Abstract
The distribution of P2X and P2Y receptor subtypes in upper lumbosacral cat dorsal root ganglia (DRG) has been investigated using immunohistochemistry. Intensity of immunoreactivity for six P2X receptors (P2X5 receptors were immuno-negative) and the three P2Y receptors examined in cat DRG was in the order of P2Y2 = P2Y4>P2X3>P2X2 = P2X7>P2X6>P2X1 = P2X4>P2Y1. P2X3, P2Y2, and P2Y4 receptor polyclonal antibodies stained 33.8%, 35.3%, and 47.6% of DRG neurons, respectively. Most P2Y2, P2X1, P2X3, P2X4, and P2X6 receptor staining was detected in small- and medium-diameter neurons. However, P2Y4, P2X2, and P2X7 staining was present in large- and small-diameter neurons. Double-labeling immunohistochemistry showed that 90.8%, 32.1%, and 2.4% of P2X3 receptor-positive neurons coexpressed IB4, CGRP, and NF200, respectively; whereas 67.4%, 41.3%, and 39.1% of P2Y4 receptor-positive neurons coexpressed IB4, CGRP, and NF200, respectively. A total of 18.8%, 16.6%, and 63.5% of P2Y2 receptor-positive neurons also stained for IB4, CGRP, and NF200, respectively. Only 30% of DRG neurons in cat were P2X3-immunoreactive compared with 90% in rat and in mouse. A further difference was the low expression of P2Y1 receptors in cat DRG neurons compared with more than 80% of the neurons in rat. Many small-diameter neurons were NF200-positive in cat, again differing from rat and mouse.
To date, seven distinct P2X subunits (P2X1–7) have been cloned (Chen et al. 1995; Collo et al. 1996; Khakh et al. 2000) and have been found to be expressed in primary sensory neurons (Xiang et al. 1998; Dunn et al. 2001; Ueno et al. 2002). In the rat and mouse, high levels of P2X3 receptor expression have been detected in a subpopulation of primary afferent neurons, including those of dorsal root ganglia (DRG), trigeminal ganglia, and nodose ganglia (Chen et al. 1995; Cook et al. 1997; Vulchanova et al. 1997, 1998; Bradbury et al. 1998; Zhong et al. 2001). In addition to P2X3, low levels of mRNA transcripts and proteins for P2X1,2, P2X4–6 have been identified in sensory neurons of the rat (Collo et al. 1996; Xiang et al. 1998).
So far, eight mammalian P2Y receptors—P2Y1, P2Y2, P2Y4, P2Y6, P2Y11, P2Y12 (Hollopeter et al. 2001; Zhang et al. 2001), P2Y13 (Communi et al. 2001), and the more recent P2Y14 receptor (Abbracchio et al. 2003)—have been cloned and shown to be activated by extracellular nucleotides. These receptors share between 25% and 55% identity (at the amino acid level) and can be distinguished by their specific pharmacological selectivities for both purine and pyrimidine nucleotides (King and Burnstock 2002). Among the eight known P2Y receptors, mRNA for P2Y1 and P2Y2 is expressed at high levels in rat DRG sensory neurons (Nakamura and Strittmatter 1996; Molliver et al. 2002). Low levels of mRNA for P2Y4 and P2Y6 have been reported in whole DRG of rat, but neither receptor has been localized to sensory neurons (Sanada et al. 2002). We have also shown recently that P2Y1, P2Y2, P2Y4, and P2Y6 receptor mRNA is expressed in rat sensory ganglia (DRG) and nodose and trigeminal ganglia (Ruan and Burnstock 2003). P2Y1 receptor polyclonal antibodies stained more than 80% of the sensory neurons, particularly the small-diameter (neurofilament-negative) neurons. In contrast, the P2Y4 receptor antibody stained more medium- and large-diameter (neurofilament-positive) neurons than small-diameter neurons. The P2Y1 receptor has been identified on adult rat primary sensory neurons, where it has been implicated in the transduction of mechanical stimuli (Nakamura and Strittmatter 1996) and modulation of the capsaicin receptor, TRPV1 (Tominaga et al. 2001). P2Y2 receptor activation initiates sustained action potential firing and induces phosphorylation of the transcription factor CREB in isolated sensory neurons that have neurochemical characteristics of nociceptors (Molliver et al. 2002). Furthermore, stimulation of P2Y receptors increases intracellular calcium in isolated sensory neurons and facilitates release of the neuropeptide calcitonin gene-related peptide (CGRP) in vitro (Sanada et al. 2002; Zimmermann et al. 2002). Together, these studies suggest that purines and pyrimidines have multiple actions on sensory neurons mediated by both P2X and P2Y receptors; the challenge remains to delineate which actions have functional importance under various physiological and pathological conditions.
The majority of studies of purinergic signaling in mammalian sensory neurons has been carried out in rat and mouse (see Dunn et al. 2001). However, there are no reports of the distribution of the P2X and P2Y receptor protein in DRG of cat, although many important physiological studies have been carried out in the cat sensory nerves (Thor et al. 1989a,b; de Groat 1999; Jänig and Häbler 1999). In previous studies, we have described a painful bladder condition in cats termed
The DRG comprises functionally heterogeneous cell types, and the size (diameter) of their cell bodies closely correlates with their functions; a subpopulation of small-size neurons transmit nociceptive information (Bessou et al. 1971), whereas large-size neurons have been identified as being involved in the transmission of proprioceptive and tactile impulses (Nakamura and Strittmatter 1996). To examine the potential roles of P2 receptor subunits in sensory functions, we determined the subtype expression patterns of the P2X and P2Y receptors in combination with cytochemical markers of sensory neuron populations. We chose to examine the expression of the P2X3, P2Y2, and P2Y4 receptors in relation to the neuropeptide CGRP, as well as binding of the isolectin B4 from
Materials and Methods
Animals and Tissue Preparation
All procedures were conducted in accordance with Institutional Animal Care and Use Committee policies at each institution [Ohio State University, University of Pittsburgh, and Home Office (UK) regulations covering regulated procedures]. Healthy, age-matched cats obtained from commercial suppliers were evaluated at Ohio State University and determined to be free from disease and signs referable to the lower urinary tract. All cats were housed in stainless steel cages at the Ohio State University animal facilities and allowed to acclimatize to their environment for at least 3 months before study.
Upper sacral (S1,S2) DRG were dissected from deeply anaesthetized (induction with 2% halothane and then maintained with α-chloralose 60–70 mg/kg) cats (
Antisera
The following primary antisera were used in the current studies: rabbit anti-P2Y1, -P2Y2, and -P2Y4 receptor antibodies (Alomone Labs; Jerusalem, Israel, 3 μg/ml). The immunogens used for production of polyclonal P2Y1, P2Y2, and P2Y4 antibody were synthetic peptides corresponding to the carboxyl terminal of the cloned rat P2Y1, P2Y2, and P2Y4 receptors, covalently linked to keyhole limpet hemocyanin. The peptide sequences of the P2Y1, P2Y2, and P2Y4 receptors are of amino acid sequence 242–258 (RALIYKDLDNSPLRRKS), 227–244 (KPAYGTTGLPRAKRKSVR), and 337–350 (HEESISRWADTHQD), respectively. Rabbit anti-P2X1-P2X7 receptor antibodies (3 μg/ml) were provided by Roche (Palo Alto, CA) (Xiang et al. 1998). Other antibodies used were: mouse anti-neurofilament 200 (NF200, clone N52; Sigma Chemical Co., Poole, UK, 1:400), mouse anti-CGRP (Affiniti Research Products Ltd., Exeter, UK; 1:2000). Biotin-conjugated isolectin B4 from Griffonia simplicifolia (IB4) was obtained from Sigma (10 μg/ml).
Immunostaining
An indirect immunofluorescence method was used to visualize receptor expression. Briefly, the sections (eight per animal) were washed in PBS (3 X 5 min) and nonspecific binding was blocked by preincubation were 10% normal horse serum in PBS containing 0.2% Triton X-100 for 30 min. The sections were incubated overnight with the primary antibodies (P2X1-P2X7, P2Y1, P2Y2, and P2Y4) diluted to 3 μg/ml with 10% normal horse serum in PBS containing 0.05% merthiolate and 0.2% Triton X-100. Subsequently, the sections were incubated with Cy3-conjugated donkey anti-rabbit IgG, diluted 1:300 in 1% normal horse serum in PBS containing 0.05% merthiolate for 1 hr at room temperature. Slides were mounted with Citifluor (Citifluor Ltd; London, UK) and examined with fluorescence microscopy. All control experiments were performed using an excess of the appropriate homolog peptide antigen to absorb the primary antibodies and thus confirm a specific immunoreaction or omission of either the primary or secondary antibody from the staining procedure.
The sections on which counts of P2X and P2Y receptor-positive neurons had been performed were marked and then counterstained with toluidine blue (2.5% in 0.1 M phosphate buffer for 2 min, followed by dehydration through increasing grades of alcohol, cleared in xylene, and cover slipped with DPX mounting medium). This enabled the total neuron numbers to be determined by counting all neurons in the marked sections under bright-field illumination. The number of immunopositive neurons in a marked section was counted and divided by the total number of neurons, to give a percentage. A mean ± SE mean from the sections was then calculated.
Immunofluorescence Double Labeling
To demonstrate colocalization of the P2X3, P2Y2, and P2Y4 receptors with a high molecular-weight neurofilament marker (NF200) or CGRP, sections were incubated with P2X3, P2Y2, or P2Y4 antibody overnight and detected with Cy3-conjugated donkey anti-rabbit IgG, then incubated with monoclonal NF200 antibody (1:400; overnight) or monoclonal CGRP antibody (1:2000; overnight) and FITC-conjugated mouse antibody (raised in goat) (Sigma; 1:200, 1 hr). For colocalization with the IB4 sections were immunostained for the P2X3, P2Y2, and P2Y4 receptor as mentioned previously and then incubated with biotin-conjugated IB4 (10 μg/ml, 16 hr), and streptavidin-FITC (1:200; 1 hr). The sections were washed and mounted in Citifluor.
Photomicroscopy
Images of immunofluorescence labeling were taken with a Leica DC 200 digital camera (Leica; Heerbrugg, Switzerland) using the automatic exposure setting, attached to a Zeiss Axioplan microscope (Zeiss; Oberkochen, Germany), fitted with the following filters: Cy3, 510–550 nm excitation, 590 nm; FITC, 470 nm excitation, and 525 nm emission. Images were imported into a graphics package (Adobe Photoshop 5.0; San Jose, CA), where images were not manipulated with the exception of producing multiple images per figure, the addition of text and labeling, and for the double-labeling studies in which the individual images of the single receptors stained green or red are merged to reveal areas of colocalization in yellow/orange.
Analysis
All analyses were performed at X20 objective magnification. P2X and P2Y receptor expression in ganglia was determined by counting all P2X and P2Y receptor-positive neurons in every sixth section throughout the ganglia (per animal). A total of three individual observers independently judged the slides for positive/negative immunoreactivity. The number of neurons (only counted if a nucleus and cytoplasm was present) immunopositive for each P2 receptor subtype was counted for each section and this number was divided by the total number of neurons per section to give a percentage number of cells immunopositive. The mean percentages ± SE mean were calculated for each P2 receptor subtype.
To calculate percentages of P2X3, P2Y2, and P2Y4 receptor colocalization with cytochemical markers, four randomly selected ganglia sections were chosen for each pair of markers for each animal. For each section, counts were made of the number of positive neurons for P2X3, P2Y2, or P2Y4 receptors, the number of positive neurons for the other marker (NF200, IB4, or CGRP) and the number of neurons expressing both antigens and percentages were calculated (±SE mean).
Results
P2X and P2Y Receptor-staining in DRG of Cat
The polyclonal antibodies for the seven P2X and three P2Y receptor subtypes labeled different subpopulations of neurons (with the exception of P2X5) (Figure 1) and the number of neurons positive for the different P2 receptor subtypes expressed as a percentage of the total number on neurons counted (±SE mean) are given in Table 1. In control experiments, no signal was observed when the preimmune sera were used. All P2X receptor antibodies, except that for the P2X5 subtype, labeled neurons of cat DRG. P2X3 polyclonal antibodies moderately stained 33.8% of the neurons (Table 1; Figure 1C). The expression of P2X2 and P2X7 receptors (Figures 1B and 1G) in DRG was lower than that of P2X3 receptors, but higher than that of P2X1, P2X4, or P2X6 receptors (Table 1; Figures 1A, 1D, and 1F). Most P2X1, P2X3, P2X4, and P2X6 receptor staining was detected in small- and medium-diameter neurons. However, P2X2 and P2X7 polyclonal antibodies stained not only small-diameter neurons, but also large-diameter neurons. No immunoreactivity was observed for P2X5 receptors (Figure 1E). The immunoreactivity intensity of the six P2X receptors in cat DRG was in the order of P2X3>P2X2 = P2X7>P2X6> P2X1 = P2X4.
Neurons in DRG also expressed P2Y2 and P2Y4 receptor subtypes (Table 1; Figure 1). P2Y2 and P2Y4 receptor antibodies strongly stained 35.3% and 47.6% of the neurons, respectively. The staining was evenly distributed throughout the cytoplasm of these cells and positively stained cells were randomly distributed throughout the ganglia. P2Y2 receptor staining was localized to the small-diameter neurons (Figure 1I). P2Y4 antibodies stained not only small-diameter neurons, but also some large-diameter neurons (Figures 1J and 1K). The expression of P2Y1 receptors was quite low in cat DRG, but did label glial cells of the DRG (Figure 1H).
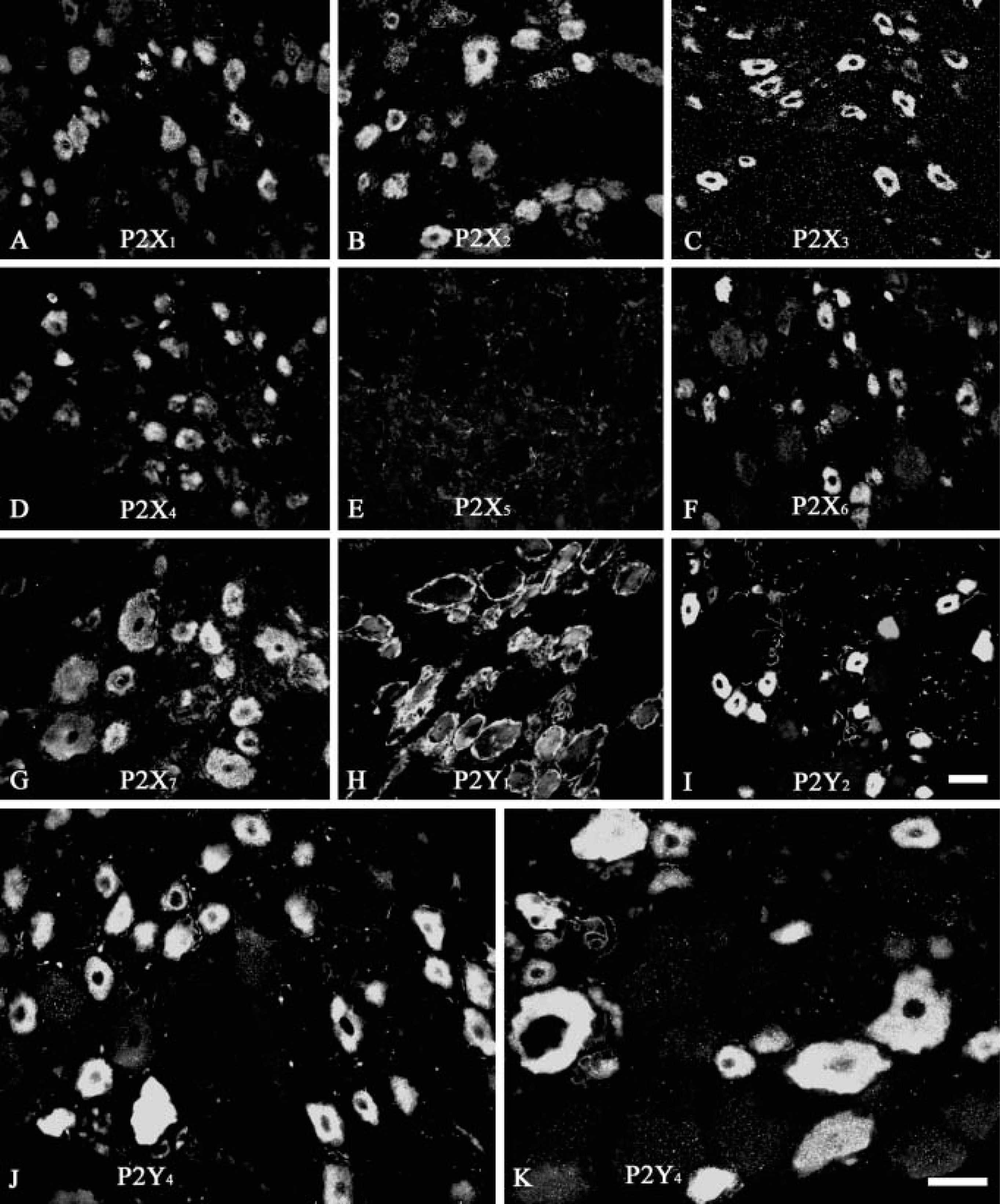
Localization of P2X and P2Y receptor immunoreactivity (IR) in cat dorsal root ganglia (DRG).
Expression of P2X1–7 and P2Y1,2,4 receptor immunoreactivity in cat dorsal root ganglion (DRG)
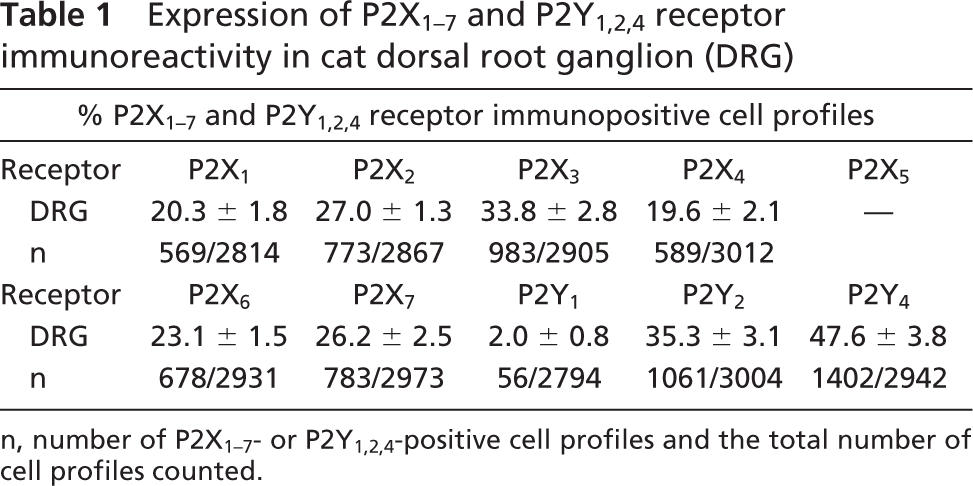
n, number of P2X1–7- or P2Y1,2,4-positive cell profiles and the total number of cell profiles counted.
Coexpression of P2X3, P2Y2, and P2Y4 Receptors and Other Neuronal Markers
Discussion
In this study, we have used immunohistochemistry to study the expression of P2X and P2Y receptors in cat DRG. The results showed that neurons in the DRG express a variety of P2X and P2Y receptor subtypes. Differences in expression level of each receptor subtype exist in different subpopulations of the neurons, which suggests that the diversity of receptor subtypes may be associated with different physiological functions of different neurons.
P2X receptors are ligand-gated ion channels activated by extracellular ATP that mediate rapid cation permeability and fast excitatory neurotransmission in both the central and peripheral nervous systems (reviewed by Ralevic and Burnstock 1998). DRG of rat and mouse are well studied in regard to the function of P2X receptors (see Dunn et al. 2000). ATP depolarizes DRG neurons by eliciting fast- and slow-inactivating inward currents. The fast-inactivating ATP currents are mediated by homomeric P2X3 receptors; the slow-desensitizing currents are mediated by heteromeric P2X2/3 receptors (Bradbury et al. 1998; Burgard et al. 1999). After the cloning of P2X3 receptors from DRG (Chen et al. 1995; Lewis et al. 1995), more attention has been paid to these ganglia because of the possible involvement of P2X receptors in nociception (Burnstock and Wood 1996; Burnstock 2001). We found that ≃33% of cat DRG neurons were P2X3-immunoreactive with moderate intensity staining. The P2X3-immunoreactive neurons were almost exclusively small to medium diameter in size. In a study of P2X3-receptor staining of the rat DRG, 90% of the total number of neurons were found to be immunopositive for P2X3 receptors to a greater or lesser degree, although the small- and medium-diameter neurons were the most intensely stained (Xiang et al. 1998). The percentage of cells that this population represented was not mentioned. In a separate study of P2X3 receptor immunoreactivity of the rat DGR, a similar percentage of neurons were also stained for P2X3 receptors (Bradbury et al. 1998). In addition to P2X3 receptors, mRNA transcripts and proteins for P2X1, P2X2, P2X4, P2X5, P2X6, and P2X7 receptors have also been detected in rat DRG (Xiang et al. 1998; Ueno et al. 2002). In our present study of cat DRG, immunoreactivity of P2X1–7 receptor subtypes, except the P2X5 receptor, was shown to be present. The lack of immunoreactivity to P2X5-receptor antibodies might reflect that this antibody does not recognize cat proteins. However, this antibody has been successfully used to visualize P2X5 receptors in the cat bladder urothelium (Birder et al. 2004), suggesting that the lack of staining for P2X5 receptors in the DRG is the result of an absence of receptors.
Colocalization of P2X3, or P2Y2 or P2Y4 receptor immunoreactivity with IB4, or CGRP, or NF200 in cat dorsal root ganglion (DRG)
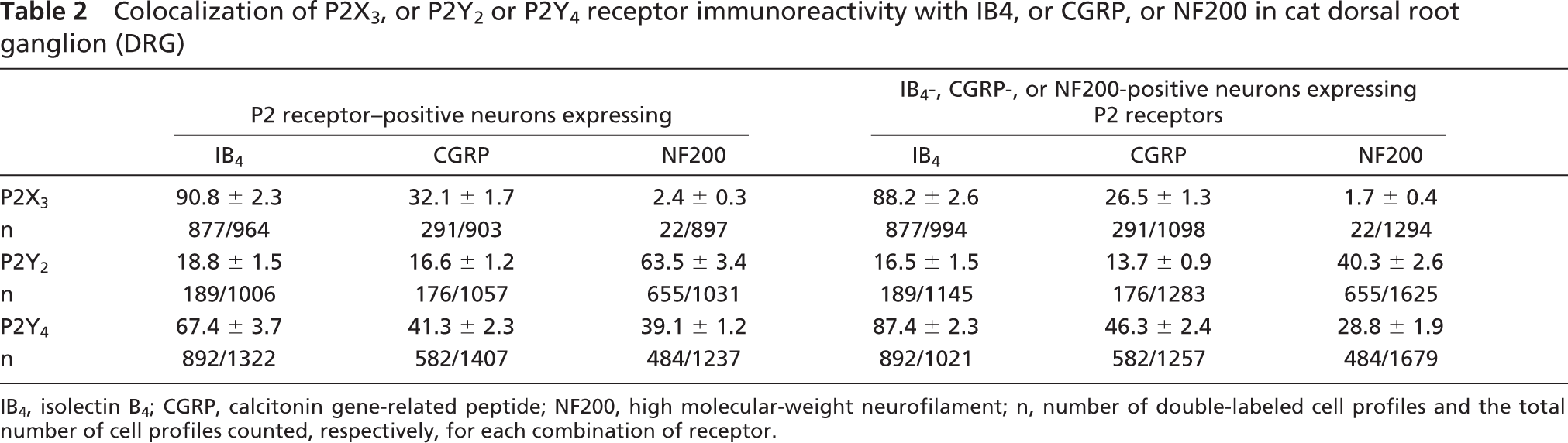
IB4, isolectin B4; CGRP, calcitonin gene-related peptide; NF200, high molecular-weight neurofilament; n, number of double-labeled cell profiles and the total number of cell profiles counted, respectively, for each combination of receptor.
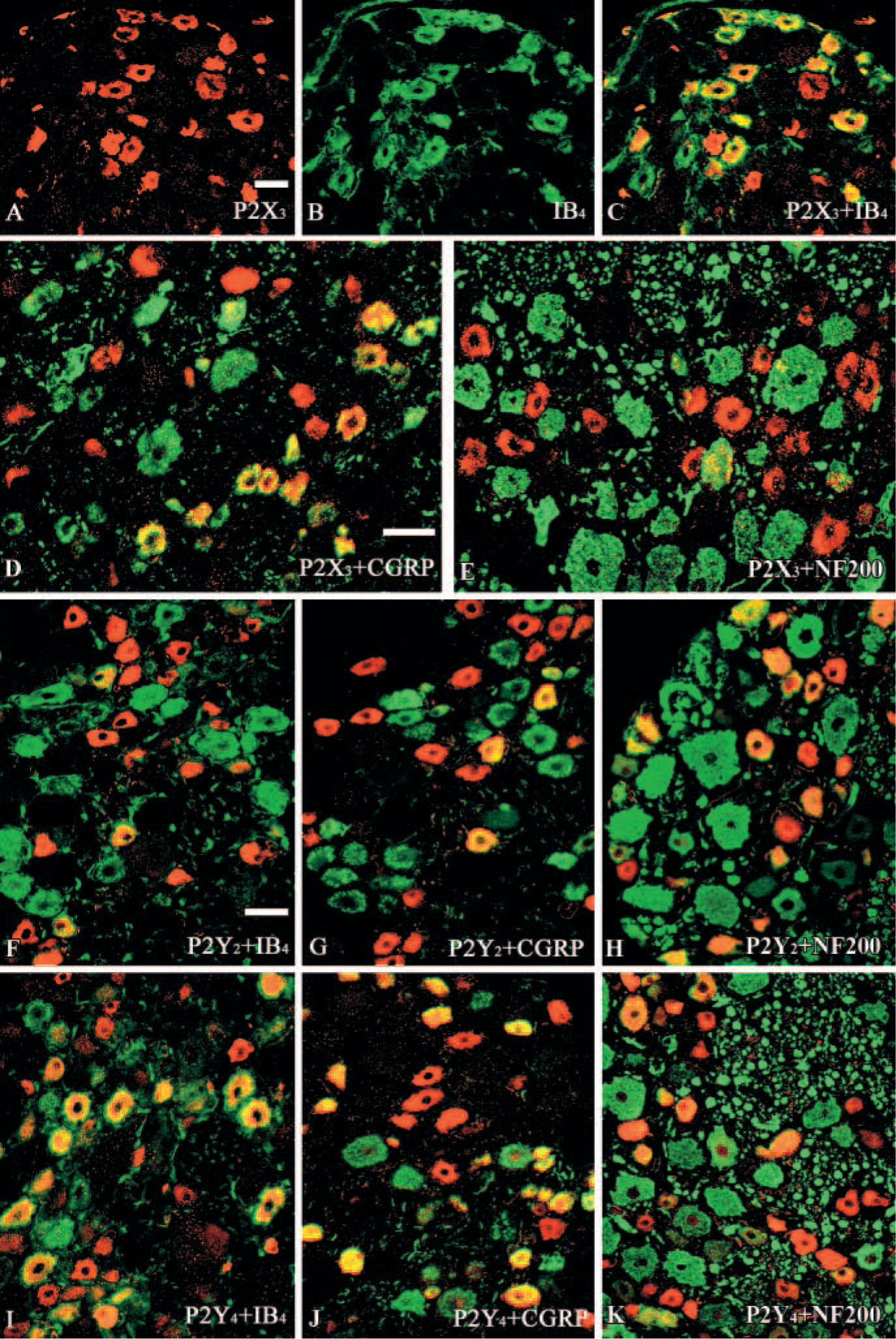
Colocalization (yellow/orange) of P2X3, or P2Y2 or P2Y4 receptor immunoreactivity (IR) (red) with IB4, or CGRP or NF200 (green) in the cat dorsal root ganglia (DRG).
ATP, in addition to other purines and pyrimidines, also evokes responses in nociceptors through P2Y metabotropic receptors. The P2Y receptors couple through G proteins to various second messenger pathways mediating slower metabotropic responses. Among the eight known P2Y receptors, mRNA for P2Y1 and P2Y2 are expressed at high levels in rat DRG neurons (Nakamura and Strittmatter 1996; Molliver et al. 2002). Low levels of mRNA for P2Y4 and P2Y6 have been reported in whole DRG of rat, but neither receptor has been localized to sensory neurons (Sanada et al. 2002).
We have also shown recently that P2Y1, P2Y2, P2Y4, and P2Y6 receptor mRNA is expressed in rat sensory ganglia (DRG, nodose ganglia, and trigeminal ganglia; Ruan and Burnstock 2003). P2Y1 receptor polyclonal antibodies stained more than 80% of the sensory neurons, particularly the small-diameter (neurofilament-negative) neurons. The P2Y4 receptor antibody stained more medium- and large-diameter (neurofilament-positive) neurons than small-diameter neurons.
In the present study, P2Y2 and P2Y4 receptor polyclonal antibodies stained ≃35% and 47% of the neurons, respectively, and labeled the neurons very strongly. However, the expression of P2Y1 receptors was quite low in cat DRG neurons, which differs from the results obtained in rat DRG using in situ hybridization and RT-PCR (Nakamura and Strittmatter 1996; Molliver et al. 2002; Sanada et al. 2002) or immunohistochemistry (Ruan and Burnstock, 2003).
To examine the potential roles of P2 receptor subtypes in sensory function, we determined the expression pattern of subtypes in combination with cytochemical markers of sensory neuron populations. We chose to examine the expression of the P2X3, P2Y2, and P2Y4 receptors in relation to CGRP, IB4, and NF200.
In the rat, monkey, and human, information about tissue damage is normally transmitted from the periphery to the spinal cord via the classically defined “small dark cells” of the DRG, which comprise 70% of all lumbar DRG neurons (Snider and McMahon 1998). These cells predominantly have unmyelinated C-fiber axons. Electrophysiological studies have shown that most of these cells (≃90%) are nociceptive in function (Snider and McMahon 1998). These small cells can be further subdivided into two minimally overlapping groups based on anatomical and functional properties. About one-half of the C-fiber population does not express NGF receptors (McMahon et al. 1994; Averill et al. 1995), but can be identified by a number of markers: they bind the lectin IB4, express the enzyme TMP, and stain with the antibody LA4 (Bradbury et al. 1998; Petruska et al. 2000). They also express the heat-responsive vanilloid receptor, TRPV1 (Caterina et al. 1997), which implicates them in thermal nociception. The central terminals of these neurons project to lamina IIi of the superficial dorsal horn of the spinal cord, a region where many fine afferents terminate. In vivo studies demonstrated that IB4 binding sites are selectively expressed by nociceptive afferents (Gerke and Plenderleith 2001). Physiological studies demonstrated that some IB4-binding DRG neurons innervate visceral organs (Yoshimura et al. 2003) and therefore may be high-threshold mechanoreceptors or polymodal nociceptors.
The other population of C-fiber afferent neurons expresses the NGF receptors trkA and p75. Virtually this entire group contains the neuropeptide CGRP, and other neuropeptides, such as substance P, are also found in this population. The central terminals of these neurons project to lamina I/IIo of the superficial dorsal horn of the spinal cord, a region where many fine afferents terminate.
The large cells within the cat sensory ganglia were rich in neurofilament. These cells were identified with the antibody neurofilament 200 (NF200). NF200 is a marker of A-fiber sensory neurons in the rat (Perry et al. 1991). Aδ-fibers together with C-fibers are capable of detecting noxious stimuli and initiating pain sensation and are considered as nociceptors (see Wu and Pan 2004). The normal pain threshold in humans is the threshold of Aδ-fiber nociceptors. The large-diameter neurons that are neurofilament-positive are known to possess myelinated axons and to be predominantly responsive to mechanical stimuli (Lawson and Waddell 1991; Perry et al. 1991). As in other mammals, such as the rat, many small-diameter neurons were NF200-positive in the cat DRG.
We have found in the current work that 90.8%, 32.1%, and 2.4% of P2X3 receptor-positive neurons coexpress IB4, CGRP, and NF200, respectively, which are similar to results obtained from the rat DRG (Bradbury et al. 1998; Vulchanova et al. 1998). Because IB4 has been shown to label almost all FRAP-positive neurons (Wang et al. 1994), and because we found some colocalization of P2X3 receptors with CGRP, but virtually no P2X3 receptor colocalization with NF200 neurons, our results indicate that P2X3 is predominantly expressed by FRAP-containing DRG neurons with unmyelinated axons. This conclusion is supported by the extensive colocalization of P2X3 and LA4-IR, because LA4 also labels almost all FRAP-positive cells (Dodd and Jessell 1985). It has been proposed that FRAP-containing and peptide-containing sensory neurons represent two parallel systems for processing of nociceptive information (Hunt and Rossi 1985). A nociceptive role for FRAP-positive neurons is also suggested by the localization of the enzyme activity in nerve fibers in cornea (Silverman and Kruger 1988) and in tooth pulp primary afferents (Fried et al. 1989), because these peripheral targets are thought to be innervated primarily by nociceptors. In contrast to the P2X3 receptor, the P2Y4 receptor was detected in 67.4%, 41.3%, and 39.1% of neurons expressing IB4, CGRP, and NF200, respectively. Thus P2Y4 receptor-expressing neurons are likely to have unmyelinated and myelinated axons and include peptidergic and nonpeptidergic subpopulations. Thus P2Y4 receptors may modulate the release of the sensory transmitter, CGRP. In addition, the presence of P2Y4 receptors in peptidergic and nonpeptidergic neurons suggests that peripheral ATP release may excite a broader population of nociceptors in the cat DRG.
We have found that 18.8%, 16.6%, and 63.5% of P2Y2 receptor-positive neurons coexpress IB4, CGRP, and NF200, respectively. These results suggest that most P2Y2 receptor-positive neurons are likely to have myelinated axons and may participate in the conversion of mechanical stimuli into nerve impulses. P2Y receptors are heptahelical G protein-coupled receptors, mediating signal transduction via the induction of inositol triphosphate, leading to intracellular Ca2+ release (Strobaek et al. 1996). P2Y receptors may directly mediate purinergic synaptic transmission or may play a modulatory role.
Colocalization of P2X3, P2Y2, and P2Y4 receptors was not examined directly in the present study. However, these receptors have been colocalized with various other markers of subpopulations of primary afferent neurons. A total of 88.2% and 87.4% of IB4-positive neurons stained for P2X3 and P2Y4 receptors, respectively, and 63.5% of P2Y2 and 39.1% P2Y4 receptor-positive neurons coexpress NF200. Therefore, some overlapping expression between the P2X3 and P2Y4, P2Y2, and P2Y4 receptors must be occurring.
In conclusion, our results suggest that sensory neurons in the cat DRG are likely to respond to ATP and other purines and pyrimidines, not only through ionotropic P2X receptors, but also through metabotropic P2Y receptors. Substantial differences in the distribution of P2 receptor subtypes have been identified in cat DRG, compared with rat and mouse DRG. It will be of interest to clarify the relative physiological and pathophysiological roles of these two receptor family subtypes.
Footnotes
Acknowledgements
This study was funded by grants from the NIH (RO1 DK-57282) and Roche Palo Alto (LAB).
The authors thank Dr. Gillian E. Knight for editorial assistance.
