Abstract
Few systematic studies have been published comparing the expression and distribution of endothelial cell (EC) markers in different vascular beds in normal human tissues. We investigated by immunohistochemistry the expression of CD31, CD34, von Willebrand factor (vWF), and Fli-1 in EC of the major organs and large vessels. Tissue samples obtained from autopsies and biopsy specimens were routinely processed and stained immunohistochemically for CD31, CD34, and vWF. Biopsy material was also stained immunohistochemically for Fli-1, D2-40, and Lyve-1. The expression pattern of the markers was heterogeneous in some of the organs studied. In the kidney, fenestrated endothelium of the glomeruli strongly expressed CD31 and CD34 but was only focally positive or completely negative for vWF. Alveolar wall capillaries of the lung strongly stained for CD31 and CD34 but were usually negative for vWF. The staining intensity for vWF increased gradually with the vessel caliber in the lung. Sinusoids of the spleen and liver were diffusely positive for CD31. They were negative for CD34 in the spleen and only expressed CD34 in the periportal area in the liver. Fli-1 was expressed in all types of EC but also in lymphocytes. D2-40 stained lymphatic endothelium only. Lyve-1 immunostaining was too variable to be applied to routinely processed tissues. The expression of EC markers CD31, CD34, and vWF in the vascular tree is heterogeneous with a specific pattern for individual vessel types and different anatomic compartments of the same organ. D2-40 labels lymphatic EC only.
Keywords
E
The few studies published have mostly been restricted to the lung. Müller et al. (2002a) showed that CD31 is usually strongly and homogeneously expressed by all human pulmonary EC. The expression pattern of CD34 and vWF was found to be more heterogeneous. The strongest expression of CD34 was found in EC of capillaries, followed by arteries, veins, arterioles, and venules. Two studies have been performed comparing the expression of vWF in human pulmonary EC (Kawanami et al. 2000; Müller et al. 2002b). Also in these studies, CD31 showed homogeneous intense staining in all pulmonary EC. In contrast, vWF-specific antibody yielded negative or weakly positive staining reactions in capillary EC. The staining intensity increased with the vessel caliber. Veins exhibited the strongest expression of vWF, followed in intensity by arteries, arterioles, capillaries, and venules.
CD31, a member of the immunoglobulin superfamily, is a 130-kDa transmembrane glycoprotein also designated as PECAM-1 (platelet endothelial cell adhesion molecule 1). It is present on the surface of platelets, monocytes, macrophages, and neutrophils and is a constituent of the endothelial intercellular junction. It plays a major role in the adhesion cascade between EC and the inflammatory cells during inflammation in facilitating leucocyte migration and between EC during angiogenesis. It has recently been recognized for its angiogenic role (DeLisser et al. 1997; Matsumura et al. 1997; Zhou et al. 1999). It is one of the best markers for benign and malignant vascular tumors (De Young et al. 1993) but also stains plasma cells, monocytes, and megakaryocytes.
CD34 is a 110-kDa transmembrane glycoprotein present on leukemic cells, EC, and stem cells. In addition, it is localized on cells of the splenic marginal zone, dendritic interstitial cells around vessels, nerves, hair follicles, muscle bundles, and sweat glands in a variety of tissues and organs. Its function is still unclear. It is used for leukemia diagnosis and subclassification and for diagnosis of vascular tumors. Antibodies to CD34 also strongly label gastrointestinal stromal tumors, and the antigen is invariably found in solitary fibrous tumor and dermatofibrosarcoma protuberans (Kutzner 1993; Westra et al. 1994).
vWF (factor VIII-related antigen) is a glycoprotein that mediates platelet adhesion to subendothelium at sites of vascular injury and binds and stabilizes factor VIII in the circulation (Alles and Bosslet 1988; Yamamoto et al. 1998). It appears to be expressed exclusively on EC where it shows a granular pattern of reactivity. It is also expressed in the cytoplasm of megakaryocytes. It is stored in Weibel-Palade bodies (Warhol and Sweet 1984; Cines et al. 1998; Zhou et al. 1999). Although it is commonly used as an immunohistochemical marker for EC, little is known about the relative level of expression of vWF in EC of the same or different vascular beds in vivo (Weiss 1991; De Young et al. 1993; Yamamoto et al. 1998; Müller et al. 2002c).
Fli-1 is a nuclear transcription factor involved in cellular proliferation and tumorigenesis. It appears to be a relatively sensitive and specific marker for Ewing sarcoma and primitive neuroectodermal tumors that are characterized in 90% of cases by a specific translocation t(11;22), resulting in the fusion of the EWS gene on chromosome 22 to the Fli-1 gene on chromosome 11. The Fli-1 immunoreactivity in vascular tumors appears to equal or exceed that for CD31, CD34, and vWF (Folpe et al. 2001). As a transcription factor, Fli-1 immunoreactivity is nuclear in location. The protein is ubiquitously expressed by all endothelia (Hewett et al. 2001) but is also expressed on hematopoietic cells. It was claimed to be the first reliable nuclear marker of endothelial differentiation (Folpe et al. 2001; Rossi et al. 2004).
The present study was undertaken to immunohistochemically compare the expression of commonly used cytoplasmic EC markers (CD31, CD34, vWF) and the new nuclear EC markers like Fli-1 in different vascular beds of normal human tissues. To provide a comprehensive picture, we did not restrict the study to one particular organ but included different parenchymatous organs and large vessels. Moreover, knowing that the expression of CD31, CD34, vWF, and Fli-1 is not restricted to blood vessels, we also used D2-40, a monoclonal antibody that has recently been recognized to be highly specific for lymphatic endothelium (Fukunaga 2005), for lymphatic vessel identification in the parenchymatous organs.
Materials and Methods
Normal tissues were obtained from biopsies and autopsy cases. Tissues were formalin fixed, paraffin embedded, and stained immunohistochemically for CD31, CD34, and vWF. In addition, biopsy specimens were stained for Fli-1 protein and D2-40. The use of human material for this study was approved by the local medical ethics committee.
Case Selection
Biopsy Specimens. Blocks of paraffin from surgical specimens or needle biopsies of normal liver, spleen, kidney, lung, heart, skin, bone marrow, and lymph node were retrieved from our files. In total, five needle biopsies of the kidney, five surgical biopsies of the spleen, four endomyocardial biopsies of the heart, five biopsies of the liver (four needle and one surgical), six surgical biopsies of the lungs, five biopsies of the skin, five biopsies of bone marrow, and five biopsies of lymph node were selected. Sections were stained with hematoxylin-eosin (H-E) for routine histology. Only normal tissues were retained for immunohistochemical analysis.
Autopsy Specimens. Kidney, lung, and spleen parenchyma and major vessels (thoracic and abdominal aorta, pulmonary artery and vein, renal artery, femoral artery and vein, and inferior vena cava) were sampled systematically from nine autopsy cases. All samples were obtained <24 hr after death. Patients ages ranged from 34 to 93 years. Three patients had a history of high blood pressure. Specimens were fixed in 4% formalin and paraffin embedded. They were then stained with H-E and Elastica Van Giesson. For bone marrow samples, routine decalcification was done prior to staining.
Immunohistochemistry
Antibodies used for this study are listed in Table 1. As lymphatic-specific marker, we initially performed immunostaining with Lyve-1. Unfortunately, the antibody gave poor results, and the immunostaining could not be interpreted due to high background staining. We then used D2-40 monoclonal antibody with reliable and reproducible results.
Antibodies used in the study
The 4-μm paraffin sections were deparaffinized in xylol and rehydrated in graded alcohol series. Endogenous peroxidase was inhibited using 3% H2O2 in methanol. The sections were then washed in distilled water and heated in a microwave oven (in citrate buffer 10 mM, pH 6, for CD31, CD34, Fli-1, and Lyve 1 and EDTA 1 mM, pH 7.5, for factor VIII) 15 min for epitope retrieval. No pretreatment was needed for D2-40. Slides were incubated first with normal horse serum (1/30 avidin 10%) for CD31 and CD34 and with normal goat serum (10% avidin) for factor VIII and Fli-1 for 5 min and then in biotin for 10 min. Endogenous biotin was inhibited with a Vector Blocking kit (Vector Laboratories; Burlingame, CA). The slides were then incubated at 20C for 40 min with monoclonal antibodies for CD31, CD34, and D2-40 and polyclonal antibody for factor VIII and Fli-1. For CD31 and Lyve-1, the slides were incubated overnight at 8C. The slides were incubated with anti-mouse/rabbit biotinylated bridging antibodies (dilution 1/200) for 30 min. Sections were then washed and incubated with standard avidin-biotin complex (ABC; DakoCytomation, Glostrup, Denmark) for 30 min. Antibody binding was revealed using H2O2 as a substrate and diaminobenzidine as chromogen. Counterstaining was performed with hematoxylin.
Results
The expression pattern for CD31, CD34, vWF, and Fli-1 in the different tissues we studied was strikingly heterogeneous. The differences concerned especially the small vascular beds including continuous and fenestrated capillaries and sinusoids. Major differences were seen in the parenchymatous organs and are summarized in Table 2. There were no significant differences between the autopsy and the biopsy tissues.
Kidney
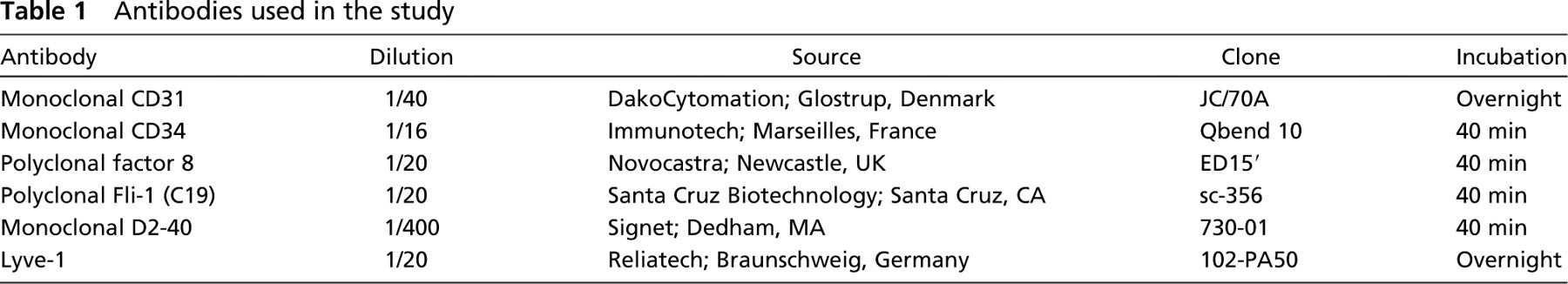
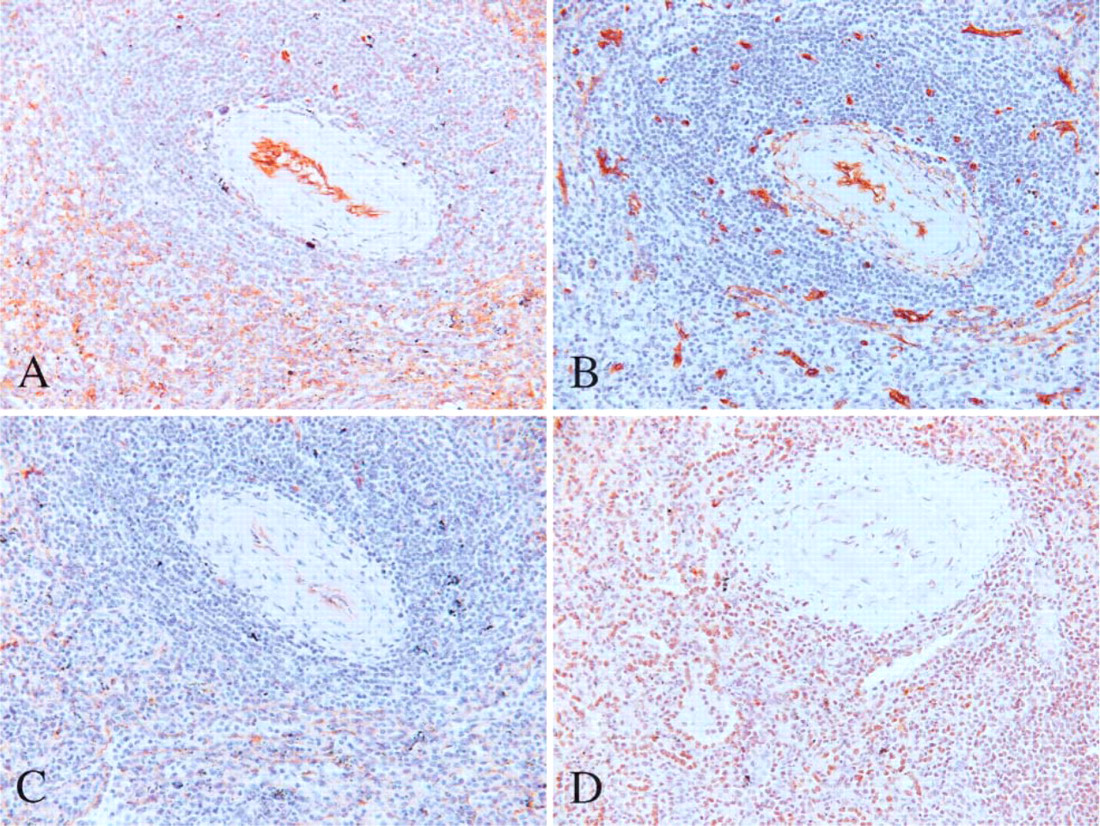
The fenestrated endothelium of the glomeruli always strongly expressed CD31 and CD34 but was only focally positive or completely negative for vWF (Figures 1A-1C). In one biopsy case, the glomeruli were diffusely positive for vWF. In the three autopsy cases from hypertensive patients, the glomeruli were weakly or moderately positive for vWF as compared with the autopsy cases from patients without hypertension. More interstitial capillaries stained for CD34 than for CD31 or vWF. Fli-1 usually stained the nuclei of EC in the glomeruli (Figure 1D), making it easier to differentiate between EC and other types of cells also present in the glomeruli, such as podocytes or mesangial cells. In addition, EC of arterioles, venules, and interstitial capillaries were stained. There were, however, some cases in which the interpretation was difficult as a result of important background staining.
Lungs
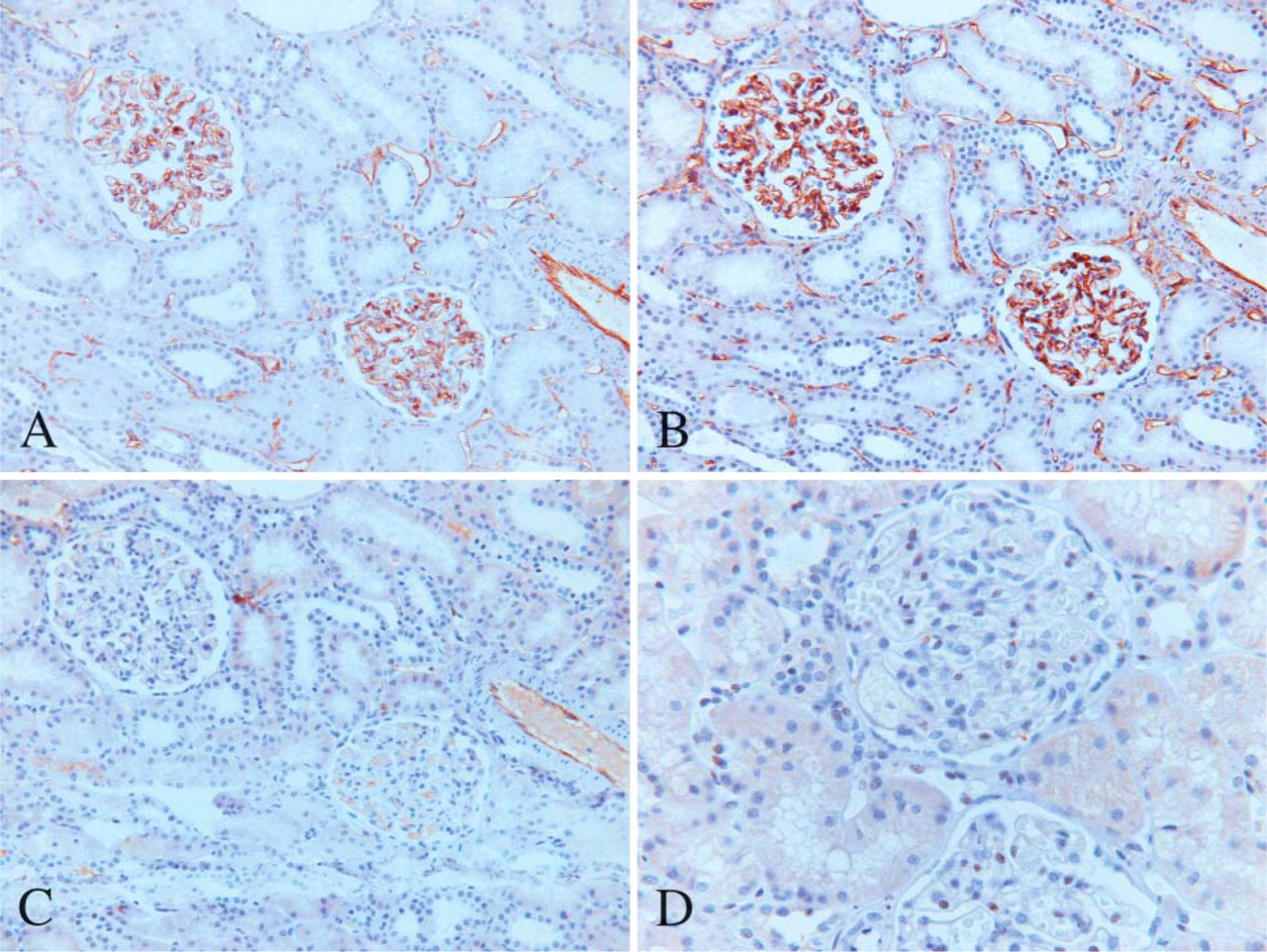
Capillaries in the alveolar wall strongly stained for CD31 and CD34 but were usually negative for vWF (Figures 2A-2C). The staining intensity for vWF also increased gradually with the vessel caliber. Small arterioles and venules showed light staining but larger arteries and veins showed intense staining. The staining intensity did not vary according to vessel type or caliber for CD31 and CD34. Alveolar macrophages were positive for CD31 with a membranous pattern of staining but were negative for CD34 and vWF. Fli-1 diffusely stained the nuclei of EC of the different vessel types in the lungs (Figure 2D). However, the nuclei of lymphocytes were also stained, as well as the cytoplasm of some alveolar wall cells probably corresponding to mastocytes.
Staining of endothelial cells for CD31, CD34, vWF, and Fli-1 in various norma l adult tissues
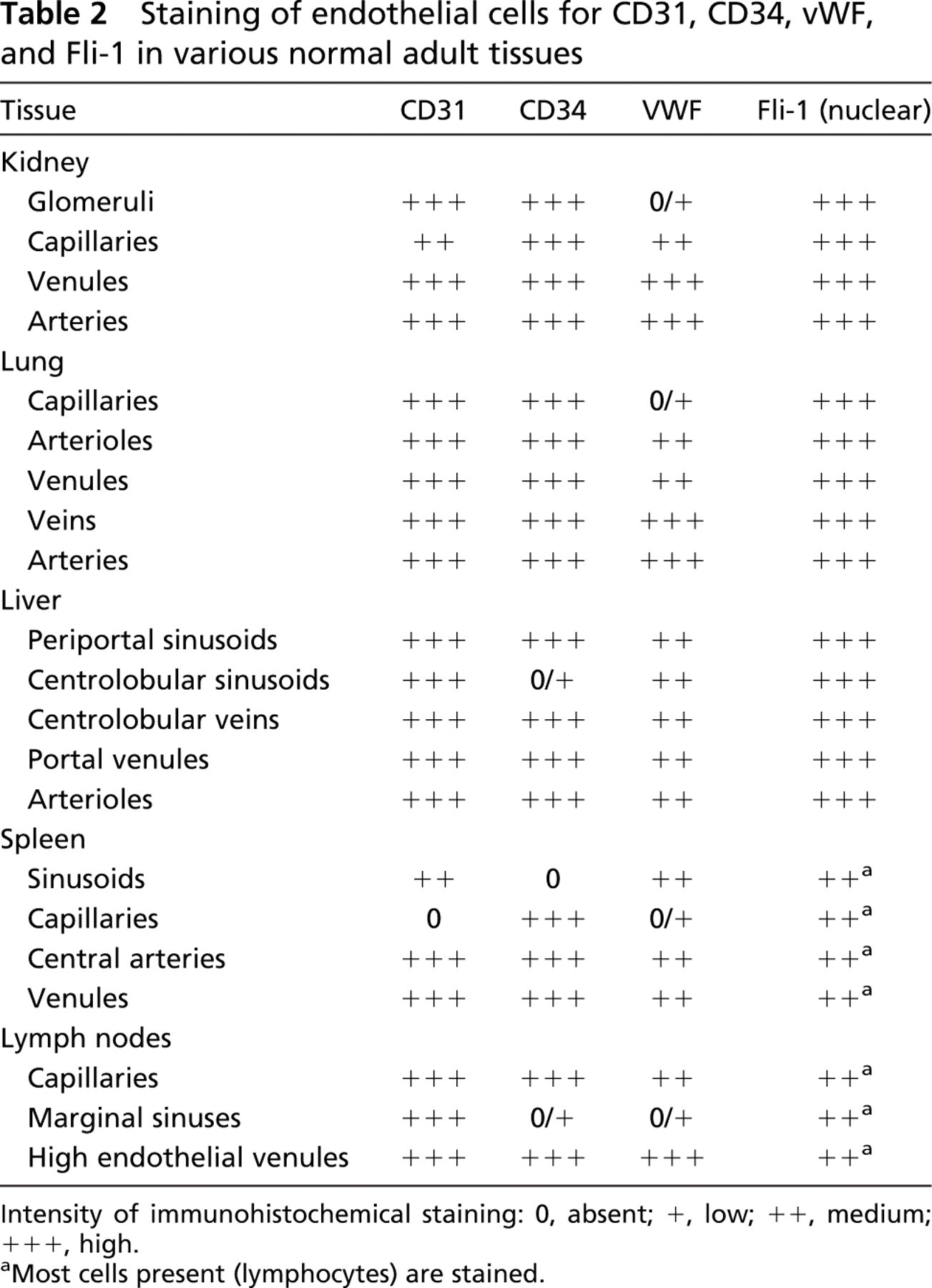
Intensity of immunohistochemical staining: 0, absent; +, low; ++, medium; +++, high.
Most cells present (lymphocytes) are stained.
(
Spleen
Sinusoidal EC were diffusely positive for CD31 but negative for CD34 (Figures 3A and 3B). In the red pulp, CD34 only marked capillaries.
vWF staining was similar to that for CD31 but slightly less intense (Figure 3C). There were no differences in the staining pattern of the central arteries, veins, and other vessels present in the spleen. Fli-1 stained the nuclei of EC present in the sinusoids but also the lymphocytes present in the white pulp. The larger vessels including the central arteries also stained (Figure 3D).
Liver
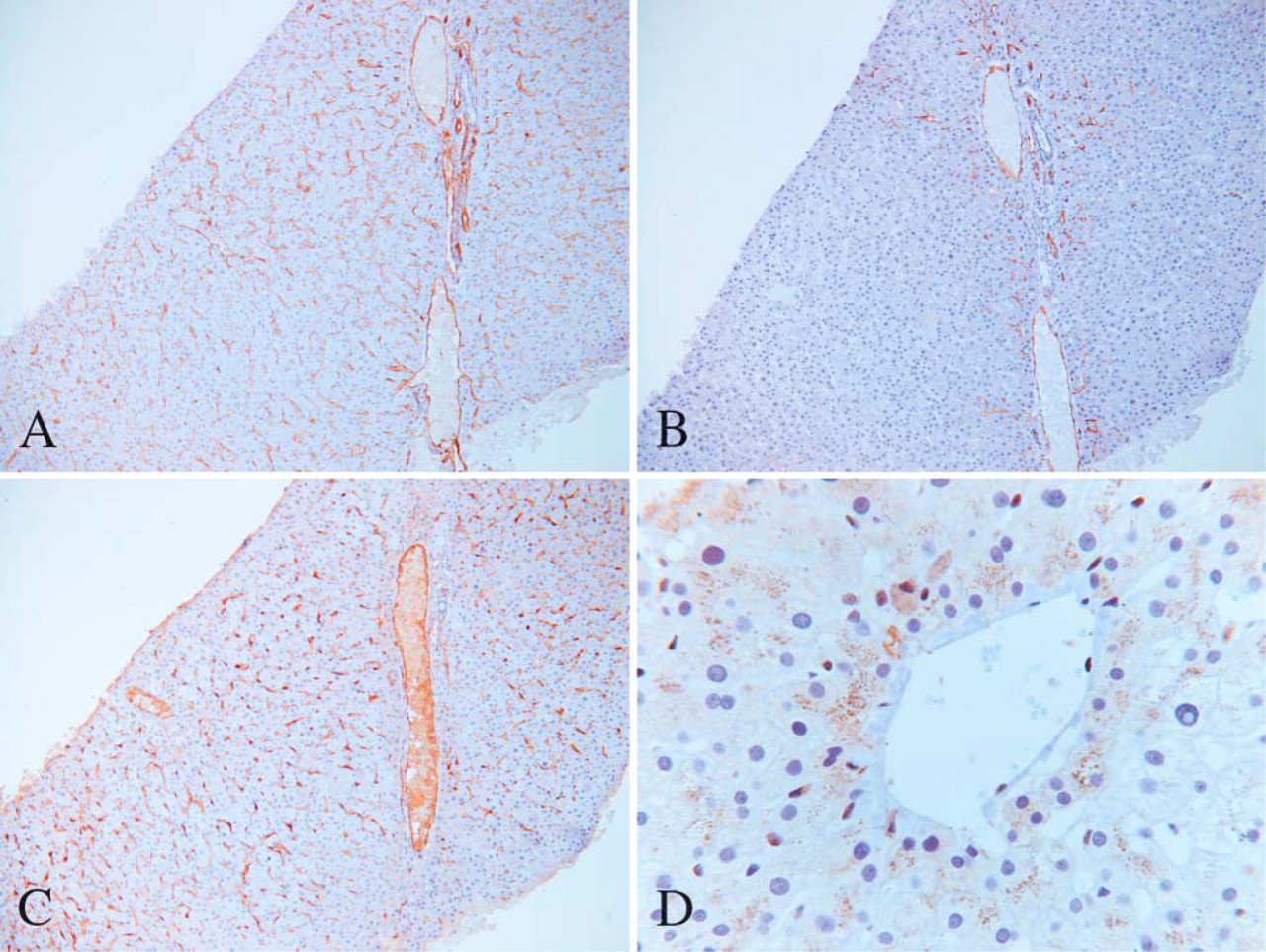
Sinusoidal EC continuously expressed CD31 from the portal space to the centrolobular vein (Figure 4A). In contrast, CD34 was expressed only in the periportal area, the centrolobular sinusoids being mostly negative (Figure 4B). Expression of vWF was similar to that of CD31 (Figure 4C). Fli-1 usually stained the nuclei of EC present in the sinusoids, portal vein, centrolobular vein, and hepatic artery (Figure 4D). A granular cytoplasmic reactivity was also occasionally seen in hepatocytes.
Heart
The staining pattern of the intramyocardial capillaries was different between CD31 and CD34. More capillaries showed CD34 than CD31 immunoreactivity. The pattern of staining for vWF was similar to that for CD31. The endocardial endothelium strongly stained for CD31 but irregularly and less intensely for CD34 and vWF. Capillaries in fat tissue were positive for CD34. EC from interstitial capillaries, arterioles, and venules present in the myocardium also stained for Fli-1. A diffuse cytoplasmic background staining of cardiomyocytes was also seen with Fli-1.
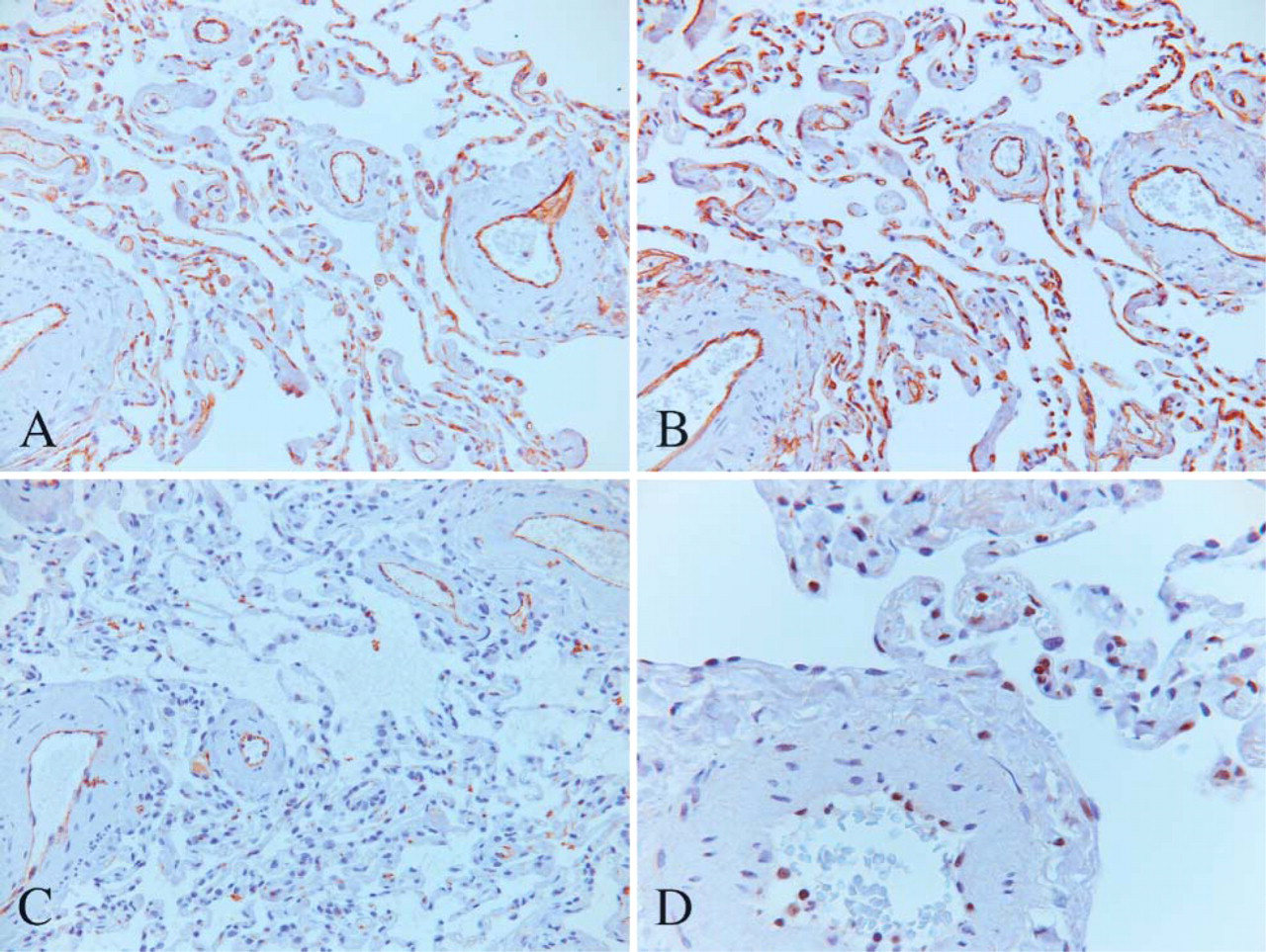
Alveolar capillaries and arterioles in the lung showing strong expression of CD31 (
Skin
Small arteries, arterioles, venules, and capillaries present in the skin were all stained with CD31, CD34, vWF, and Fli-1 in a similar fashion. Hair follicle cells were also occasionally positive for CD34. In the dermis, a reticular pattern of staining was seen with CD34, probably corresponding to CD34-positive mesenchymal cells (fibroblasts) and/or components of the extracellular matrix. Fli-1 also stained some lymphocytes present in the upper dermis and epidermis (granulation tissue).
Bone Marrow
Bone marrow sinuses, characterized by a discontinuous EC lining, were positive for CD31, CD34, and vWF. Capillaries, small arterioles, and venules present in the bone marrow in small amounts were also positive and did not show any significant differences for the above markers. Megakaryocytes were strongly positive for CD31 and vWF but negative for CD34. Myeloid and erythroid lineages cells were usually negative for the above markers, some cells occasionally being positive for CD31. In some cases a slight background staining was seen, especially with vWF. Fli-1 immunostaining in the bone marrow did not show nuclear staining, presumably due to the decalcification process. Only in one or two cases, the nuclei of some megakaryocytes and of the myeloid lineage cells were positive.
Lymph Nodes
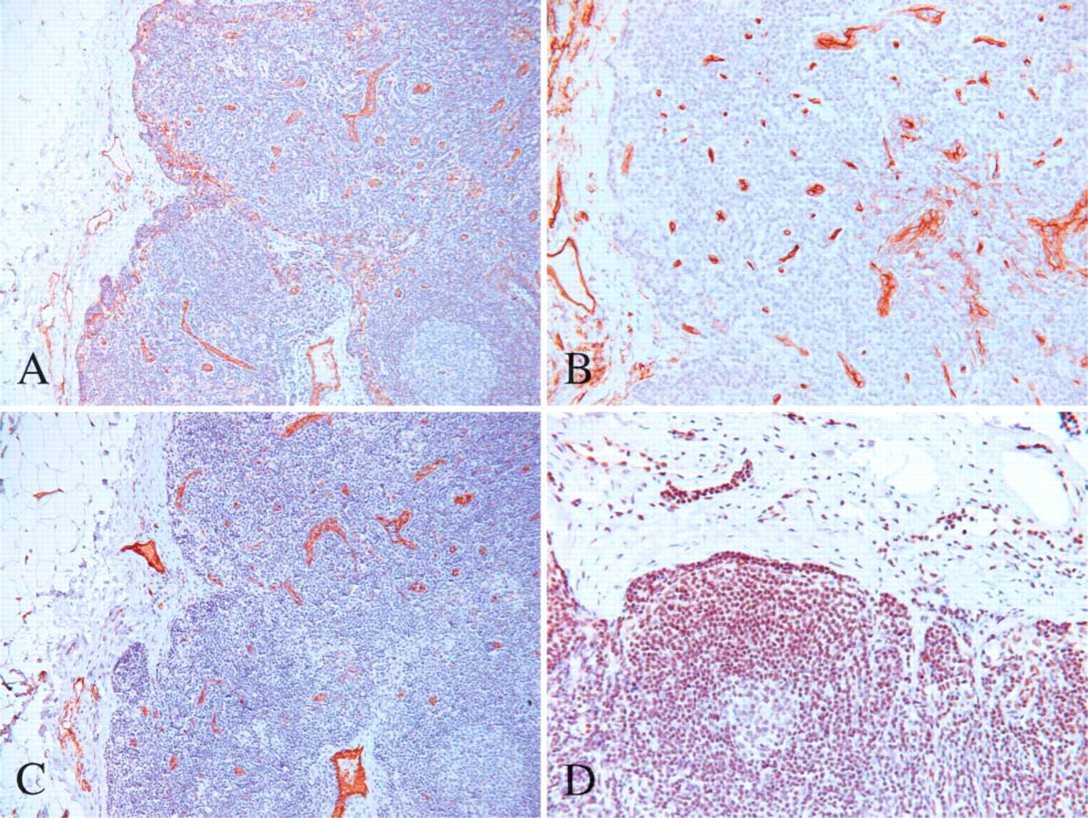
Sinusoidal EC of the marginal (subcapsular) and medullary sinuses were positive for CD31 but usually negative or focally and faintly positive for CD34 and vWF (Figures 5A-5C). In one case, sinusoidal EC of the marginal sinuses were positive for CD34 with the same intensity as for CD31. High endothelial venules were positive for CD31, CD34, and vWF. In the lymph node capsule, a reticular pattern of staining was also seen with CD34, probably corresponding to CD34-positive mesenchymal cells. Sinus histiocytes were positive for CD31 but negative for CD34 and vWF. Because Fli-1 stained most lymphocytes present in the lymph node, evaluation of EC staining was difficult, but EC at all levels seemed immunopositive for Fli-1 (Figure 5D).
(
Large Vessels
Large Arteries (Thoracic and Abdominal Aorta, Pulmonary Artery). The staining pattern of the endothelium was similar for CD31 and CD34 in the various large arteries we investigated. Due to loss of endothelial cell integrity, staining of the endothelium was often discontinuous for CD31 and CD34. This was not the case with vWF, which showed a continuous pattern of staining with the regions depleted of EC still being stained for vWF. CD34 also stained some components of the media, probably corresponding to smooth muscle cells.
Middle-sized Arteries (Renal Artery, Femoral Artery). Renal arteries taken from autopsy cases showed a continuous and strong staining for vWF. The staining pattern was usually discontinuous for CD31 and CD34, the intensity being less for vWF. CD34 also stained some components of the media, probably corresponding to smooth muscle cells.
Large Veins (Inferior Vena Cava, Pulmonary Vein) and Middle-sized Veins (Femoral Vein). The staining pattern of the endothelium was similar for CD31, CD34, and vWF for the different veins investigated.
Lymphatic Endothelium
In all parenchymatous organs, small vascular spaces morphologically consistent with lymphatics were positive for D2-40. Such vessels were present in variable numbers in different organs. They were rare in the spleen (around central arteries and in the capsule), liver (in portal spaces and the capsule), kidney, and bone marrow. In the lungs, they were usually seen around veins, arterioles, and bronchioles but were very rare or absent in the alveolar wall. In the heart, they were present in small numbers in between cardiomyocytes. Endocardial endothelium was negative for D2-40. Surprisingly, the number of D2-40-positive vessels present in lymph nodes was also low. The marginal and medullary sinuses were negative for D2-40. In the pericapsular region, D2-40-positive afferent and efferent lymph channels could rarely be identified. Few lymphatic vessels were present around arteries and veins in the adipous tissue around lymph nodes.
(
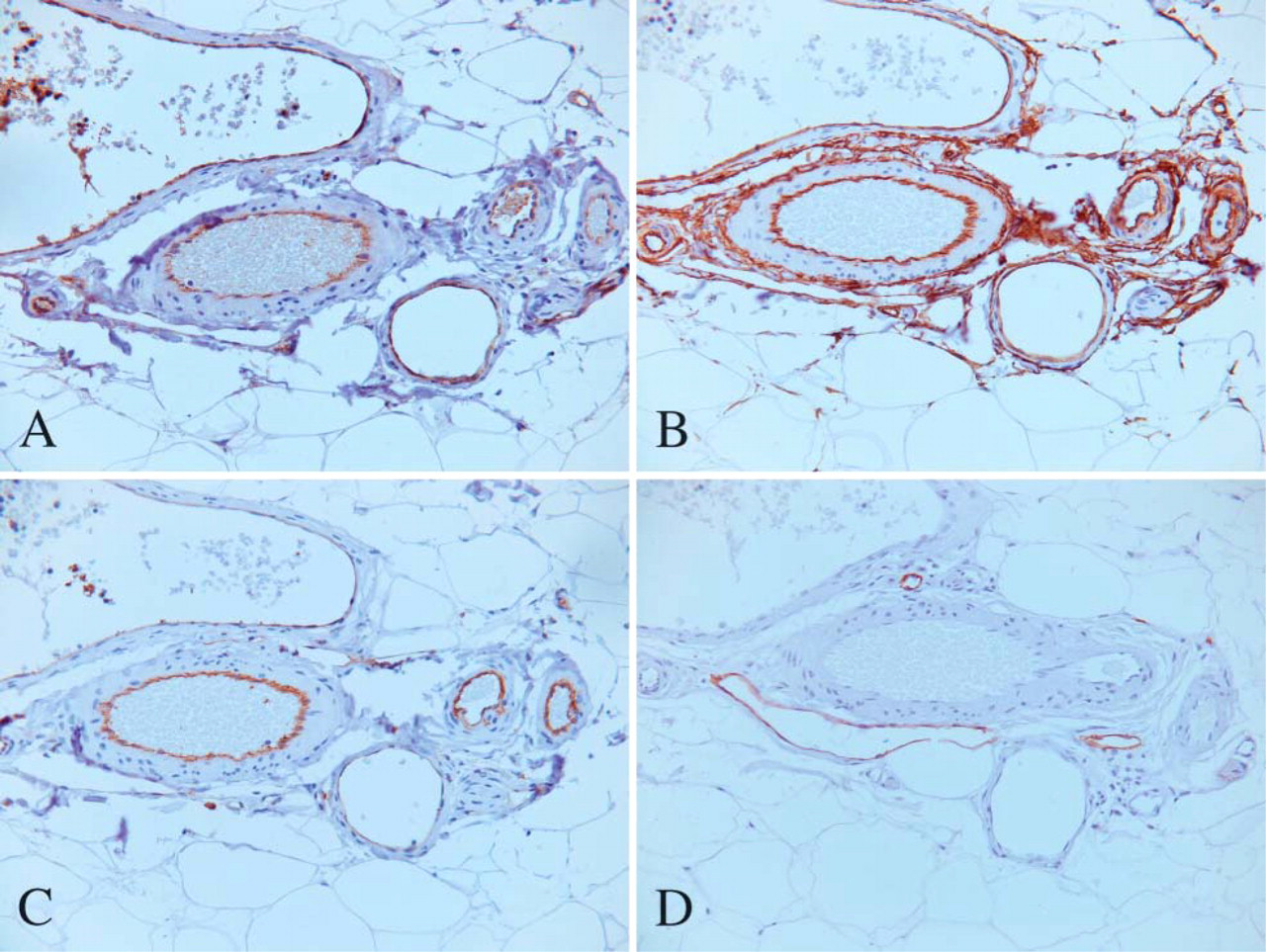
EC in arteries and veins were always negative for D2-40. CD31, CD34, vWF, and Fli-1 were occasionally and irregularly expressed in lymphatic endothelium, but the staining intensity was usually lower than in vascular endothelium and was very weak when compared with D2-40. Hence, D2-40 immunostaining gives a mirror image when compared with the other EC markers (Figures 6A-6D). Mesothelial cells from the pleura or spleen capsule were occasionally positive for D2-40. This was also the case with osteocytes in the bone and bronchial wall chondrocytes, as well as basal cells from sebaceous glands and hair follicle cells, in a pattern similar to that of CD34. In bone marrow, hematopoietic stem cells including megakaryocytes were negative for D2-40.
Discussion
This study confirms that EC in different vessel types of various organs express EC antigens heterogeneously. Variations in expression patterns were most striking in the small capillaries of the continuous, fenestrated, and sinusoid type. We could confirm studies in humans (Gorog et al. 1998; Yamamoto et al. 1998; Müller et al. 2002c) and in animals (Rand et al. 1987) that show that the level of expression of vWF in the lung vasculature is higher in larger vessels (from being almost negligible in EC in alveolar capillaries but prominent in the major vessels). We also confirmed earlier findings in the kidney concerning immunoreactivity of fenestrated endothelium of the glomeruli for CD31 and CD34 but not for vWF and show, therefore, that antigen expression differences exist between EC in the same organ (Fleming and Jones 1989; Kinjo et al. 1989). We found that vWF expression was higher in renal glomeruli of patients with hypertension. This again confirms other studies that show upregulation of vWF expression on EC in hypertension and conditions such as acute respiratory failure (Carvalho et al. 1982), acute and chronic renal insufficiency, diabetic nephropathy (Lufkin et al. 1979), vasculitis, scleroderma, and Raynaud's phenomenon (Kahaleh et al. 1981). Increased levels of circulating vWF have also been described in patients with angina pectoris, coronary thrombotic events (Gorog et al. 1998), or pulmonary hypertension (Lopez and Maeda 1998).
(
In large vessels, discontinuous immunolabeling was found for CD31 and CD34, in spite of continuous labeling for vWF. This might be explained through loss of integrity of the EC lining, which was suggested by the presence of EC positive for CD31, CD34, and vWF inside the vascular lumen. Subendothelial matrix-bound vWF could explain the continuous staining seen with vWF (Rand et al. 1991). Smith et al. (1996) showed that vWF is differentially expressed in canine large vessels. We were not able to confirm these findings in normal human blood vessels.
CD34 also stained some components in the media of large vessels. Indeed, it has been shown in earlier studies that aortic smooth muscle cells and some fibroblasts also stain for CD34 (Miettinen et al. 1994).
We found D2-40 to be a sensitive and relatively specific marker for lymphatic endothelium in all parenchymatous organs. CD31, CD34, vWF, and Fli-1 did not discriminate between vascular and lymphatic EC and are therefore panvascular EC markers. However, other cells (including mesothelial cells) also stained for D2-40. This is in agreement with a study showing that D2-40 is expressed in normal and reactive mesothelial cells and in most (93-96%) mesotheliomas (Ordonez 2005; Ordonez in press). D2-40 is also expressed in lymphangiomas, Kaposi's sarcomas, and a subgroup of angiosarcomas but not in endothelial cells of blood vessels. To our knowledge, the fact that osteocytes and chondrocytes also occasionally stain for D2-40 has not been reported previously. We can hypothesize that these D2-40-positive cells might express sialoglycoproteins similar to those of lymphatic EC and with which the antibody cross-reacts.
EC from arterioles and venules present in the skin are positive for CD31 (
An important application of EC marker immunohistochemistry is the classification of human vascular tumors. CD31 is generally regarded as the single best marker of EC differentiation because it is expressed in ~90% of EC tumors but only very rarely in carcinomas, lymphomas, and mesotheliomas (DeYoung et al. 1998; Folpe et al. 2001). However, CD31 is also expressed in macrophages, which calls for careful interpretation of CD31 expression in poorly differentiated tumors (McKenney et al. 2001). CD34 is expressed in ~90% of vascular tumors. It is, however, also expressed in many other neoplasms, including dermatofibrosarcoma protuberans, malignant peripheral nerve sheath tumors, gastrointestinal stromal tumors, and epitheloid sarcomas. Notably, CD34 negative is the littoral cell angioma of the spleen, which correlates with the lack of expression of CD34 in the sinusoids of the spleen (Ben-Izhak et al. 2001). Korkusuz et al. (2002) identified the distinctive antigenic profile of sinus lining cells, in sharing both endothelial cell and macrophage lineage characteristics. In embryogenesis and vasculogenesis, vascular and hematopoietic tissues develop together from the early mesoderm (Cines et al. 1998). Hence, it is not surprising that the antigen expression pattern of hematopoietic cells and EC overlap; both cell types express CD31, CD34, vWF, and Fli-1. A common stem cell has been postulated that can differentiate into hematopoietic or vascular cells or retain characteristics of both, such as the littoral cells of the spleen. Likewise, some tumors with EC stem cell-like phenotype or poorly differentiated angiosarcoma might not express all of the commonly used EC markers (Poblet et al. 1996) and/or might express some of the leukemia/lymphoma markers like CD34.
Under pathological conditions, EC can modify their antigen expression. In the liver, capillarization of hepatic sinusoids is a well-recognized phenomenon that occurs in long-standing liver disease and hepatic cirrhosis as well as in hepatocellular carcinoma (HCC). EC of the sinusoids that usually do not express CD34 can alter their phenotype and express this marker. On the contrary, Lyve-1, thought to be restricted to lymphatic vessels, is expressed in liver sinusoids but down-regulated in liver cirrhosis and HCC (Carreira et al. 2001). CD34 and Lyve-1 might constitute valuable tools to distinguish well-differentiated HCC from non-neoplastic liver (Ruck et al. 1995; Di Carlo et al. 2002). Consequently, EC markers (vascular and lymphatic) can also aid in the diagnosis of non-vascular tumors and of pathological processes other than tumors. The usefulness of Lyve-1 as a marker for diagnostic purposes is limited because the performance of the antibody on routinely processed tissues was very variable. We propose D2-40 for this purpose.
For diagnostic purposes, vWF is a problematic marker because it is not expressed by all EC and is retained in the adjacent extracellular matrix, which limits its reliability for identification of the presence of endothelial cells. In vascular tumors, it is expressed in only 50-75% of cases.
In our study, Fli-1 was expressed in all types of EC but was also expressed by other cell types, especially lymphocytes. The expression of Fli-1 did not change among different vessel types or organs. This marker can thus be a valuable tool in diagnosing both benign and malignant vascular tumors, especially when used in conjunction with membranous EC markers such as CD31 or CD34. The cytoplasmic reactivity seen in hepatocytes, renal tubular cells, and cardiomyocytes could be explained either by cross-reactivity with a cytoplasmic protein or by the presence of Fli-1 protein in the cytoplasm of these cells.
In conclusion, this study confirms that EC antigen expression varies among different vessel subtypes and different anatomic compartments of the same organ. The antigen expression pattern in a given organ or vascular bed is dynamic and can change due to a variety of pathological processes or simply in senescence (Müller et al. 2002b). Phenotypic differences of EC found in different vascular beds suggest highly differentiated site-specific functions of EC. This may be the result of specific characteristics of EC subtypes and a tissue-specific microenvironment.
Footnotes
Acknowledgments
We thank Magalie Cornavon, Christine Brueger, Solange Gros, and Marie-Martine Bertholet for expert immunochemistry support.
