Abstract
Current cytogenetic approaches in noninvasive prenatal diagnosis focus on fetal nucleated red blood cells in maternal blood. This practice may be too restrictive because a vast proportion of other fetal cells is ignored. Recent studies have indicated that fetal cells can be directly detected, without prior enrichment, in maternal blood samples by fluorescence in situ hybridization (FISH) analysis for chromosomes X and Y (XY-FISH). In our blinded analysis of 40 maternal blood samples, we therefore examined all fetal cells without any enrichment. Initial examinations using conventional XY-FISH indicated a low specificity of 69.4%, which could be improved to 89.5% by the use of two different Y-chromosome-specific probes (YY-FISH) with only a slight concomitant decrease in sensitivity (52.4% vs 42.9%). On average, 12–20 male fetal cells/ml of maternal blood were identified by XY- and YY-FISH, respectively.
A
Not only has the suitability of both enrichment strategies been questioned but also the use of the fetal NRBC as a sole target cell, because this cell appears to be refractory to fluorescence in situ hybridization (FISH) analysis (Babochkina et al. unpublished data). For this reason, the question has been raised whether a different target cell should be chosen or whether the analysis should be performed on all fetal cell types present in the maternal circulation. Evidence that the latter approach may be feasible was described in non-blinded studies for the first time by Hamada et al. (1993) and more recently by Krabchi et al. (2001). Hamada et al. (1993) published parallel investigations by FISH and polymerase chain reaction (PCR) in 50 pregnancies in which both methods used repetitive and single-copy sequences specific for the Y-chromosome. Analyzing ~100,000 nucleated cells per maternal blood sample, these investigators reported an overall sensitivity of 82.8% for Y-chromosome-specific FISH as well as increasing fetal cell numbers during pregnancy. At term, they detected up to 1 nucleated fetal cell in 10,000 maternal nucleated cells.
Krabchi et al. (2001) described the quantification of the entire fetal nucleated cell population in maternal blood in a morphology-independent manner using FISH. Their study indicated that 2–6 fetal nucleated cells were present per ml of maternal blood in 12 healthy pregnancies. Their procedure, which required minimal manipulation of cells, was based on a hypotonic treatment combined with Carnoy's fixation. This treatment removes the cell cytoplasm and causes swelling of the nuclei, thereby rendering all cell types readily amenable to FISH analysis.
To test this procedure in a larger study population and to assess the number of fetal cells per ml of maternal blood, we performed a blinded analysis of 40 samples from normal pregnancies with unknown fetal gender using two different sets of FISH probes.
Materials and Methods
Five hundred μl of whole maternal peripheral EDTA-blood was processed within 6 hr after blood sampling. After washing with RPMI 1640 (Invitrogen; Basel, Switzerland) the samples were incubated with prewarmed 0.4% KCl for 20 min at 37C, fixed several times with freshly prepared precooled Carnoy's fixative (methanol-glacial acetic acid, 3:1), and washed in 1% BSA/PBS.
Equal volumes of the cell suspension representing the entire cell population of 125 μl maternal whole blood were cytocentrifuged (four slides per case). Dual-color FISH using the chromosome enumeration probe cocktail CEPX spectrum green/CEPY [alpha (α)-satellite] spectrum orange (Vysis/Abbott Diagnostics; Baar, Switzerland), further referred to as XY-FISH, was performed on two slides per case. Pretreatment of slides encompassed 0.005% pepsin digestion (10 min) and fixation in 1% formaldehyde (10 min). The FISH probe cocktail was diluted 1:100 in cDenHyb-1 (Insitus Biotechnologies, Albuquerque, NM) and 10 μl per slide was applied. Co-denaturation at 72C for 8 min was followed by hybridization at 37C for 6 hr. Posthybridization washes were performed in 50% formamide/1.5 × SSC at 45C (5 min) and in 1.5 × SSC at 45C (2 min). After a final wash with distilled water, slides were counterstained with 0.01% DAPI (Sigma; Fluka Chemie, Buchs, Switzerland)/glycerol.
In addition, we performed dual-color FISH with two different Y-chromosome probes [CEPY α-satellite (spectrum orange) and CEPY III-satellite (spectrum aqua) (Vysis/Abbott), further referred to as YY-FISH] on the remaining two slides per case. Per cytospin, 1 μl of the centromeric Y-chromosome probe was applied, together with 1 μl of a 1:10 dilution of the III-satellite probe in chromosome enumeration probe (CEP) hybridization buffer (Vysis/Abbott). One microliter of distilled water and 7 μl CEP hybridization buffer were added, to a final volume of 10 μl. Co-denaturation was performed at 72C for 8 min, and hybridization followed at 37C for 16 hr.
In every hybridization, a male cord blood control slide was run in parallel to monitor the quality of the FISH performance. According to the manufacturer, the efficiency of the Y-chromosome-specific probe in the XY cocktail is 97.5%. In our hands, the efficiency on male cord blood control slides was calculated to be 98.5%, on average. Likewise, the manufacturer-described 99.5% efficiency of both single Y-chromosome probes was obtained in our controls.
Blinded evaluation of the FISH signals was performed by manually screening two cytospins per FISH probe combination using single bandpass filters. In the case of XY-FISH, the identification of any cell with a Y-chromosome α-satellite signal prompted us to check for the further presence of an X-chromosome signal. Identification of any cell with a Y-chromosome α-satellite signal in the case of YY-FISH was followed by checking for a second Y-chromosomal III-satellite signal. The latter hybridizations were also re-scanned to detect first the Y-chromosomal III-satellite signal and then re-checked for the presence of the Y-chromosomal α-satellite-signal. A cell was accepted to be of male origin only in cases of two Y-chromosomal signals, one of each color.
In general, all identified spots were counterchecked for nonspecificity in all other filters at a magnification of X1000. FISH results predicting fetal gender were compared with birth outcome only after all slides had been evaluated. For seven cases, the birth outcome was not available due to delivery outside of our clinic. Therefore, real-time PCR for glyceraldehyde 3-phosphate dehydrogenase (forward primer 5'CCCCACACACATGCACTTACC3', reverse primer 5'CCTAGTCCCAGGCCTTTGATT3', probe 5'AAAGAGCTAGGAAGGACAGGCAACTTGGC3') and SRY (forward primer 5'TCCTCAAAAGAAACCGTGCAT3', reverse primer 5'AGATTAATGGTTGCTAAGGACTGGAT3', probe 5'CACCAGCAGTAACTCCCCACAACCTCTTT3') on maternal plasma was performed as described by Zhong et al. (2001).
Results
To investigate all fetal cells in maternal whole blood irrespective of their morphology, we analyzed 40 healthy pregnancies in a blinded manner using two different FISH probe combinations. To monitor the quality of these FISH experiments, we correlated the predicted outcome with the fetal gender stated at birth or determined by real-time PCR on maternal plasma.
The results for the XY-FISH in Table 1 show a high percentage of false-positives in the case of female fetuses (31.6%) together with a low sensitivity (52.4%) in the pregnancies with male fetuses.
To test a possible improvement of the detection system for fetal cells, we performed FISH hybridizations in which a second Y-chromosome probe was substituted for the X-chromosome probe. Both Y-chromosome probes are complementary to different regions of the Y-chromosome and should provide a 1250-fold increase in FISH efficiency when applied together (0.5/100 for single probe to 0.0004/100 for the combination). The underlying FISH efficiency of 99.5% for both Y-chromosome probes described by the manufacturer was achieved in our experiments as well. For the evaluation, we considered all cases showing cells with only one Y-chromosome signal to be female, because insufficient indication (two Y-chromosome signals of different colors) had been obtained to indicate male status. Therefore, these pregnancies with an actual male fetus can be regarded as similar to false-negatives, whereas female pregnancies are correctly identified, despite false-positive FISH signals. On this basis, the results of the YY-FISH presented in Table 2 show a significant increase in specificity to 89.5% but also a slight reduction in sensitivity to 42.9% compared with XY-FISH.
FISH results using the XY-probe combination a

aResults of FISH analysis using the CEPX spectrum green/CEPY(α) spectrum orange cocktail. To predict the fetal gender, the results of both slides per case were taken into account. For the evaluation, a sample with at least one cell with one X- and one Y-chromosome signal on one slide was considered to represent a male fetus.
FISH results using the YY-probe combination a

aResults of FISH analysis with CEPY(α) spectrum orange/CEPY(III) spectrum aqua. For the prediction of fetal gender, the results of both slides per pregnancy were taken into account. A cell is considered to be of male origin only in a case of two Y-chromosome signals/cell with different colors. All cells with no Y-chromosome signal or one Y-chromosome-specific signal of either color are categorized as descending from female parentage with correct or incorrect FISH result.
In correctly identified male pregnancies, the number of fetal cells was used to calculate a total number of fetal cells per unit of maternal blood. Between one and five NRBCs with one X- and one Y-chromosome signal were identified on two slides in the first FISH experiments, which accounts for 4–20 fetal cells/1 ml of maternal blood. In the second FISH experiments, one to nine fetal NRBCs with two different Y-chromosome signals could be detected on two slides, indicating a concentration of 4–36 fetal cells/ml of maternal blood.
In regard to the quality of FISH signals, we obtained easily identifiable spots for all fluorochromes on male cord blood control slides and for the maternal X-chromosomes in the patients' samples. In contrast, the signals in the fetal cells were much smaller and significantly less intense, which renders both identification and counterchecking more difficult (Figures 1 and 2).
Discussion
Comparing our data to those of similar earlier studies, Hamada et al. (1993), using Y-chromosome-specific FISH, reported an overall sensitivity of 82.8% and specificity of 93.3% in 50 pregnancies, as well as 75% sensitivity and 100% specificity in another 30 pregnancies (Hamada et al. 1995). Krabchi et al. (2001) showed 100% sensitivity in female control preparations and 100% specificity for 12 male pregnancies, 10 of which were analyzed by XY-chromosome FISH and two by XY-chromosome PRINS. Both studies were evaluated in a non-blinded manner.
Applying the described cell treatment protocol of hypotonic treatment and Carnoy's fixation after MACS enrichment (CD13-, CD5-, CD 71+), Hromadnikova et al. (2002) reported complications in evaluation. These were caused by lack of cells or damage to the isolated cells in seven of eight analyzed cases. Because we used the same experimental set-up for the preparation of the FISH slides as Krabchi et al. (2001), we suggest that the lower values we obtained for specificity and sensitivity (89.5% and 42.9%, respectively) might be due to procedure-inherent factors influencing the FISH efficiency (Hromadnikova et al. 2002). However, the lower sensitivity and specificity levels in our experimental conditions mirror much better the situation of a real noninvasive diagnostic setting, because all samples were evaluated in a blinded manner, which is in contrast to the studies performed by Hamada et al. (1993) and Krabchi et al. (2001). In our more realistic scenario, a significant increase in specificity has been achieved by substituting for the X-chromosome probe a second Y-chromosome probe from a different region of the Y-chromosome. Notwithstanding the slight reduction in sensitivity, this probe combination renders the assay for detection of male fetal cells much more reliable. This positive effect is also shown by the low correlation of pregnancy outcome to the predicted outcome of cases with cells showing one Y-chromosome signal of either color only (Table 2). Owing to the incorporation of the second Y-chromosome probe, 78.9% of the female pregnancies are correctly identified; on the basis of only one Y-chromosome probe of either color, these would have been wrongly counted as male pregnancies.
Because this study comprised not only erythroblasts but all kinds of fetal cells, it is possible that fetal cells persisting from previous pregnancies could contribute to some of the false-positive cases. Contamination of the samples with Y-chromosome material during handling of the probes can be excluded because only female examiners processed the samples.
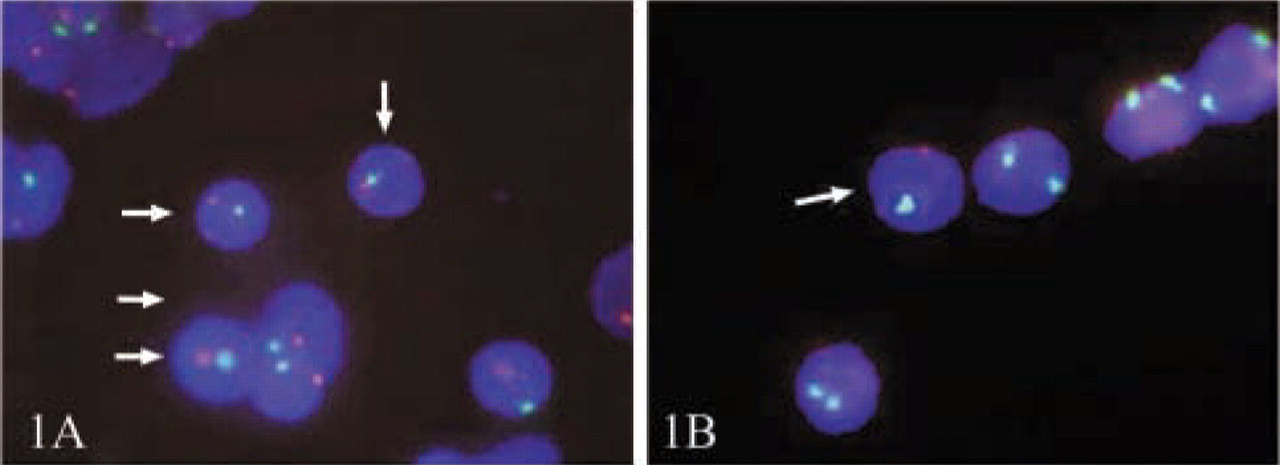
Comparison of XY-FISH quality in male cord-blood control cells and male fetal cells on maternal slides. (
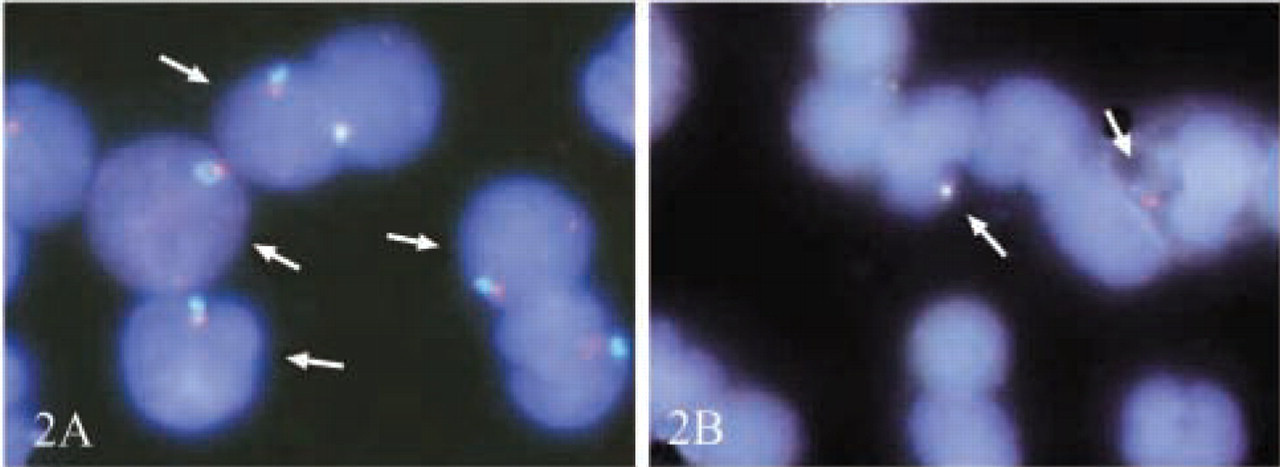
Comparison of YY-FISH efficiency in male cord-blood control cells and male fetal cells on maternal slides. (
In an interstudy comparison regarding fetal cell numbers, the average concentration of 2 fetal cells/1 ml maternal blood found by Krabchi et al. (2001) matches approximately the concentration of 1.2 fetal cell equivalents per 1 ml of maternal blood found by Bianchi et al. (1997) using PCR without prior enrichment. However, our results exceed these average cell numbers to a rather large extent. The reasons for these differences might be the much broader range of gestational age represented in our patients than in the study populations of Krabchi et al. (2001) and Bianchi et al. (1997), as well as the differences in our treatment protocol compared with the procedure described by Bianchi et al. (1997).
In our study, no male fetal cells could be detected in 9/21 (42.8%) pregnancies with a male fetus. Therefore the amount of 500 μl of maternal whole blood does not appear to be sufficient to guarantee the presence of a small but reliable number of fetal cells of any morphology for use in noninvasive prenatal diagnosis.
Because the nuclei of fetal NRBCs are tightly condensed, the observed differences in the quality of the FISH signals between control cells and maternal cells versus fetal cells were not completely unexpected. However, because in the present cell preparation, fetal cells of any origin and morphology are present, we also expected to find some XY- and YY-chromosome signals of normal size and fluorescence intensity. The lack of these cells suggests that all fetal cells that migrate into the maternal circulation undergo physiological changes that leave the cells in a status not amenable to reliable FISH analysis.
