Abstract
Obscurin is a newly identified giant muscle protein whose functions remain to be elucidated. In this study we used high-resolution confocal microscopy to examine the dynamics of obscurin localization in cultures of rat cardiac myocytes during the assembly and disassembly of myofibrils. Double immunolabeling of neonatal and adult rat cells for obscurin and sarcomeric α-actinin, the major protein of Z-lines, demonstrated that, during myofibrillogenesis, obscurin is intensely incorporated into M-band areas of A-bands and, to a lesser extent, in Z-lines of newly formed sarcomeres. Presarcomeric structural precursors of myofibrils were intensely immunopositive for α-actinin and, unlike mature myofibrils, weakly immunopositive or immunonegative for obscurin. This indicates that most of the obscurin assembles in developing myofibrils after abundant incorporation of α-actinin and that massive integration of obscurin occurs at more advanced stages of sarcomere assembly. Immunoreactivity for obscurin in the middle of A-bands and in Z-lines of sarcomeres bridged the gaps between individual bundles of newly formed myofibrils, suggesting that this protein appears to be directly involved in their primary lateral connection and registered alignment into larger clusters. Close sarcomeric localization of obscurin and titin suggests that they may interact during myofibril assembly. Interestingly, the laterally aligned striated pattern of obscurin formed at a stage when desmin, traditionally considered as a molecular linker responsible for the lateral binding and stabilization of myofibrils at the Z-bands, was still diffusely localized. During the disassembly of the contractile system in adult myocytes, disappearance of the cross-striated pattern of obscurin preceded the disorganization of registered alignment and intense breakdown of myofibrils. The cross-striated pattern of desmin typical of terminally differentiated myocytes disappeared before or simultaneously with obscurin. During redifferentiation, as in neonatal myocytes, sarcomeric incorporation of obscurin closely followed that of α-actinin and occurred earlier than the striated arrangement of desmin intermediate filaments. The presence of obscurin in the Z-lines and its later assembly into the A/M-bands indicate that it may serve to stabilize and align sarcomeric structure when myosin filaments are incorporated. Our data suggest that obscurin, interacting with other muscle proteins and possibly with the sarcoplasmic reticulum, may have a role as a flexible structural integrator of myofibrils during assembly and adaptive remodeling of the contractile apparatus.
O
During differentiation, the architecture of the cytoskeleton and the contractile system in cardiac muscle cells undergoes progressive development and structural maturation, which is accompanied by intense formation of new myofibrils and cytoskeletal elements (Rumyantsev 1991). Terminal differentiation of cardiomyocytes has long been considered an irreversible process. However, over the past two decades it has become apparent that adult cardiac myocytes are plastic, able to remodel their structure profoundly both in vivo and in vitro (for literature see Borisov 1991; Bugaisky 1991; Claycomb 1991; Eppenberger et al. 1994; Swynghedauw 1999). To better understand the potential functions of obscurin in cardiac muscle, it is important to determine its role during myocyte differentiation and remodeling.
Cardiac cell cultures are an interesting and convenient object for studies of myofibrillar dynamics (for review see Ehler and Perriard 2002). This study was undertaken to elucidate the dynamics of spatial and temporal patterns of obscurin expression during myofibrillogenesis, remodeling, and redifferentiation in primary cultures of neonatal and adult rat cardiomyocytes.
Z-lines are important structures of striated muscle cells that provide the stability and functional integration of sarcomeres and play an important role in myofibril assembly. Because the mechanisms of cardiac myocyte differentiation and remodeling involve significant modifications and changes in the areas of Z-lines of myofibrils (for discussion see Borisov 1991; Sanger and Sanger 2002), we investigated the localization of obscurin in relation to α-actinin and desmin, two functionally different proteins topographically associated with Z-lines in terminally differentiated striated muscle.
α-Actinin, a major structural protein of Z-lines that provides a proper orientation and mechanical fixation of actin filaments within the sarcomeres, is indispensable for normal progression of all stages of myofibrillogenesis. This is one of the first proteins detected in structural precursors of myofibrils. Movement of α-actinin-containing bodies (Z-bodies) along the long axis of nascent myofibrils underlies the formation of Z-lines, apparently through their coalescence and lateral alignment with closely apposed neighboring nascent myofibrils (for literature see Borisov 1991; Shimada et al. 2002).
Desmin is a muscle-specific intermediate filament protein that shifts from filamentous to striated pattern of localization at terminal stages of myogenic differentiation, binding myofibrils together at the level of Z-lines and structurally integrating the contractile apparatus during contraction-relaxation cycles (e.g., Saetersdal et al. 1989; Borisov 1991; Opie 1998). Taking into account the recent reports concerning the capacity of obscurin to bind titin in solution (Bang et al. 2001; Young et al. 2001) and the data concerning the direct involvement of titin in the assembly and function of sarcomeres (reviewed by Shimada et al. 2002), we also compared the patterns of obscurin and titin localization within nascent and newly formed sarcomeres. Titin is believed to link myosin and, probably, actin filaments to Z-lines and provide the elasticity to sarcomeric structure (for discussion see Sanger and Sanger 2002; Shimada et al. 2002). With technical advances of confocal microscopy, it has become feasible to analyze the co-localization of individual proteins in differentiating and dedifferentiating cells at high resolution. Our goal was to analyze the localization of obscurin in relation to α-actinin and desmin during the processes of assembly and disassembly of myofibrils using confocal microscopy and high-resolution digital imaging.
Materials and Methods
Primary Cardiac Muscle Cell Cultures
Primary cell cultures of neonatal and adult cardiac myocytes were isolated from the hearts of 2–3-day-old and adult rats by enzymatic dissociation of the myocardial tissue as previously described (Borisov et al. 1985; Westfall 2003) and plated on glass coverslips in 60-mm petri dishes. The cells were cultivated in a CO2 incubator (5% CO2) in F-12 medium continuing 10% of bovine fetal serum. Neonatal and adult cells were collected for immunolabeling every 24 hr beginning on day 2 after plating.
Immunocytochemistry and Confocal Microscopy
Cell fixation, processing, and immunostaining were performed as previously described (Borisov et al. 1989). We previously reported the development and characterization of the monoclonal antibody (MAb)EA- 53 to the sarcomeric isoform of α-actinin (Borisov et al. 1989; Fridlianskaia et al. 1989) which is now available commercially (Sigma Chemical; St Louis, MO). A polyclonal antibody recognizing the carboxy region of obscurin was prepared in a rabbit host using standard methods, and its specificity has been examined (Kontrogianni-Konstantopoulos et al. 2003). An anti-titin MAb (clone 9D10) binding to the PEVK segment of titin in the I-bands near the I-A interfaces of sarcomeres (Trombitás et al. 1998) was obtained from the Developmental Studies Hybridoma Bank, University of Iowa (Iowa City, IA). Monoclonal anti-desmin and all secondary antibodies were obtained from Sigma. Immunostained samples of cultured cardiac myocytes were mounted on slides and examined with a Carl Zeiss LSM 510 Meta confocal microscope using the ×40 and ×63 objectives.
Results
To determine the spatial and temporal dynamics of obscurin localization in progressively differentiating cardiac myocytes, we used indirect immunofluorescent labeling for α-actinin as a structural marker of mature myofibrils and their nascent structural precursors. This enabled us to localize the sites of myofibrillogenesis and to examine the dynamics of the maturation of the contractile apparatus at different time periods in culture. In our experiments, cardiac myocytes isolated from neonatal animals became elongated, developed cytoplasmic processes, and started to spread on the substrate on days 2–3 after plating. During cultivation, the cells actively assembled new contractile structures such that by days 4–5 in culture most of them formed clusters of mature myofibrils, typically located in the central sarcoplasm (Figure 1). Myofibrillogenesis advanced towards the cell periphery, resulting in the progressive expansion of the central cluster of well-differentiated myofibrils. At the periphery, the contractile system typically consisted of bundles of nascent myofibrils at different stages of maturation and their non-striated precursors that resembled stress fibers (Figures 1–3). The cultures were heterogeneous in terms of the development of the contractile apparatus of individual myocytes and the abundance of different structures immunopositive for α-actinin. We found the immunoreactivity of α-actinin in three distinct types of structures: (a) Z-bands of nascent and mature myofibrils with clearly visible, well-developed cross-striations; (b) non-striated bundles with typically dense distribution of α-actinin located in stress fiberlike structures; and (c) rounded bodies organized linearly in a dotted pattern with a periodicity of 0.3–1.5 μm (Figures 2 and 3). The presence of the latter two structures decreased during the course of differentiation as mature myofibrils assembled and accumulated. By days 10–11 in vitro, cell spreading was nearly complete and clusters of well-aligned, longitudinally oriented myofibrils typically occupied the greater part of the cytoplasm. Slower accumulation of new contractile structures continued in well-developed cytoplasmic processes. Simultaneously, the phenotypical polymorphism of the cell population significantly diminished so that nearly all of the myocytes contained well-developed striated contractile structures. Therefore, cardiac myocytes undergo progressive differentiation of their contractile system in this cell culture model.
Comparison of the patterns of sarcomeric α-actinin and obscurin localization in differentiating myocyte cultures revealed significant differences in the cytoplasmic distribution of these two proteins. Double indirect immunofluorescent labeling demonstrated that obscurin was most prominent in the areas of the central myofibrillar clusters. The areas of intense immunoreactivity for obscurin progressively expanded as mature myofibrillar clusters developed. At the same time, large numbers of dispersed nascent myofibrils containing α-actinin, located at the periphery of the cytoplasm, did not express detectable amounts of obscurin (Figures 2A–2F and 3A–3F). To identify the earliest contractile structures that contain obscurin, we examined its localization in the myofibrillar apparatus at different stages of structural maturity. As shown in Figures 2, 3, 4A, and 4B, mature myofibrils are intensely immunopositive for obscurin located in the middle of A-bands (M/H-line regions) and, to a lesser extent, in the Z-lines of assembled sarcomeres. The intensity of immunoreactivity for obscurin in the mid A-band areas of myofibrils progressively increased during differentiation (compare Figures 2A, 2C, and 2E with 2B, 2D, and 2F). Interestingly, the series of nascent myofibrils did not contain significant amounts of obscurin in the A-bands despite the clear presence of a periodic pattern of α-actinin (Figures 3A, 3B, and 3I). Nascent myofibrils and non-striated stress fiberlike structures were weakly immunopositive for obscurin despite an intense immunopositivity for α-actinin (Figures 2, 4A, and 4B). This was especially evident where striations developed from non-striated regions within nascent myofibrillar bundles (Figures 2, 3, 4A, and 4B). Interestingly, several small rounded bodies of α-actinin were typically visible at the points of transition from sarcomeric to non-sarcomeric regions of nascent myofibrils (Figures 2B and 2E). Some structures containing α-actinin were immunonegative for obscurin, especially in the spreading ruffled edges and processes of peripheral sarcoplasm (Figures 2A, 2B, 2D, 2E, and 3). Therefore, the initial assembly of myofibrils, as revealed by the cross-striated pattern of sarcomeric α-actinin, occurs before large amounts of obscurin assemble into sarcomeres. This suggests that the accumulation of obscurin in the middle of sarcomeres occurs soon after or simultaneously with the differentiation of developed Z-lines. Interestingly, the abundant incorporation of obscurin into myofibrils coincided with the process of lateral fusion of newly formed contractile structures into large myofibrillar bundles and clusters (Figures 2 and 3). We found that during the process of lateral alignment of myofibrillar bundles at advanced stages of differentiation, obscurin localized in the M-bands and Z-lines progressively developed a continuous transverse striated pattern that extended across large areas of the sarcoplasm of cardiac myocytes (Figures 2 and 3). Therefore, active incorporation of obscurin into myofibrils and significant intensification of the immunoreactivity for this protein in the middle part of A-bands is associated with the stage of lateral alignment and fusion of myofibrils into larger bundles. This suggests that obscurin may aid the elastic alignment of myofibrils across the cardiomyocyte as an integrating linking protein at advanced stages of differentiation.
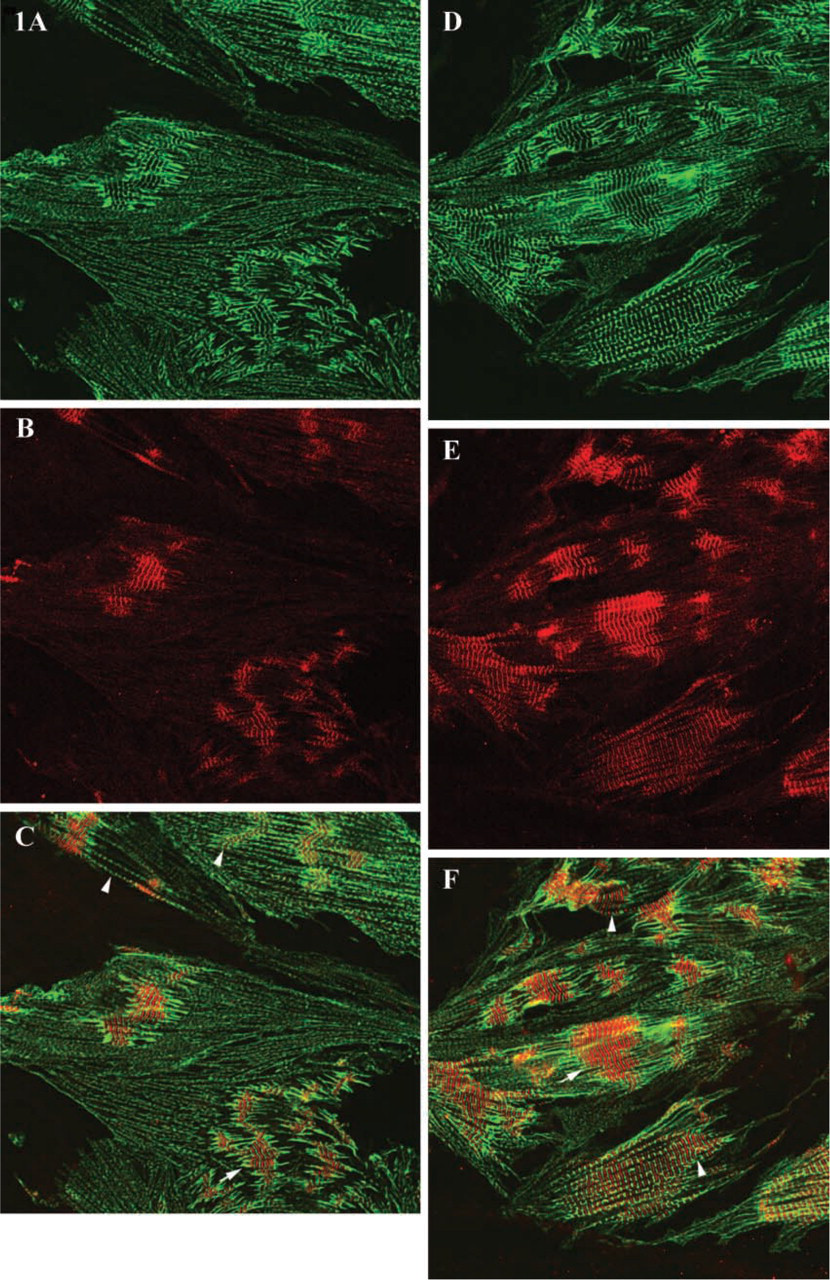
Confocal microscopy of cardiac myocytes immunolabeled for sarcomeric α-actinin and obscurin on day 4 (
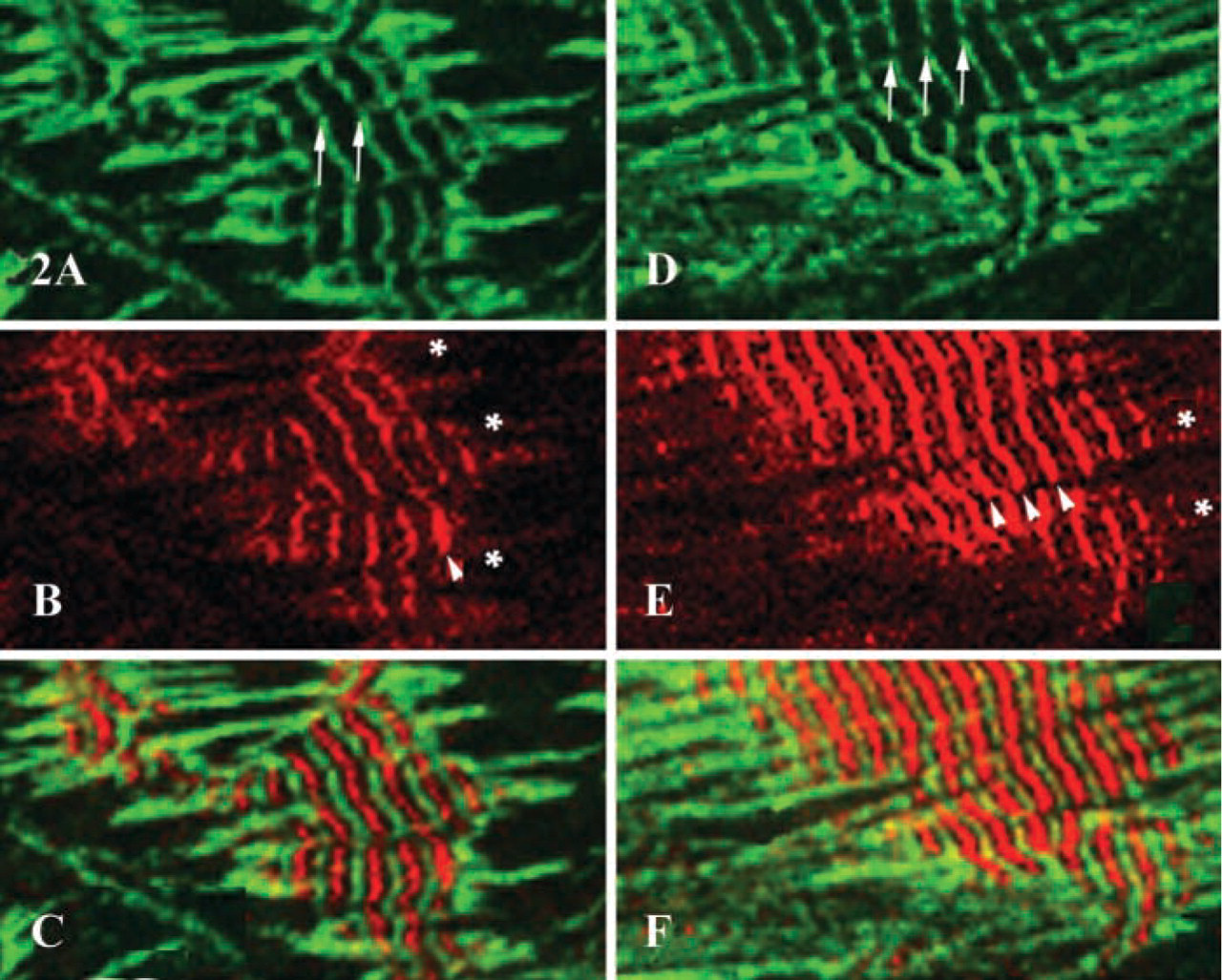
Localization of sarcomeric α-actinin and obscurin in clusters of laterally aligning myofibrils on days 4 (
Given obscurin's reported ability to interact with titin (Bang et al. 2001; Young et al. 2001), we examined the localization of titin in remodeling cardiac myocytes using an antibody that recognizes the PEVK segment of titin. We found that the immunolabeling of titin was concentrated in the I-bands in mature myofibrils and that some positive reactivity was present in nascent myofibrils (Figures 4C and 4D). Therefore, close topographical association of obscurin and titin (Figure 4) may allow their direct functional interaction in nascent and mature sarcomeres.
To further examine the possible role of obscurin in binding individual myofibrils into larger functional units, we examined its co-localization with desmin, an intermediate filament protein known to link myofibrils in register at the periphery of Z-lines in terminally differentiated cardiac muscle. We found that obscurin and desmin did not co-localize in differentiating cardiac myocytes (Figures 5A–5C). Instead, the transverse striated pattern of obscurin was already well developed and the fusion and lateral alignment of myofibrils was in progress while desmin was still in diffuse filaments typical of developing muscle. This demonstrates that obscurin is primarily associated with laterally aligning myofibrils before they associate with desmin and that obscurin-mediated primary alignment of the contractile apparatus precedes the maturation of the intermediate filament system.
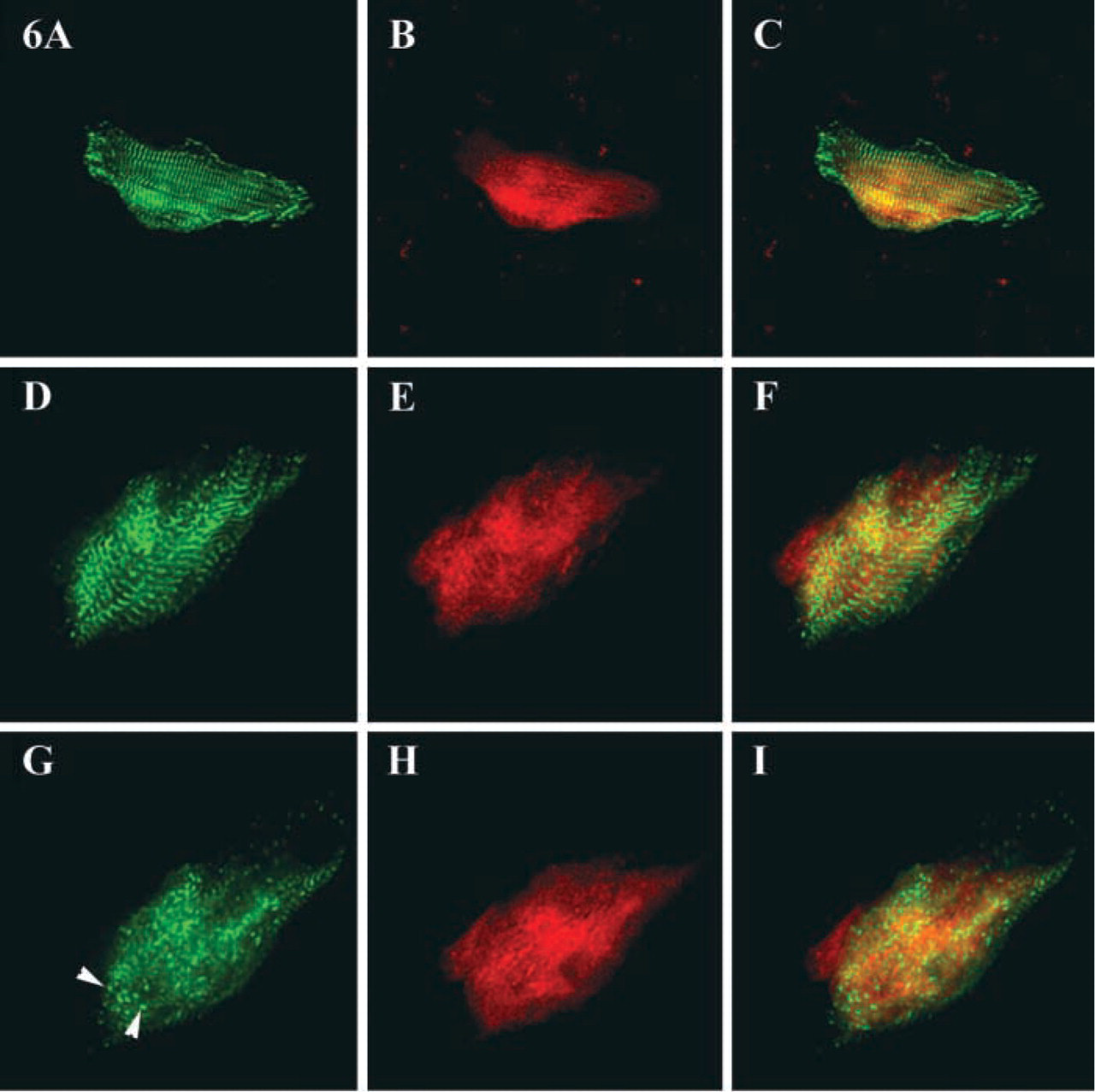
To better understand the possible functional role of obscurin in stabilizing the contractile apparatus, we examined the dynamics of its localization during the disassembly of myofibrils induced by treating terminally differentiated adult cardiac myocytes with fetal bovine serum. We found that, during induced dedifferentiation and remodeling of adult cells, obscurin undergoes profound reorganization. Progressive loss of obscurin from the sarcomeres, as evidenced by the disappearance of the striated pattern of localization, was noticeable within 24–26 hr after the addition of fetal serum and was one of the earliest events in the disassembly process. Nevertheless, many cells with diffuse sarcoplasmic reactivity for obscurin still demonstrated a striated pattern of α-actinin localization (Figures 6A–6I), indicating the persistence of Z-lines, at least transiently, after the removal of obscurin. In significantly dedifferentiated cells, both obscurin and α-actinin were localized as granular particles diffusely distributed in the sarcoplasm (Figures 6G–6I). Some of the granular structures appeared to contain both proteins, whereas others contained only one (Figures 6G–6I). As redifferentiation proceeded, the development of a striated pattern of obscurin localization closely followed the assembly and differentiation of Z-lines and M/H-bands as described above for the neonatal cells.
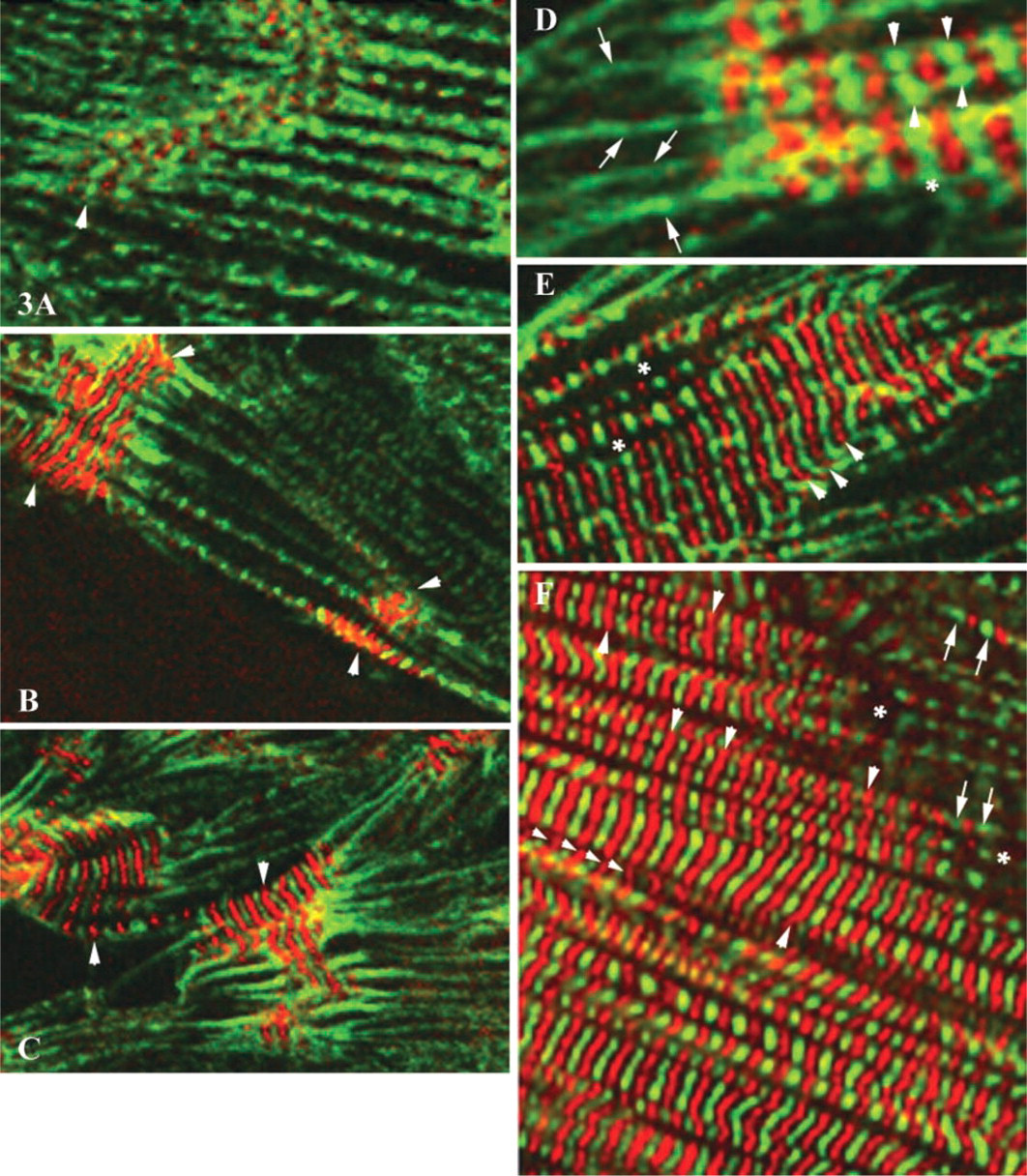
Co-localization of α-actinin (green fluorescence) and obscurin (red fluorescence) at different stages of myofibrillogenesis. (
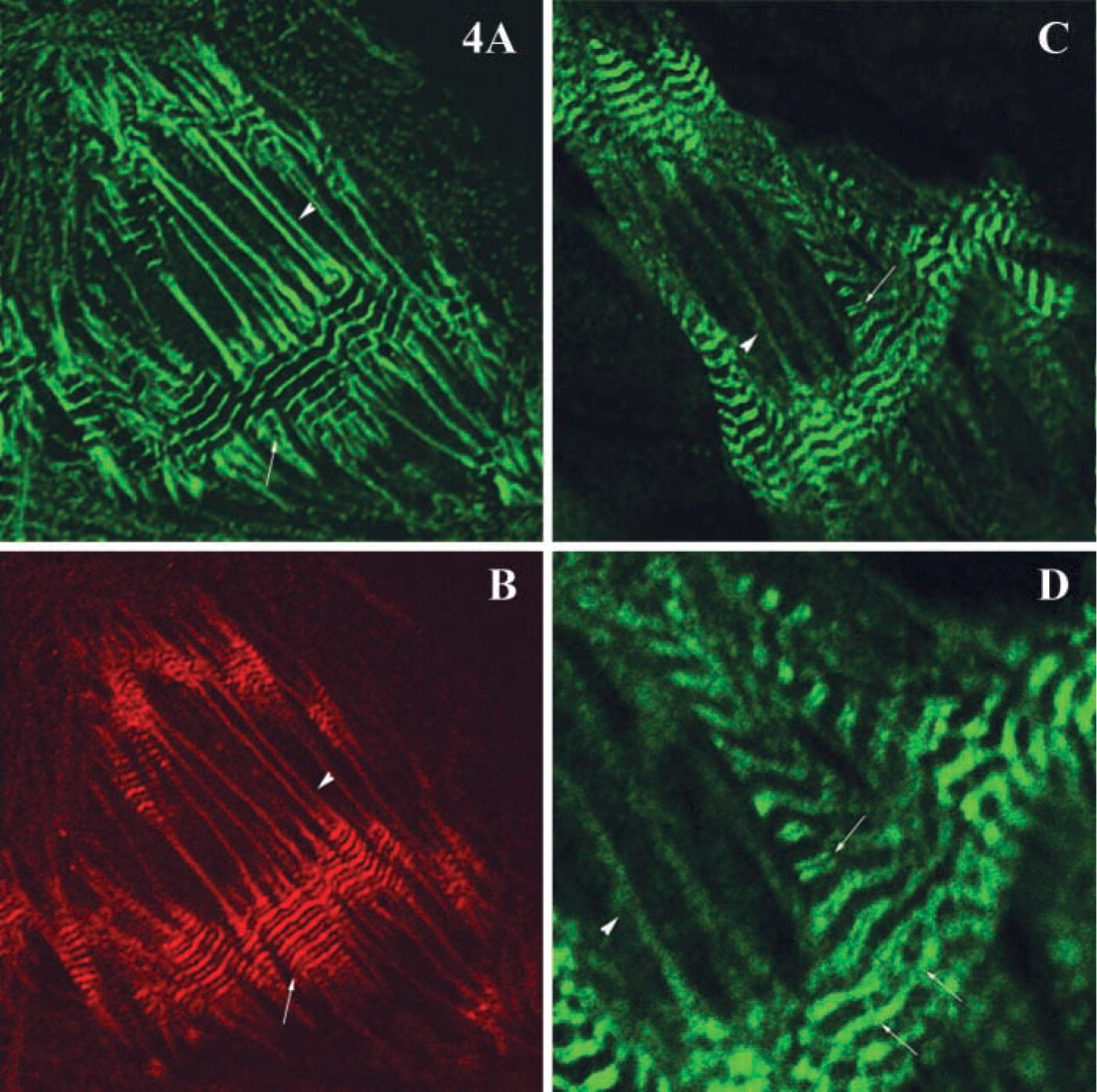
Immunolocalization patterns of α-actinin, obscurin, and the PEVK region of titin in differentiated and nascent myofibrils. α-actinin (
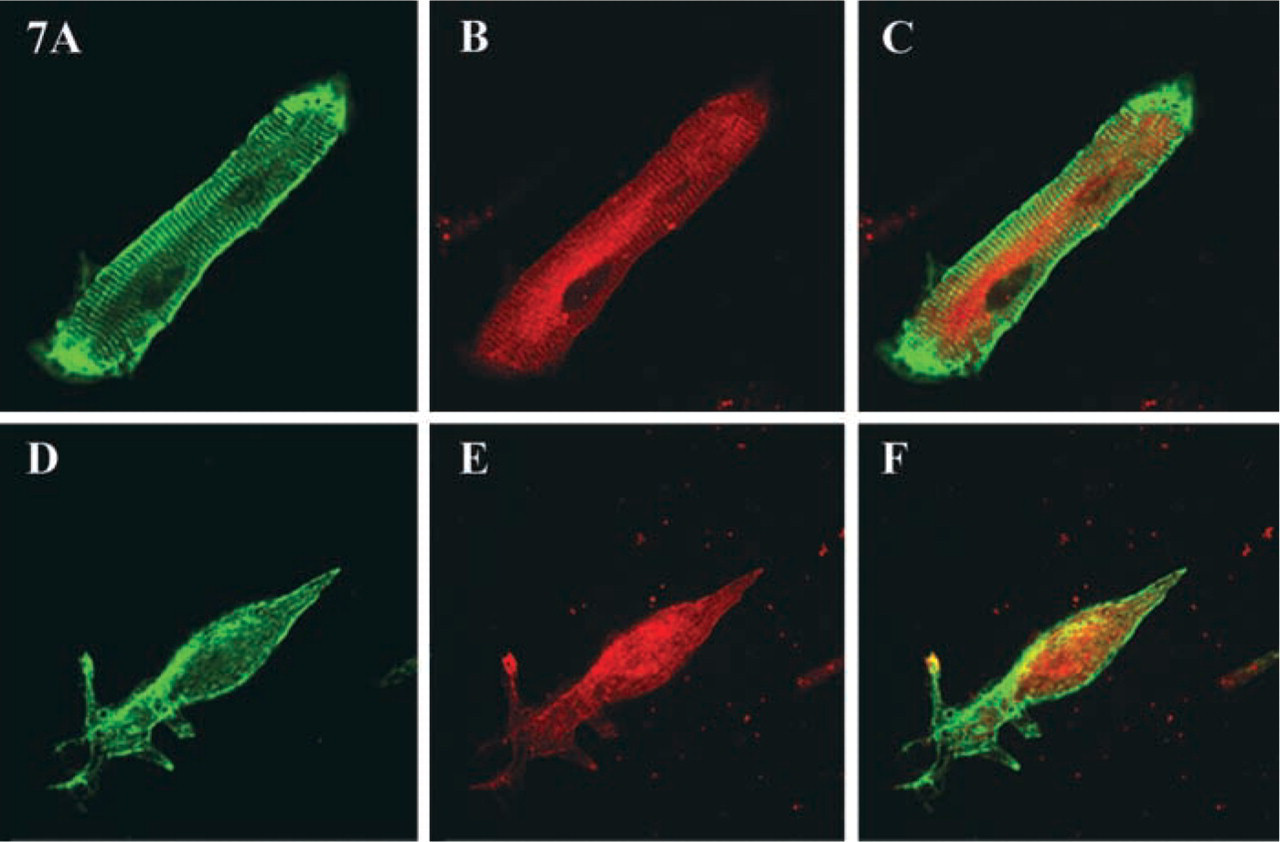
Double immunolabeling for obscurin and desmin showed that, during dedifferentiation, the cross-striated pattern of desmin disappeared simultaneously with or before that of obscurin (Figures 7A–7F). As in neonatal cells, the incorporation of obscurin into myofibrils of redifferentiating adult myocytes occurred before the arrangements of desmin intermediate filaments into a striated pattern of terminally differentiated muscle. Therefore, the kinetics of obscurin re-incorporation during redifferentiation are similar to the patterns of de novo assembly of myofibrillar clusters.
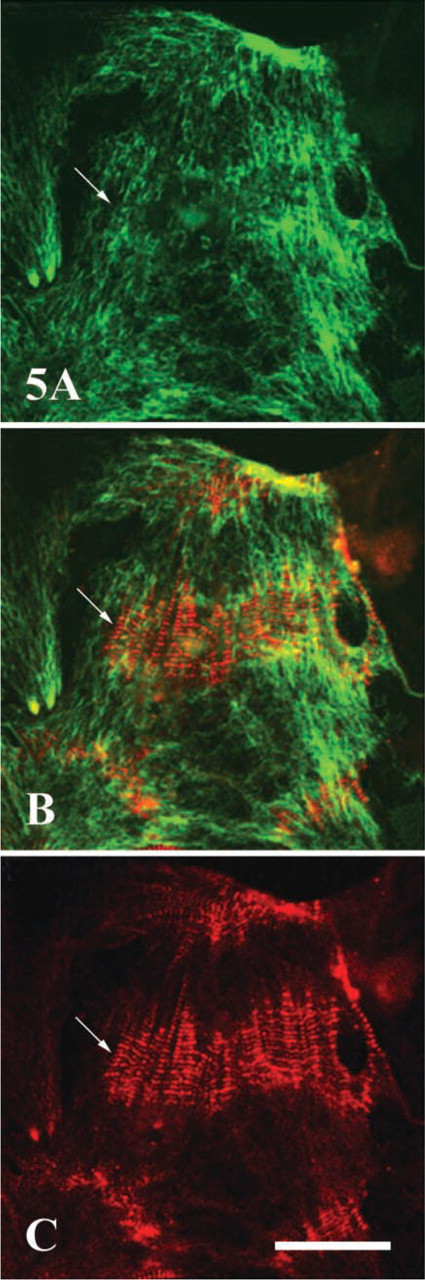
Double immunofluorescent labeling of desmin and obscurin in 6-day-old culture of neonatal rat cardiac myocytes. Note a clearly visible, non-striated filamentous distribution of desmin (
Discussion
Our results show that most of obscurin incorporates into myofibrils primarily at relatively advanced stages of sarcomeric assembly, as the contractile apparatus becomes aligned laterally to form cross-striations typical of differentiating cardiac muscle. Temporally, this coincides with the incorporation of myosin filaments and assembly of the A-bands and M-bands. Electron microscopic and immunocytochemical studies of M-band differentiation have shown that these structures form at advanced stages of myofibrillogenesis (Markwald 1973; Brook et al. 1983; Ehler et al. 1999). This suggests that the concentration of obscurin in A-bands at advanced stages of myofibril assembly might be structurally and functionally associated with the proper positioning of myosin filaments and differentiation of myofibrils into larger structural units, which is indispensable for rhythmic contractile function.
We found that the primary structure of obscurin bears a striking similarity to Unc-89, a protein required for assembly of myofibrils and M-bands in Caenorhabditis elegans (Russell et al. 2002). Given its localization in maturing myofibrils and structural similarity to Unc-89, obscurin may play a similarly vital role in the completion of sarcomere differentiation in mammalian myocytes. Obscurin and its possible embryonic isoforms may perform additional functions at different stages of myofibrillogenesis. Young and co-authors (2001) described the presence of obscurin, which appears to be a fetal isotype, in Z-lines of myofibrils in chick and mouse embryos (<10-somites stage in chick, E9.5 in mouse) and observed noticeable staining of the M-band at later stages. Bang et al. (2001) found the presence of this protein both in the Z-lines and the areas of M-bands of mature sarcomeres, which agrees with our data. We recently reported elevated expression of obscurin and its intense localization to the new sites of myofibrillogenesis after pharmacological induction of hypertrophy in neonatal rat cardiomyocytes in vitro (Borisov et al. 2003).
Obscurin binds a small form of ankyrin and co-localizes with it in skeletal muscle cells, suggesting that it may serve as a molecular link between the sarcoplasmic reticulum and the myofibrils (Bagnato et al. 2003; Kontrogianni-Konstantopoulos et al. 2003). If obscurin plays a similar role in the heart, as seems likely, it may help to align the sarcoplasmic reticulum and, probably, T-system with M- and Z-lines in differentiating cardiomyocytes. The presence of a calmodulin-binding domain in obscurin would be consistent with its involvement in control of calcium homeostasis and calcium-regulated signaling vital for rhythmic contractile function.
Localization of α-actinin and obscurin during remodeling of adult cardiac myocytes. α-actinin (
Localization of desmin and obscurin during the remodeling of adult cardiac myocytes. Note the striated pattern of desmin with enhanced immunoreactivity in the areas of the intercalated disks (A). Note also the preserved striated distribution of obscurin with diffuse labeling of the central sarcoplasm (B) in a cardiomyocyte on day 3 in culture. By day 6, desmin is found in filaments (D) and the reaction for obscurin is diffuse (E). C and F show little colocalization of desmin (green fluorescence) and obscurin (red fluorescence) on days 3 and 6, respectively. Magnifications: A–C × 470; D,E × 240.
Based on our data and previous studies, we propose that obscurin functions as an integrator of myofibrillar structure. Its similarity to Unc-89 supports this model. Like Unc-89, obscurin is a large, muscle-specific protein containing numerous tandem immunoglobulin domains and tandem SH3 and Rho guanine nucleotide exchange factor domains. Mutation of Unc-89 caused marked disorganization of muscle in C. elegans. In particular, the thick (myosin) filaments were not organized into A-bands and there were no M-bands (Waterston et al. 1980). It was suggested that Unc-89, through interactions with other muscle proteins and the sarcolemma, enabled the organization and stabilization of the contractile apparatus (Benian et al. 1996). As the only known vertebrate orthologue of Unc-89, obscurin may perform similar functions during myogenic differentiation in mammals (Sutter et al. in press). Obscurin may also play a regulatory, signaling role during the assembly, disassembly and remodeling of myofibrils. The involvement of the Rho-GTPase in myofibril formation and organization (Hoshijima et al. 1998) supports the possibility that obscurin's Rho-GEF domain is involved in regulating myofibrillar structure.
Of special interest are the changes in obscurin localization after induction of disassembly of myofibrils in terminally differentiated heart cells with fetal bovine serum. Early decrease of immunoreactivity for obscurin after induction of myofibril disassembly demonstrates that this protein leaves sarcomeres earlier than the contractile system disintegrates during reversible dedifferentiation. Its early dissociation from myofibrils supports the view that at least some domains of this protein may be located on the external surfaces of the M- and Z-lines as demonstrated recently in skeletal muscle (Kontrogianni-Konstantopoulos et al. 2003).
An important role of obscurin may be linked to its structural and functional interactions with another giant muscle protein, titin, that extends through several sarcomeric regions. One of the major functions of titin in mature sarcomeres is to link the thick (myosin) filaments to the Z-lines (Sanger and Sanger 2002; Shimada et al. 2002). Titin is believed to function as an elastic spring-like element during muscle contraction and relaxation, and is postulated to define the spatial positions of other contractile proteins during myofibril assembly (Freiburg et al. 2000; Hein et al. 2002). In developing muscle, titin plays a role as a molecular blueprint for the structural layout of sarcomeric elements (Gregorio et al. 1999; Trinick and Tskhovrebova 1999; Tskhovrebova and Trinick 2003). Early in myofibrillogenesis, titin co-localizes with α-actinin at Z-lines (Tokuyasu and Maher 1987; Komiyama et al. 1993), and may integrate myosin filaments with I–Z–I complexes of nascent myofibrils (Barral and Epstein 1999; Shimada et al. 2002). The comparison of the localization of obscurin and titin in nascent and mature myofibrils suggests that these two proteins may function together to coordinate sarcomeric assembly.
A similar linkage is unlikely to exist between obscurin and desmin, one of the earliest muscle-specific proteins expressed during development of skeletal muscle and the heart. Earlier, we did not find co-localization of desmin and obscurin after induction of cardiac cell hypertrophy (Borisov et al. 2003). In terminally differentiated cardiac myocytes, the major muscle-specific intermediate filament protein, desmin, binds bundles of myofibrils at the level of their Z-lines, thus helping to align them into striations (for discussion and illustrations see Saetersdal et al. 1989; Borisov 1991; Opie 1998). Desmin is also believed to play a role in stabilizing the myofibrillar apparatus during muscle contraction (Capetanaki and Milner 1998). Because obscurin assembles into striations and associates with aligning myofibrils before desmin, it may play a more important role than desmin at early stages of myofibrillar integration in the developing muscle. This may provide a partial explanation for the surprising fact that the desmin null (–/–) mice generated through homologous recombination by Capetanaki and co-workers (1997) are viable and fertile. Because cardiac muscle starts intense contractile activity early in embryonic development, the presence of an elastic support system reversibly uniting myofibrils during contractions provides an important functional advantage that permits easy spatial remodeling during heart growth under conditions of increasing hemodynamic load. Indeed, its size (≃720 kD), distribution, and multidomain structure suggest that obscurin may interact with several sarcomeric elements. Together with titin, it may serve to integrate and stabilize the contractile apparatus at the Z-lines and the A- and M-bands. Therefore, giant molecules of obscurin and titin may work as elastic staples or springs, integrating and stabilizing different structural elements of the sarcomeres, the sarcoplasmic reticulum and, possibly, the T-system during the cycles of contraction and relaxation in cardiac muscle.
Footnotes
Acknowledgements
Supported by NIH 5RO3 HD39693–02 to MWR and NIH 1R01 HL64304 to RJB.
We wish to thank Chris Edwards and Bruce Donohoe, the members of Micropscopy and Image Analysis Laboratory, for support of this work. We also thank Pavel Borisov for help with preparation of the manuscript.
