Abstract
Four distinct genes encode tropomyosin (Tm) proteins, integral components of the actin microfilament system. In non-muscle cells, over 40 Tm isoforms are derived using alternative splicing. Distinct populations of actin filaments characterized by the composition of these Tm isoforms are found differentially sorted within cells (Gunning et al. 1998b). We hypothesized that these distinct intracellular compartments defined by the association of Tm isoforms may allow for independent regulation of microfilament function. Consequently, to understand the molecular mechanisms that give rise to these different microfilaments and their regulation, a cohort of fully characterized isoform-specific Tm antibodies was required. The characterization protocol initially involved testing the specificity of the antibodies on bacterially produced Tm proteins. We then confirmed that these Tm antibodies can be used to probe the expression and subcellular localization of different Tm isoforms by Western blot analysis, immunofluorescence staining of cells in culture, and immunohistochemistry of paraffin wax-embedded mouse tissues. These Tm antibodies, therefore, have the capacity to monitor specific actin filament populations in a range of experimental systems.
T
Because of the vast array of Tm isoforms, a number of studies have addressed the functional significance of individual isoforms. Tm3, but not Tm5NM1, is involved in intracellular granule movements (Pelham et al. 1996). γTm isoforms are associated with a subpopulation of vesicles derived from the Golgi apparatus (Heimann et al. 1999; Percival et al. 2004). In the yeast Saccharomyces cerevisae, TPM1 and TPM2 have isoform-specific functions and are implicated in polarized cell growth and secretion (Drees et al. 1995; Pruyne et al. 1998). In fission yeast Schizosaccharomyces pombe, cdc8 tropomyosin is essential for cytokinesis (Bala-subramanian et al. 1992). High-molecular-mass tropomyosins, mainly Tm1, 2, and 3, have been implicated in transformation (Pawlak and Helfman 2001). Gene transfection studies have demonstrated that tropomyosin isoforms can differentially regulate the organization of actin filaments (Bryce et al. 2003).
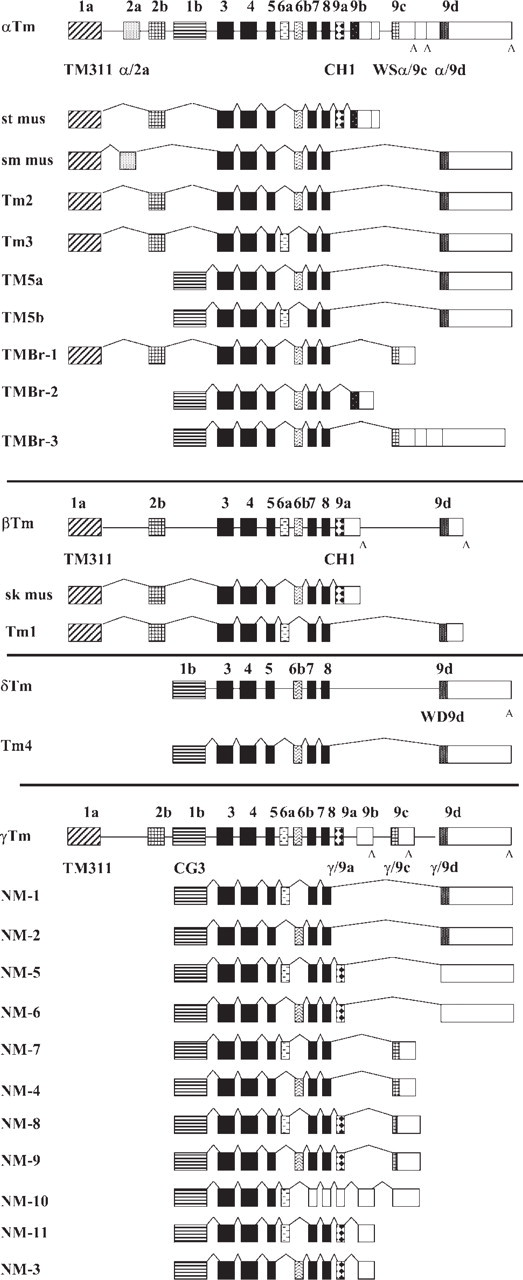
Schematic representation of the organization of the α, β, δ, and γ mammalian Tm genes and the respective isoforms generated following alternative splicing. Exons are numbered 1–9 as described by Lees-Miller and Helfman (1991), and the same numbering between genes is used but is not indicative of homology. Exons are shaded boxes, unshaded boxes correspond to 3’ untranslated ∗∗∗sequences, and introns are shown as lines. The black-shaded exons are common to all genes. The name of the Tm antibody is indicated in bold below the exon where the epitope is found.
Subcellular localization studies using isoform-specific antibodies further support isoform-specific functions of Tms. Immunohistochemical studies, both in vivo and in cells in culture, have clearly demonstrated that Tm isoforms mark distinct populations of actin microfilaments (reviewed in Gunning et al. 1998a,b). Such distinct intracellular compartments defined by the association of Tm isoforms may allow for independent regulation of specific microfilaments. A comprehensive characterization study of both actin and Tm antibodies is described in this report, using Western blotting, immunohistochemistry of mouse tissues, and immunofluorescence staining of mouse fibroblasts in culture. These antibodies should have broad utility in the evaluation of tropomyosin function and regulation of the actin cytoskeleton.
Materials and Methods
Antibodies
The primary rabbit polyclonal antibodies were used for Western blots at the following dilutions: WSα/9c antiserum at 1:250 and WD4/9d (Hannan et al. 1998) at 1:400. The mouse monoclonal antibodies were used at the following dilutions: β-actin (clone AC-74) (Gimona et al. 1994) at 1:1000 (Sigma Aldrich; Castle Hill, Australia); CG3 (IgM class) at 1:250 (kind gift of Dr. Jim J. Lin); C4 total actin at 1:500 (kind gift of Dr. Jim Lessard); TM311 (T-2780; Sigma Aldrich) (Nicholson-Flynn et al. 1996) at 1:500; monoclonal anti-tropomyosin (sarcomeric, clone CH1) (Lin et al. 1985) at 1:50 (T-9283; Sigma Aldrich); and monoclonal anti-α-smooth-muscle actin (clone 1A4) (Skalli et al. 1986) at 1:400 (Sigma Aldrich). The primary sheep polyclonal antibodies were used at the following dilutions: α/9d at 1:400 (Schevzov et al. 1997), γ/9a at 1:100, γ/9c at 1:100 (Vrhovski et al. 2003), α/2a at 1:100, γ/9d at 1:100, and γ-actin at 1:1000. The α/2a, γ/9d, and γ-actin antibodies were made essentially as described by Vrhovski et al. (2003). In brief, the specific peptides were synthesized (see Table 1) and conjugated to diphtheria toxin (Mimotopes; Melbourne, Australia). The peptides were injected into sheep in four equal doses of 1.3 mg at the Institute for Medical and Veterinary Sciences, Veterinary Services Division (Adelaide, Australia) following their standard protocol. A total of 5 ml of serum was affinity purified (Mimotopes) using sepharose coupled to the peptide column. For immunofluorescence staining and immunohistochemistry, the following dilutions were used: WSα/9c at 1:250, α/9d at 1:400, WD4/9d at 1:400, β-actin (clone AC-74) at 1:1000, CG3 at 1:250, C4 total actin at 1:500, TM311 at 1:500, α/2a at 1:100, γ/9a at 1:50, γ/9c at 1:50, γ/9d at 1:100 (Percival et al. 2004), and γ-actin at 1:1000. The secondary antibodies used for immunofluorescent staining were Alexa Fluor 488 donkey anti-mouse IgG (H + L) or IgM (H + L) conjugate, Cy3 donkey anti-sheep IgG (H + L), and Cy3 donkey anti-rabbit IgG (H+L) (Jackson ImmunoResearch Laboratories, Inc.; Westgrove, PA) at 1:1000.
List of tropomyosin antibodies
Culturing of Primary Mouse Embryonic Fibroblasts
A 13.5 embryonic day (ED) pregnant ARC mouse was euthanized by cervical dislocation; five embryos were isolated, and their limbs, heads, and internal organs removed. The embryos were transferred into a sterile 15-ml tube with sufficient 0.25% Trypsin/EDTA (Invitrogen Life Technologies; Melbourne, Australia) to cover them, finely minced, and incubated for15 min at 37C. The tissue was dissociated by passing it several times through a glass Pasteur pipette. Large pieces of cellular debris were allowed to settle and the supernatant was transferred to a 154-cm2 flask with media (DMEM, 10% fetal calf serum, 1% penicillin/streptomycin, 1% l-glutamine, 0.2% β-mercaptoethanol) and cultured for ∼3–4 days at 37C with 5% CO2.
Gel Electrophoresis and Immunoblotting
Six-month-old ARC mice were euthanized by cervical dislocation. The major organs were quickly dissected and frozen in liquid nitrogen. The stomach was cut open and its contents removed, and rinsed several times with cold PBS before being frozen. The tissue was dissociated in a sufficient volume of 50 mM Tris-Cl, pH 8, using a Polytron blender. Protein concentration was estimated using a BCA protein detection kit (Pierce; Rockford, IL). The proteins were precipitated as previously described (Wessel and Flugge 1984) and solubilized in SDS solubilization buffer (1x buffer contains 0.125 M Tris, pH 6.8, 0.5% SDS, 5% glycerol, 5% 2-mercaptoethanol, and 0.005% bromophenol blue). SDS-PAGE was performed according to Laemmli (1970) on 12.5% acrylamide and 0.1% bis-acrylamide. Prestained molecular-mass markers were used (BenchMark PreStained Protein Ladder; Invitrogen). Proteins were transferred to Immobilon-P PVDF (Millipore Corp.; Bedford, MA) for 2 hr at 80 V according to Towbin et al. (1979). A 5% low-fat skim milk in TBS (100 mM Tris-Cl, pH 7.5, 150 mM NaCl) solution was used to block nonspecific binding on the blot. Primary and secondary (anti-rabbit, anti-sheep, and anti-mouse Ig-conjugated horseradish peroxidase; Amersham Biosciences Pty, Ltd.; Sydney, Australia) antibodies were incubated for 1 to 2 hr each, and 4 × 15 min washes with TTBS (TBS with 0.05% Tween 20) were carried out following each antibody incubation. Blots were developed with the Western Lighting Chemiluminescence Reagent (PerkinElmer Life Sciences; Boston, MA) and exposed to Fuji X-ray Film (Kodak; Rochester, NY). Equal protein loading on the Western blots was verified by staining the polyvinylidene fluoride membrane, following transfer, with 0.5% Ponceau-S red/1% acetic acid solution for 1 min. Blot was destained with Milli-Q-grade water (Millipore) to the desired contrast.
Immunofluorescence Staining
Primary mouse embryonic fibroblasts were prepared for immunofluorescence staining by first culturing them in eight-well glass chamber slides (∼5000 cells/well). Cells were fixed in 4% paraformaldehyde in PBS for 15 min (except for γ/9a, see method below), rinsed three times with PBS, and then permeabilized with chilled methanol for 15 min. For the γ/9a antibody, cells were fixed for 45 min in a mixture of 4% paraformaldehyde and 0.1% glutaraldehyde (Sigma Aldrich) in PBS followed by permeabilization with chilled methanol as described above. The primary fibroblasts were then incubated in blocking solution/wash solution (2% fetal bovine serum in PBS) for 30 min and then incubated with the primary antibody diluted in PBS for 1 hr at room temperature. The cells were rinsed three times with wash solution and then incubated with the secondary antibody for 1 hr. Cells were finally rinsed three times in PBS before mounting a coverslip onto the chamber slides with 1,2-diazabicyclo (2.2.2) octane (Sigma Aldrich). Slides were examined with a Leica TCS SP2 laser scanning confocal microscope (Leica Microsystems Pty, Ltd; Wetzlar, Germany) using the x63 objective. Primary fibroblasts were optically sectioned in the x-y plane, and serial sections of 0.5 μm-thickness were taken with 10-scan averaging. Images were processed using the Image Pro Plus 4.1 (Media Cybernetics; Singapore, Republic of Singapore).
Immunohistochemistry
Immunohistochemistry was performed essentially as described by Weinberger et al. (1996). Major mouse organs were dissected from 6-month-old ARC mice, fixed in 10% neutral-buffered formalin, and embedded in paraffin, and 8-μm parasagittal sections were cut onto poly-l-lysine-coated glass slides (Lomb Scientific; Sydney, Australia). The sections were blocked with normal goat serum for 30 min or preimmune serum as a negative control. Primary antibody was incubated at the same dilution as that used for immunofluorescence staining for 2 hr, followed by secondary antibody for 1 hr at 1/1000 (donkey anti-sheep; goat anti-rabbit, or goat anti-mouse conjugated to alkaline phosphatase; Jackson ImmunoResearch). Immunoreactivity was visualized by nitroblue tetrazolium/ 5-bromo-4-chloro-3-indolyl phosphate (Invitrogen, La Jolla, CA) for 40–60 min. Slides were counterstained using nuclear fast red for 1 min prior to dehydration. A number of serial sections were analyzed, and the different antibodies were incubated and developed concurrently. General tissue morphology was visualized by hematoxylin and eosin stain. Phase-contrast images were captured using a Spot II cooled CCD digital camera (Diagnostic Instruments Inc.; Sterling Heights, MI) mounted on an Olympus Bx50 microscope (Olympus Australia Pty, Ltd, Melbourne, Australia).
Tropomyosin Expression Constructs
The pPROEX HT prokaryotic expression system (Gibco BRL, Invitrogen; Melbourne, Australia) was used for the production of recombinant Tm isoform proteins. The Tm cDNAs (Tm2, 3, 5a, 5b, and NM1 in pGEX plasmids originally supplied by D. Helfman and Tm1, Tm4, TmBr1, Br2, Br3, and Tm5NM2) were PCR amplified and cloned into the pPROEX HT vector using restriction enzymes engineered into the PCR primers. All Tm constructs were verified by sequencing. Induction of recombinant pPROEX HT clones was done according to the protocols outlined by Gibco BRL, Invitrogen; Melbourne, Australia. In brief, DH5α strain of Escherichia coli carrying the Tm constructs was cultured in the presence of 100 μg/ml of ampicillin to a density of A590 of 0.5–1.0. To induce expression of the recombinant Tm proteins, isopropyl-β-d-thiogalactopyranoside (IPTG) was added to a final concentration of 0.6 mM. A 1-ml aliquot of cells was removed after 2 hr induction.
Preparation of Recombinant Tm Isoforms
IPTG-induced bacterial cells were pelleted, resuspended in lysis buffer (50 mM Tris, pH 8, 1 mM EDTA, 100 mM NaCl), and sonicated. The suspension was centrifuged to remove cell debris. The supernatant was heated at 95C for 10 min and cooled on ice. The denatured proteins were removed by centrifugation, and the remaining supernatant was enriched for recombinant Tm proteins. Protein concentration was estimated using a BCA protein detection kit (Pierce).
Two-dimensional Gel Electrophoresis
Human acute lymphoblastic leukemia CCRF-CEM cell pellets were resuspended in lysis buffer (7 M urea, 2 M thiourea, 2% CHAPS, 1% sulfobetaine 3–10, 1% amidosulfobetaine-14, 2 mM tributyl phosphine, 65 mM dithiothreitol, 1% carrier ampholyte 3–10, 1% carrier ampholyte 4–6, 0.01% bromophenol blue) as previously described (Verrills et al. 2003). Narrow-range immobilized pH gradient strips, pH 4.5–5.5 (Pharmacia; Uppsala, Sweden) were rehydrated in 500 μl lysis buffer, cup-loaded with 100 μg of total cellular protein, and isoelectrically focused before separation in the second dimension as previously described in detail (Verrills et al. 2003). Proteins were either transferred onto membranes and probed with the corresponding antibodies, β-actin, γ-actin, and C4 (total actin antibody), or stained with SYPRO Ruby.
Results
Specificity of the Tm Antibodies
The exon/intron structure of the four Tm genes is shown in Figure 1, together with the different Tm isoforms derived from each gene. The Tm isoforms can be divided into two major groups, the high-molecular-mass group containing 284 amino acids (apparent molecular mass of 32–40 kDa) and the low-molecular-mass group containing 248 amino acids (30 kDa). Sequence diversity is restricted to regions encoded by the amino and carboxyl end exons, as well as one internal exon. The amino terminal exons 1a and 2b have 88% homology between the Tm genes. The internal exons 6a and 6b have 44% and 76% homology between the genes, respectively. Finally, the carboxyl terminal exons 9a, 9c, and 9d have 82%, 60%, and 37% homology between the genes, respectively. Exons 3, 4, 5, 7, and 8 are common to all the genes with 84% to 88% homology among the exons between all the genes.
Ten Tm antibodies have been generated and are listed in Table 1. Figure 1 shows the name of the antibody below the exon where the epitope is found. To confirm the specificity of the antibodies, recombinant Tm proteins were used. As described in Materials and Methods, the cDNA of each corresponding Tm isoform was cloned into the pPROEX HT prokaryotic expression system. Expression of the recombinant Tm proteins was induced, and the total bacterial lysate was heated at 95C for 10 min to enrich for the recombinant Tm proteins.
The mouse monoclonal TM311 antibody detects the amino terminal exon 1a of the αTmfast, βTm, and βTm genes; hence, it detects recombinant Tm1, 2, 3, and Br-1 (Figure 2A) but none of the products with an exon 1b N terminus. The sheep α/9d antibody was raised against a peptide corresponding to the entire exon 9d of the αTmfast gene (Figure 1). Consequently, the antibody detects Tm1, 2, 3, 5a, and 5b and exhibits no cross-reactivity with exon 9d containing Tm isoform from the δ (Tm4) gene but very low cross-reactivity with the isoforms from the γ (Tm5NM1, Tm5NM2) Tm gene (Figure 2B). The rabbit WSα/9c antibody raised against a peptide corresponding to exon 9c of the αTmfast gene only detects TmBr-1 and TmBr-3 (Figure 2C). The rabbit WD4/9d antibody raised against 18 amino acids of exon 9d of the δTm gene preferentially detects Tm4 but exhibits cross-reactivity with Tm1 from the βTm gene, Tm2 and 3 from the αTmfast, and very weak reactivity with Tm5NM1 and Tm5NM2 (Figure 3D). The epitope for the mouse monoclonal CG3 antibody has been mapped to exon 1b amino acids 29–44 of the γTm gene (Novy et al. 1993), and it detects all products from this gene including Tm5NM1 and NM2 (Figure 2E). The sheep γ/9d antibody raised against a peptide corresponding to the entire exon 9d of the γTm gene was found to detect both Tm5NM1 and NM2 (Figure 2G). The two bands seen correspond to the his-tagged bacterial products. The γ/9d antibody was also found to show slight cross-reactivity with the other exon 9d containing isoforms from both the αTmfast and βTm gene, including Tm1, 2, 3, 5a, and 5b. Due to the high degree of homology among exon 9d of the α and γTm genes, the α/9d and γ/9d antibodies recognize the same set of Tm isoforms but with different avidity. Finally, the sheep γ/9c antibody raised against exon 9c of the γTm gene was found to show slight cross-reactivity with the other exon 9c containing isoforms from the αTmfast, TmBr-1, and TmBr-3, 34, and 30 kDa, respectively (Figure 2F). However, it does detect the appropriate product in brain extracts of 30 kDa (Vrhovski et al. 2003).
Expression Profile of Tm Isoforms in Mouse Tissues
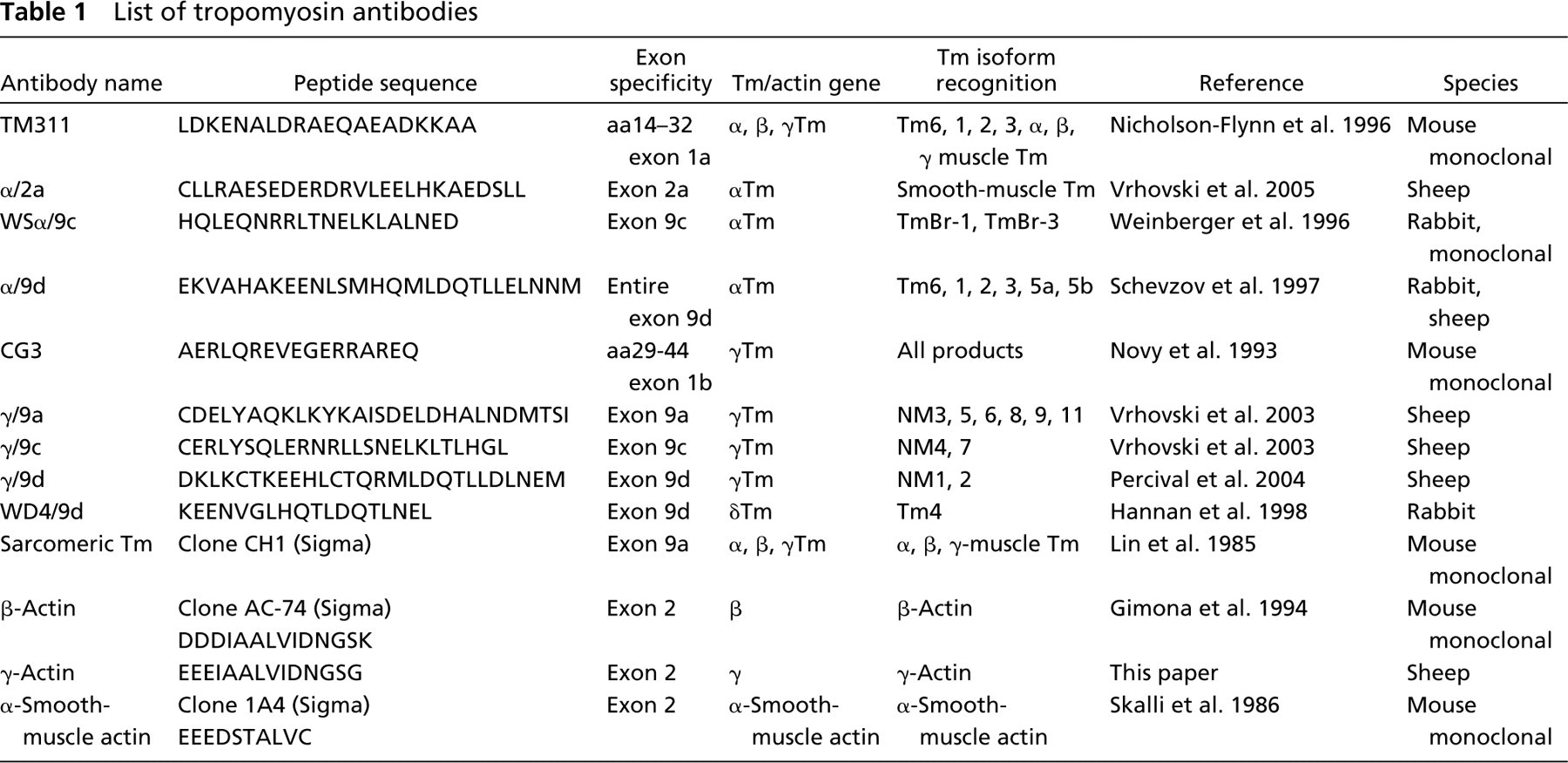
The Tm antibodies were used to identify the repertoire of isoforms expressed in different mouse tissues. Although individual mouse tissues are composed of more than one cell type, Western blot analysis allows for the relative comparison of the major Tm isoforms expressed in different tissues. Figure 4 shows representative Western blots probed with five Tm antibodies generated to detect isoforms from the αTmfast and βTm genes. The TM311 detects a number of bands migrating between 34 and 40 kDa. In heart and quadriceps muscle, the major products are the sarcomeric Tms α and β. Heart expresses only α, whereas quadriceps muscle expresses both α and β (Figure 3A). The same result is obtained with the monoclonal anti-sarcomeric tropomyosin antibody (clone CH1) (Figure 3E). Although the epitope of this antibody (anti-sarcomeric Tm antibody) is yet to be mapped, the antibody is thought to detect exon 9a of the α, β, and γTm genes. Accordingly, the anti-sarcomeric tropomyosin antibody detects the muscle Tm isoforms α, β, and γTm. However, the presence of non-muscle exon 9a containing isoforms from the γTm gene is undetected by this antibody in any of the non-muscle tissues examined, even after longer exposure of the Western blot to the X-ray film (data not shown). This suggests that it has limited reactivity with the 9a exon of the γTm gene (see below).

Testing the specificity of the Tm antibodies with bacterially produced Tm proteins. Expression of the Tm isoforms was induced with isopropyl-β-d-thiogalactopyranoside (IPTG). The bacterial sample was induced for 2 hr with IPTG, and the cell lysate was enriched for recombinant Tm proteins by heating at 95C for 10 min. Two hundred fifty ng of recombinant Tm proteins was electrophoresed on 12.5% low-bis SDS gel, followed by immunoblotting with the respective Tm antibodies. (
In the lung, spleen, and stomach. the TM311 antibody detects Tm6 and 1, together with a low-molecular-mass band (36 kDa) that can correspond to either smooth-muscle αTm or a non-muscle Tm isoform, Tm2. To confirm the identity of these additional bands, we compared this Western blot with that probed with the α/9d antibody (Figure 3B). Both the TM311 and α/9d antibodies detect the non-muscle Tms6, 1, 2, and 3, with the TM311 antibody also detecting TmBr-1. Hence, in the brain, a very faint band corresponding to TmBr-1 is seen (Figure 3A). The lung, spleen, and stomach express Tm6 and 1 as detected with the TM311 and α/9d antibodies (Figures 3A and 3B). The lung, spleen, and stomach also express Tm2, which has the same molecular mass as α-striated Tm. Kidney and brain express much lower levels of Tm6 and 1. Relative to Tms 6 and 1, kidney expresses a significant amount of Tm2 (Figure 3B). The α/9d antibody also detects the low-molecular-mass isoforms Tm5a and 5b, with Tm5a being preferentially expressed only in the brain and Tm5b being the predominant isoform in kidney, liver, and lung relative to Tm6, 1, and 2. Tm5b was undetectable at this sensitivity in heart and quadriceps muscle but was present in spleen, stomach, and, to a lesser extent, in embryonic primary fibroblasts. Tm3 was detected only in embryonic fibroblasts and (possibly) stomach.
Expression of Tm isoforms from the αTm and βTm gene in different mouse tissues. Equal loading (20 μg) of total cellular protein (except for quadriceps muscle, 10 μg) isolated from mouse tissues was electrophoresed on 12.5% low-bis SDS gels, and individual blots were immunoblotted with the (
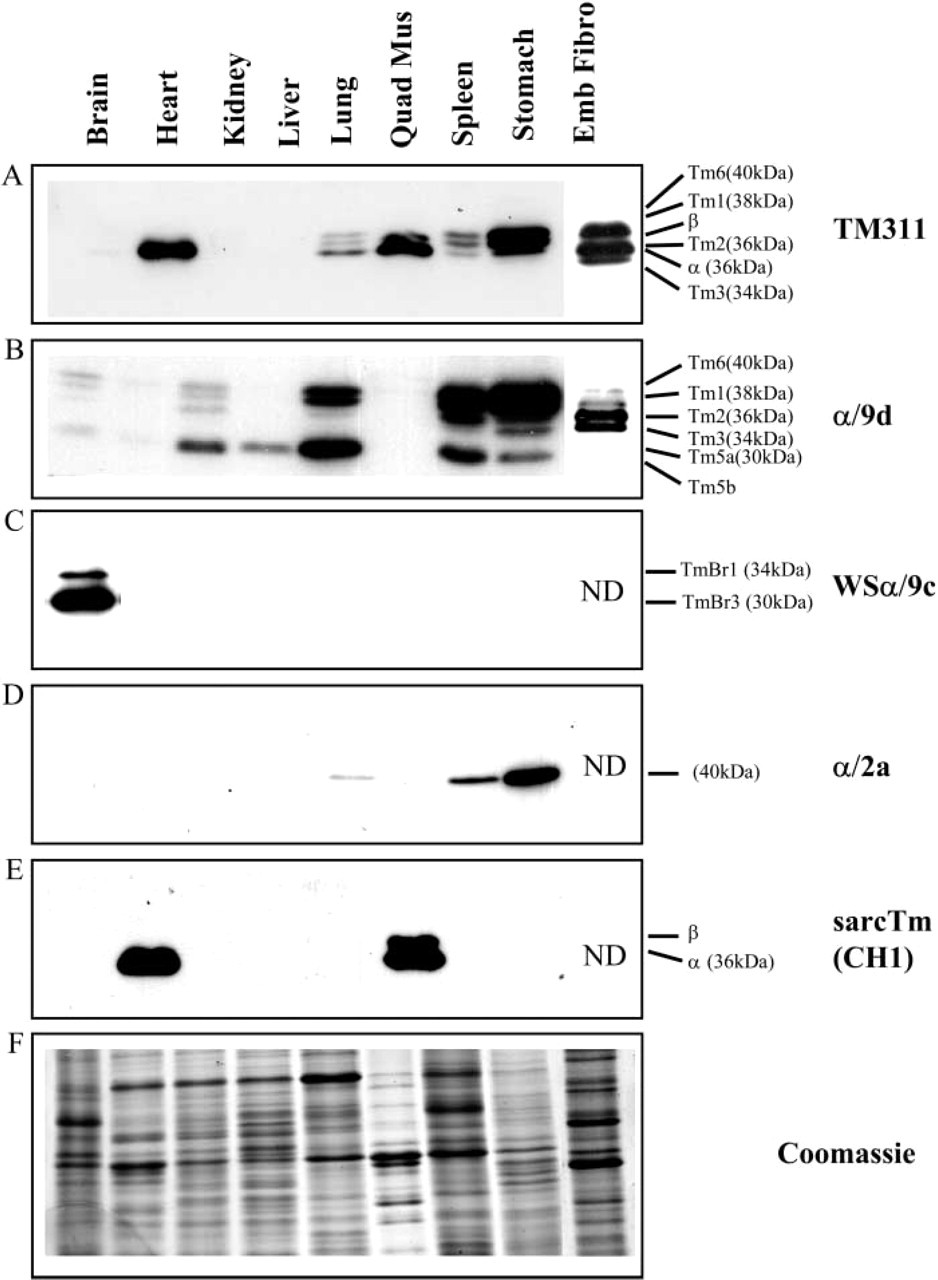
Confirmation of the apparent molecular mass of smooth α Tm isoform. Mouse stomach sample, 10 μg, was electrophoresed on 12.5% low-bis SDS gels and immunoblotted with Lane 1, α/9d; Lane 2, α/2a; and Lane 3, TM311.
The WSα/9c antibody detected the brain-specific isoforms TmBr-1 and Br-3 only in the brain, with apparent molecular mass of 34 and 30 kDa, respectively (Figure 3C).
The α/2a antibody was raised against a peptide corresponding to the unique exon 2a of the αTmfast gene, not found in any other Tm isoform. It detects α-smooth Tm isoform (apparent molecular mass, 40 kDa) expressed at high levels in the stomach but also seen in the spleen and, to a lesser extent, in the lung (Figure 3D). In Figure 4, a sample of stomach was probed with the α/9d, α/2a, and TM311 antibodies. The α/2a antibody unambiguously recognizes Tm6, also known as α-smooth Tm, (40 kDa) in the stomach but not Tm1 seen with both the α/9d and TM311 antibodies. The Tm6 band was found to be heat stable (data not shown). This does not absolutely prove that there is only α-smooth Tm in the Tm6 band, but it does unambiguously show that the α-smooth Tm migrates at the position of Tm6. A complete characterization of the α/2a antibody is published separately (Vrhovski et al. 2005) because this is the first time that such an antibody has become available. The conclusion reached from the Vrhovski et al. data is that both Western blots and immunohistochemistry on human and mouse lung with the α-smooth Tm antibody (α/2a) showed an identical profile and tissue colocalization with α-smooth actin.
The antibody specific to Tm4, WD4/9d, detects Tm4 (30 kDa) in all the mouse tissues examined except for the sample of quadriceps muscle, where Tm4 was undetectable at this sensitivity (Figure 5A). A higher-molecular-mass band expressed in lung, spleen, stomach, and embryonic fibroblasts was also detected. This band is proposed to be either Tm1, inasmuch as a similar pattern is seen with the α/9d antibody that also detects Tm1, (Figure 3A) or an unknown isoform. To confirm the identity of this 38-kDa band, protein extracts from a Tm1 stably transfected cell line (Yager et al. 2003) was probed with the WD4/9d and α/9d antibodies. The WD4/9d antibody failed to detect the 38-kDa Tm1 band (Figure 5B) seen with the α/9d antibody in the Tm1-expressing cells (Figure 5C).
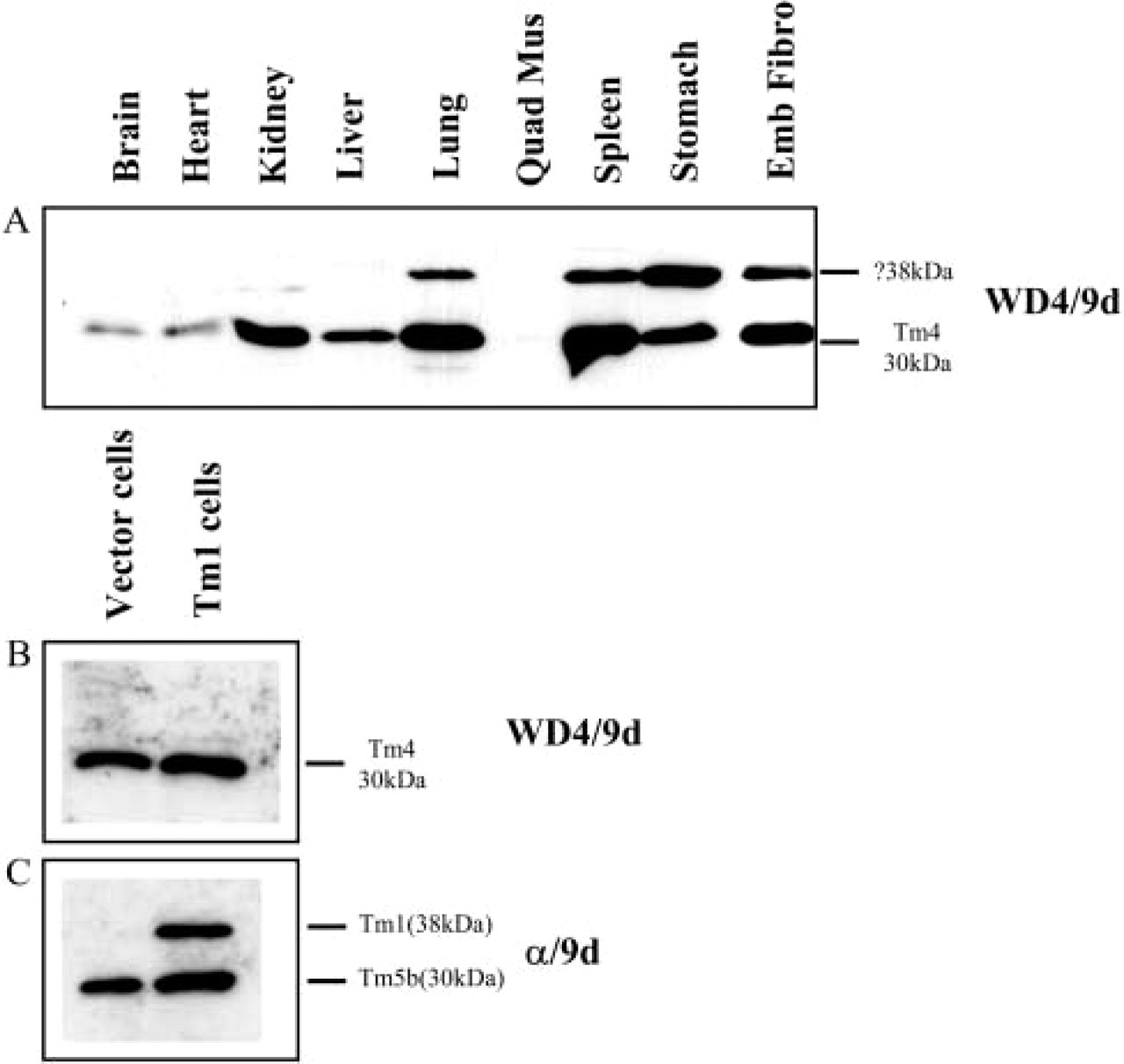
Expression of Tm4 from the δTm gene in different mouse tissues. (
A total of four antibodies have been generated to identify Tm isoforms derived from the γTm gene. The CG3 antibody generated by Lin and co-workers (Novy et al. 1993) recognizes exon 1b of the γTm gene and thus detects all non-muscle products from the γTm gene. A band of apparent molecular mass of 30 kDa is detected in all the mouse tissues including the heart seen on a longer exposure (not shown here) but was undetected in the quadriceps muscle (Figure 6A). The peptide used to generate the γ/9a antibody is derived from exon 9a of the γTm gene. This exon is highly homologous (81.5%) among the different Tm genes. Therefore, it is predicted that the γ/9a antibody would detect the muscle Tm isoforms derived from the α, β, and γTm genes, together with the non-muscle Tm isoforms from the γ Tm gene, NM5 NM6, NM8, NM9, NM11, and NM3 (Figure 1). As shown in Figure 7B, the two muscle samples, heart and quadriceps muscle, express a predominant band, 36 kDa, corresponding to the αTm muscle isoform, with the quadriceps muscle also expressing βTm, seen in a shorter exposure (not shown). The lung and stomach also express the αTm muscle isoform but to a lesser extent, with the βTm muscle isoform faintly present in the stomach. A similar result was obtained with both the TM311 and sarcomeric Tm (CH1) antibodies (Figures 3A and 3E) that can also detect the α and β muscle Tm isoforms containing exon 9a. The brain, lung, and spleen also expressed a lower-molecular-mass band, 30 kDa, corresponding to the non-muscle exon 9a containing isoforms from the γTm gene. Once again, on a longer exposure the 30-kDa band was also detected in kidney, liver, and embryonic primary fibroblasts (data not shown). Interestingly, CH1 does not detect these 30-kDa products, even on long exposures. This suggests that CH1, although recognizing the 9a exons from the α and β genes, shows only poor reactivity with the same exon from the γ gene. This also strongly supports the conclusion that these products are indeed from the γ gene. RT-PCR studies have previously confirmed the expression of 9a containing products from this gene (Dufour et al. 1998).
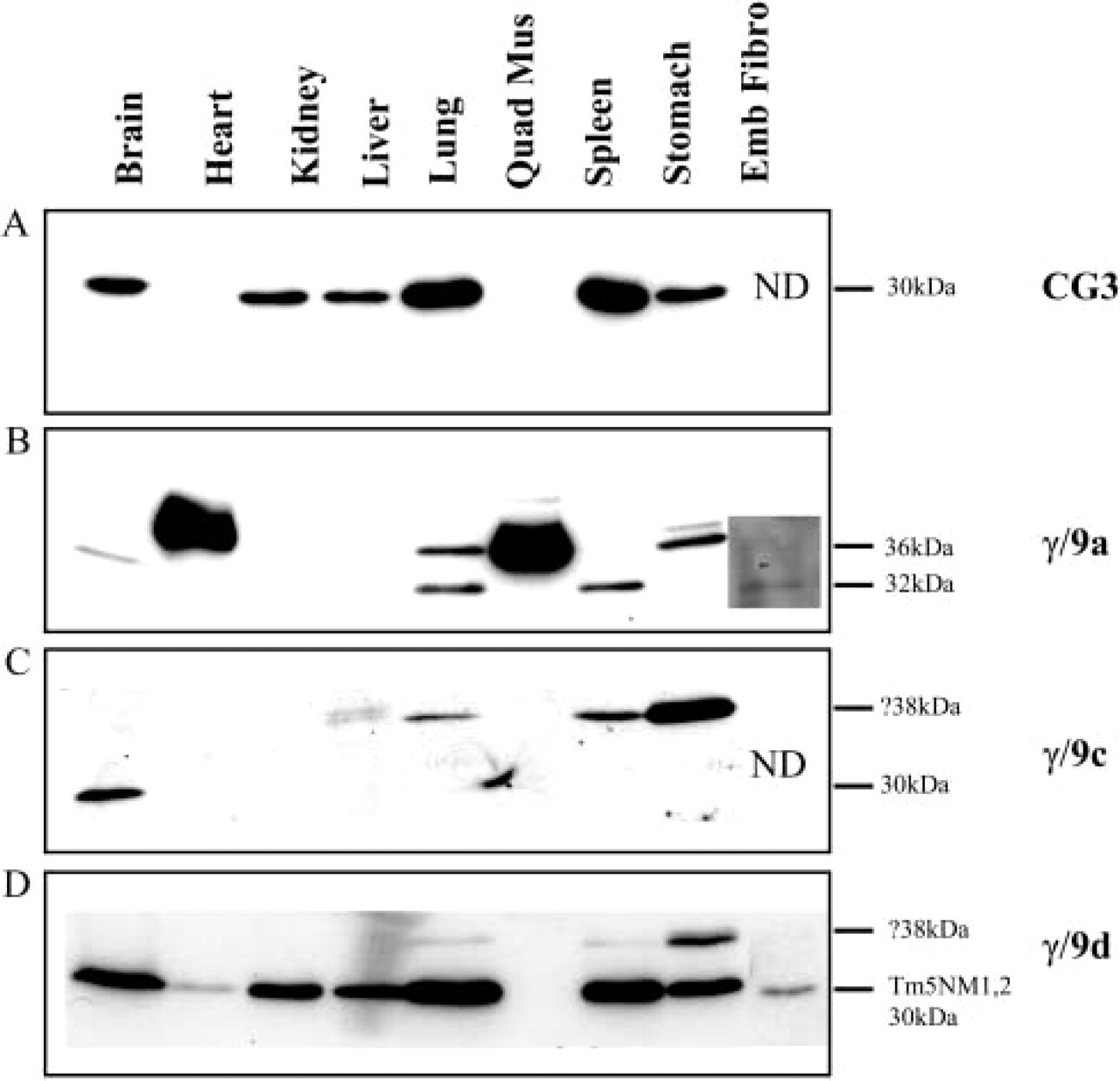
Expression of Tm isoforms from the γTm gene in different mouse tissues. Equal loading (20 μg) of total cellular protein isolated from mouse tissues was electrophoresed on 12.5% low-bis SDS gels, and individual blots were immunoblotted with the (
The γ/9c antibody also detected two proteins of apparent molecular mass of 38 and 30 kDa (Figure 6C). The 30-kDa band present only in the brain corresponds to the exon 9c containing isoforms from the γTm gene, either NM7, 4, 8, or 9. Due to the slight cross-reactivity of the γ/9c antibody to Tm1 and the considerable levels of Tm1 seen in the lung, spleen, and stomach (Figure 3B), it is possible that this band is either Tm1 or an unknown tropomyosin isoform.
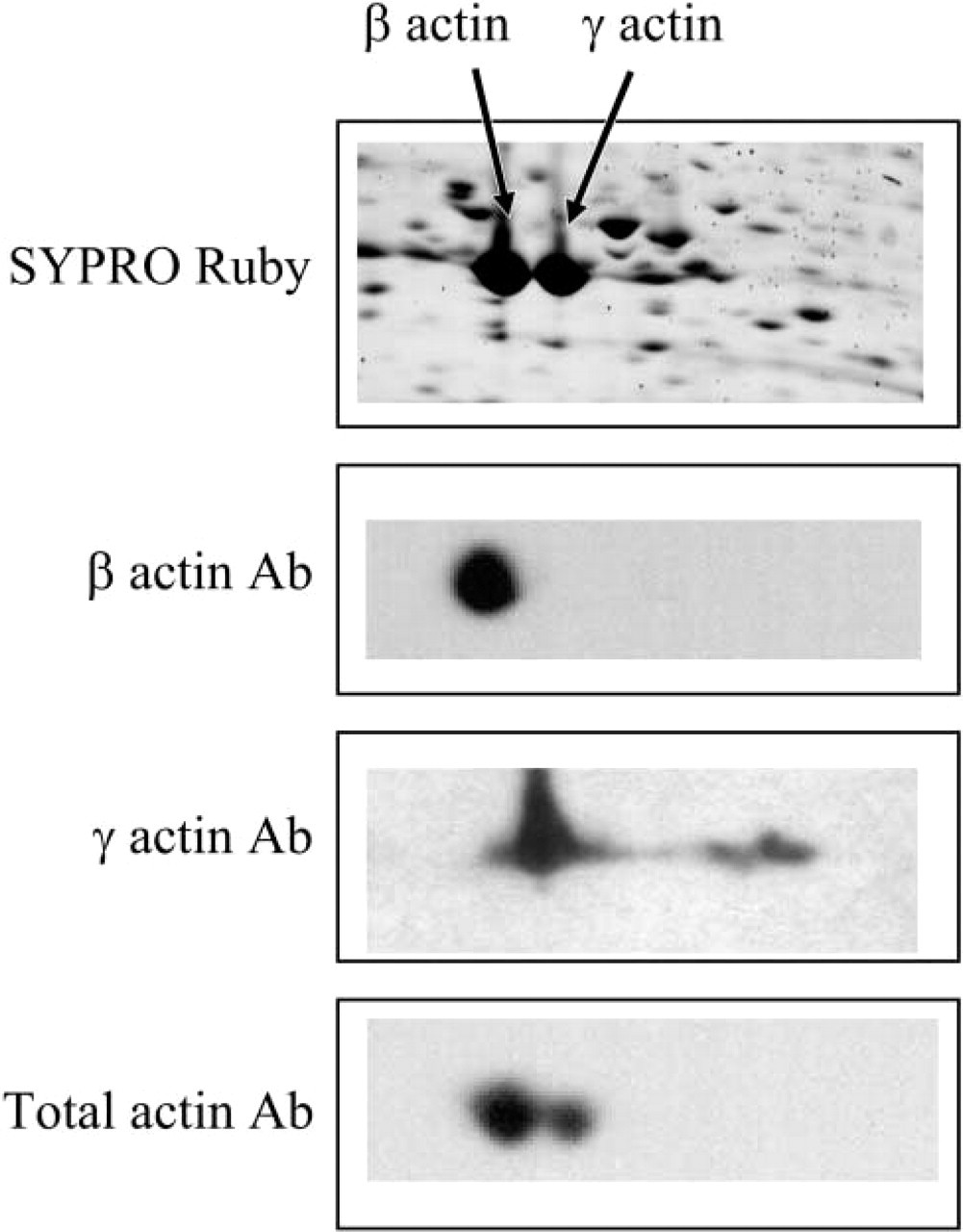
Specificity of the γ-actin antibody. Two-dimensional immunoblots of 100 μg of CCRF-CEM total cellular protein, probed with the β-actin, γ-actin, and total actin antibody (C4).
The γ/9d antibody detects a significant level of a 30-kDa protein in most tissues, with even quadriceps muscle showing a very faint band, seen in a longer exposure (not shown) (Figure 6D). In addition, a higher-molecular-mass band is also detected, 38 kDa, in lung, spleen, and stomach (Figure 6D). Interestingly, the high-molecular-mass bands detected with the γ/9a, γ/9c, and γ/9d antibodies are not seen by the CG3 antibody. This observation means that these isoforms lack exon 1b; however, they may contain exons 1a or 2b of the γTm gene. Due to the slight cross-reactivity of the γ/9d antibody with Tm1 (Figure 2G) and the relatively high levels of expression of Tm1 in these tissues as seen with the α/9d antibody (Figure 3B), it is also possible that this 38-kDa band is Tm1.
Expression Profile of Actin Isoforms in Mouse Tissues
The non-muscle actin isoforms β- and γ-actin differ by four amino acids within the first 10 coding amino acids. Both the β- and γ-actin antibodies have been generated with peptides that span these 10 amino acids. The specificity of the γ-actin antibody was confirmed by Western blotting of two-dimensional SDS poly-acrylamide gels (Figure 7) and mass spectrometry as previously reported (Verrills et al. 2003). Both the β-actin and the γ-actin antibodies are specific for the corresponding actin isoforms.
In the different mouse tissues, γ-actin was found to be the most widely expressed non-muscle isoform as compared with β-actin (Figures 8A and 8B). The presence of β-actin was undetected in the muscle samples like heart, quadriceps muscle, and stomach under these conditions (Figure 8A). In contrast, detectable levels of γ-actin were seen in all tissues examined. The stomach sample showed numerous lower-molecular-mass bands (Figure 8B). These bands were also detected with the α-smooth-muscle actin antibody and may correspond to degraded actin (Figure 8C).
Smooth-muscle actin was detected only in the lung, spleen, and stomach and not cardiac or striated quadriceps muscle or the other non-muscle tissues (Figure 8C). The expression of α-smooth-muscle actin seen only in the lung, spleen, and stomach parallels the expression of α-smooth Tm (α/2a antibody) in these tissues (Figure 3D). This further confirms that the α/2a antibody is specific for α-smooth Tm, also known as Tm6.
Sorting of Tm Isoforms in Mouse Spleen
The 10 non-muscle Tm antibodies were tested on a range of paraffin wax sections of different mouse tissues. Previously published data have shown that these Tm antibodies have been successfully used to identify distinct cellular compartments, by immunohistochemistry, on a range of different tissues, including mouse kidney (Ashworth et al. 2004), mouse skeletal muscle (Kee et al. 2004), mouse brain (Vrhovski et al. 2003), human colon (Percival et al. 2000), and rat duodenum (Dalby-Payne et al. 2003). In this study, we report observations made in the spleen with four of the Tm antibodies and two non-muscle actin antibodies. The mouse spleen consists of three distinctive components: the trabecule, white pulp, and red pulp. The trabecule includes mainly fibrous tissues, the white pulp consists of lymphocytes, and the red pulp contains mainly red blood cells and macrophages. If different Tm isoforms are sorted to different cell types, the localization of these isoforms in the spleen should be different. The WD/49d and γ/9d were the most widely distributed and gave the strongest staining in all three components of the spleen (Figures 9B and 9D). In contrast, the α/9d and γ/9a antibodies were mainly enriched in only one component, the trabecule (Figures 9A and 9C). The β-actin antibody was found to stain the red and white pulp but not the trabecule, whereas the γ-actin antibody was found in all three components (Figures 9E and 9F). In conclusion, the distribution of Tm isoform expression between the different cell populations in the spleen is not equivalent; in particular, products of the α and β Tm genes containing exon 9d are restricted, as are exon 9a containing products from the γTm gene.
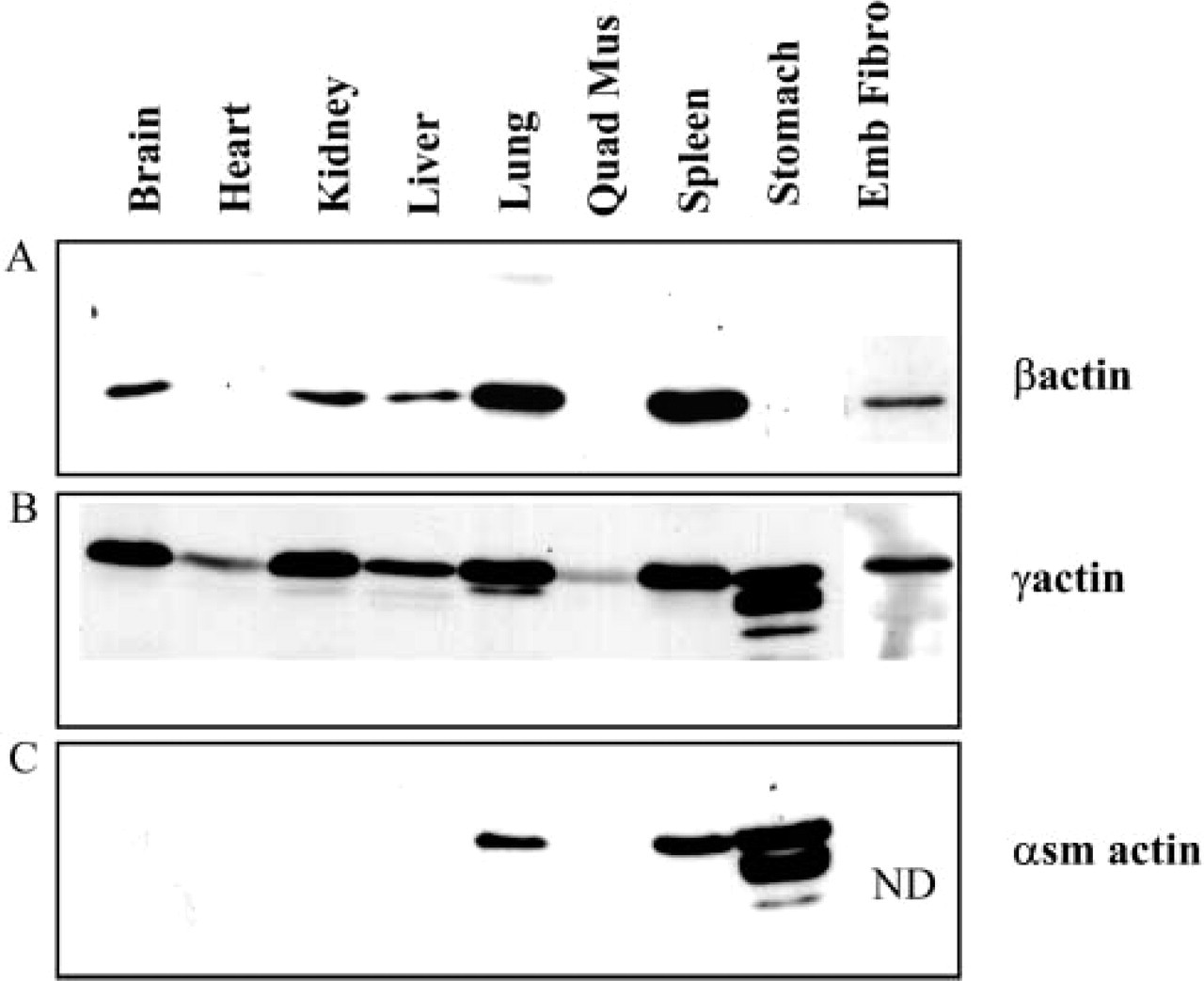
Expression of actin isoforms in different mouse tissues. Equal loading (20 μg) of total cellular protein isolated from mouse tissues was electrophoresed on 12.5% low-bis SDS gels, and individual blots were immunoblotted with (
Subcellular Localization of Tm Isoforms in Cultured Mouse Primary Fibroblasts
The subcellular localization of the non-muscle Tm isoforms was investigated in mouse primary fibroblast by immunofluorescent staining. In this study and the study published by Hook et al. (2004), primary embryonic fibroblasts were shown to express Tm6, 1, 2, 3, 4, 5a, and 5b and products from the γTm gene, including those containing exon 9a and Tm5NM1/ NM2. The TM311 antibody, which detects Tm6, 1, 2, and 3, predominantly stained stress fibers, most of which colocalized with γ-actin and did not stain the cell periphery (Figure 10D, arrow). The α/9d antibody, which detects Tm5a and Tm5b, with the addition of the high-molecular-mass Tm isoforms seen by TM311, was also found to stain stress fibers that colocalized with β-actin (Figures 10A-10C). Furthermore, the α/9d antibody also clearly stained the cell periphery and ruffling membranes (Figure 10A, arrow). We therefore conclude that Tm5a and Tm5b are the isoforms present in the cell periphery and ruffling membranes. This correlates well with the apical enrichment of these isoforms in epithelial cells (Dalby-Payne et al. 2003). The WD4/9d antibody, which detects Tm4 and Tm1, was also predominantly found in stress fibers (Figure 10G) and more resembles α/9d than TM311 staining.
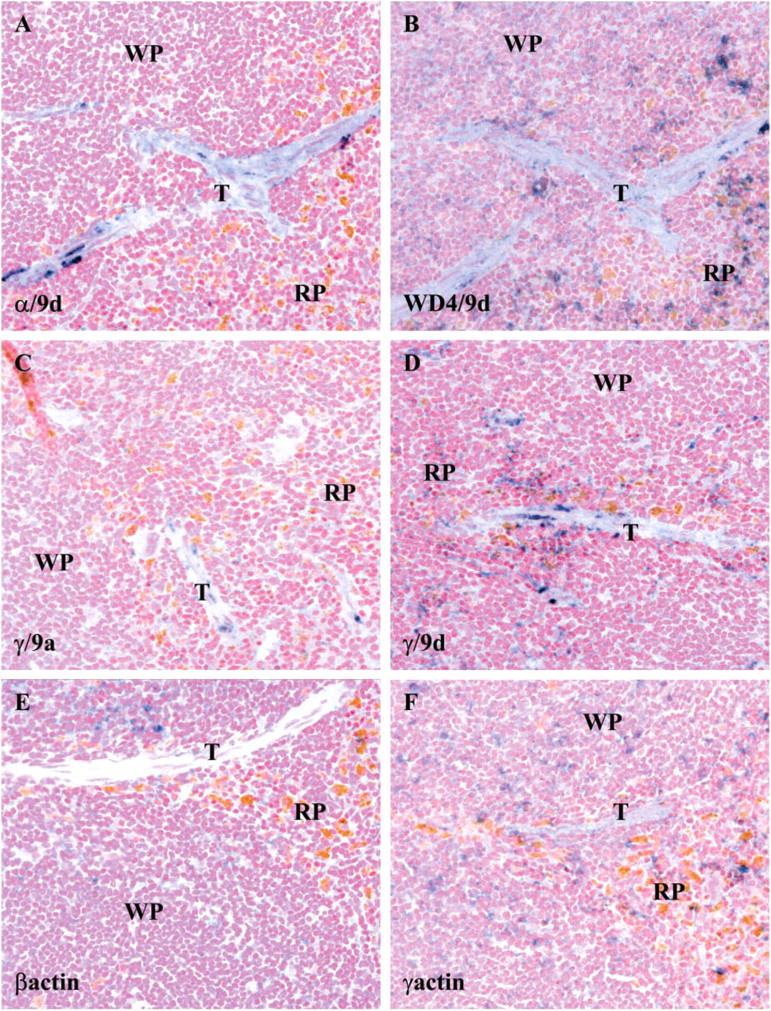
Localization of Tm isoforms in the spleen. Parasagittal sections of mouse adult spleen stained with the (
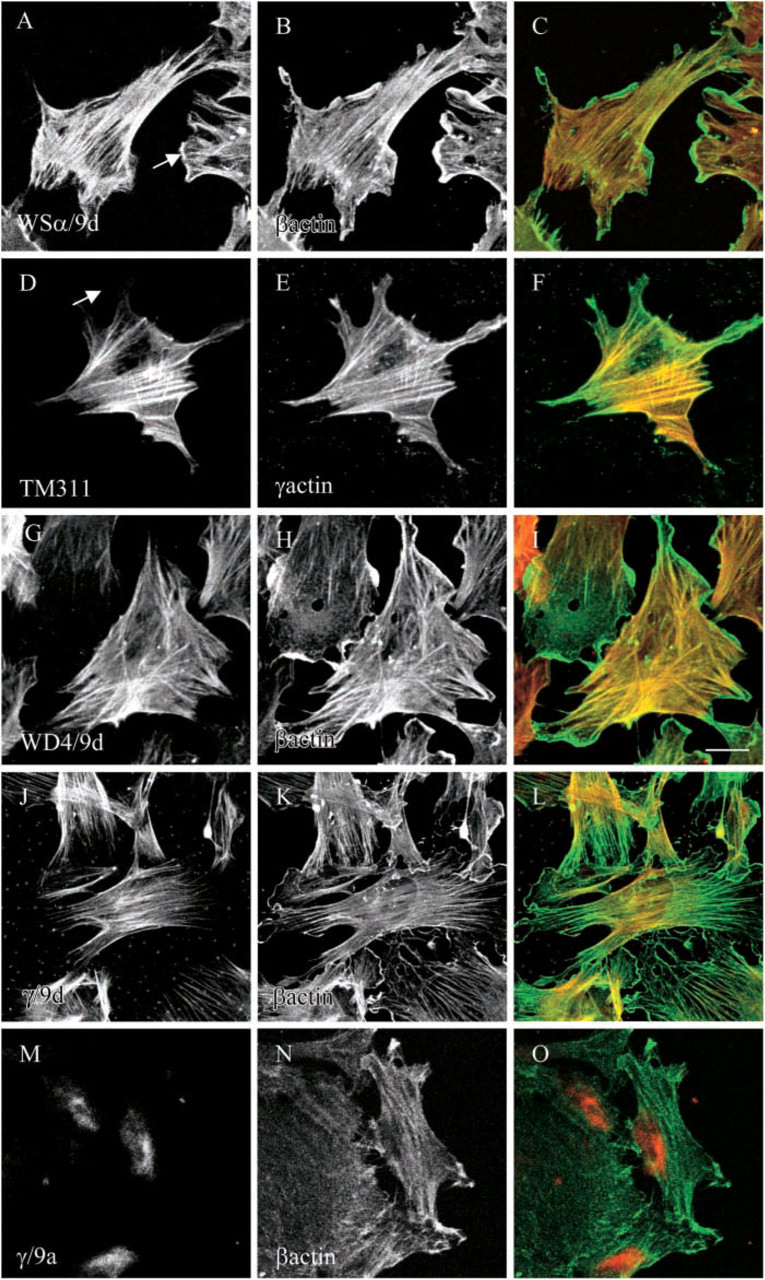
Subcellular localization of Tm isoforms in cultured mouse primary fibroblasts. Embryonic mouse primary fibroblasts were cultured in vitro and double immunofluorescence stained with (
We also investigated the subcellular localization of Tm isoforms derived from the γTm gene. The γ/9d antibody that detects Tm5NM1 and NM2 was clearly sorted to stress fibers that colocalize with β-actin (Figures 10J-10L) but not the ruffling membranes, and staining was more similar to TM311 than α/9d. The γ/9a antibody, which detects the non-muscle exon 9a containing isoforms from the γTm gene, preferentially stained the perinuclear region (Figures 10M-10O).
These results demonstrate that different Tm isoforms can potentially sort to three different compartments within mouse primary fibroblasts, stress fibers, cell periphery/ruffling membranes, and the perinuclear region.
In summary, our bank of tropomyosin antibodies allows both the quantitation and visualization of different actin filament populations in mammalian cells, tissues, and organs.
Discussion
The actin cytoskeleton plays a fundamental role in many aspects of eukaryotic cell behavior, including cell morphology, division, and motility, and organelle and vesicle trafficking. Consequently, dysregulation of proteins known to regulate actin dynamics, such as tropomyosin isoforms, may be key indicators of altered cell behavior and hence disease.
In this study, a cohort of 10 Tm isoform-specific antibodies were fully characterized and tested on Western blotting, immunohistochemistry, and immunofluorescence staining of cells in culture. This is the first comprehensive study that clearly demonstrates that different normal mouse tissues express a very restricted repertoire of Tm isoforms (Table 2). The expression of different Tm isoforms may therefore cater for the specific functions of actin filaments within different cells. Immunohistochemical distribution of these Tm isoforms was studied in different mouse tissues, and different isoforms were found to sort to distinct regions. At the subcellular level, these Tm isoforms were found to be sorted to specific compartments within embryonic primary fibroblast cells in culture (Figure 10), including stress fibers, ruffling membranes, and the actin cytoskeleton present in the perinuclear region. Consequently, this degree of resolving power allows one to detect fundamentally distinct populations of actin filaments as opposed to visualizing and quantitating the entire actin cytoskeleton.
To date, multiple Tm isoform-specific compartments have been identified in a number of different cell types, including neurons (Weinberger et al. 1996; Schevzov et al. 1997), fibroblasts (Percival et al. 2004), and epithelial cells (Percival et al. 2004). Recently, Tm antibodies have also been used to identify a novel non-muscle cytoskeletal compartment in muscle cells that might be involved in the pathology of muscular dystrophy (Kee et al. 2004). In addition, Tm5a/Tm5b have been shown to mark an apical population of microfilaments that can regulate the insertion and/or retention of an apical chloride channel, CFTR (cystic fibrosis transmembrane conductance regulator) (Dalby-Payne et al. 2003).
Tm expression in adult mouse tissues
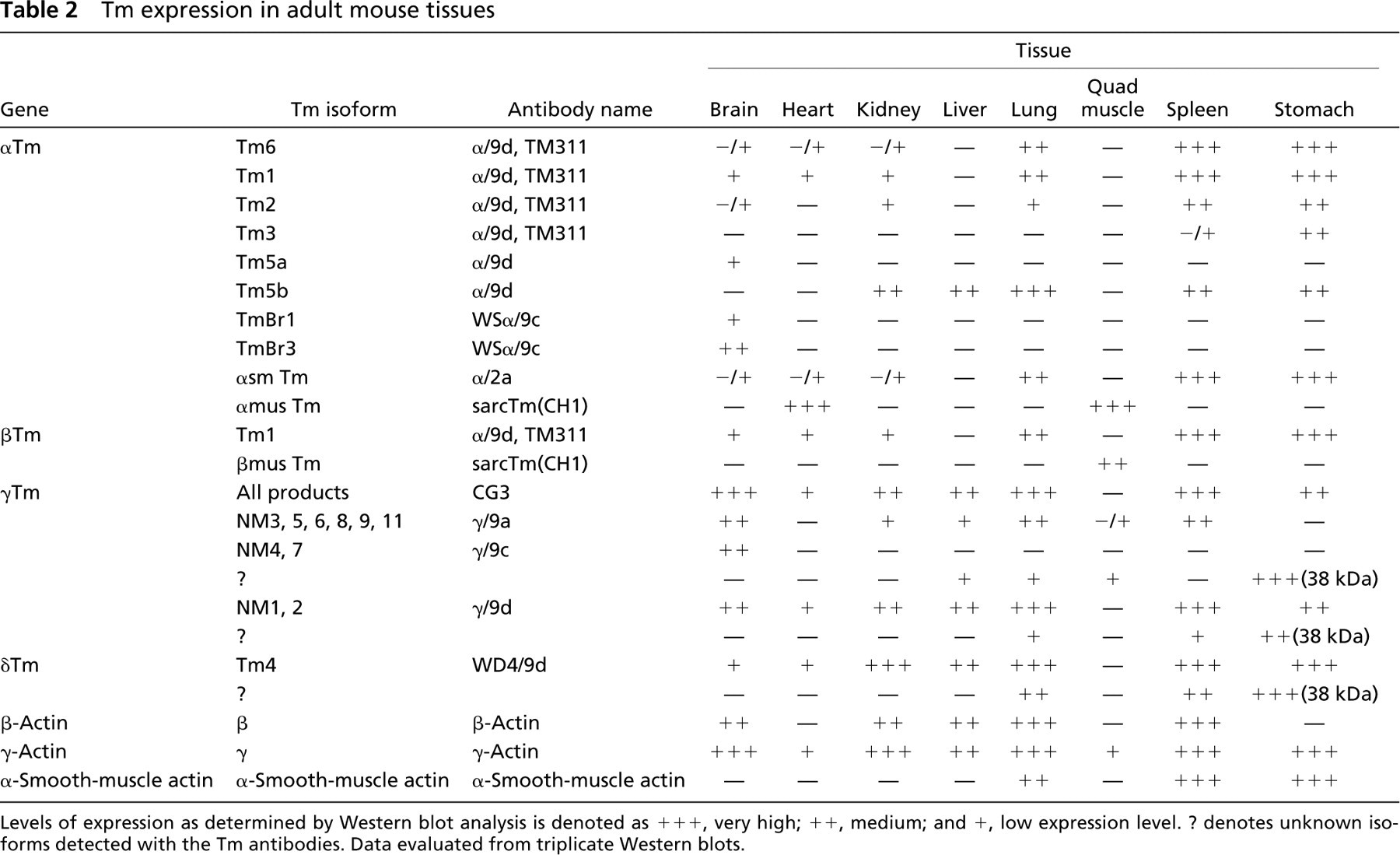
Levels of expression as determined by Western blot analysis is denoted as + + +, very high; + +, medium; and +, low expression level. ? denotes unknown isoforms detected with the Tm antibodies. Data evaluated from triplicate Western blots.
It is therefore envisaged that these Tm antibodies could potentially be used as diagnostic markers of tissue dysfunction. Indeed, modifications in the expression and/or subcellular localization of specific non-muscle Tm isoforms are slowly emerging as markers of human disease. For example, alterations in the expression of specific Tm isoforms have been shown in transformed rat fibroblasts cell lines and hence have implicated Tm as suppressors of transformation (Pittenger et al. 1994). Further studies in a range of human cancers have indicated a predominant decrease in the expression of high-molecular-mass Tm isoforms, Tm1, 2, and 3. These include human breast cancer, prostate cancer, and gastric cancer (Franzen et al. 1996,1997; Wang et al. 1996; Jung et al. 2000; Raval et al. 2003). In addition, human transitional cell carcinoma of the urinary bladder showed not only a marked reduction in the expression of Tm1 and Tm2 but also changes in the expression of isoform(s) from the γTm gene (Pawlak et al. 2004). Similarly, in colon cancer, aberrant expression of Tm isoform (s) derived from the γTm gene has also been reported (Lin et al. 2002).
Other human defects known to have altered Tm expression include ulcerative colitis, in the pathogenesis of which an autoimmune response toward hTm5 is thought to be involved (Biancone et al. 1998,2003). And during the induction of renal ischemia-induced cell injury, breakdown of the apical microvilli in the kidney proximal tubule cells occurs, which is in turn correlated with the dissociation of specific Tm isoforms from microfilaments present in these microvilli (Ashworth et al. 2004). Finally, Tm expression has also been linked to essential hypertension. Altered expression of Tm isoforms derived from both the α and γTm genes in blood cell membranes of a subgroup of patients implicate these isoforms in the pathogenesis of this disease (Dunn et al. 2003). Consequently, this cohort of Tm antibodies could potentially be used as candidate markers for early detection and prognostic indication of a wide range of pathophysiological defects.
Footnotes
Acknowledgements
This work was supported by Australian National Health and Medical Research Council (NHMRC #117409) grants to PWG. PWG is a Principal Research Fellow of the NHMRC #163626. GMO is the NSW Cancer Council Research Fellow.
