Abstract
Elevated levels of extracellular ATP have been observed in many tumors. We have localized NTPDase1/CD39, one of the principal extracellular nucleotide-hydrolyzing enzymes, in normal and cancerous human pancreas. NTPDase/E-ATPDase activity was demonstrated with an enzyme histochemical technique on cryosections of human pancreas. Acinar and duct epithelial cells were devoid of E-ATPDase activity in both normal and transformed tissue. Endothelial cells and smooth muscle around blood vessels and larger ducts showed strong activity. Nerves, connective tissue, and the β-cells of the islets were also stained. In cancerous tissue this activity was diminished in the smooth muscle around the ducts and was absent from newly formed connective tissue. Immunostaining for CD39 supported these results but revealed the presence of inactive CD39 in the duct epithelial cells. We hypothesize that the significantly diminished activity of NTPDase1 in the tissues surrounding the ducts may be associated with the processes that lead to tumor formation in human pancreas.
T
Few data are available regarding extracellular ATP and NTPDase activity in the pancreas. In pig pancreas, two isoforms of NTPDase1 have been characterized, purified, and identified (Laliberte et al. 1982; Sévigny et al. 1995). One of their putative roles is to modulate the extracellular concentrations of purines.
According to the authors, the E-ATPDase is strategically positioned to modulate purine-mediated actions such as fluid and electrolyte secretion, smooth muscle contraction, and blood flow (Sévigny et al. 1998). There are data (in the guinea pig) suggesting that, in the exocrine pancreas, luminal nucleotides may act in an autocrine or paracrine fashion to enhance duct secretion and that basolateral nucleotides, perhaps released from nerve terminals, may have an inhibitory effect (Ishiguro et al. 1999). However, no information is available on the regulatory role of ATP or on the physiological-pathophysiological role of NTPDase1 in human pancreas. Our goal was to describe the distribution of NTPDase1 in human pancreas in order to examine the possible correlation between enzyme activity and tumor transformation.
Materials and Methods
All reagents were purchased from Sigma (St Louis, MO) unless otherwise specified. Affinity-purified rabbit anti-CFTR (cystic fibrosis transmembrane conductance regulator) was prepared in the lab of A. Nairn (Rockefeller University; New York, NY). LP2K, a mouse monoclonal anti-cytokeratin 19, was obtained from Dr. E. B. Lane (University of Dundee; Dundee, Scotland).
Preparation of Cryosections
Human pancreatic tissue blocks were obtained at surgery (IMIM; University of Barcelona). A certified clinical pathologist at the location characterized each sample. Tissue blocks were frozen in isopentane cooled by liquid nitrogen. Three- to 5-μm cryosections were cut and, after drying, fixed with acetone (-20C) for 10 min. Each slide contained both normal and tumor sections. Staining procedures were carried out in a Shandon immunostainer (Shandon Scientific; Cheshire, UK).
Enzyme Histochemistry
A lead precipitation enzyme histochemical technique was used for demonstration of NTPDase (E-ATPDase) activity (Kittel 1997). After fixation and several rinses in 0.07 M Tris-maleate buffer, pH 7.4, the sections were incubated in a medium containing ATP or ADP (1 mM) as substrate, 2 mM Pb(NO3)2 (capturing agent for the liberated phosphate), 1 mM levamisole (inhibitor of alkaline phosphatases; Amersham, Poole, UK), 1 mM ouabain, (Na+, K+-ATPase inhibitor; Merck, Darmstadt, Germany), 50 mM a, β-methylene-ADP (5′-nucleotidase inhibitor), and KCl (5 mM) in Tris-maleate buffer (70 mM, pH 7.4) for 30 min at room temperature (RT). Incubation was followed by three rinses in Tris-maleate buffer. The precipitate was converted to PbS with 1% (NH4)2S (1-min incubation).
Immunohistochemistry
After the enzyme histochemical reaction, immunostaining was performed to identify the main cell types in the human pancreatic cryosection. α-LP2K (1:4 dilution) was used to identify epithelial cells, amylase antibody (in undiluted solution) identified the acinar cells, or a chromogranin antibody (Biomeda; Foster City, CA; 3:1 dilution) was applied to distinguish the islet B-cells.
After blocking endogenous phosphatase activity (3 mM levamisole, 0.01% NaN3 in PBS, 10 min) and washing, nonspecific binding sites were blocked with 5% dried milk and 0.04% Tween-20 in PBS for 20 min. The primary antibody was diluted in 1% BSA-PBS and the incubation time was overnight at 4C. After washing with PBS, the secondary antibody (alkaline phosphatase-conjugated IgG, diluted in 1% BSA-PBS) was applied for 60 min at RT. After washing and preincubation with Tris (0.1 M, pH 8.2), staining was visualized with Fast Red (0.1 M, pH 8.2, 5 min). Sections were counterstained in 20% Papanicolau in PBS and, after washing with tapwater, mounted in Aquatex. Images were obtained with an Olympus BP 50 digital camera (Olympus Optical (Europa); Hamburg, Germany).
Immunostaining was also performed with α-CD39 monoclonal antibody (Ancell; Bayport, MN) on the human pancreatic cryosections. Fixation was carried out as above. After rinsing with PBS, the nonspecific binding sites were blocked with 5% BSA in PBS for 30 min and the sections were incubated overnight with CD39 antibody in 1:400 dilution at 4C. The ABC method was used and Vector VIP as chromogen (Vector Laboratories; Burlingame, CA) was applied according to the manufacturer's instructions. After washing with distilled water, sections were mounted in Aquatex and imaged on a Nikon Eclipse E 600 microscope with a Spot digital camera (Nikon; Kumagaya City, Japan) using a ×20 objective.
Results
The enzyme histochemical reaction used in the present work demonstrates the active NTPDase1 enzyme. Whether ATP or ADP was used as a substrate, the distribution of enzyme activity indicated by the precipitate was similar. Ecto-ATPase/E-ATPDase activity was detected as a brown lead sulfide (PbS) deposit, which is generated at sites where phosphate is liberated from the extracellular ATP/ADP during hydrolysis. Immunohistochemical staining performed after enzyme histochemistry was used to identify the different cell types. Immunostaining was also done to localize the DC39/NTPDase1-positive cells. As controls for the enzyme histochemical reaction, the substrate ATP or ADP was omitted from the reaction mixture (Figure 1A). For the immunostaining, the incubation step with the first antibody was omitted (Figure 1I).
As described in Materials and Methods, certified clinical pathologists identified the origin of the tissue blocks used. We studied only one type of pancreatic cancer, duct adenocarcinoma. The general appearance of this cancerous pancreas differs from the normal tissue in many details. We observed proliferation and disorganization of duct epithelial cells, an increased amount of connective tissue, and no staining in the acini when amylase immunostaining was applied (Figure 1D, inset).
Endothelial cells and smooth muscle cells around blood vessels showed the strongest E-ATPDase activity in cryosections of human pancreas (Figure 1B). Acini were unstained. When CD39/NTPDase1 immunostaining was applied, the result was the same for these types of cells (Figure 1C). Each presented staining was performed on normal tissue, but, as shown in Figure 1D, the E-ATPDase activity in the endothelial cells and smooth muscle cells was not altered in cancerous pancreas. The cryosection shown in Figure 1D was cut from a tissue block of tumor origin. The vessel endothelial and smooth muscle cells are covered with the brown PbS deposit because of the active ectoenzyme. Duct epithelial cells are negative for the enzyme reaction; only the connective tissue around the group of ducts shows some very weak staining. Although amylase staining was applied to identify the acini, they were unstained because of the cancerous transformation of the tissue. In normal tissues, when amylase immunostaining was applied (Figure 1D, inset) the acini were positive and showed the red color of the Fast Red. The brown color of the endothelial cells demonstrated E-ATPDase activity.
In normal pancreas, the smooth muscle cells around the larger ducts were also stained (Figure 1E) but the intensity of this staining decreased in the cancerous tissue and the number of ducts was increased because of proliferation (Figure 1E, inset). LP2K staining confirmed the identification of the duct cells (Figure 1E and inset). Although according to the result of enzyme histochemical staining the duct cells did not bear ATPDase enzyme activity, immunostaining showed strong immunoreactivity for CD39 (Figure 1F). Some E-ATPDase activity was also detected in nerves (Figure 1G). The presence of CD39/NTPDas1 was proved with immunostaining (Figure 1G, inset). We have also demonstrated for the first time the E-ATPDase activity of the β-cells of the islets of Langerhans (Figure 1H). β-Cells were identified with chromogranin staining (Figure 1H, upper inset). Immunostaining for CD39 confirmed that the E-ATPDase activity demonstrated by enzyme histochemistry belongs to NTPDase1 (Figure 1H, lower inset).
Discussion
NTPDase1, one of the most intensively studied members of the ecto-NTPDase enzyme family, is considered to play an important role in purinergic signaling, in thromboregulation, and in cell-protective processes by converting ATP released by damaged cells to AMP. The source of extracellular ATP can also be nerve terminals and other types of cells. ATP can be released in response to endogenous ligands and is involved in a special mode of signal transmission, so-called cascade transmission (Nitahara et al. 1995; Sperlágh et al. 1995; Vizi et al. 1997).
Among the nucleotide receptors, P2 receptors are selective for ATP and ADP. In 1981, when the stimulatory effect of ATP on insulin secretion in isolated perfused rat pancreas was demonstrated, it was concluded that a purinoceptor of the P2-type is likely to be present on the β-cells of the rat pancreas (Chapal and Loubatieres-Mariani 1981). There are now data indicating the presence and functional role of both P2X and P2Y receptors on islet cells (Chapal et al. 1984; Bertrand et al. 1987,1989,1990; Hillaire-Buys et al. 1992,1994,1999; Stam et al. 1996). α-Cells possess adenosine receptors involved in the stimulation of glucagon secretion and β-cells have P2 purinoceptors (for ATP and/or ADP) which increase insulin secretion (Loubatieres-Mariani and Chapal 1988; Hillaire-Buys et al. 1994; Tang et al. 1996). The P2Y1 gene is expressed in placenta, skeletal muscle, platelets, and pancreas, and another G-protein-coupled P2 receptor is encoded by an intronless single-copy gene that is expressed exclusively in pancreas (Ayyanathan et al. 1996). However, there are no data concerning the presence or absence of NTPDase1 on islet cells, although its distribution might be expected to correlate with the P2 receptors. On the other hand, the presence of 5'-nucleotidase was shown in rat pancreas about 20 years ago (Chapal et al. 1984) and the substrate of 5′-nucleotidase is AMP, the degradation product of NTPDase activity. Our histochemical staining is the first to demonstrate a strong NTPDase/E-ATPDase activity on the islet cells of human pancreas (Figure 1H). The presence of the entire hydrolysis chain for ATP in the human pancreas may therefore correlate with the presence of both adenosine and P2 receptors.
The presence of E-ATPDase activity has previously been demonstrated on several cell types in the exocrine pancreas of rat and pig (LeBel et al. 1980; Sévigny et al. 1998; Beaudoin et al. 1999). Both basolateral and apical membranes of the acinar cells were positive, as were the membranes of zymogen granules in the acinar cells. In those studies a strong signal was also observed on the duct epithelium. In contrast, we could not demonstrate any active enzyme in either acini or duct cells in normal human pancreas. Endothelial and smooth muscle cells served as intrinsic positive controls for NTPDase1 and showed the expected strong activity. It is important, however, to emphasize the difference between the immuno- and the enzyme histochemical method. The immunostaining used by Sevigny and co-workers is useful for identifying the localization pattern of the target protein molecule but usually does not reveal anything about its activity (Sévigny et al. 1998). In contrast, our enzyme histochemical technique demonstrates the active enzyme only. When immunostaining for CD39 was applied, the localization pattern of this protein in endothelial, smooth muscle, neural, and β-cells was almost identical to the results of enzyme histochemical staining. In agreement with the enzyme histochemical staining, the acinar cells and especially the zymogen granules were devoid of reaction product. However, the duct epithelial cells, which did not show enzyme activity, did show CD39 immunoreactivity. This result may indicate that duct epithelial cells contain an inactive form of the enzyme.
In the tumor samples examined in this study, the transformation was of duct adenocarcinoma origin. Given the overexpression of NTPDase1 observed previously in human melanoma cell lines (Dzhandzhugazyan et al. 1998), ovarian tumor of rats (Slivinskii et al. 1993), and multidrug-resistant breast carcinoma cells (MCF-7-AdR) (Kittel et al. 2000), duct cells might be expected to show some NTPDase activity. Our observation that neither ductal nor acinar cells had E-ATPDase activity in the human pancreas may indicate a significant difference in the distribution and functions of NTPDases in different species.
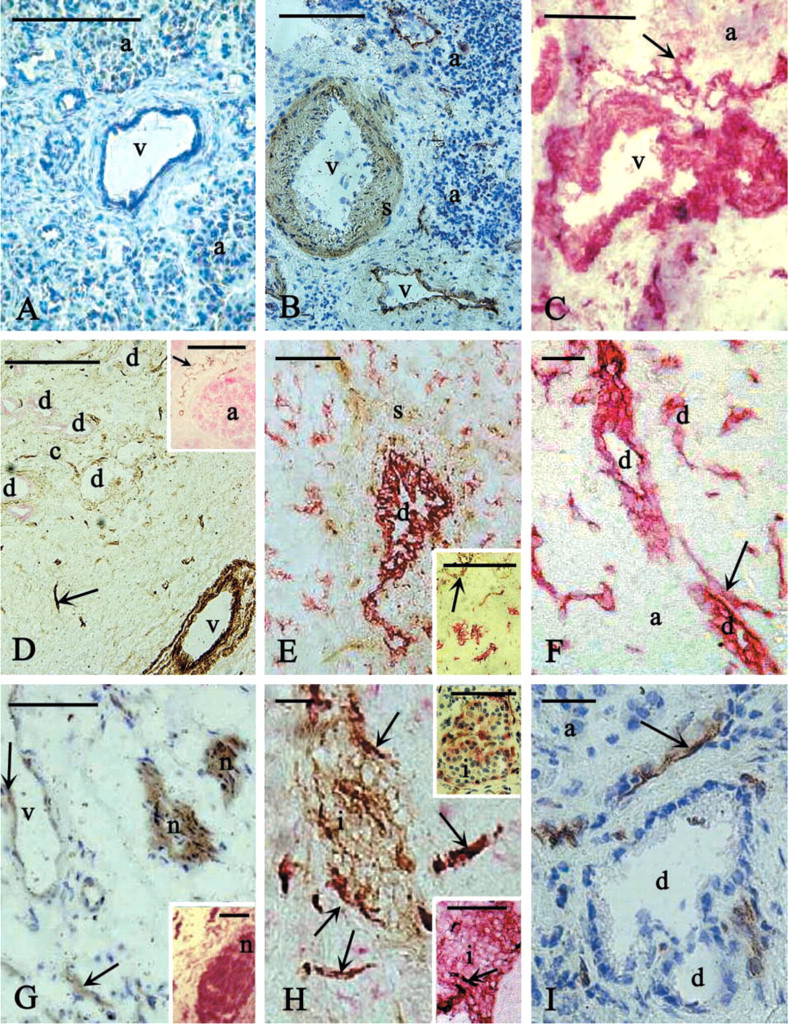
Enzyme histochemistry for E-ATPDase activity and CD39/NTPDase1 immunostaining in cryosections of human pancreas. Brown deposit (lead sulfide) indicates the active enzyme and two kinds of red color (Fast Red for amylase, LP2K, and chromogranin and Vector-VIP for CD39 staining) show the immunoreactive cells. (
Other cells in the human pancreas, such as endothelial cells, smooth muscle cells, nerves and, to a lesser extent, fibroblasts, showed both E-ATPDase activity and CD39 immunoreactivity. This finding is in agreement with previous experience in other types of tissue. Endothelial cells and smooth muscle cells were among the first cell types in which E-ATPase/E-ATPDase activity was demonstrated (Côté et al. 1992; Cunningham et al. 1993). In addition, the role of E-ATPases in the nervous system has been extensively studied (Zimmermann 1996). Our present work reveals that, in the newly formed regions of cancerous tissue, E-ATPDase activity is diminished in the smooth muscle cells around the ducts and is absent from the connective tissue. Only few and controversial data are available about the relation between NTPDases and tumor formation (e.g., Gutensohn and Jahn 1988; Agteresch et al. 1999). The cascade of ectonucleotidases, including NTPDase1, may have a protective function by keeping extracellular ATP and adenosine levels within physiological limits. Therefore, we hypothesize that the significantly diminished activity of NTPDase1 in the tissues surrounding the ducts may be associated with tumor formation in human pancreas. This hypothesis can be supported by another observation. NTPDase1 is generally said to be evenly distributed in the membranes of the cells. However, we have observed that in cell types which possess caveolae, specialized membrane invaginations involved in signaling, NTPDase1 is preferentially targeted to these membrane microdomains (Kittel and Bácsy 1994; Kittel 1997,1999). Several molecules involved in signaling (receptors, eNOS, G-proteins), which are targeted to the caveolae, undergo post-translational modifications (palmitoylation). It is the same for NTPDase1 (Kittel et al. 1999; Koziak et al. 2000), suggesting a possible role of NTPDase1 in signal transmission. Smooth muscle cells, for example, possess many caveolae. The diminished E-ATPDase activity in the smooth muscle cells around the ducts in cancerous pancreas may therefore be significant. It may affect the communication between neighboring cells and contribute to the process of malignant transformation. Conversely, it is also possible that the loss of enzyme activity is a consequence of tumor transformation.
In conclusion, our findings suggest that NTPDase1 is not expressed in acinar cells of the human pancreas and that the duct cells express only an inactive form of the enzyme. This may indicate a different role of NTPDase1 in human pancreas compared with other species, and its differential distribution in the surrounding tissues may be associated with the processes that lead to tumor transformation.
Footnotes
Acknowledgements
Supported by INCO program of the European Comission (ERB IC20-CT98-0202), Hungarian Committee for Technical Development (EU-98-D9-065), and Hungarian Medical Research Council 282/2000 grants, as well as Biomed contract BMH4-CT98-3805 and by grant PM97-0077 from the Direccion General de Enseñanza Superior e Investigación Científica (Spain) Grant.
We thank Drs F.X. Real and Anouchka Skoudy (IMIM; Barcelona, Spain) for their valuable help during the experimental work. We also thank Drs S.C. Robson, (Harvard BIDMC; Boston), J. Sevigny (Harvard BIDMC; Boston), and M.C. Steward (Manchester Univ., UK) for critical reading of the manuscript.
