Abstract
The human enteric nervous system (ENS) derives from migrating neural crest cells (NCC) and is structured into different plexuses embedded in the gastrointestinal tract wall. During development of the NCC, a rearrangement of various cytoskeletal intermediate filaments such as nestin, peripherin, or alpha-internexin takes place. Although all are related to developing neurons, nestin is also used to identify neural stem cells. Until now, information about the prenatal development of the human ENS has been very restricted, especially concerning potential stem cells. In this study the expression of nestin, peripherin, and alpha-internexin, but also of neuronal markers such as protein gene product (PGP) 9.5 and tyrosine hydroxylase, were investigated in human fetal and postnatal gut. The tissue samples were rapidly removed and subsequently processed for immunohistochemistry or immunoblotting. Nestin could be detected in all samples investigated with the exception of the 9th and the 12th week of gestation (WOG). Although the neuronal marker PGP9.5 was coexpressed with nestin at the 14th WOG, this could no longer be observed at later time points. Alpha-internexin and peripherin expression also did not appear before the 14th WOG, where they were coexpressed with PGP9.5. This study reveals that the intermediate filament markers investigated are not suitable to detect early neural crest stem cells.
N
In humans, the development of the intestine is largely completed at about the end of the first trimester, a time when the ENS is readily identifiable (Montgomery et al. 1999) and characterized by a significant increase in the diameter of the myenteric ganglia (Bagyánszki et al. 2002). These developmental changes are accompanied by cytoskeletal rearrangements inside each single ENS cell. An important part of the cytoskeleton is formed by intermediate filament proteins, intercellular filaments measuring ∼10 nm in diameter. They appear to be cell type and tissue specific, and there seems to be a developmental pattern in the expression of intermediate filament proteins. During neural maturation a progressive expression of nestin, alpha-internexin, and neurofilaments, the first predominantly expressed by neural stem cells, can be observed. Alpha-internexin is supposed to be the first intermediate filament expressed during neural development (Kaplan et al. 1990). Another filament protein found in the developing peripheral nervous system, including enteric ganglion cells, is peripherin, which together with alpha-internexin signals the commitment of primitive neuroepithelial cells to the neuronal lineage. All these filaments could be detected in the human ENS retrieved from surgically removed specimens (Eaker 1997) or from autopsy material (Vanderwinden et al. 2002) of various postnatal ages: 62–93 years (Eaker 1997) and 2 months to 26 years (Vanderwinden et al. 2002).
In this study, the human ENS development concerning intermediate filaments, as well as neuronal markers [protein gene product (PGP) 9.5, tyrosine hydroxylase (TH)] were investigated in an earlier time frame. So far, the expression of these filaments and of TH in the human fetal gut is unknown. To obtain not only a time-specific expression pattern, but also a segment-specific pattern, samples from all gut segments, if available, were examined.
Materials and Methods
Patients and Tissue Specimens
Thirty four bowel samples (Table 1) were obtained from either stillborn or aborted human fetuses [9th to 33rd week of gestation (WOG)]; gestational age of the fetuses was determined by crown-rump length, head circumference, and foot length, from children (4 days to 14 years) or from adults (63 to 81 years). All tissues were removed rapidly during surgery or examination and subsequently processed as described below. Tissues from abortion or postmortem examination included segments of all or most sections of the digestive tract (at least small intestine and colon). Gut received from stillborn or aborted fetuses suffering from chromosomal disorders like Tris 21, placental insufficiency, or maternal inflammation was morphologically inconspicuous, and the samples used showed age-related similarities.
All samples were collected after informed consent from patients or parents, in accordance with the Helsinki Declaration and with the approval of the local ethical committee.
Classification of samples

WOG, week of gestation.
Immunofluorescence
All tissues were fixed in 2–4% formaldehyde in PBS and embedded in paraffin. Adjacent sections of 5–6 μm were cut on a microtome (R. Jung; Nussloch, Germany). After dewaxing, rehydrating, and antigen retrieval (citrate buffer, pH 6.0, 95C, 15 min), the slides were allowed to cool for 30 min at room temperature (RT). After washing in PBS, the slides were incubated in 0.5% Triton X-100 (Roth; Karlsruhe, Germany) in PBS for 10 min, rinsed in PBS, blocked with 10% NGS (normal goat serum; DAKO, Hamburg, Germany) in PBS for 60 min, and incubated in a humid chamber overnight with the primary antibodies at RT (Table 2). After repeated washing in PBS, the antigens were visualized using appropriate fluorescent secondary antibodies (1 hr RT, see Table 2), washed several times in PBS, and counterstained with DAPI (Sigma; Taufkirchen, Germany) for 10 min. Negative controls lacking the primary antibody were included in each series.
Immunohistochemistry
All tissues were fixed in 2–4% formaldehyde in PBS and embedded in paraffin. Adjacent sections of 5–6 μm were cut on a microtome (R. Jung). After dewaxing and rehydrating, the sections were washed in TBST (Tris-buffered solution + 0.05% Tween 20) for 5 min. After antigen retrieval (citrate buffer, pH 6.0, 95C, 30 min), the slides were allowed to cool for 30 min at RT and washed again in TBST for 5 min. The detection of the PGP9.5 antigen was carried out with the EnVision + System-HRP for use with rabbit primary antibodies (DakoCytomation; Hamburg, Germany), whereas the detection of TH was carried out with the Vectastain Elite ABC kit (Linaris; Wertheim-Bettingen, Germany) according to the manufacturer's protocol. TBST was always used as a buffer. Incubation with the primary antibodies (Table 2) was carried out in a humid chamber overnight at RT and DAB was used as a chromogen to localize the peroxidase in our tissue sections. After the DAB reaction, the sections were dehydrated and mounted in Neo-Mount (Merck; Darmstadt, Germany).
List of antibodies
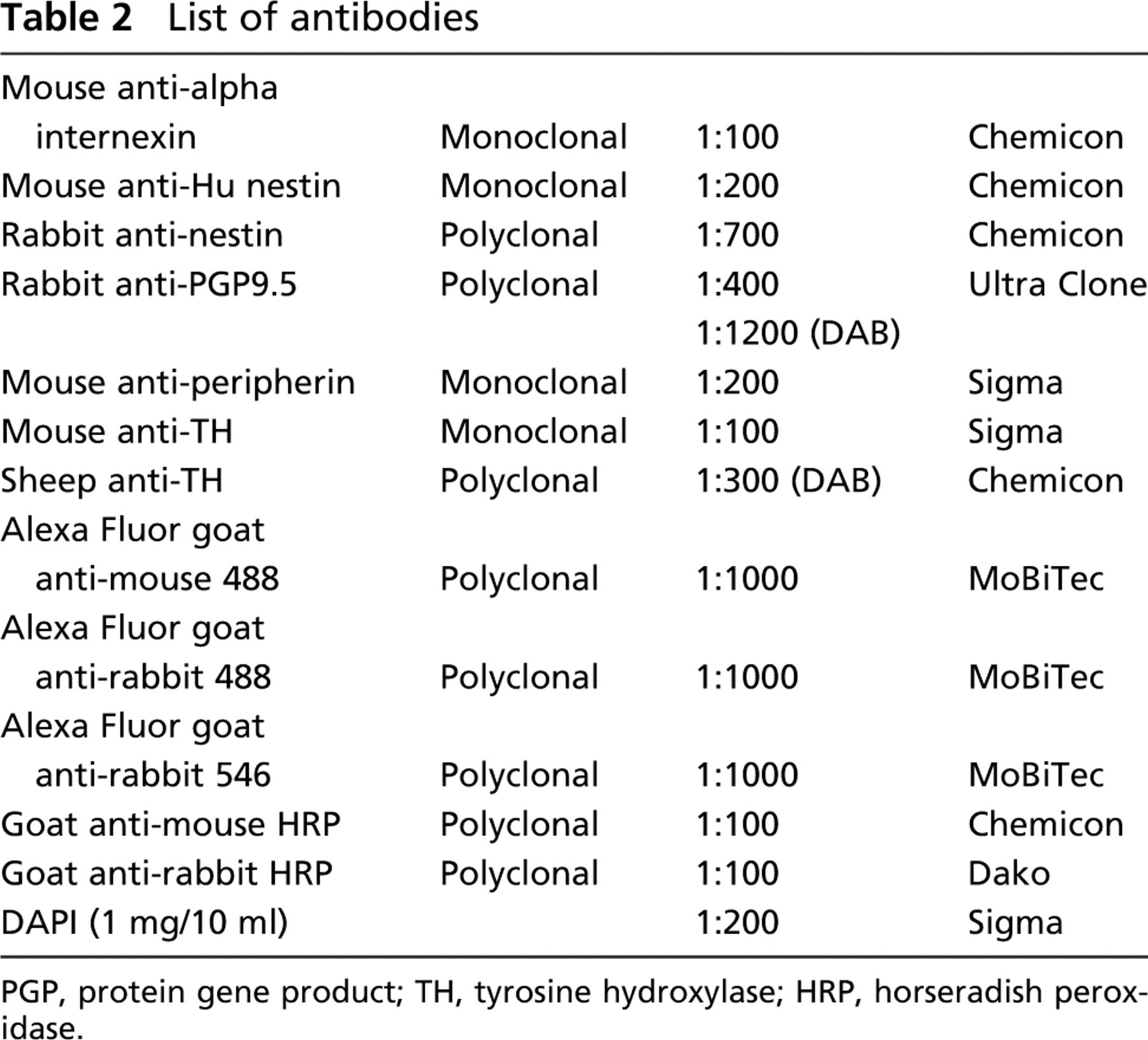
PGP, protein gene product; TH, tyrosine hydroxylase; HRP, horseradish peroxidase.
To analyze the proportion of PGP9.5- and TH-stained areas of myenteric ganglia in human gut, selected intestinal sections were subjected to morphometric analysis. An inverted light microscope (IX50; Olympus, Hamburg, Germany) equipped with a CCD camera (ColorView XS, SiS; Münster, Germany) in combination with the analySIS software (analySIS 3.2; SiS) was used for measurements. We made photos of the stained sections, defined regions of interest (myenteric ganglia), and selected a set of pixels whose red, green, blue (RGB) values define a phase. Finally, the software determined the area of the phase inside the ganglionic area, which represents PGP9.5- and TH-stained areas. To compare PGP9.5 and TH stainings, we analyzed following sections of the same tissue.
Immunoblotting
The muscle and submucous layers were parted by manual dissection. Tissue samples from the muscle and submucous layer were homogenized separately in PBS containing a protease inhibitor cocktail (Roche; Mannheim, Germany). Proteins were solubilized in sample buffer (Invitrogen; Karlsruhe, Germany), heated at 75C for 5 min, and centrifuged at 12,000 × g for 10 min. Samples were separated by electrophoresis on a 4–12% gradient polyacrylamide gel (Invitrogen) together with standard proteins (Mark12; Invitrogen) and transferrred to a nitrocellulose membrane (Invitrogen) in a tank blot unit (Amersham; Freiburg, Germany) at 400 mA for 1 hr. Membranes were cut into single-sample strips. After blocking with blot buffer (PBS/Tween 0.5% containing 5% dry milk powder), the sample strips were incubated with the primary antibody directed against PGP9.5 (Table 1) for 2 hr in blot buffer. The strips were then washed three times, 5 min each, in blot buffer and incubated for 1 hr with an alkaline-phosphatase (AP)-conjugated antibody against IgG from rabbit (Sigma). After washing, the reactions were visualized with the AP substrate 5-bromo-4-chloro-3-indolyl phosphate/nitroblue tetrazolium (BCIP-NBT; Calbiochem, San Diego, CA).
Results
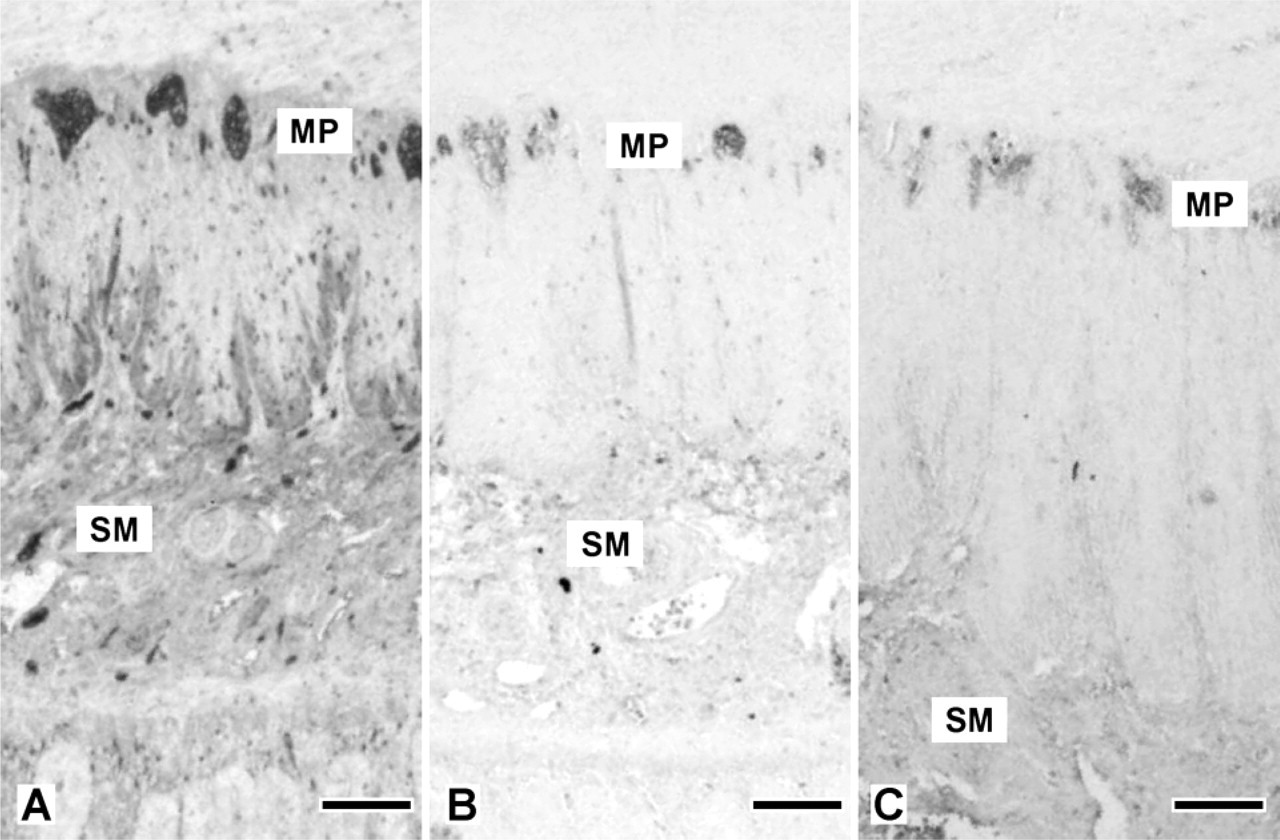
The intermediate filaments nestin, alpha-internexin, and peripherin, together with the pan-neuronal marker PGP9.5, could be detected by immunohistochemistry in all samples of gut from the 14th WOG and older fetuses, whereas only PGP9.5 also showed a strong staining in the earliest stages investigated (9th WOG). At that time (9th WOG) an increasing density of the PGP9.5 staining along the gut axis from esophagus to the distal colon, as well as from the mesentery toward the antimesenterial side of the gut, could be noticed (Figure 1). In contrast to more distal regions of the gut, there seemed to be cells migrating from the MP toward the submucosa in the esophagus and the stomach, whereas in the other sections only a developing MP could be stained. At the 12th WOG, a readily developed MP can be found throughout the gut, whereas the SMP is only partially developed in the small intestine and could not be detected in the colon.
An initial overall expression of PGP9.5 in the MP and the SMP was detected from the 14th WOG up to and including adulthood. Analysis of the ganglionic area showed that the proportion of PGP9.5-positive tissue inside the myenteric ganglia of all gut sections, except the esophagus, increased significantly when comparing pre- and postnatal samples (Table 3).
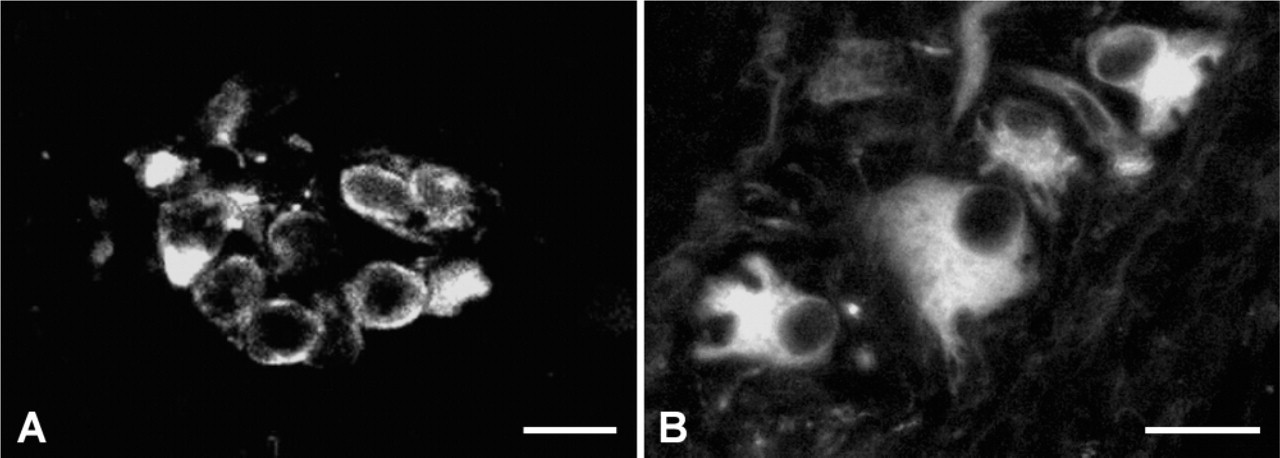
At the 14th WOG, every PGP9.5-positive cell in the MP also showed a strong nestin expression, which decreased with age (Figure 2). Nevertheless, as was shown for PGP9.5, nestin could still be detected after birth and even in adult tissue.
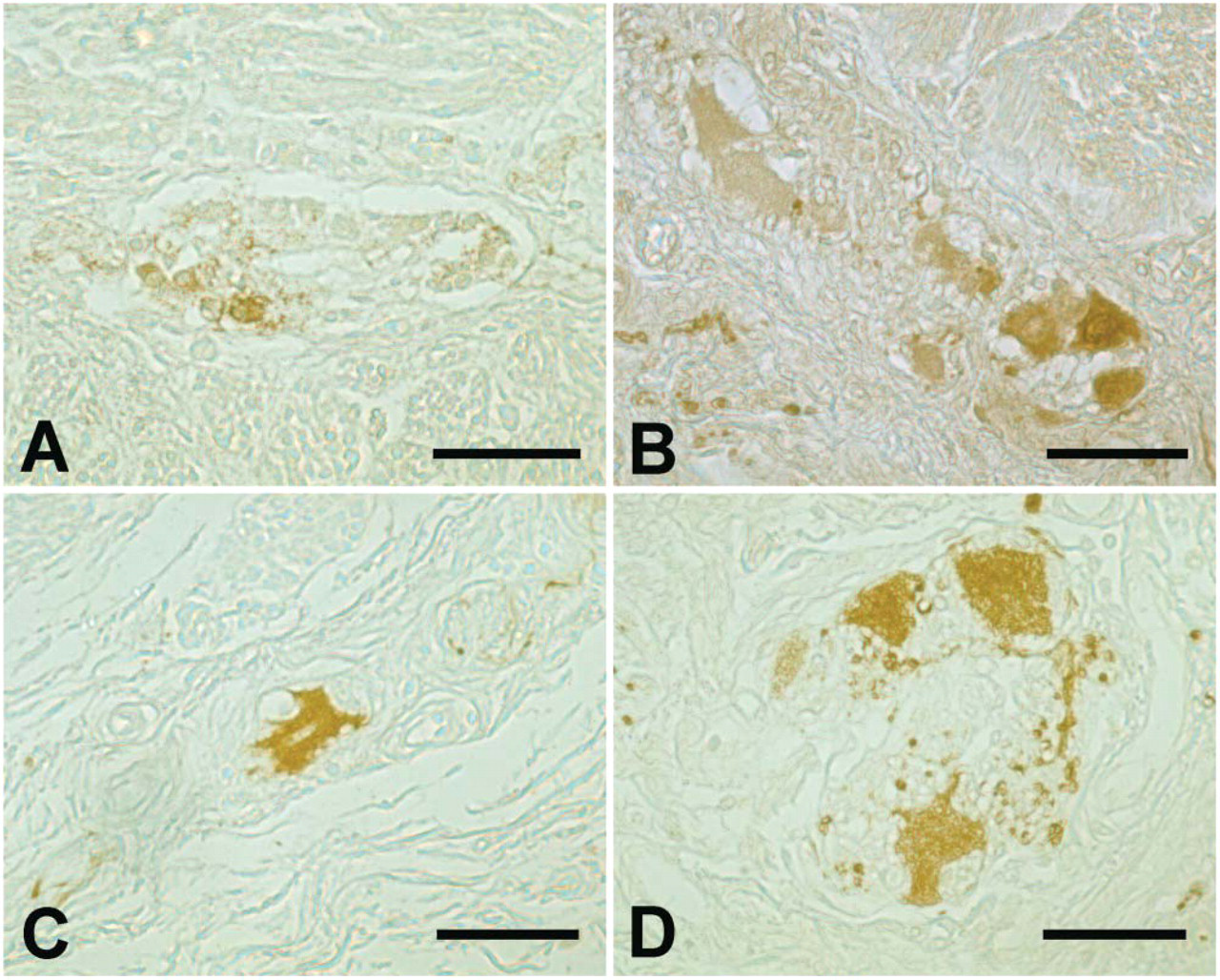
Peripherin and alpha-internexin could only be detected in the MP at the 14th WOG (Figure 3). At the 21st WOG and from that time until birth, peripherin and alpha-internexin could also be seen in the submucous layer. After birth and in adults, peripherin and alpha-internexin could be detected mainly in ganglia of the MP. In addition to the different expression patterns, there were also morphological differences concerning internexin-positive cells between younger and older samples, reflecting the maturation of the ENS that could be seen. Whereas the cells stained at the 14th WOG appeared as a homogeneous population of small and rounded cells, with increasing age the morphology of the neuronal cell bodies became larger and polygonal, thus demonstrating a more differentiated, neuron-like appearance (Figure 4).
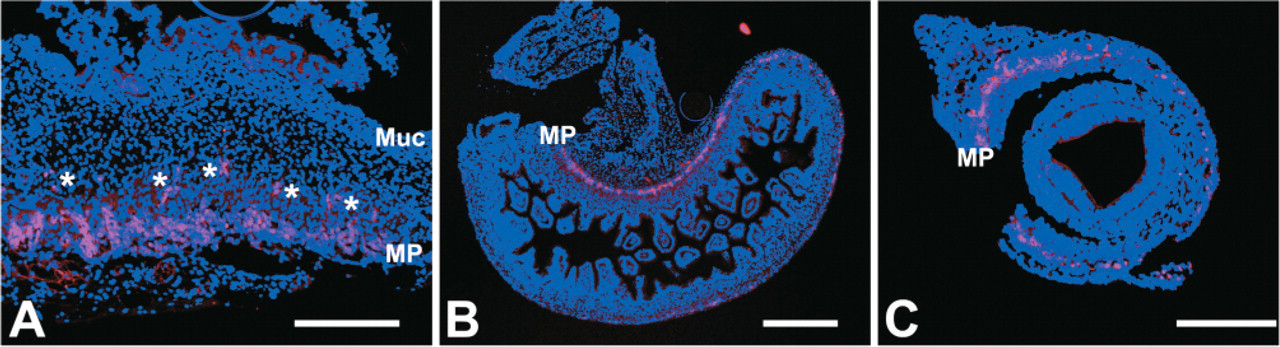
Protein gene product (PGP) staining pattern at the 9th week of gestation (WOG). (
Proportion of PGP9.5- and TH-stained area in myenteric ganglia depending on age and localization
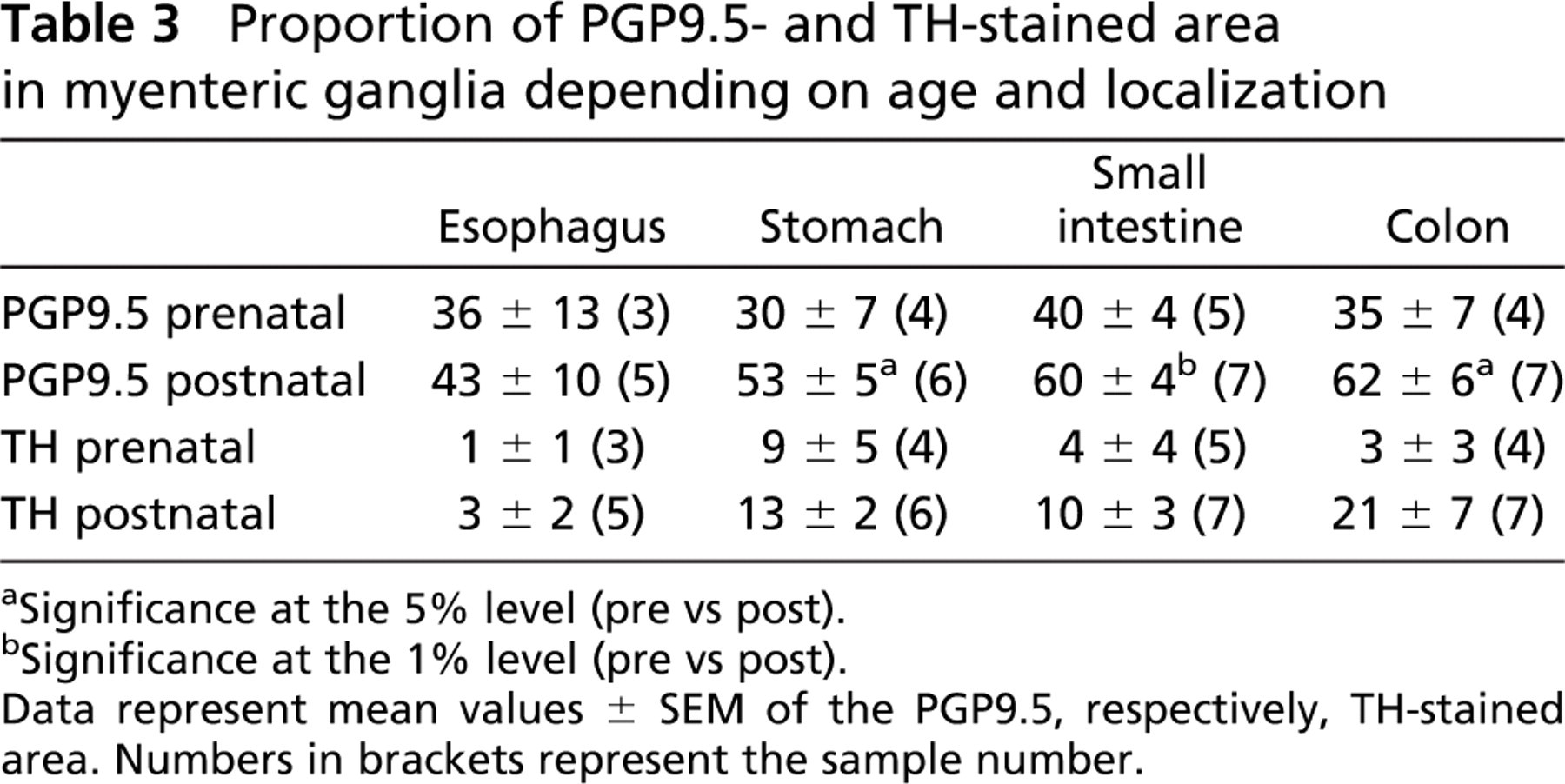
Significance at the 5% level (pre vs post).
Significance at the 1% level (pre vs post).
Data represent mean values ± SEM of the PGP9.5, respectively, TH-stained area. Numbers in brackets represent the sample number.
TH could be detected from the 24th WOG on. At that time its expression was restricted to the stomach, whereas it could be seen in the MP of all gut sections from the 32nd WOG on. Concerning the expression pattern of TH, we observed an insignificant increase in TH-positive tissue inside the myenteric ganglia in all gut sections when comparing pre- and postnatal samples (Table 3 and Figure 5).
To estimate the amount of neuronal tissue at various time points, immunoblots from muscular and submucous tissues at corresponding developmental stages were made. These immunoblots show aberrant results from what was seen in immunohistochemistry. Whereas in situ, a PGP9.5 staining of the SMP can already be seen at the 14th WOG, the corresponding immunoblot showed a strong PGP9.5 signal in the muscle layer only. Also during late development (33rd WOG), PGP9.5 could be found only in the MP, whereas in postnatal gut both muscle and submucous layer gave a strong signal of PGP9.5.
Discussion
In this study we examined the expression of intermediate filaments, as well as that of neuronal markers during development of the human ENS (Figure 6). Nestin, a marker for various progenitor cells, has been found in all samples and at all ages studied so far, with the exception of the sample at the 9th WOG. Nestin expression after birth has already been demonstrated by Vanderwinden et al. (2002); however, they never found any cell that simultaneously expressed nestin and a neuronal marker. In contrast to their observation, we found a clear overlap in nestin and PGP9.5 expression at earlier stages, which vanished with ongoing development. This decrease in coexpression seems to demonstrate a process of maturation; however, there is still no available information about the function of nestin in the adult ENS (Vanderwinden et al. 2002). As most of the nestin-positive cells in the adult ENS exhibit glial properties (Vanderwinden et al. 2002), one may presume that at least a subpopulation of these cells may serve as neuronal progenitors as is discussed for the CNS as well (Fukuda et al. 2003). Although nestin does not seem to be an early neuronal stem-cell marker in the human ENS, it has to be investigated as to whether it might serve as a marker for more mature neural crest-derived neuronal progenitor cells. In contrast to nestin, PGP9.5 is expressed from the earliest samples on, corresponding to findings in the developing rodent gastrointestinal tract. PGP9.5 has been shown to be expressed in ENS and other neurons and is discussed as a tool to identify NCC and neuronal and glial precursors in mice (Sidebotham et al. 2001). In our study it could be detected from the 9th week of gestation on, when the organogenesis of the human intestine is more or less complete. Whereas the MP at that age can clearly be identified throughout the gut length, the SMP can only be seen in the most proximal segments. From the 14th WOG on, traces of SMP can be found throughout the gut. Nevertheless, neuronal staining of the ENS at that time (14th WOG) does not show a fully developed SMP. This is in contrast to the findings of Burns and Le Douarin (1998), who described that NCC primarily colonize the submucosal region of the chick colorectum before the MP forms but is in accordance with the situation in murine large intestine, where neuron precursors colonize the MP prior to the submucosal region (McKeown et al. 2001). This indicates a species-specific difference between mammals and birds with regard to the development of the ENS in the large intestine.
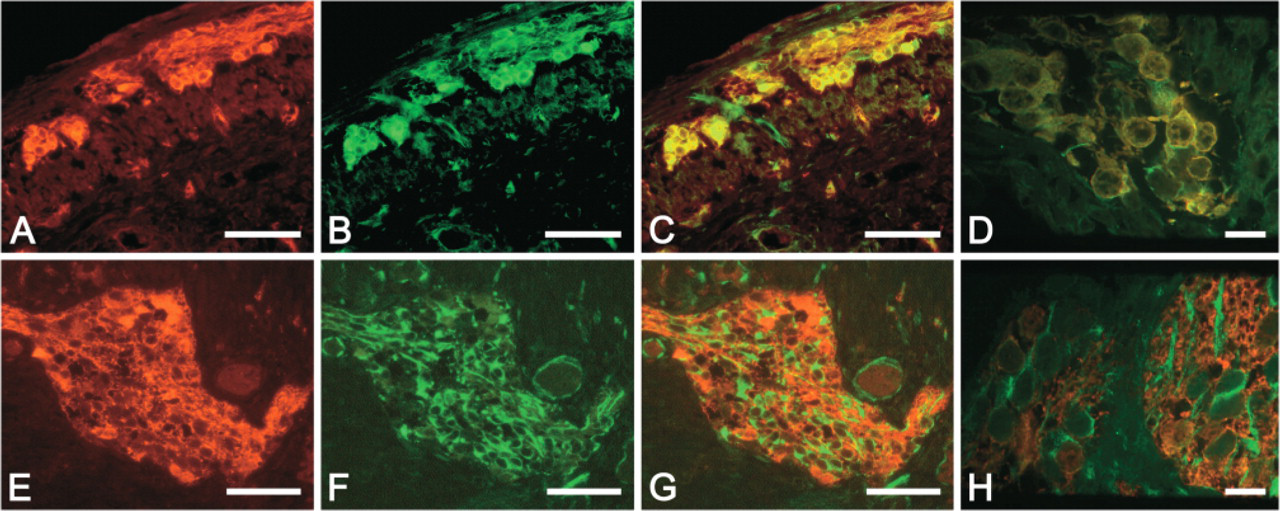
PGP and nestin immunolabeling. Ganglion of the human MP at the 14th (
At the 14th WOG, PGP-positive cells (
(
Tyrosine-hydroxylase (TH) immunolabeling. TH-positive cells can be found in myenteric ganglia throughout life. (
Western blots from submucous layers do not show a clear PGP9.5 expression before birth (Figure 7), whereas immunohistochemical stainings already show a partially developed SMP, with only smaller neuronal aggregates. This might reflect the immaturity of the ENS before birth, or in prematurely born children, respectively, where the SMP is still not fully developed. These children often suffer from digestive and motility problems as well as from acquired infectious diseases such as necrotizing enterocolitis.
The discrepancy between immunohistochemistry and Western blots in the prenatal stages might be evidence for the quantity of neuronal tissue at various time points. Obviously the amount of neuronal tissue in prenatal samples, in contrast to the older ones, is not sufficient to be detected in the Western blot.
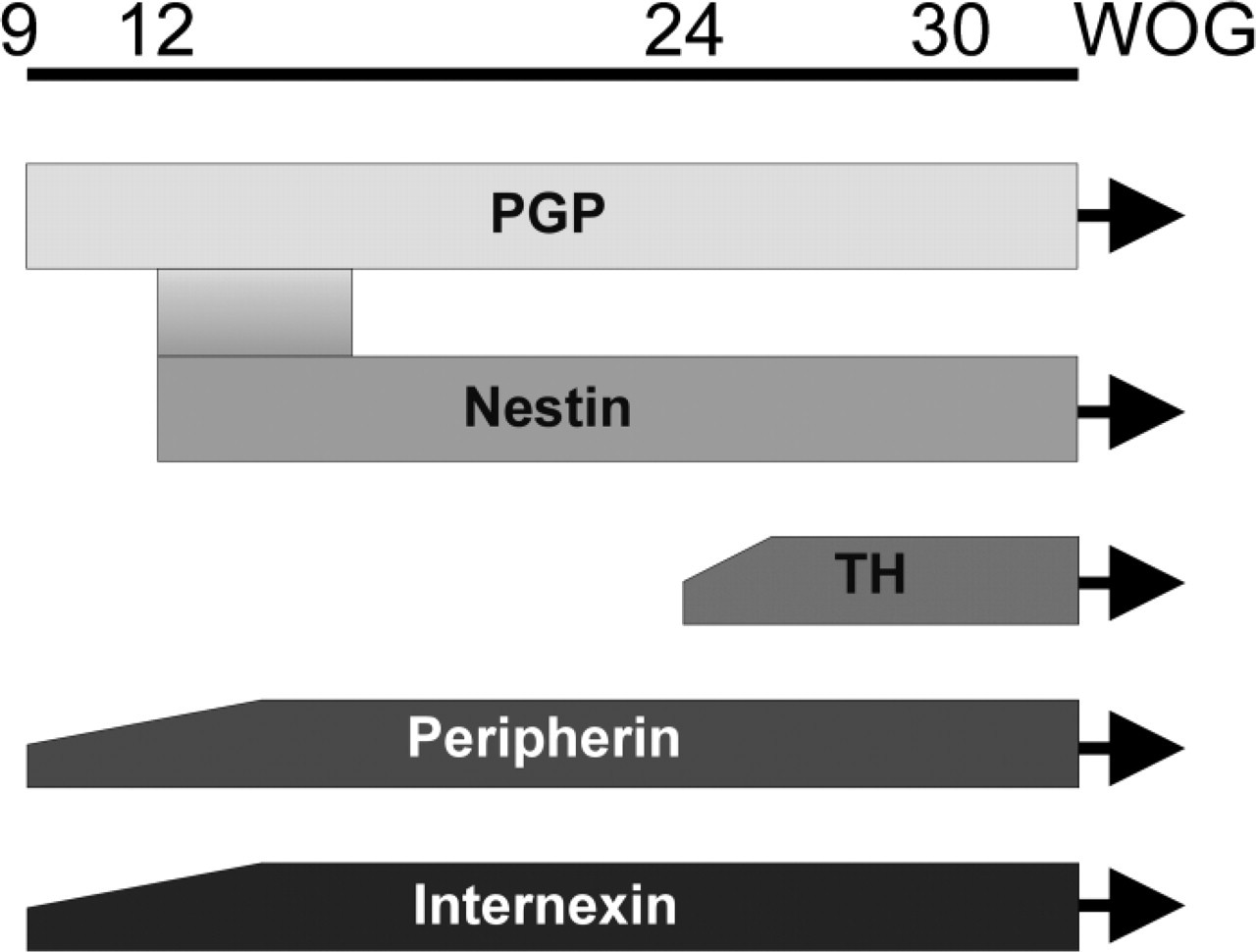
Expression profile of neuronal and intermediate filament proteins depending on the WOG (9th–30th and beyond).
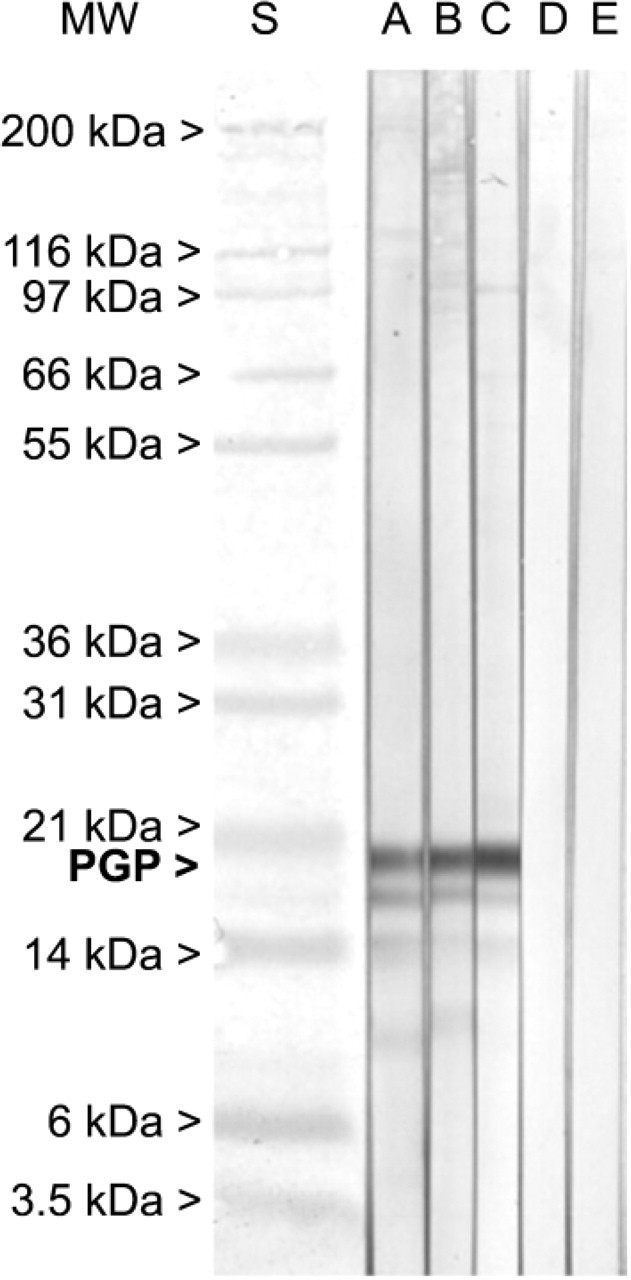
Immunoblot PGP-immunoblot of muscle (
Internexin and peripherin were both detected in all of our prenatal samples as was already described for postnatal specimen in rodent gut by others (Matini et al. 1997; Faussone-Pellegrini et al. 1999). We observed a slight developmental difference between peripherin, internexin, and PGP9.5, showing a discrete head start of PGP9.5 in front of the others. We were not able to detect a clear temporal difference in the expression of internexin and peripherin as it was described in rat intact intestine, where internexin was first detectable at embryonic day 18 (E 18), whereas peripherin could be seen from the day of birth on (Matini et al. 1997). In this case our results resemble those obtained in the mouse gut, where internexin and peripherin are both expressed from E 12.5 on until birth (Faussone-Pellegrini et al. 1999). In contrast to mouse gut, internexin was still present in adult human gut, as could be shown in this study.
In addition to these filamentous markers, we were also able to detect TH-positive cells in the human ENS. Similar results were obtained by Anlauf et al. (2003) who found that up to 20% of all intrinsic neurons in the gastrointestinal tract are TH+. They examined human tissue from 36- to 79-year-old patients that also shows that these cells are present throughout life, in accordance with our own results/observations. In contrast to humans, TH is only transiently expressed in rodents during embryonal life but not in adulthood, which led to the concept of the transient catecholaminergic innervation of the gut (Baetge et al. 1990). We did not see such a decrease or even a disappearance of TH+ tissue in myenteric ganglia of human gut when comparing pre- and postnatal samples.
The present study demonstrates, for the first time, the correlation of nestin and PGP9.5 expression, as well as the expression patterns of periphern, internexin, and TH in the fetal and prematurely born human gut. Further studies are needed to show whether in postnatal or adult ENS the nestin expression also indicates the presence of adult “enteric stem cells.”
Concerning “early enteric stem cells” PGP9.5 so far seems to be a more appropriate marker. Due to the fact that PGP9.5 is also a pan-neuronal marker, strong efforts have to be undertaken to identify other stem-cell markers that will be able to selectively identify neuronal stem cells, ideally at all ages. Interesting candidates such as p75, Phox2b (Young et al. 1999), and SOX10 (Bondurand et al. 1998; Kapur 1999; Sham et al. 2001) were found in various animal models and will be tested in our human samples.
Footnotes
Acknowledgements
This study was supported by Grant Deutsche Forschungs-gemeinschaft (DFG) SCHA 878/1-1.
We thank the Department of Pathology, Homburg, and the Department of Obstetrics at the University of Heidelberg for their logistic support. We thank Ailsa Boyce for proofreading the manuscript.
