/min respectively via a micro-infusion pump during hemorrhagic shock. After 24 h, blood samples and jejunal tissues were collected for analysis of serum ANP levels, intestinal reactive oxygen species (ROS), tight junction proteins ZO-1 and Claudin-1, and jejunal apoptosis.
Effect of inhibition of NAPDH oxidase (NOX) and p38 MAPK on ROS and NPR expression in rats treated with ANP following THS
To assess the impact of NOX and p38 MAPK pathway inhibition on ROS production and NPR family expression, 32 rats were randomly divided into one of four groups (n = 8): sham, control, ANP/NOXs inhibitor, and ANP/p38 MAPK inhibitor. The ANP/NOXs inhibitor group received 1 mg/kg of diphenyleneiodonium chloride (DPI, NOXs inhibitor, HY-100965, MedChemExpress, Monmouth Junction, NJ, USA) intraperitoneal during hemorrhagic shock. The ANP/p38 MAPK inhibitor group received 1 mg/kg of SB203580 (MedChemExpress, p38 MAPK inhibitor, Monmouth Junction, NJ, USA) intraperitoneally during hemorrhagic shock. After 24 h, jejunal tissues were harvested to measure ROS and expressions of NF-κB, NPR-A, and NPR-C.
Regulation of PLC-γ1 on ROS production in ANP and hypoxia/re-oxygenation-treated IECs
We modulated PLC-γ1 expression to explore its effect on ROS production. On the one hand, IECs were incubated with PI3K inhibitor LY294002 at 20 μM (8250, ApexBio, Houston, TX, USA) or PLC inhibitor U73122 (28922, MedChemExpress, Monmouth Junction, NJ, USA) at 0.5 × 10−6 (defined as U1), 1.0 × 10−6 (U2), 1.5 × 10−6 (U3) and 2.0 × 10−6 mmol/L (U4) respectively during cell hypoxia in along with ANP at 50 mol/L. On the other hand, PLC-γ1 cDNA was amplified with the following primers: F: 5′- TCTACTCCAAGAAGTCGCAGC-3′ and R: 5′- CCTTCGTTCCCGACTGTCAA-3’. The vector pcDNA3.1/CT-GFP TOPO (Invitrogen) was used to optimize the transfection of pRP-EGFP plasmids (2 μg/mL) into IECs. The pRP-EGFP plasmid to target PLC-γ1 was constructed which was confirmed by DNA sequencing. Transfection was performed using Lipofectamine 2000 reagent (Invitrogen, Carlsbad, CA) according to the manufacturer’s instructions. After 48 h of transfection (at 37°C), cells were used for testing ROS production.
Enzyme-linked immunosorbent assay (ELISA)
Serum and intestinal ANP and ROS were measured using the ELISA method. Briefly, whole blood was obtained from the heart at 24 h. After standing for 30 min undering 4°C, the whole blood samples were centrifuged at 3000 rpm (4°C) for 10 min. Similarly, the small intestinal tissues were harvested and homogenized in sterile PBS. The homogenates were centrifuged at 3000 rpm (4°C) for 20 min. All the supernatants were measured via corresponding ELISA kit (ANP, MM-21097R2; ROS, MM-21264R2) that were purchased from MeiMian Biotechnology (Yangcheng, Jiangsu, China) on a microplate reader (CMax Plus, Molecular Devices). The detailed experimental procedures were following the manufacturer’s instructions.
Western blot analysis
Intestinal tissues and IECs were lysed with RIPA buffer and were centrifuged at 12,000 g at 4°C for 5 min. Bicinchoninic acid assays were used for detection of protein concentration. GAPDH was used as the loading control. The resolved proteins (20 μg per lane) were separated by 10% sodium dodecyl sulfate-polyacrylamide gel electrophoresis and then transferred to polyvinylidene difluoride membrane. After being blocked with 5% nonfat milk in phosphate buffered saline for 1.5 h at room temperature, membranes were incubated with diluted primary antibodies such as anti-Bcl-2 (1:1000; ab196495; Abcam), anti-Bax (1:1000; 14796S; CST), anti-NF-κB (1:1000; 8242S; CST), anti-PLC-γ1 (1:1000; 5690S; CST), anti-ZO-1 (1:1000; sc-33725; Santa Cruz), anti-Claudin-1 (1:500; ab15098; Abcam), anti-NPR-A (1:500; ab14356; Abcam), and anti-NPR-C (1:500; ab14355; Abcam) at 4°C overnight. Blots were washed in tris-buffered saline with 0.1% Tween twice over 30 min, and then were incubated with horseradish peroxidase (HRP) conjugated goat-anti-rat immunoglobulins (1:1000, 7076, Cell Signaling Technology) or goat-anti-rabbit immunoglobulins (1:1000, 7074, Cell Signaling Technology) for 1 h at room temperature. Membrane images were gained using ChemiDocTM MP imaging system (Bio-Rad Laboratories, Hercules, CA). The density of each band was quantified using Quantity One software (Bio-Rad Laboratories, Hercules, CA).
Immunohistochemical assay
Small intestinal tissues were fixed with 10% neutral formalin overnight at room temperature and processed for paraffin embedding. All samples were sectioned at 5 mm of thickness. Sections were deparaffinized and then rehydrated through a graded series of ethyl alcohol. The sections were then blocked with 5% normal goat serum (MD911695, MDL, Beijing, China) at room temperature for 30 min and incubated with ZO-1 (GB11195, 1:50 dilution, Servicebio, Wuhan, China) or Claudin-1 (GB11032, 1:50 dilution, Servicebio, Wuhan, China) antibodies at 4C overnight. The next morning, the tissues were incubated for 1 h at room temperature with an HRP-conjugated secondary antibody (1780935A, Invitrogen, Carlsbad, CA). Hematoxylin was used for counterstaining (room temperature, 5-10 min). Image Pro-Plus (version 6.0) was used to analyze average integrated optical density (IOD) of positive expression (brown stain) for all sections (six random visual fields for each section under microscope) (DM3000, Leica, Wetzlar, Germany).
Quantitative real-time PCR
Total RNA was extracted using the TRIzol reagent (B511311, Sangon Biotech, Shanghai, China) according to the manufacturer’s protocol. Complementary DNA (cDNA) was synthesized using HiFiScript cDNA Synthesis Kit (CW2569 M, Cwbio, Beijing, China) according to the manufacturer’s instrument. The following specific primers were used in the study:Bcl-2 forward 5′- CCGGGAGAACAGGGTATGATAA-3′ and reverse 5′-CCCACTCGTAGCCCCTCTG-3′, Bax forward 5′-AAACTGGTGCTCAAGGCCCT-3′ and reverse 5′-AGCAGCCGCTCACGGAG-3′, NF-κB forward 5′-TGGCAGACGACGATCCTTTC-3′ and reverse 5′-GGTATGGGCCATCTGTTGAC -3, NPR-A forward 5′-ACTCCTGGGGCAAGCG-3′ and reverse 5- AAATTGGGAGCGTCCGAGAG-3′, NPR-C forward 5′-GCTCTCCTTGCAAATCATGTGG-3′ and reverse 5′-TGATGGAATCTTCCCGCAGC-3′, GAPDH forward 5′-TGCCACTCAGAAGACTGTGG-3′ and reverse 5′- TTCAGCTCTGGGATGACCTT-3’.
A quantitative real-time polymerase chain reaction (PCR) was performed using the UltraSYBR Mixture (CW2601, Cwbio, Beijing, China) through LightCycler® 96 instrument (Roche, Basel, Switzerland) in accordance with the manufacturer’s protocol. PCR conditions were started at one step initial denaturation (95°C for 10 min), followed by 40 cycles (95°C for 10 s, and 60°C for 60 s). Gene expression was normalized to GAPDH. The relative fold change for each gene was calculated using the 2
Flow cytometry analysis of apoptosis
Detection of IECs apoptosis was performed using the Annexin V-FITC/PI Apoptosis Detection Kit (CW2574, Cwbio, Beijing, China). IECs were cultured in 6-well plate at a concentration of 1106 cells/mL, and then were harvested, washed twice with PBS. A 100 μL binding buffer containing 10 μL propidium iodide (PI) and 5 μL Annexin V-FITC was utilized to incubate and resuspend the cell pellets for 15 min (at room temperature). Apoptosis of IECs was tested using Accuri™ C6 Flow cytometer (BD). Each test was carried out in triplicate.
Statistical analysis
Data are presented as mean ± standard deviation unless otherwise stated. Comparisons of results between two independent groups were analyzed by t test. Differences among multiple groups were analyzed by one-way analysis of variance followed by post hoc Tukey’s test. Statistical analyses were performed through IBM SPSS Statistics (Version 19.0). A value of p < .05 was considered statistically significant.
Results
Effects of exogenous ANP on ROS production, tight junction protein expression, and IECs apoptosis in rats after THS and H/R-treated IECs
In animal study, serum and intestinal ANP levels, along with intestinal ROS production in the control group, were significantly higher than those in the sham group (all p < .01) (Figure 1(a) and (c)). Administration of exogenous ANP at 0.025 or 0.05 μg/kg/min resulted in notably increased serum and intestinal ANP and intestinal ROS production at 24 h post-injection compared to the control group (all p < .01). Furthermore, exogenous ANP at these dosages significantly upregulated the mRNA expression of Bcl-2 and downregulated that of Bax in the intestinal tissues (Bcl-2: p < .01; Bax: p < .05, compared to the control group) (Figure 1(d) and (e)). Protein expression analyses through Western blot revealed increased levels of Bcl-2 and decreased levels of Bax in groups B and C when compared to the control group; however, there was no significant difference between groups B and C (Figure 1(g) and (h)). ANP treatment markedly elevated ZO-1 and claudin-1 protein expressions in groups B and C compared to the control group (both p < .01) but not in group A (Figure 1(f), (i), and (j)). Nonetheless, the ANP and ROS levels and the ZO-1 and claudin-1 protein expressions in groups B and C did not differ significantly (all p > .05). Immunohistochemical analyses showed that ZO-1 and claudin-1 protein expressions in the control group were significantly lower than in the sham group (both p < .01) (Figure 2(a), (b), (d), (e)). In the ANP-treated group (0.025 μg/kg/min), the expressions of these proteins were significantly higher than in the control group (both p < .01) (Figure 2(c), (f)). Effects of exogenous ANP on ROS production, expression of tight junction protein, and apoptosis in THS rats. Panel A and B, testing serum and intestinal ANP level through ELISA in various groups. Panel C, intestinal ROS production with or without ANP treatment. Panel D, E, G, and H, effect of exogenous ANP on mRNA and protein expression of Bcl-2 and Bax. Panel F and I, Effect of exogenous ANP on expression of intestinal tight junction protein ZO-1 and claudin-1. Panel J represents semiquantitative comparisons of band intensities (normalized to GAPDH) which correspond to blots in panel F, G, H, and I derived from western blot experiments. S, sham group; c, control group; A, group A (ANP at 0.0125 g/kg/min); B, group B (ANP at 0.025 g/kg/min); C, group C (ANP at 0.05 g/kg/min); THS, traumatic hemorrhagic shock; ANP, atrial natriuretic peptide; ROS, reactive oxygen species. #p < .01 versus sham group, ##p < .05 versus sham group, *p < .01 versus control group, **p < .05 versus control group. Immunohistochemical locations of expression of gut tight junction protein in THS rats (original magnification, 200). Panel A-C, ZO-1 expression in sham group (A), control group (B), and ANP group (ANP at 0.025 g/kg/min) (C). Panel D-F, claudin-1 expression in sham group (D), control group (E), and ANP group (ANP at 0.025 μg/kg/min) (F). IOD, integrated optical density. *p < .01 versus sham group, #p < .01 versus control group.

Effects of U73122 and PLC-γ1 overexpression on ANP expression and ROS production in hypoxia/reoxygenation-treated IECs.

*p < .01 versus H/R group, #p < .05 versus H/R group, **p < .05 versus ANP + H/R + U4 group, ##p < .05 versus control group. U1, U73122 at 0.5 × 10−6. mmol/L; U2, U73122 at 1.0 × 10−6. mmol/L; U3, U73122 at 1.5 × 10−6. mmol/L; U4, U73122 at 2.0 × 10−6. mmol/L; ANP, atrial natriuretic peptide; ROS, reactive oxygen species; H/R, hypoxia/reoxygenation.

Flow cytometric analysis of the effects of regulation of PLC-γ1 on apoptosis of H/R-treated IECs. AR, apoptosis rate. Panel A, effects of ANP on apoptosis of H/R-treated IECs. Panel B, effects of PLC inhibitor U73122 on apoptosis of H/R-treated IECs. Panel C, effects of PLC-γ1 over-expression on apoptosis of H/R-treated IECs. *p < .01 versus control group, **p < .05 versus control group, #p < .01 versus H/R group, ##p < .05 versus ANP + H/R group. ANP, atrial natriuretic peptide; IECs, intestinal crypt epithelial cells; H/R, hypoxia/re-oxygenation; PLC: phospholipase C.
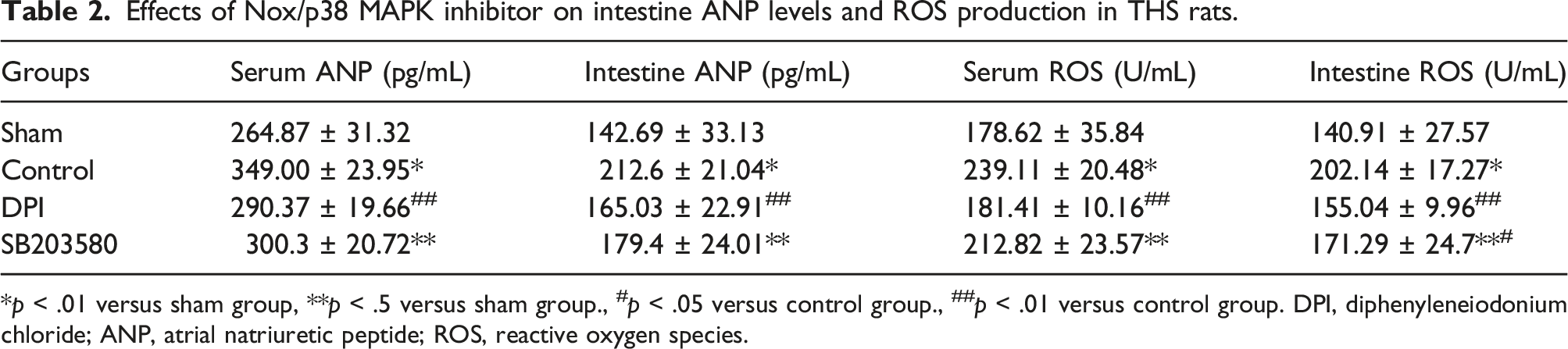
Effects of NOXs/p38 MAPK inhibitor on intestinal NF-κB and NPR-A/C expression, and ANP and ROS production in rats following THS
In THS rats, the mRNA and protein levels of NF-κB and NPR-C in the intestine were significantly higher compared to the sham group (Figure 4(a), (c)–(f)). Treatment with exogenous ANP and NOXs inhibitor or p38 MAPK inhibitor led to a significant reduction in mRNA and protein levels of NF-κB and NPR-C in THS rats. However, mRNA and protein levels of NPR-A were not significantly affected (p > .05) (Figure 4(b)). Both NOXs inhibitor and p38 MAPK inhibitor markedly decreased intestinal ANP levels and ROS production in comparison to the control group (Table 2). Protein (Panel B, D, and F) and mRNA expression (Panel A and E) of NF-κB and NPR-A/C in intestine following intraperitoneal injection of NOXs/p38 MAPK inhibitor in THS rats. Panel B, D, and F are semiquantitative comparisons of band intensities (normalized to GAPDH) which correspond to blots in panel C derived from western blot experiments. The expression of NPR-A mRNA was not integrated into this graph as between-group differences were not significantly different. *p < .01 versus sham group, **p < .05 versus sham group, #p < .01 versus control group. Effects of Nox/p38 MAPK inhibitor on intestine ANP levels and ROS production in THS rats. *p < .01 versus sham group, **p < .5 versus sham group., #p < .05 versus control group., ##p < .01 versus control group. DPI, diphenyleneiodonium chloride; ANP, atrial natriuretic peptide; ROS, reactive oxygen species.
Effect of U73122 on ANP expression, ROS production, and apoptosis in H/R-treated IECs
In IECs treated with exogenous ANP and H/R, U73122 at concentrations of 1.0, 1.5, and 2.0 × 10−6 mmol/L (but not 0.5 × 10−6 mmol/L) significantly reduced ROS production and ANP expression compared to treatment with ANP alone, as shown in Table 1 Variable set 1. Co-incubation of U73122 and ANP at 0.5/1.0 × 10−6 mmol/L (but not at 1.5/2.0 × 10−6 mmol/L) significantly increased Bcl-2 expression and decreased Bax levels in relative to the H/R-treated group (p < .01 and p < .05, respectively) (Figure 5(a), (b), and (e)). However, U73122 at 1.5/2.0 × 10−6 mmol/L (not at 0.5/1.0 × 10−6 mmol/L) significantly reducecd Bcl-2 expression and increased Bax expression compared to ANP-only treatment. Additionally, co-incubation with U73122 and ANP significantly increased the apoptosis rate of IECs in a dose-dependent manner relative to ANP-only treatment (Figure 3(b)). Effects of inhibition and over-expression of PLC-γ1 on Blc-2 and Bax expression in hypoxia/re-oxygenation-treated IECs. Panel A and B, semiquantitative comparison of band intensities (normalized to GAPDH) corresponding to the blots in E. Panel C and D, semiquantitative comparison of band intensities (normalized to GAPDH) corresponding to the blots in F. *p < .01 versus control group, **p < .05 versus control group, #p < .01 versus H/R group, ##p < .05 versus H/R group. U1, U73122 at 0.5 × 10−6 mmol/L; U2, U73122 at 1.0 × 10−6 mmol/L; U3, U73122 at 1.5 × 10−6 mmol/L; U4, U73122 at 2.0 × 10−6 mmol/L; ANP, atrial natriuretic peptide; H/R, hypoxia/re-oxygenation; PLC, phospholipase C.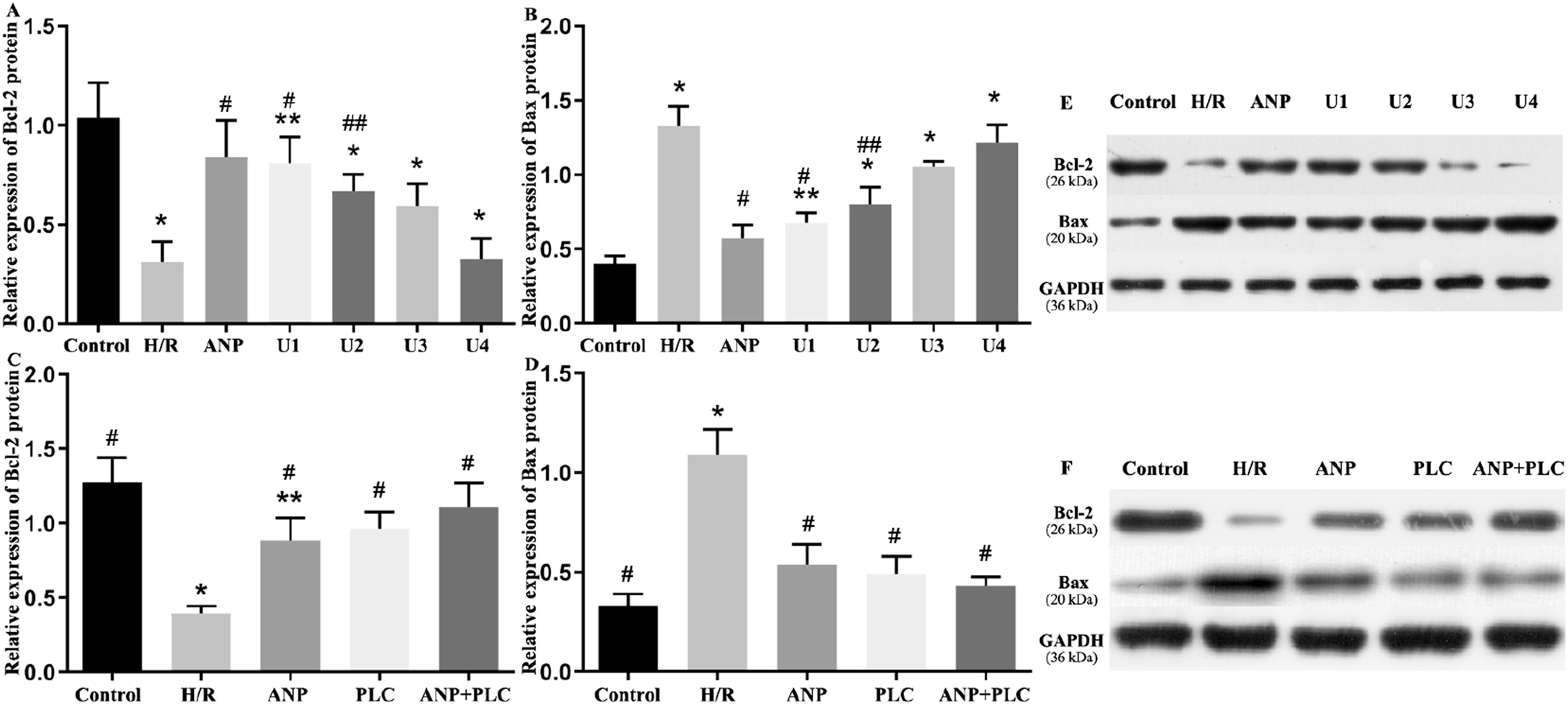
Effect of over-expression of PLC-γ1 on expression of ANP, ROS production, and apoptosis in H/R-treated IECs
In H/R-treated IECs, overexpression of PLC-γ1 did not affect ANP expression but did increase ROS production, which was attenuated by H/R treatment (p < .05). IECs with PLC-γ1 overexpression and concurrent ANP treatment did not exhibit a significant increase in ROS production compared to PLC-γ1 overexpression alone (p > .05) (Table 1 Variable set 2). The expression of Bcl-2 protein was significantly lower, and Bax protein expression was significantly higher in the H/R group compared to the control group (p < .01). ANP treatment or PLC-γ1 overexpression significantly upregulated Bcl-2 protein expression and downregulated Bax protein expression in H/R-treated IECs (both p < .01) (Figure 5(c), (d), and (f)). However, no significant differences were observed between the ANP treatment group and the PLC-γ1 overexpression group in terms of Bcl-2 and Bax protein expression. PLC-γ1 overexpression significantly decreased the apoptosis rate of H/R-treated IECs (p < .05 compared with the H/R modeling group) and appeared to further reduce apoptosis in IECs co-treated with ANP and H/R, although this reduction was not statistically significant. The apoptosis rate of IECs showed no significant difference between the ANP treatment group and the PLC-γ1 overexpression group (Figure 3(c)).
Effect of PI3K inhibitor on expression of PLC-γ1 protein in H/R-treated IECs
The expression of PLC-γ1 protein in H/R-treated IECs incubated with or without exogenous ANP was significantly up-regulated compared with sham group (both p < .01). PI3K inhibitor significantly reduced the expression of PLC-γ1 protein in H/R-treated IECs compared with those without PI3K inhibitor treatment (both p < .01). However, PI3K inhibitor group still show higher expression of PLC-γ1 protein than that of sham group (p < .5) (Figure 6). Effects of PI3K inhibitor (LY294002) on expression of PLC-γ1 protein in hypoxia/re-oxygenation-treated IECs. Panel A, semiquantitative comparison of band intensities (normalized to GAPDH) corresponding to the blots in Panel B. *p < .01 versus sham group, **p < .05 versus sham group, #p < .01 versus control or ANP group.
Discussion
Our research, based on both in vivo and in vitro studies, has shown for the first time that the serum expression of ANP is regulated by the PLC-γ1/ROS feedback loop following THS. This regulation contributes to maintaining gut tight junction integrity and reducing IEC apoptosis. Additionally, we found that PLC-γ1 overexpression in H/R-treated IECs led to decreased ROS production, suggesting that IECs alone may not effectively activate the PLC-γ1/ROS feedback loop.
ANP, a 28-amino-acid peptide, has long been recognized for its role in regulating fluid and electrolyte balance as well as the activity of the hypothalamic-pituitary-adrenal axis.24,25 Its effectiveness in protecting patients with heart failure or acute kidney injury has been supported by recent clinical trials.26,27 Recent studies, however, have highlighted ANP as an influential cytokine in modulating the immune system. 28 ANP enhances innate immunity by stimulating the activities of natural killer cells, promoting neutrophil migration, and increasing ROS release. It also suppresses the expression of TNF-α, nitric oxide, and cyclooxygenase-2. Additionally, ANP influences adaptive immunity by upregulating IL-10 and Th2/Th17 dendritic cells. 28 These properties are critically important for developing new trauma treatments.29–31
The safety of ANP during hemorrhagic shock after severe trauma was uncertain, given its blood pressure-lowering diuretic effect. Our recent preliminary study showed that administering ANP at a rate of 0.025 μg/kg/min did not affect arterial pressure during hemorrhagic shock, likely due to anuria from inadequate renal perfusion. In our current study, ANP treatment markedly elevated serum and intestinal ROS production, in parallel with increased expression of intestinal tight junction proteins and reduced IEC apoptosis in THS rats. This supports the concept that ROS predominantly acts as a signaling molecule regulating various physiological functions rather than as an oxidative stress mediator.15,32 The link between endogenously overexpressed ANP and these findings warrants further investigation.
ANP exerts its biological effects by binding to specific membrane receptors, such as NPR-A and NPR-C. Previously, NPR-C was thought to function mainly as a clearance receptor, inactivating ANP through endocytosis. However, recent research has demonstrated that ANP’s interaction with the NPR-C receptor lowers intracellular cAMP levels and stimulates PLC activity. 33 Crosstalk between PLC and ROS has been the subject of other studies. For instance, inhibiting phosphatidylcholine-specific PLC reduced ROS levels in vascular endothelial cells. 34 Licochalcone A has been shown to trigger apoptosis in HepG2 hepatocellular carcinoma cells by inducing endoplasmic reticulum stress through a PLC-γ1-calcium-ROS-dependent pathway. 35 Additionally, prolonged exposure to oxidized low-density lipoprotein-associated phospholipids can lead to platelet activation via a CD36- and PLC-dependent pathway mediated by NOX-2-derived ROS. 36 ROS itself can further stimulate PLC production. H2O2 activates PLC-γ1 dose-dependently, while U73122 completely inhibits H2O2-induced calcium oscillations in rat cortical astrocytes. 32
To investigate the involvement of ROS and NPR-A or NPR-C in the regulation of ANP’s endogenous expression, we treated rats with inhibitors targeting NPR-A/C and ROS during ANP administration. We observed that inhibiting ROS significantly decreased the mRNA and protein levels of NF-κB and NPR-C, as well as the intestinal ANP level. However, this had no effect on NPR-A expression. In contrast, blocking ANP production with a p38 MAPK inhibitor led to a reduction in intestinal ROS production. These findings suggest that ROS is critical for the endogenous overexpression of ANP, and that ANP-induced ROS production is dependent on NPR-C, not NPR-A.
The relationship between PLC and ROS in regulating ANP expression requires further elucidation. ROS promotes the translocation of NF-κB p65 to the nucleus, which in turn drives gene expression, including the ANP gene via the p38 MAPK pathway. 20 Additionally, ROS can activate PLC by stimulating phosphatidylinositol 3,4,5-trisphosphate (PIP3), which is produced by the phosphorylation of phosphatidylinositol 4,5-bisphosphate (PIP2) following PI3K activation.37,38 The activation of PLC-γ1 then further promotes ROS production through a calcium-dependent pathway. In this study, the inhibition of PLC-γ1 expression in intestinal epithelial cells (IECs) using U73122 resulted in a significant decrease in both ROS production and ANP expression. Similarly, suppressing the PI3K pathway with LY294002 in IECs also reduced PLC-γ1 expression. However, overexpression of PLC-γ1 did not significantly affect ANP expression or ROS production. This finding suggests that IECs alone are insufficient to initiate the ROS/ANP pathway without the involvement of immune cells.
The findings from the present study suggest that atrial natriuretic peptide (ANP) plays a protective role in maintaining intestinal barrier function following severe trauma. This is achieved through increasing the expression of tight junction proteins in the gut and inhibiting the apoptosis of intestinal epithelial cells (IECs), which are crucial for preserving the structural integrity and function of the gut barrier. Following severe trauma, the body’s stress response includes the centralization of blood flow to vital organs, which unfortunately results in splanchnic vasoconstriction. This reduced blood flow can lead to gut hypoxia and ischemia, compromising the integrity of the intestinal barrier. 29 The associated damage to IECs can manifest as a reduction in tight junction connections, cellular swelling, and an increase in cell death through apoptosis and necrosis. 39
The disruption of the gut barrier following trauma can have significant systemic consequences, as it can lead to increased permeability of the gut-blood barrier. This increased permeability can facilitate the translocation of bacteria and endotoxins from the gut lumen into the systemic circulation, contributing to the fluid shift from the intravascular space to the gut lumen. The result is exacerbated hypovolemia, which can further impair hemodynamic stability in patients with multiple traumas. 5 Clinical studies have shown that gut barrier dysfunction following trauma is associated with a higher incidence of infectious complications and organ failure. 40 Therefore, maintaining the integrity of the gut barrier is of paramount importance in the management of severely traumatized patients. The ability of ANP to enhance tight junction protein expression and reduce IEC apoptosis highlights its potential therapeutic benefits in the early management of severe trauma patients. By protecting the gut barrier, ANP could help to mitigate the development of multiple organ dysfunction and improve clinical outcomes. This reinforces the importance of further research into ANP and similar agents that could be used to bolster gut barrier function as part of a comprehensive approach to trauma care.
Nevertheless, there are several key issues left to be declared. On the one hand, the ideal duration of ANP therapy after severe THS requires to be elucidated. The current study administered ANP only during the shock state, and the long-term effects (24 h or longer) of ANP on gut barrier and remote organ function are unknown. It’s critical to determine the optimal duration of ANP treatment to maximize therapeutic benefits while minimizing potential side effects. On the other hand, caution is advised when extrapolating results from small animal models to humans. Differences in physiology, dosage effects, and treatment responses can vary significantly between species. Therefore, a large animal study is recommended to better assess the role of ANP in THS before proceeding to human clinical trials.
Our study has limitations. Firstly, we did not calculate the sample size in animal studies. A sample of 8 rats for each group may be insufficient. Secondly, elimination of NPR-A involvement in NPR-mediated pathway was not using the NPR-A inhibitor. As a result, whether exogenous ANP increased ROS production through NPR-A receptor is unknown, which needs to be defined further by introducing knock-down study. Thirdly, as we found in vitro study that PLC-γ1 over-expression in IECs did not influence the expression of ANP and ROS production, further research should base on co-culture of ICEs and immunocytes (i.e., macrophagocytes) to understand its actual effect. Lastly, we did not assess the quality of the standard samples included in the ELISA kits, which may be associated with potential biases concerning the relevant results.
Conclusions
In THS, ANP expression is regulated by the PLC-γ1/ROS feedback loop, which maintains gut tight junction integrity and reduces IEC apoptosis. The independence of this mechanism from the NPR-A-mediated pathway and the requirement for immune cell involvement warrant further investigation. More research, particularly in large animal models, is needed to evaluate the benefits of extended ANP treatment on gut barrier function and remote organ health following THS.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Natural Science Foundation of China (81701933).
Ethical statement
Animal welfare
Animals received humane care in accordance with the Guide for the Care and Use of Laboratory Animals: Eighth Edition (National Institutes of Health publication, 2011).

