Abstract
Hyaluronan is an unsulfated glycosaminoglycan (GAG) that is ubiquitously expressed in the extracellular matrix (ECM) of all vertebrates, where hyaluronan rich matrices constitute a particular permissive environment for the development of complex biological structures and also for tumor progression. Because of its conserved structure and ubiquitous expression, antibodies for its histochemical detection cannot be produced. We have engineered a fusion protein, neurocan–GFP, and expressed it as a secreted molecule in mammalian cells. Neurocan–GFP fusion protein specifically binds to hyaluronan and directly visualizes hyaluronan on tissue sections, revealing a very detailed picture of hyaluronan distribution. The fluorescent fusion protein can be used in combination with antibodies and nuclear markers for double or triple staining. In addition, it is suitable to visualize hyaluronan on living cells by time-lapse video microscopy. The successful production and application of the neurocan–GFP fusion protein opens up new perspectives for using GFP fusion proteins as detection tools in histological and cytological studies complementing conventional antibody and biotin/avidin techniques.
H
Because of its conserved structure and ubiquitous expression, histological detection of hyaluronan requires specific methods other than application of antibodies. The currently used method utilizes hyaluronan-binding proteins and protein fragments modified with biotin. To generate these probes, hyaluronan and its binding proteins are co-purified from animal tissue, and the proteins are biotinylated and separated from hyaluronan (Ripellino et al. 1988). These biotinylated probes are then visualized by avidin/strepavidin conjugated with enzymes or other detection aids.
Here we introduce a simple one-step method to detect hyaluronan with high spatial resolution. It is based on a fusion protein consisting of a GFP module and the hyaluronan-binding domain of neurocan, a PG of the lectican family (Rauch et al. 2001). The fusion protein was expressed and secreted by mammalian cells. To validate the fusion protein as a histochemical detection aid, mammalian eyes were used because their sections comprise many different types of tissues in a relatively small area. Furthermore, the distribution of hyaluronan in this organ appeared well characterized (Chan et al. 1997; Gong et al. 1997; Hollyfield et al. 1997, 1998; Lerner et al. 1997, 1998). Our analysis demonstrated the suitability of the fusion protein for affinity histochemical applications. In addition, we found that the fusion protein was also able to visualize hyaluronan on living cells using time-lapse video microscopic observation.
Materials and Methods
Production of Neurocan–GFP Fusion Proteins
The N-terminus of the rat neurocan hyaluronan-binding domain, starting with aspartic acid 23, was linked to the BM 40 signal peptide via an APLGRGSHHHHHHGGLA linker sequence (Oohashi et al. 2002), and the C-terminus ending with histidine 359 was fused via a GSSGA linker to methionine 1 of EGFP (Clontech; Palo Alto, CA). The cDNA encoding this fusion protein was cloned into an episomal expression vector with a CMV promoter, SV 40 polyadenylation site, and a puromycin resistance cassette (Kohfeldt et al. 1996). The plasmid was transfected into HEK 293 EBNA cells with lipofectamine (Invitrogen; Carlsbad, CA), and cells were maintained with 1 mg/liter puromycin in DMEM/F12 medium containing 10% fetal calf serum. Conditioned serum-free medium was collected, supplemented with 0.05% NaN3 and 1 mM N-ethylmaleimide, and concentrated by centricon 30 (Amicon) centrifugation. For staining of living cells, medium lacking puromycin was conditioned for 24 hr.
SDS-PAGE and Blotting
SDS-PAGE was performed on slab gels using the Bio-Rad (Hercules, CA) mini gel system. Proteins in conditioned medium were precipitated with trichloroacetic acid, washed with ice-cold acetone, and dissolved in SDS-PAGE sample buffer containing 2% SDS with no reducing agents. Blots were performed in Tris/glycine buffer containing 10% methanol for 1 hr at 100 V using the Bio-Rad mini gel system. For immunodetection, proteins were transferred to PVDF membrane (Amersham; Arlington Heights, IL), which was blocked with 5% dry milk in 150 mM NaCl, 20 mM Tris/HCl, pH 8, with 0.1% Tween-20 (TBST) overnight, and incubated with an antiserum against the N-terminal neurocan domain in TBST (1:7000). This rabbit antiserum was raised against the N-terminal neurocan fragment 359H recombinantly expressed in HEK 293 cells and purified by ConA affinity and consecutive Superose 12 chromatography (Retzler et al. 1996). After a further incubation with horseradish peroxidase-linked donkey anti-rabbit antibody (Jackson; West Grove, PA) in TBST (1:20 000), the blot was developed with the ECL plus detection system (Amersham).
Tissue Preparation
Procedures for handling mice were approved by the Committee for Ethics in Animal Experiments in Malmö and Lund. Mice (129SW) were anesthetized by CO2 and decapitated. The eyes were enucleated and immersion-fixed in cold Bouin fixation solution for 12–18 hr at 4C. The eyes were embedded in paraffin, sectioned at 6 μm and stored at −80C.
Fusion Protein Staining
Sections were dewaxed with xylene (three times for 5 min) and an ethanol series (100%, 90%, 70%, and 50%) 5 min for each. After washes in phosphate buffer (20 mM NaxH3-xPO4, 150 mM NaCl, pH 7.4) three times for 5 min, the sections were blocked in blocking solution (2% normal goat serum in phosphate buffer) for 1 hr at room temperature. The fusion protein was diluted 1:3 in blocking solution and briefly centrifuged at 14,000 × g before it was applied to sections. The fusion protein was incubated on sections overnight at 4C. The next day unbound fusion protein was washed away with phosphate buffer (three times for 5 min). Slides were counterstained with propidium iodide and then coverslipped with Vectashield (Vector Laboratories; Burlin-game, CA). For triple staining the fusion protein was incubated together with an anti-laminin-1 antiserum (455, 1:500; kindly provided by Dr. Lydia Sorokin, Lund) in blocking solution overnight at 4C. Omitting the fusion protein, the secondary antibody (Cy3-conjugated goat anti-rabbit antibody; Jackson) was incubated at RT for 30 min in phosphate buffer. The slides were washed in phosphate buffer and counterstained with DAPI.
Hyaluronan Digestion In Situ
Deparaffinized sections were incubated for 3 hr at 37C with 100 U/ml Streptomyces hyaluronate lyase (Sigma; St Louis, MO) in 50 mM Na acetate buffer, pH 5, in the presence of 5 mM EDTA, 5 mM benzamidine/HCl, 5 mM N-ethylmaleimide, and 1 mM freshly added phenylmethylsulfonylfluoride. Control samples were incubated under the same conditions without the enzyme.
Probe Preabsorption
The fusion protein was preincubated with 1 mg/ml hyaluronic acid (Sigma H5388) for 30 min at RT and then applied to adjacent sections.
Fluorescence Microscopy
Specimens were viewed with an epifluorescent microscope (Axiophot; Zeiss, Oberkochen, Germany) and images were taken with digital cameras AxioCam (Zeiss) or C4742–95–12NRG (Hamamatsu; Tokyo, Japan) with Openlab software (Improvision; Tucson, AZ). The images were processed using Openlab and Photoshop software (Version 6.0; Adobe, Tucson, AZ).
Time-lapse Video Microscopy
Specimens were viewed every 2 min by phase-contrast and epifluorescent microscopy (Axiovert 200M; Zeiss) at 37C and 5% CO2 in a humidified chamber. Images were taken with the digital camera C4742–95–12ERG (Hamamatsu) and merged with Openlab software (Improvision). The images were combined using Photoshop software (Version 6.0; Adobe).
Results
The cDNA coding for the rat neurocan hyaluronan-binding domain was fused to the GFP cDNA (Figure 1A) and transfected into HEK 293 cells. Conditioned serum-free medium was analyzed by Western blotting with an antiserum against the hyaluronan-binding domain of neurocan (fragment 359H). Under non-reducing conditions, a major protein band at 70 kD was detected (Figure 1B), which is the expected combined size of 45 kD from the neurocan hyaluronan-binding domain and 26 kD from the GFP module (Retzler et al. 1996; Mahajan et al. 1998).
To validate the fusion protein as a histochemical probe, we used tissue sections from the mouse eye. In inner-ocular structures such as the ciliary body and the retina, hyaluronan is particularly evident one week after birth (Figures 2A and 2C), although even at this stage hyaluronan was more prominent in surrounding tissues such as muscle, episclera, sclera, and conjunctiva, as has been observed in adult mice (Hollyfield et al. 1997). To verify the binding specificity of the fusion protein to hyaluronan, the two controls established for conventional hyaluronan detection were performed: preincubation with excess hyaluronan, and hyaluronidase treatment of the section (Wang et al. 1996; Hollyfield et al. 1998). For the preincubation, the conditioned medium containing the fusion proteins was incubated with soluble hyaluronan before it was applied to the sections. This procedure completely abrogated staining of the tissue sections (Figures 2A and 2B). For the hyaluronidase treatment, the tissue sections were incubated with Streptomyces hyaluronidase to selectively eliminate hyaluronan in the sections. This specific hyaluronan digestion in tissue sections completely abolished the staining (Figures 2C and 2D).
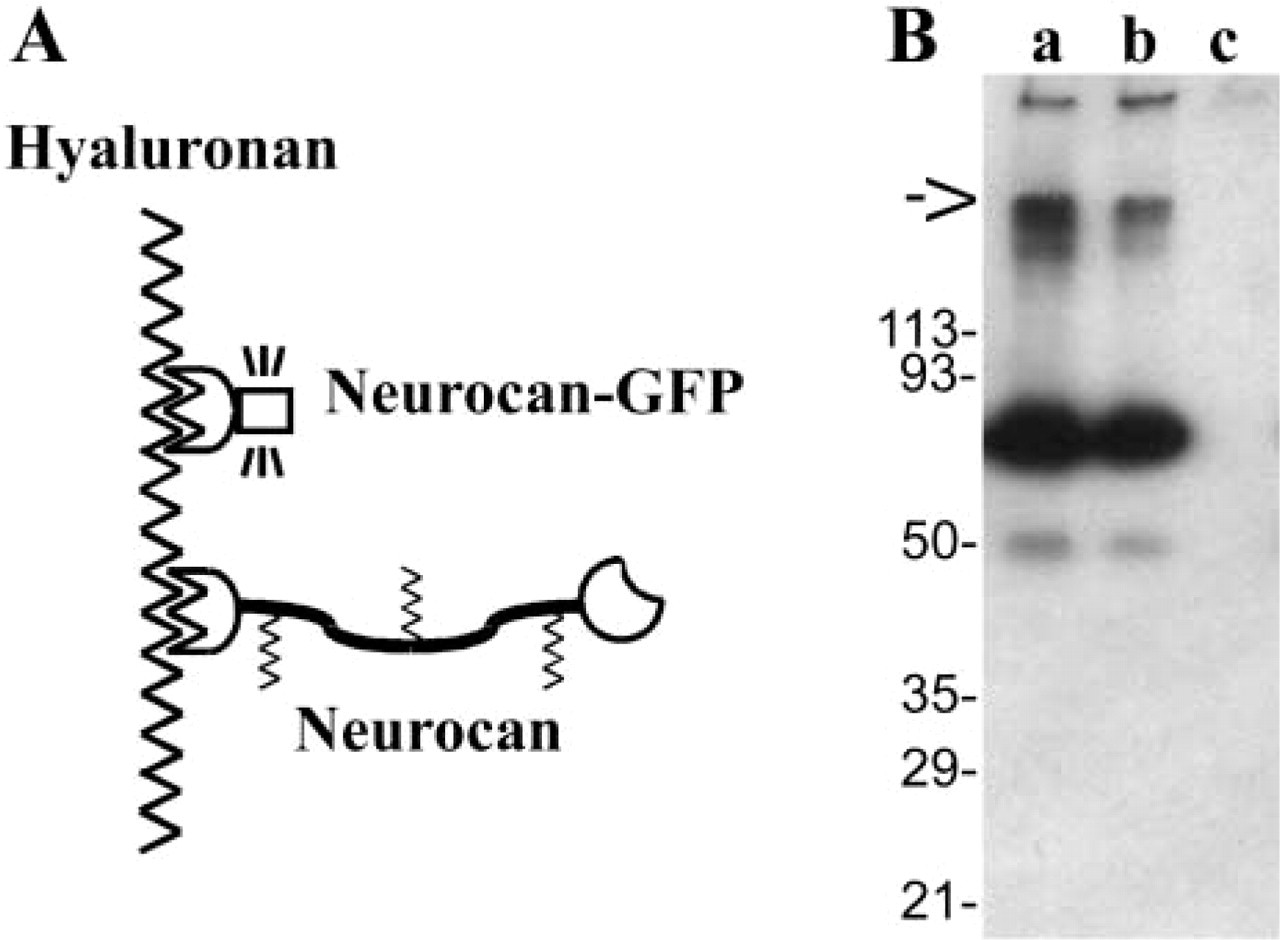
Design (
The suitability of the fusion protein for triple staining was demonstrated in the ciliary body by a co-incubation with anti-laminin antibodies and consecutive nuclear counterstaining (Figures 2E–2J). At postnatal day 6, hyaluronan and laminin are located along the basement membrane of the inner and outer epithelial cells (Figures 2E–2G). Hyaluronan is predominantly located on the surface of the inner epithelial cells, outlining their profiles (Figures 2E–2G). In the adult eye, both molecules are dramatically reduced at this site but appear essentially unchanged at the basement membrane of the outer epithelial cells (Figures 2H–2J).
In the dog eye, hyaluronan staining was seen clearly in the vitreous (Figure 3A), where hyaluronan was first identified and commonly is considered as reference for hyaluronan deposition in mammals. In addition, this staining, which extended in radial strands into the ganglion cell layer (Figure 3C), was no longer evident after treatment with hyaluronidase (Figures 3B and 3D).
In general, the probe could be applied to sections processed in different ways, such as Bouin-fixed paraffin sections and paraformaldehyde-fixed cryostat sections, without any obvious difference in the intensity of staining. In addition, we found that in sections mounted in fluorescence preserving medium the staining retained its intensity after several months of refrigeration.
Because it is possible to apply the fusion protein without any secondary incubation or color reaction, it was tested for its suitability in a real-time application such as time-lapse video microscopy. The culture medium of native, untransfected HEK 293 cells was changed to medium that had been conditioned by cells secreting the fusion protein. Consecutively distinct fluorescent patches appeared on the cells. Time-lapse video microscopy revealed a dynamic redistribution of the fluorescence signal in a reproducible pattern. The staining frequently accumulated at sites where cells showed a tendency to physically segregate, but parts of the cell membrane were still attached to each other (Figure 4).
Discussion
The hyaluronan-binding domain of neurocan was fused to a GFP module to serve as a detection probe for hyaluronan. This fusion protein represents a new class of fluorescent detection tools combining the properties of carbohydrate-binding domains and fluorescent protein modules. The hyaluronan-binding domain of neurocan consisting of three modules, one immunoglobulin-like module and two link modules, has previously been shown to be efficiently produced in eukaryotic cells and to interact with hyaluronan (Retzler et al. 1996; Talts et al. 2000). Homologous hyaluronan-binding domains are present in the three other lectican family members, aggrecan, versican, and brevican, and constitute the four link proteins (Yamaguchi 2000; Spicer et al. 2003). However, problems encountered in attempts to recombinantly express equivalent domains of aggrecan and versican have been reported (Cao and Yang 1999; Yang et al. 2000), and cartilage link protein, the best characterized of the four link proteins, was found to have a tendency to oligomerize (Neame and Barry 1993; and our unpublished observations). These observations rendered especially the neurocan N-terminal domain as a promising candidate for the preparative production of hyaluronan-binding–GFP fusion proteins.
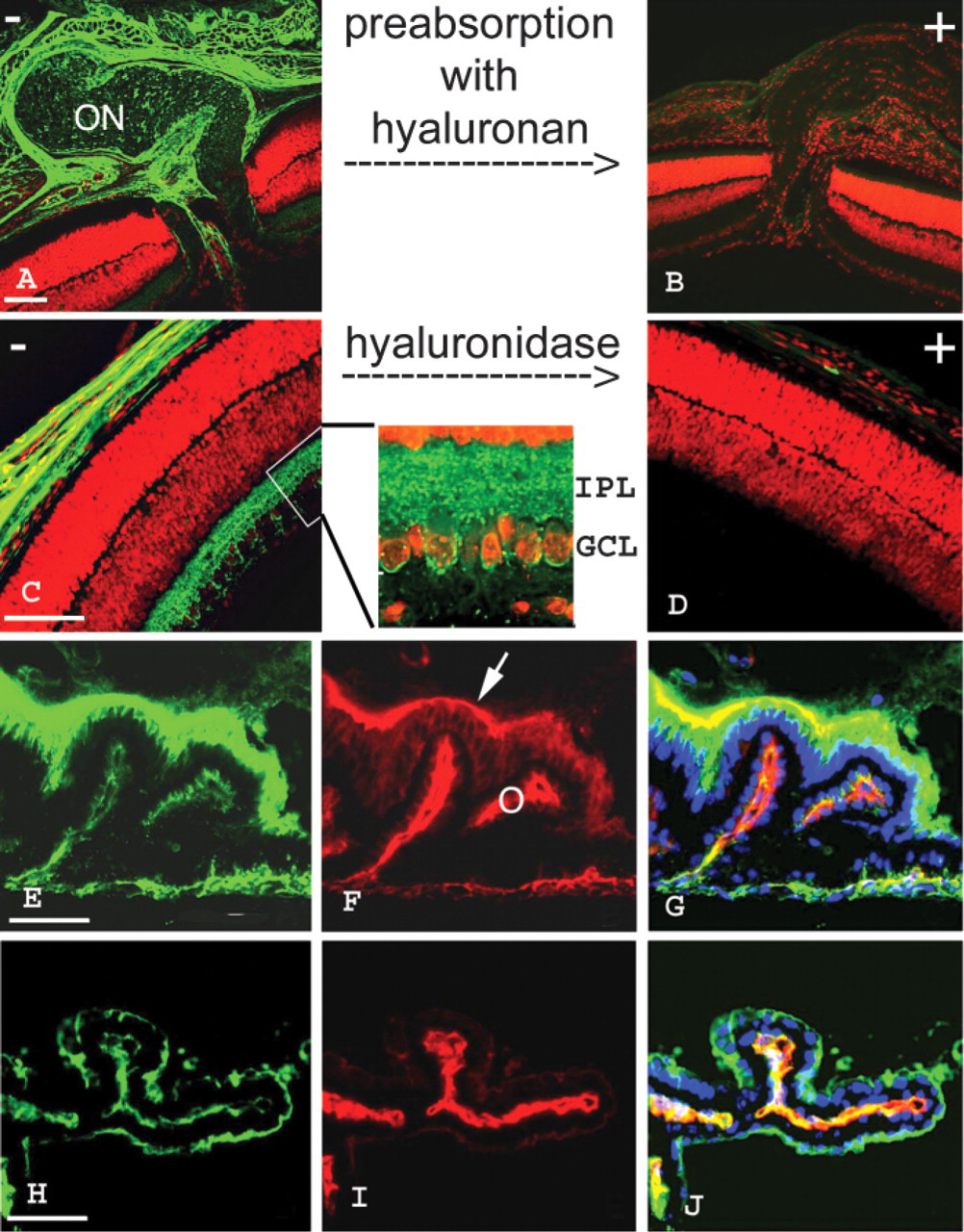
Histochemical staining of hyaluronan in mouse eye sections with the neurocan-GFP fusion protein. (
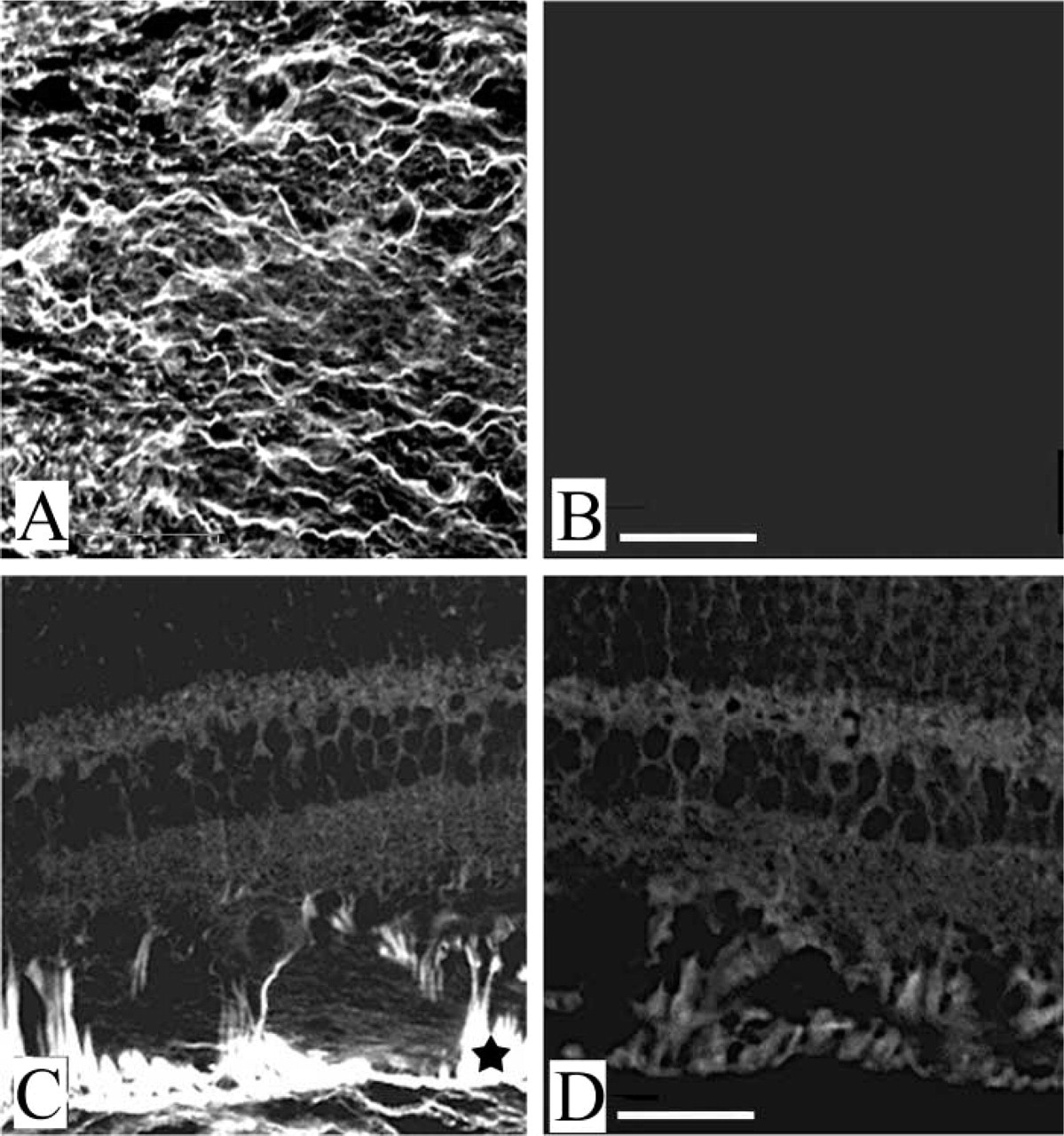
Hyaluronan expression in dog retina and vitreous. Hyaluronan is clearly evident in the dog vitreous (
To ensure that this fusion protein would bind to hyaluronan, proper folding of this molecule, especially of the hyaluronan-binding domain, was considered to be very important. We chose to express the fusion protein in mammalian cells, where molecules in the secretory pathway are usually subjected to an endogenous “proofreading” system that does not allow the export of misfolded proteins (Vashist et al. 2001). The hyaluronan-binding domain of neurocan alone has been previously successfully expressed in this system (Retzler et al. 1996). An analysis of the medium conditioned by fusion protein-secreting HEK 293 cells by Western blotting confirmed that it was possible to obtain distinct monomeric neurocan–GFP fusion proteins. Because the antiserum used was raised against the N-terminal neurocan domain, which represents the major part of the fusion protein, the observation of a dominant band at the expected size of the fusion protein and only a minor band of a size expected for the hyaluronan-binding domain alone indicated that no extensive degradation had occurred. Moreover, because the SDS-PAGE was performed under non-reducing conditions, this observation also excludes extensive aberrant covalent associations of the fusion proteins via disulfide bonds.
The direct fusion of the fluorescent detection module to the target recognition domain should reveal the localization of hyaluronan with the highest possible resolution. To fully exploit the spatial resolution of the fusion protein as probe, we used Bouin fixation, which is considered to maintain the tissue structure very well. As presented in this study, the probe gives a detailed staining pattern, e.g., showing the localization of hyaluronan between cells within the retinal inner plexiform/ganglion cell layer. It should be noted that those signals were obtained without any amplification mediated by secondary detection aids. Those are likely to cause a broadening of the stained area, thereby obscuring the exact spatial distribution. Furthermore, compared with the conventional detection method, this probe offers the possibility to be used together with biotinylated probes, such as lectins, for double or triple staining.
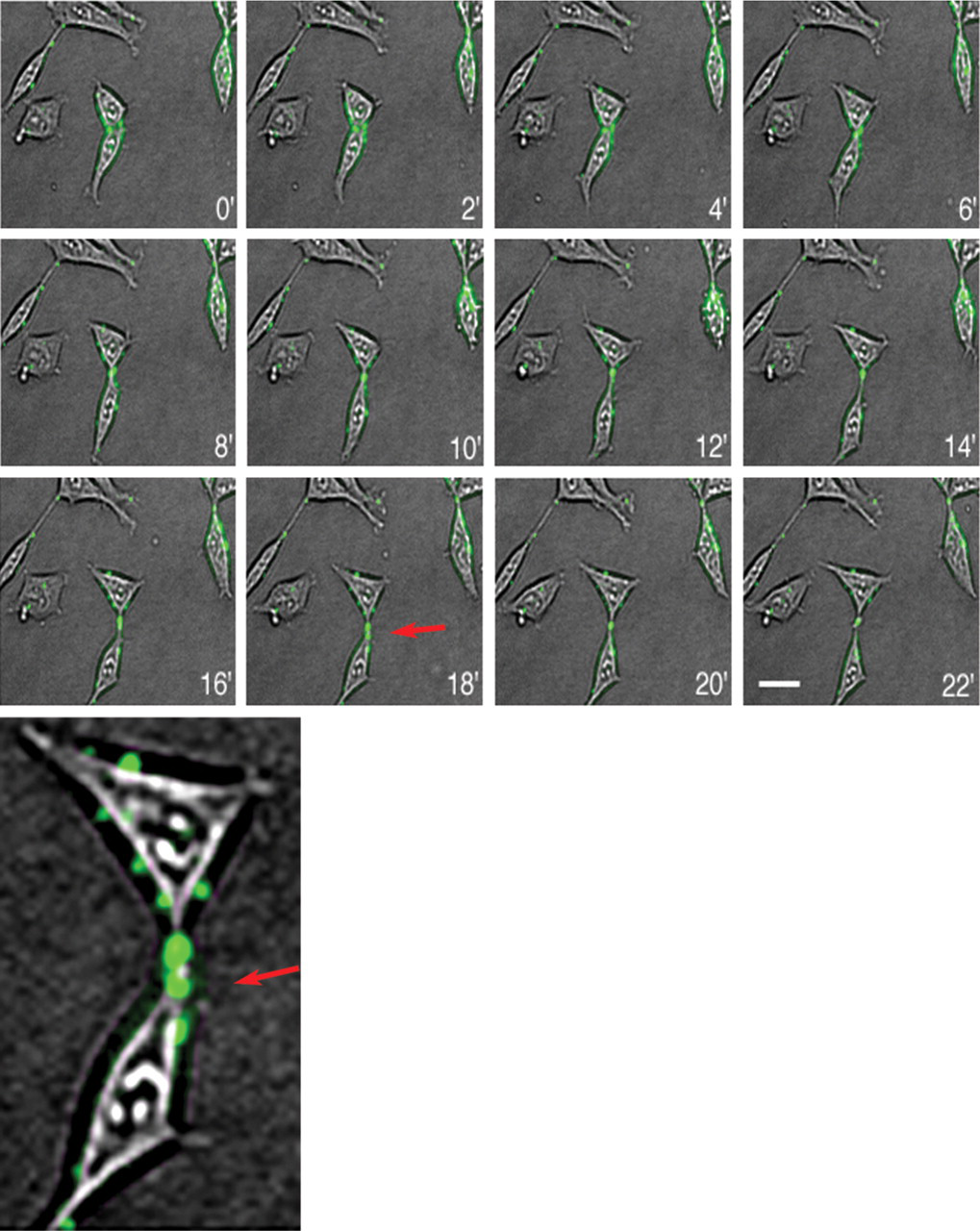
Time-lapse video observation of hyaluronan on cultured HEK 293 cells. Individual phase-contrast and fluorescence images were taken every 2 min over a 22-min period and merged. Note the frequent localization of hyaluronan deposits at connecting cytoplasmic processes between marginally interacting cells (red arrows in the 18-min picture and in the enlarged part of this picture. (The low resolution in this figure is caused by the low resolution of the original pictures due to technical limitations from the time-lapse setup.) Bar = 20 μm.
The specificity of hyaluronan detection was demonstrated by abolition of the binding after preincubation of the probe with excess hyaluronan, which specifically absorbed the probe from the concentrated conditioned medium. A second demonstration of the specificity was the lack of binding of the probe after treatment of the tissue sections with Streptomyces hyaluronidase, which selectively digests hyaluronan. Both experiments indicate that the fusion protein is a specific probe for the detection of hyaluronan comparable to the established method with hyaluronan-binding proteins isolated from bovine cartilage or sheep brain (Wang et al. 1996; Victor et al. 1999). Because the international exchange of products derived from animals is prone to restrictions, fusion proteins, which can be produced in any laboratory culturing cells, may become a reliable source for affinity histochemical probes.
Considering that hyaluronan was first identified as a component of the vitreous of the eye, a surprisingly low level of hyaluronan was evident in the mouse vitreous. To be sure that the lack of hyaluronan in the mouse vitreous is not due to the detection method, we analyzed also the vitreous of dog eyes, in which hyaluronan was clearly apparent. The low hyaluronan levels might be related to the small size of the mouse vitreous compared with that of other mammalian species, and might indicate that, for this structure, hyaluronan is not of crucial functional importance.
The observation of fluorescent patches on living cells rather than a general staining of the entire cell surface indicates a focal concentration of hyaluronan to distinct areas. This distribution was possibly mediated by hyaluronan-binding cell surface molecules. Investigations of cultured periodontal ligament fibroblasts, which were fixed with methanol, indicate indeed a localization of CD44 in focal deposits at the edges and in cytoplasmic processes of cells (Zohar et al. 2000).
GFP fusion proteins produced in mammalian cells appear to be especially suitable for the detection of carbohydrate structures. These structures are less sensitive to denaturing and crosslinking fixation procedures and are therefore structurally better conserved in histological applications, while their usually extracellular location renders them accessible to fusion proteins supplied in the culture medium in cytological and cell biological applications. On the other hand, protein domains, which recognize carbohydrates, can be quite complex and require more than a single protein module to achieve the necessary specificity (Tu et al. 1996). These domains may display complex disulfide linkage patterns or depend for their activity on typical vertebrate glycosylations (Sgroi et al. 1996). However, this study demonstrates that it is possible to express in a preparative fashion carbohydrate-binding protein domains linked to GFP in mammalian cells.
In summary, a GFP fusion protein produced in and secreted from mammalian cells successfully detected hyaluronan in fixed and living biological material, opening up new perspectives for using GFP fusion proteins as detection tools in histological and cytological studies.
Footnotes
Acknowledgements
Supported by the Swedish Natural Sciences Research Council (UR #B650–19981127/2001), the Tage Bluchers Stiftelse, the Alfred österlunds Stiftelse, the H och J Forssmans Fond, the Greta och Johan Kocks Stiftelser, and the Crafoordska Stiftelsen.
We thank Drs Reinhard Fässler, Lydia Sorokin, and Ronny Fransson-Steen for support and Dr Berndt Ehinger for critical comments. The dog sections were kindly provided by Department of Pathology, AstraZeneca in Sweden.
