Abstract
Cystatin C, a cysteine protease inhibitor, was examined in the apical buds of rat incisors by immunohistochemistry, because in transition and maturation zones most of the dendritic cells in the papillary layer are anti-cystatin C–positive. Anti-cystatin C–labeled cells were sparse and localized to the proliferation and differentiation zones, constituting the apical bud of 5-week-old rat incisors. These cells were considered macrophages or dendritic cells, based on their reactivity with OX6 and ED1, as well as their ultrastructure. Basement membrane at the periphery of apical bud was also labeled by anti-cystatin C antibody. The apical buds included a few apoptotic fragments and weak reactivity with antibody to cathepsin L, a cysteine protease. Reactivity to anti-cystatin C and anti-cathepsin ∗∗∗
A
Most of the dendritic cells in the enamel organ also bind anti-cystatin C antibodies (Nishikawa and Sasaki 2000). Cystatin C is an endogenous cysteine protease inhibitor and is expressed ubiquitously (Chapman 1991). In particular, macrophages secrete cystatin C and are thought to regulate proteolytic activity in the extracellular space (Warfel et al. 1987; Chapman 1991). Some populations of dendritic cells are also reported to exhibit strong expression of cystatin C (El-Sukkari et al. 2003). This expression occurs in dendritic cells in the enamel organ of the rat incisor. Anti-cystatin C-labeled OX6− cells are distributed in the secretion zone of the rat incisors (Nishikawa and Sasaki 2000). These cells are speculated to be precursor cells of dendritic cells in the transition and maturation zones, invading the enamel organ initially as OX6− cells and becoming OX6+ thereafter.
It is not known whether invasion of dendritic cell precursors begins at the secretion zone or in an earlier zone, that is, in the differentiation zone or even in the proliferation zone. The aim of this study was to determine the pattern of cystatin C expression in the enamel organ and to characterize the cystatin C–positive cells that may play a role in amelogenesis.
Materials and Methods
Six 5-week-old male Wistar rats (140–150 g) (Jcl Wistar; Clea Japan, Tokyo, Japan) were used for the immunofluorescence microscopy, following institutional guidelines for animal care. The animals were perfused under sodium pentobarbital (Nembutal; Abbott, North Chicago, IL) anesthesia with 4% paraformaldehyde in 0.1 M PBS, pH 7.2, through the left ventricle at RT for 10 min. The maxillae and mandibles were dissected and were subsequently immersed in the same fixative at 4C for 2 hr. After being washed with 0.1 M PB, the maxillae and mandibles were demineralized by immersion in 5% EDTA solution adjusted to pH 7.3 with sodium hydroxide solution, at 4C for 3 to 4 weeks. Demineralized incisors were cut transversely into two segments. Both segments were immersed in 25% sucrose in PBS overnight, rapidly frozen, and then cut longitudinally (6- to 8-μm thick) using a cryotome.
Three newborn rats (5.8–6.2 g; Clea Japan) were also killed by decapitation. Mandibles were dissected and fixed with 4% paraformaldehyde in 0.1 M PB. They were subsequently immersed in the 25% sucrose solution overnight, rapidly frozen, then cut using a cryotome.
Antibodies used in this study were mouse monoclonal ED1, ED2, and OX6 antibodies (Serotec; Oxford, UK), rabbit polyclonal anti-cystatin C antibodies (Upstate Biotechnology; Lake Placid, NY), and goat polyclonal anti-cathepsin B (E-19), cathepsin L (C-18), and cathepsin S (M-19) antibodies (Santa Cruz Biotechnology; Santa Cruz, CA). ED1, ED2, OX6, and anti-cystatin C have been previously characterized (McMaster and Williams 1979; Dijkstra et al. 1985; Damoiseaux et al. 1994; Pierre and Mellman 1998; Nishikawa and Sasaki 2000).
For triple labeling, cryosections on glass microscope slides were incubated in 1% BSA-PBS at RT for 30 min. They were then labeled with ED1, ED2, or OX6 antibodies diluted 1:100 (10 μg/ml) with 1% BSA-PBS at RT for 30 min, followed by labeling at RT for 10 min with biotinylated anti-rabbit and anti-mouse immunoglobulins absorbed to abolish cross-reactivity with rat serum proteins (LINK; DAKO LSAB2 Kit, DAKO, Carpinteria, CA). Sections were visualized by labeling with 2 μg/ml Rhodamine Red-X-conjugated streptavidin (Molecular Probes; Eugene, OR) at RT for 10 min. After being washed with PBS and incubated with 1% BSA-PBS, the sections were labeled with anti-cystatin C antibodies diluted 1:100 (10 μg/ml) at RT for 30 min, followed by labeling with a mixture of Alexa 488–conjugated anti-rabbit IgG diluted 1:100 and 0.5 μg/ml Hoechst 33342 (Molecular Probes). For control sections, mouse isotype control IgG (mouse IgG1–negative control, DAKO) and rabbit immunoglobulin fraction (DAKO) were used instead of ED1, ED2, or OX6 and anti-cystatin C, respectively. For some sections, triple labeling was performed using a mixture of anti-cystatin C and ED1 or OX6, followed by labeling with a mixture of Alexa 488–conjugated anti-rabbit IgG, Alexa 647–conjugated anti-mouse IgG (Molecular Probes), and Hoechst 33342. Fluorescent images were obtained using an Olympus A X80 fluorescence microscope equipped with a Charge Coupled Device camera (Quantix KAF1401E; Photometrics, Tucson, AR) and MetaMorph software (Universal Imaging; Downingtown, PA).
For cathepsin immunohistochemistry, sections were incubated at room temperature for 30 min with anti-cathepsin B, L, and S antibodies diluted 1:20, followed by donkey anti-goat IgG conjugated with biotin (Santa Cruz Biotechnology) diluted 1:100 for 10 to 20 min. They were then incubated with streptavidin peroxidase (DAKO LSAB2), and visualized by Simple Stain DAB (Nichirei; Tokyo, Japan).
Procedures for conventional electron microscopy and preembedding immunoelectron microscopy for anti-cystatin C have been described elsewhere (Nishikawa and Sasaki 1995,2000).
Results
Labeling the Proliferation Zone of the Rat Incisor Enamel Organ with Anti-cystatin C, OX6, and ED1 Antibodies
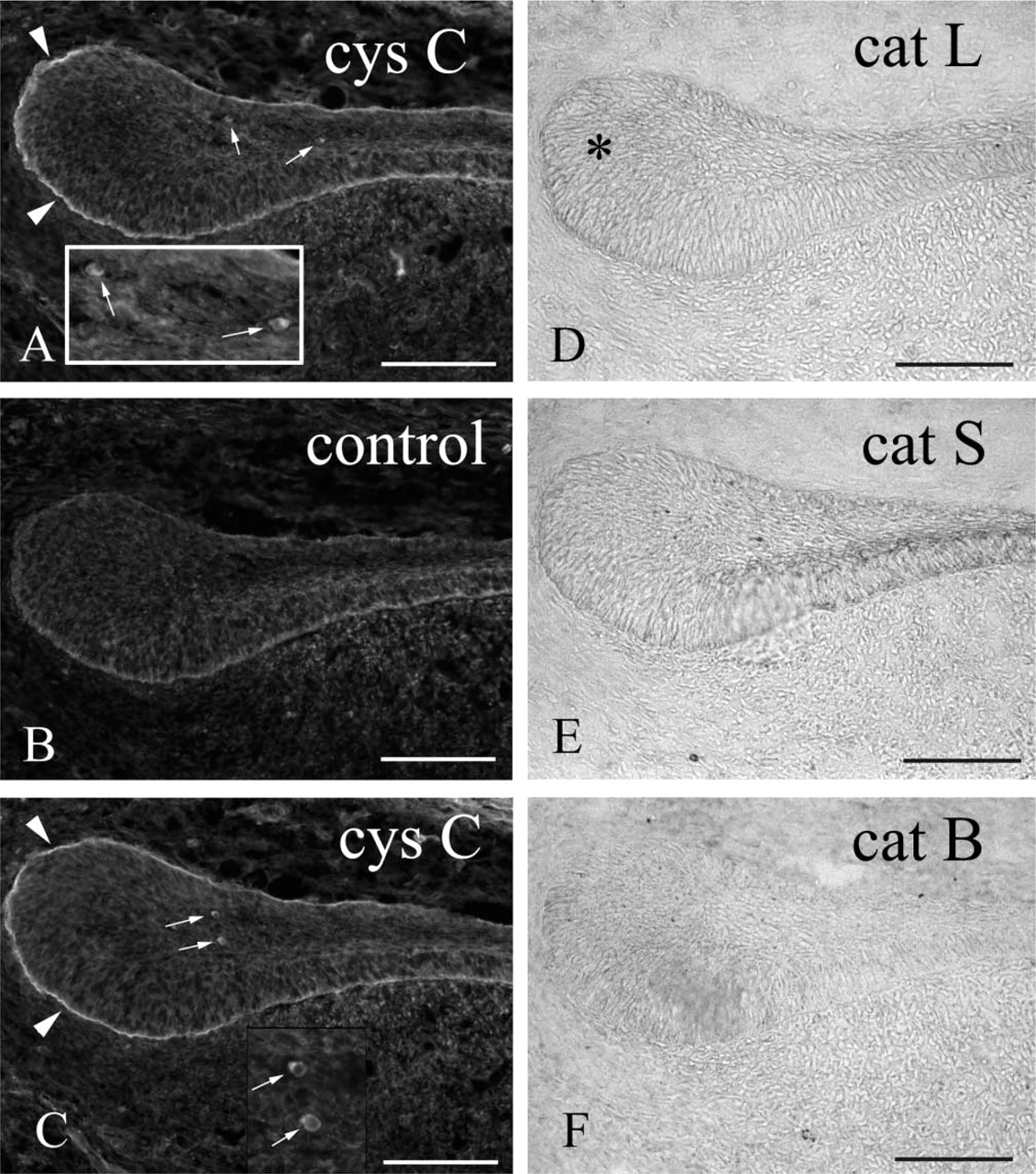
Anti-cystatin C–labeled cells were present mainly in the proliferation and differentiation zones (Figures 1A, 1C, 1D, and 1F) and were scattered sparsely in the apical bud of the incisor enamel organ. The anti-cystatin C–labeled cells varied in shape, from a simple ovoid to a dendritic morphology. Basement membrane of the apical bud was also labeled by anti-cystatin C (Figures 1A and 1D). Several anti-cystatin C–labeled cells were also OX6+ (Figures 1A and 1B), whereas ED1 antibodies labeled the majority of anti-cystatin C–labeled cells (Figures 1D–1F). None of the zones of the enamel organ were labeled with ED2 antibodies (Figure 1G). In the secretion zone, anti-cystatin C, but not OX6, antibodies labeled few cells of enamel organ (Figures 1G and 2), whereas in the maturation zone many OX6+ anti-cystatin C–labeled dendritic cells were observed (Figures 1H and 2). Ratios of average OX6+ cell numbers to average anti-cystatin C–positive cell numbers were 0.56, 0.72, 0.03, and 1.06 in the proliferation, differentiation, secretion, and late maturation zones, respectively (Figure 2). Conventional electron microscopy revealed nonepithelial cells lacking both tonofilaments and desmosomes. These cells exhibited small numbers of lysosomal granules, organelle-poor cytoplasm at the cell periphery, and deep invaginations at the plasma membrane, with an appearance of dendritic-precursor cells (Figure 3A). Some nonepithelial cells exhibited more lysosomal inclusions (Figure 3B). Anti-cystatin C immunocytochemistry revealed that cystatin C–labeled cells possessed endoplasmic reticulum including nuclear envelope (Figure 3C).
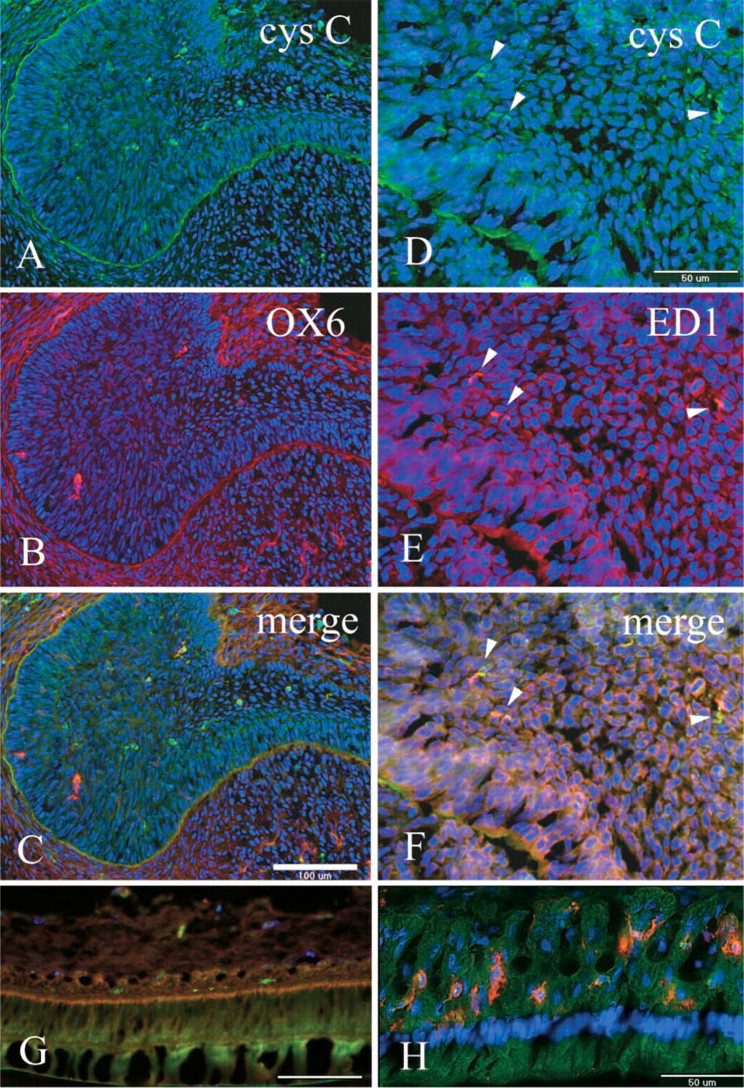
Immunohistochemical localization of anti-cystatin C (Cys C) (
Apoptotic Events of Enamel Epithelial Cells in the Rat Incisor Apical Bud
To assess the role of nonepithelial cystatin C–positive dendritic cells or macrophages in the apical bud, the terminal deoxynucleotidyl transferase–mediated deoxyuridine triphosphate nick-end labeling (TUNEL) method was employed to determine whether apoptosis occurred in the proliferation zone. In the apical bud of the enamel organ, TUNEL-positive cells were detected, although their frequency was low (Figure 4A). Conventional electron microscopy revealed the apoptotic cell fragments (Figure 3D).
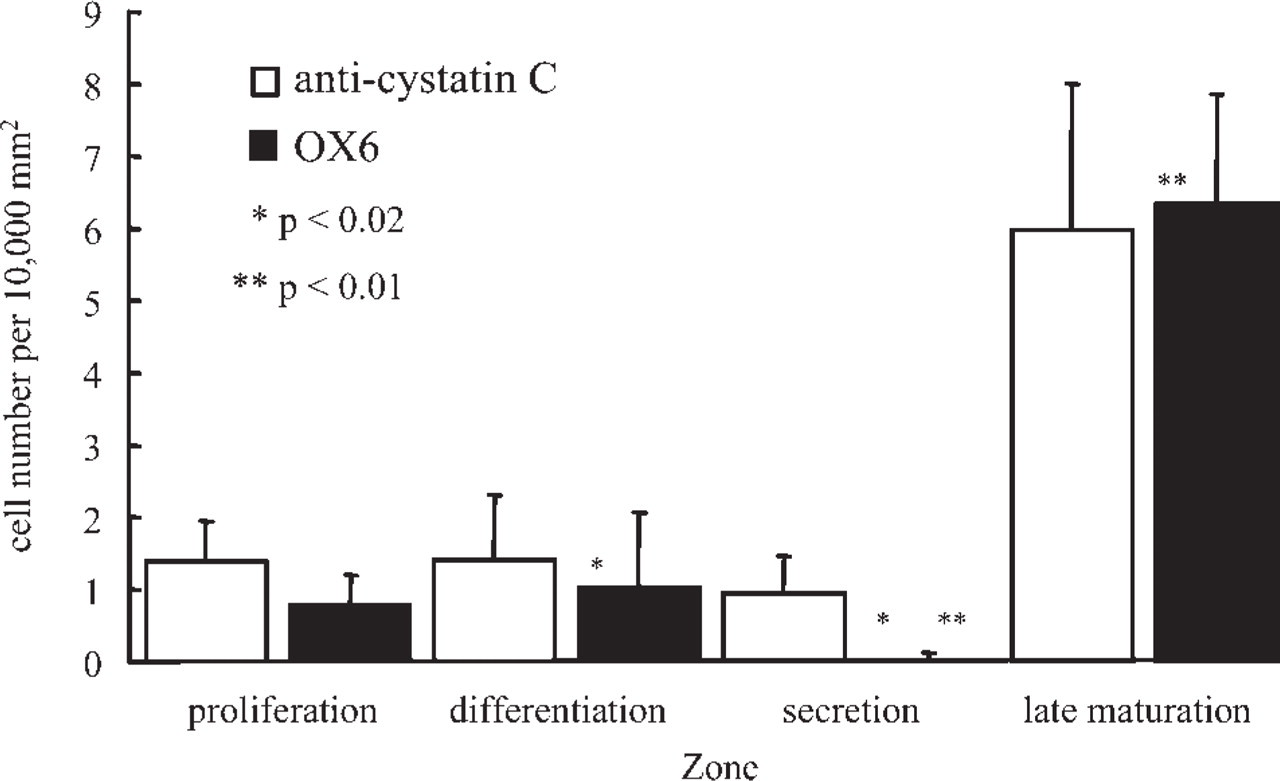
Anti-cystatin C–labeled or OX6+ cell numbers per unit area (10,000 mm2) in the rat incisor enamel organ. Mean and standard deviations were obtained from 5 to 12 sections of the proliferation (4 rats), differentiation (3 rats), secretion (2 rats), and late maturation (1 rat) zones. ∗,p < 0.02; ∗∗,p < 0.01 (Student's t-test).
Cathepsin B, L, and S Immunocytochemistry
To determine whether cysteine proteases were active in the enamel organ, cysteine protease (cathepsin B, cathepsin L, cathepsin S) immunocytochemistry was performed. Anti-cathepsin L labeled apical bud enamel organ epithelia weakly, and this labeling extended into the differentiation zone (Figure 4B). Anti-cathepsin S labeling was not detected in most specimens (Figure 4C). Anti-cathepsin B labeling was not detected in the enamel organ (Figure 4D), but the odontoblast layer of dental pulp was weakly labeled.
Cystatin C–positive Cells Present in the Apical Bud Epithelia of Neonatal Rat Incisors
To determine whether invasion of cystatin C–positive cells in the apical bud of enamel organ is related to adulthood, apical bud enamel organ of neonatal rat incisors was also examined. Anti-cystatin C–labeled cells were present in the apical bud of the neonatal incisors (Figures 5A–5C), and the basement membrane of apical bud was also labeled with anti-cystatin C (Figures 5A and 5C). Anti-cathepsin L and S, but not B, weakly labeled the apical bud epithelium (Figures 5D–5F).
Discussion
Nishikawa and Sasaki (2000) reported that anti-cystatin C–positive dendritic cells are abundant in the epithelial papillary layer of maturation zone and also exist in the secretion and transition zones in the rat incisors. In this study, anti-cystatin C–labeled cells were localized to the differentiation zone and proliferation zones of the incisor apical bud. Some of these anti-cystatin C–labeled cells were also OX6+. As in previously published results (Nishikawa and Sasaki 2000), cystatin C–positive cells seem to be distributed in the epithelial enamel organ throughout rat incisor amelogenesis. These cells seem to be dendritic cells or of macrophage lineage, based on their ultrastructure and immunohistochemistry results from the current study. It is not clear, however, whether these cystatin C–positive cells are a uniform population, because in the current study some of them also stained with OX6 antibodies in the proliferation, differentiation, transition, and maturation zones but did not stain with OX6 antibodies in the secretion zone, located between differentiation and transition zones. These results support previous findings (Nishikawa and Sasaki 2000).
It has been shown that macrophages or dendritic cells that incorporate apoptotic cell fragments are localized in the transition zone (Nishikawa and Sasaki 1996,2000). Therefore, in the current study, apoptosis was examined in the apical bud using the TUNEL method and electron microscopy. Both methods revealed the presence of a limited degree of apoptosis in the apical bud. These results are consistent with those of previous studies, in which molar tooth germs exhibited numerous apoptotic events, but apoptosis in incisor tooth germs was less common (Vaahtokari et al. 1996; Kieffer et al. 1999). The low level of apoptosis in the incisor apical bud may be related to the survival of progenitor cells of inner enamel epithelia and ameloblasts. Cystatin C–positive cells may eliminate apoptotic cell fragments, or conversely they might offer protection from apoptosis by secretion of the cysteine protease inhibitor, cystatin C.
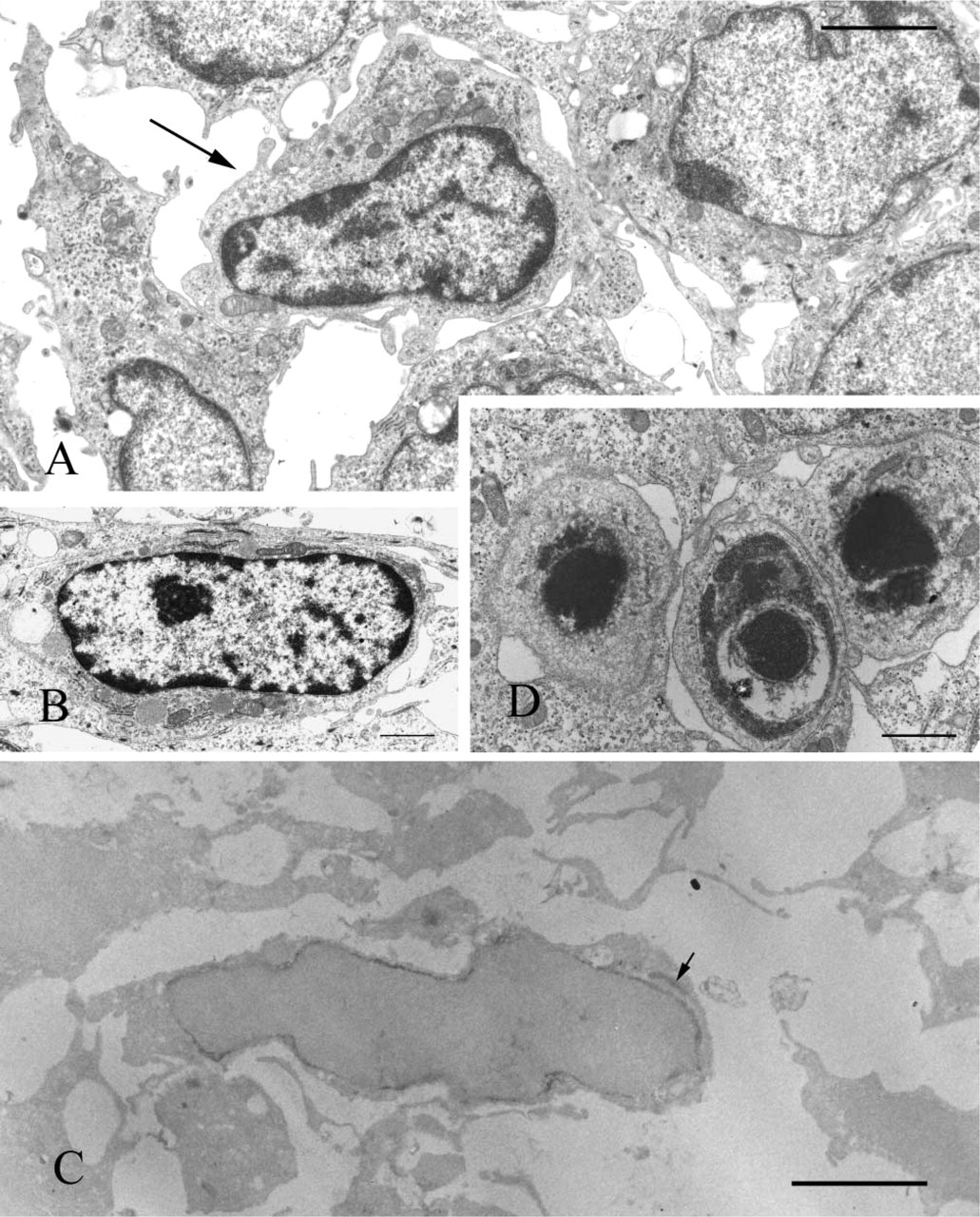
Conventional electron micrographs (
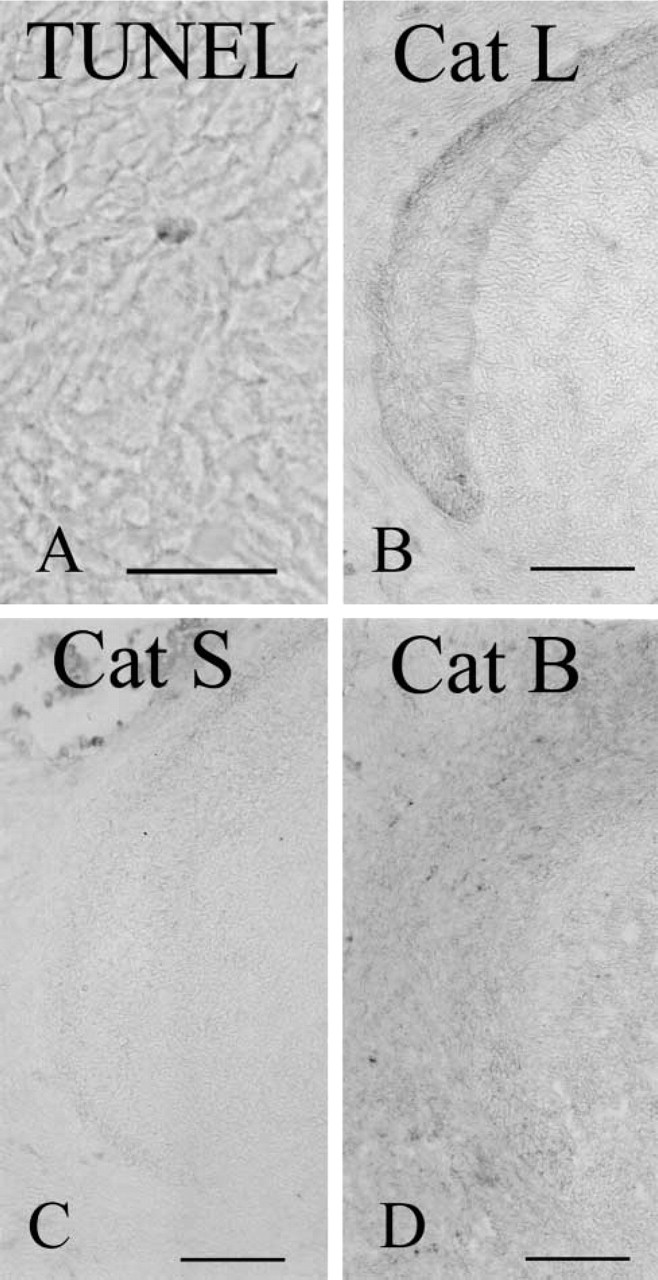
Apical bud labeled using the TUNEL method (
Cystatin C is a normal blood plasma constituent, secreted constitutively by monocytes/macrophages and ubiquitously expressed (Warfel et al. 1987; Abrahamson et al. 1990). Because cystatin C is an endogenous cysteine protease inhibitor, it may function in regulating cysteine proteases in the enamel organ. Indeed, in the current study, cathepsin L reactivity was detected at the apical bud of 5-week-old rats, and faint cathepsin L and S reactivities were found at the apical bud of newborn rats. Although the primary targets of cathepsins are cellular lysosomes, a fraction of these enzymes are thought to be secreted into the extracellular space (Chapman 1991). Proteolytic regulation by cathepsins and cystatin C has been widely reported in embryo implantation (Afonso et al. 1997), normal organogenesis (Roth et al. 2000; Tobin et al. 2002; Wright et al. 2003), and some pathological processes (Shi et al. 1999; Stypmann et al. 2002). In addition, cystatin C is reported to be involved in the proliferation of neural stem cells as a cofactor of FGF2 (Taupin et al. 2000). Serine protease and matrix metalloproteinase function in processing and degradation of enamel matrix proteins (Tanabe et al. 1992; Bartlett et al. 1996; Robinson et al. 1998; Simmer et al. 1998). This study raises the possibility that cysteine proteases and cysteine protease inhibitor, cystatin C, play a role in tooth morphogenesis and enamel formation. In the current study, anti-cystatin C also labeled basement membrane, but adjacent epithelial or mesenchymal cells lacked reactivity. Basement membrane labeling of anti-cystatin C may reveal the storage of physiologically active substances such as cytokines and enzymes there, as has been reported (Dowd et al. 1999; Kawashima et al. 2003; Wenzel et al. 2003). Although this point remains to be clarified, it is hypothesized that the secreted cystatin C is trapped at the basement membrane and may protect against attack by proteases diffusing from the epithelia, pulp region, and nearby osteoclasts. Cathepsin S-positive macrophages are localized to the early pulp (unpublished observation) and to many osteoclasts on the alveolar bone socket around apical bud. In mice incisors, strong cathepsin K expression has been reported in cells lining bone apically (Yamada et al. 2003). Osteoclasts are known to be positive for cysteine protease cathepsin K and L (Nakase et al. 2000). It is thus proposed that cystatin C plays a role in the protection from putative cysteine protease activity, but further study is needed to address this possibility.
OX6+, ED1-positive, or ED2-positive dendritic cells or macrophages are scarce in the incisor and molar pulp of newborn rats but increase in number after 2 weeks in the molar and 1 week in the incisor (Jontell et al. 1991; Okiji et al. 1996). Dendritic cells in the enamel organ of the maturation zone of rat incisors are scarce in juvenile animals, whereas numerous dendritic cells are present in the adult animals (Takano et al. 1996). This study showed the presence of round cystatin C–positive cells in the incisor apical bud of the newborn rat. Furthermore, this study showed immunoreactivity of anti-cystatin C at the basement membrane. These results indicate that cystatin C–positive cells are not unique to adult rats.
Apical buds of newborn (day 0) rats labeled with anti-cystatin C (cys C) (
In conclusion, the results of this study suggest that cystatin C–positive macrophages or dendritic cells are involved in normal incisor morphogenesis. In the apical bud, these cells seem to be related to apoptotic events and/or protection from proteolytic activity of cysteine proteases.
Footnotes
Acknowledgements
This work was supported in part by a grant from the Ministry of Education, Culture, Sports, Science and Technology of Japan to promote multidisciplinary Research Projects from 2001 to 2005.
