Abstract
The tyramide signal amplification (TSA) technique has been shown to detect scarce tissue antigens in light and electron microscopy. In this study we applied the TSA technique at the electron microscopic level to pre-embedding immunocytochemistry. This protocol was compared to the non-amplified protocol. With the TSA protocol, the labeling of GM130, a cis-Golgi matrix protein, was tested in a cell line and found to be highly sensitive and more enhanced than that with the simple protocol. Moreover, the gold particles were well localized to the cis-side of the Golgi apparatus in both the TSA and the simple protocol.
Since the tyramide signal amplification (TSA) technique was successfully introduced in immunoassays (Bobrow et al. 1989), this novel approach has widely been used for in situ hybridization and immunocytochemistry at the light (Köhler et al. 2000) and electron (Mayer and Bendayan 1997) microscopic levels. This technique is based on the ability of HRP, to which the antigen-antibody complex binds indirectly, to catalyze the deposition of labeled tyramide onto proteins surrounding the HRP. This deposition is believed to take place at the site of the enzyme reaction, thus leading to good resolution.
Here we report the use of the TSA method at the electron microscopic level using a pre-embedding nanogold-silver staining for visualization. A human melanoma cell line, G361, was purchased and cultured in RPMI supplemented with 10% FBS, and then fixed in 3.0% glutaraldehyde in 0.1 M phosphate buffer. The fixed cells were immersed in 1% sodium borohydride in PBS to block free aldehydes. Cells were quenched in 3% H2O2 in 60% methanol to inactivate endogenous peroxidase and then incubated in blocking buffer (10% normal horse serum, 1% bovine serum albumin, 0.1% gelatin in PBS). With the simple protocol, cells were incubated in anti-GM130 Ab (1.0 μg/ml or 0.1 μg/ml) (BD Transduction Laboratories; San Diego, CA), biotinylated secondary antibody (1:200), and then streptavidin-nanogold (1:100) (Nanoprobes; Stony Brook, NY) in incubation buffer (10% normal horse serum, 1% bovine serum albumin, 0.1% gelatin in PBS). With the TSA protocol, the signal was amplified by the primary antibody (0.1 μg/ml, anti-GM130 Ab), the biotinylated secondary antibody (1:200), streptavidin-HRP (1:500), biotinyl-tyramide (1:50) (PerkinElmer Life Sciences; Boston, MA), and the streptavidin-nanogold (1:100). Streptavidin-HRP was diluted in incubation buffer [10% normal horse serum, 1% BSA, 0.1% gelatin in TBS (0.15 M NaCl, 50 mM Tris-HCl, pH 7.5)] and biotinyl-tyramide diluted in the 1 × amplification diluent (PerkinElmer). Next, cells underwent HQ silver enhancement (Nanoprobes) and gold toning followed by Epon infiltration. The sections were examined with a JEOL electron microscope.
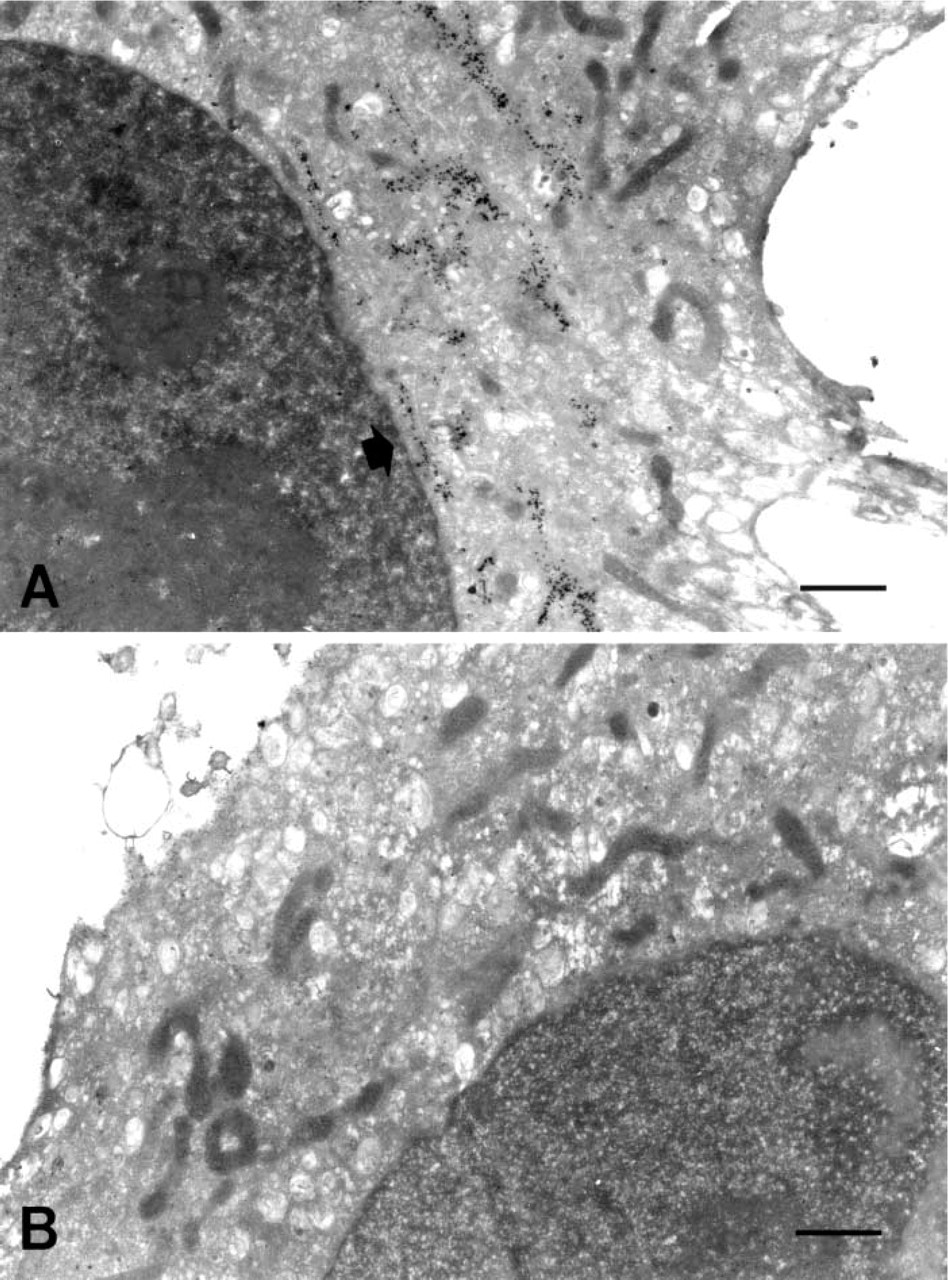
First, we performed the simple protocol with the recommended use (1.0 μg/ml) of anti-GM antibody. The simple protocol led to gold particle labeling specific over the cis-side of Golgi apparatus (Figure 1A). Omission of the primary antibody in the labeling protocol led to very low background labeling (Figure 1B). Preservation of the cellular ultrastructure was fairly good and many organelles, including nucleus, mitochondria, and Golgi apparatus, could be recognized.
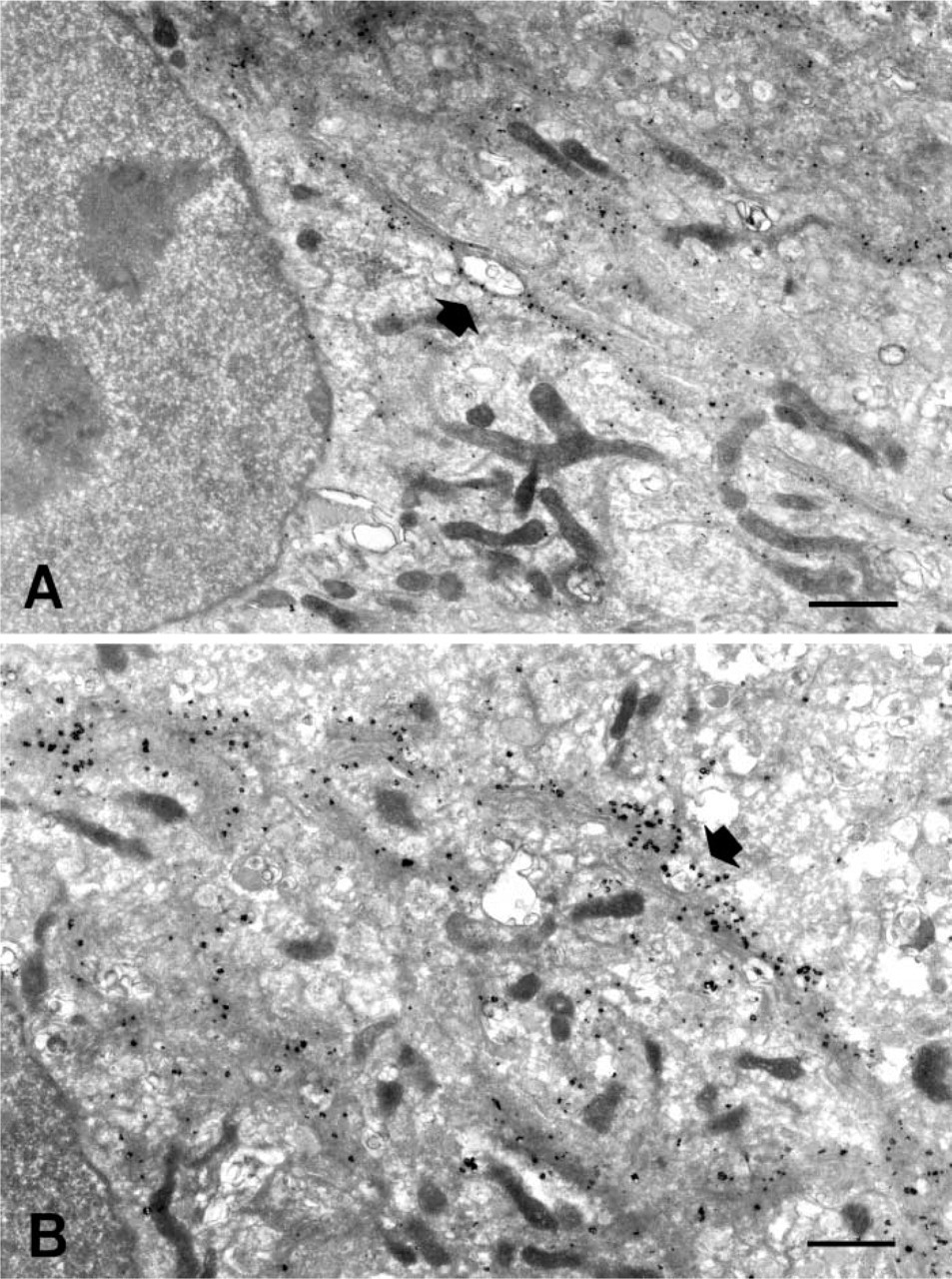
With a rather diluted primary antibody (0.1 μg/ml, 10 times less than the recommended use), we assessed the TSA application and compared it with the simple protocol. Both the TSA technique and the simple protocol yielded a particulate black signal over the Golgi apparatus. However, the signal of gold particles was somewhat more intense with the TSA technique than with the simple protocol (Figures 2A and 2B). Gold particles were quite limited within the Golgi apparatus both in the non-amplified and amplified protocols. The signal appeared as a collection of gold particles localized along the ribbons of the Golgi cisternae, contrasting with the nucleus and mitochondria, which were devoid of labeling.
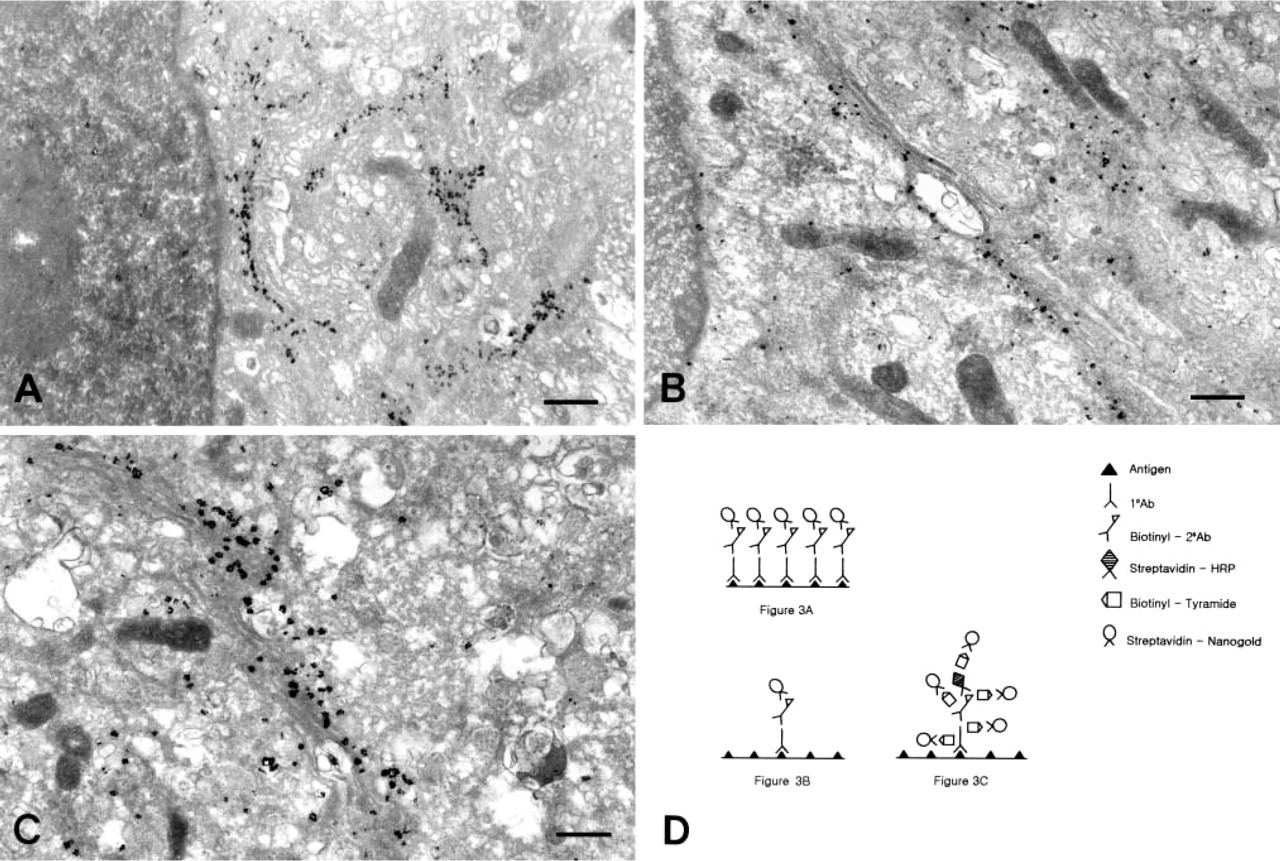
The high-magnification view of the three samples (simple protocol 1.0 μg/ml anti-GM130; simple protocol 0.1 μg/ml anti-GM130; TSA protocol 0.1 μg/ml anti-GM130) revealed the nature of the enhancement observed in the TSA protocol. The size of the particles in the Golgi apparatus was greater for the TSA protocol using 0.1 μg/ml anti-GM130 (Figure 3C) than for the simple protocol using 0.1 μg/ml anti-GM130 (Figure 3B). However, the numbers of particles in the Golgi cisternae were similar between them. This finding became quite clear when the TSA protocol using 0.1 μg/ml anti-GM130 (Figure 3C) was compared to the simple protocol using 1.0 μg/ml anti-GM130 (Figure 3A). The size of particles in the Golgi apparatus is bigger in Figure 3C but the number of particles is higher in Figure 3A. This shows that the TSA enhancement has an effect by increasing the size but not the number of gold particles bound to the antigen site.
One group has performed signal amplification using biotinylated tyramide and nanonogold-silver staining at the light microscopic level (Köhler et al. 2000). However, our present report is the first on the TSA application to pre-embedding nanogold-silver staining at the electron microscopic level.
We found that a high concentration (3%) of glutaraldehyde can be used for fixation and for obtaining good sensitivity in immunocytochemistry without yielding any nonspecific binding (Kosaka et al. 1986). This can be done by the additional step of quenching (or free aldehyde blocking) with 1% sodium borohydride.
The issue of resolution with the TSA technique also seems to be mainly related to the working mechanism of tyramide. The gold particles with the TSA protocol, bound to the biotin-tyramide, are well localized within the cis-Golgi apparatus to a degree comparable to those with the simple protocols (Figures 3A-3C). This finding is in accordance with the result that resolution of the TSA technique in post-embedding immunocytochemistry remains very good (Mayer and Bendayan 1997).
High-magnification views
Footnotes
Acknowledgements
Supported by grant no. R01-2002-000-00119-0 from the Basic Research Program of the Korea Science & Engineering Foundation.
