Abstract
The sex steroids, estrogens, progesterone, and androgens, all play a role in mammary development and function. To precisely identify the sites of action of these steroids, we studied the localization of the estrogen receptor α (ERα) and ERβ, the progesterone receptor A (PRA) and PRB, and androgen receptors (AR) in the normal human mammary gland. Immunocytochemical localization of ERα, ERβ, PRA, PRB, and AR was performed with reduction mammoplasty specimens from premenopausal women. ERα, PRA, PRB, and AR were localized mostly to the inner layer of epithelial cells lining acini and intralobular ducts, as well as to myoepithelial cells scattered in the external layer of interlobular ducts. AR was also found in some stromal cells. ERβ staining was more widespread, resulting in epithelial and myoepithelial cells being labeled in acini and ducts as well as stromal cells. These results suggest that all sex steroids can directly act on epithelial cells to modulate development and function of the human mammary gland. Estrogens and androgens can also indirectly influence epithelial cell activity by an action on stromal cells.
T
In the mouse mammary gland, ERα expression was detected in luminal epithelial cells and stromal compartments, but no staining was seen in the myoepithelium (Shyamala et al. 2002). The cells that stained positively for ERβ were in the luminal cell and basal cell population. Fat cells were also weakly stained for ERβ. PRA and PRB expression are temporally and spatially separated during murine mammary gland development from puberty through pregnancy, lactation, and involution (Zeps et al. 1999). PRA is expressed predominantly in the adult virgin mouse mammary gland, while PRB is expressed predominantly during pregnancy, mainly in alveolar epithelial cells (Aupperlee et al. 2005b).
In the rat mammary gland, ERα-immunoreactive cells were detected only in the epithelium lining terminal end buds, alveolar buds, ducts, or lobules (Russo et al. 1999), while ERβ staining was found mainly in the nuclei of all the epithelial cells and of some stromal cells (Saji et al. 2000). By using immunohistochemical methods, Aupperlee et al. (2005a) detected PRA and PRB expression in rat mammary gland. PRA expression was restricted to luminal epithelial cells, while PRB was expressed in both luminal and myoepithelial cells (Kariagina et al. 2007).
In human breast, it has been reported that ERα is expressed in a minority of luminal epithelial cells and not at all in any of the other cell types (Petersen et al. 1987; Clarke et al. 1997; Anderson and Clarke 2004). ERβ has been localized in the nuclei of luminal epithelial, myoepithelial, endothelial, and stromal cells (Speirs et al. 2000, 2002; Shaw et al. 2002). We have reported that in the human mammary gland, ERα and ERβ immunoreactivity was present in the nuclei and to a lesser degree in the cytoplasm of epithelial cells of acini and interlobular ducts (Pelletier and El-Alfy 2000), the cytoplasmic staining for ERβ being more prominent than that observed for ERα. It has been reported that PRA and PRB were both coexpressed at similar levels in normal human mammary gland throughout the menstrual cycle by dual immunofluorescence (Mote et al. 2002). All PR staining of epithelial cells was nuclear, and no cytoplasmic staining was detected. Ruizeveld de Winter et al. (1991) have reported that AR immunoreactivity is localized in nuclei of epithelial cells in acini and ducts of human mammary gland. Myoepithelial cells were not immunostained. On the other hand, Kimura et al. (1993) found staining for AR not only in epithelial cells but also in myoepithelial cells in acini and ducts.
So far, there has been no report on the cellular and subcellular expression of the different sex steroid receptors in the same human mammary gland specimens. The aim of our study was to evaluate the pattern of expression and distribution of sex steroid receptors ERα, ERβ, PRA, PRB, and AR in mammary glands of pre-menopausal women.
Materials and Methods
Mammary Gland Tissue Preparation
This study was approved by the institutional review board at Laval University Medical Center. All patients signed informed consent forms before participation in this research project. Samples of mammary gland tissue from 17 patients were obtained at surgery for reduction mammoplasty. All the patients were premenopausal (14–43 years of age) and did not receive any hormonal treatments for at least 6 months prior to surgery. The specimens used in the present immunocytochemical studies had a volume of ×2.5 cm3. They were fixed in 4% paraformaldehyde in 0.2 M phosphate buffer (pH 7.4) within 15 min after dissection. The average fixation time was 12 hr. The tissues were then dehydrated through increasing concentrations of ethanol, cleared in toluene, and embedded in paraffin. In each case, at least three separate tissue specimens were studied, and the results were consistent.
Immunocytochemistry
Paraffin sections (5-μm thick) were deparaffinized, hydrated, and treated with 3% H2O2 in methanol (pH 7.6) for 15 min. Sections were then heated in a microwave oven for antigen retrieval, using citrate buffer (pH 5.5) as previously described (Tacha and Chen 1994). The sections were then incubated overnight at 4C with ERα, ERβ, PRA, PRB, and AR antibodies at the dilutions indicated in Table 1. After samples were incubated with the appropriate biotinylated anti-immunoglobulin serum (anti-mouse for ERβ, PRA, and PRB; anti-rabbit for ERα and AR) for 2 hr at room temperature, they were incubated with peroxidase-labeled streptavidin (Signet Laboratories, Inc.; Dedham, MA) for 2 hr at room temperature. Control sections were incubated with antibodies preabsorbed with an excess of corresponding antigens (10−6 M). Sections were lightly counterstained with hematoxylin and dehydrated through an ethanol series, followed by exposure to toluene and mounting.
Scoring of Immunoreactivity
Data were generated from independent observations by three of the present authors (SL, BH, and GP). Divergences were resolved by joint examination of the slides. Since the cells located in the external layer of acini and ducts are likely myoepithelial cells, we analyzed only positive cells belonging to the inner layer of acini and ducts. We counted the number of immunostained cells from 300 to 400 cells in three randomly chosen fields of each tissue regardless of the intensity of reactivity. We then calculated the mean percentage of labeled cells (labeling index).
Primary antibodies
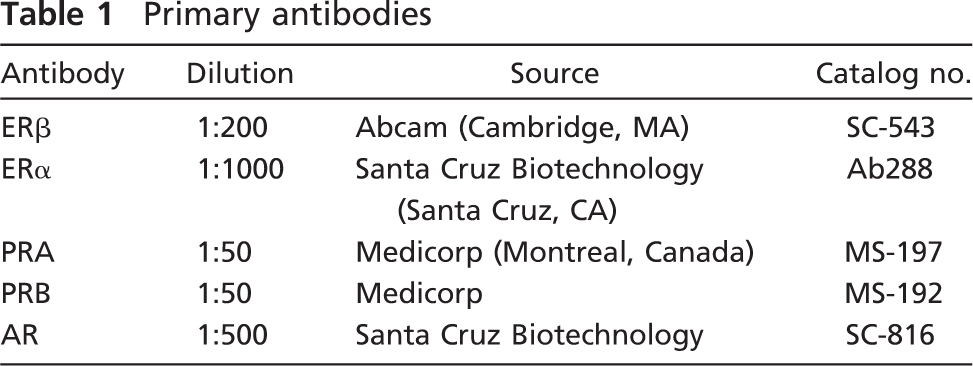
Immunolocalization of ERα (
Results
Localization of ERα and ERβ
ERα immunoreactivity was detected in the nuclei of ×10% (range, 5–28%) of epithelial cells in acini and interlobular ducts. Positive cells were located mostly in the inner layer of epithelial cells in acini. They were distributed as scattered cells or formed a continuous layer in contact with the acinar lumen (Figure 1A). In the interlobular ducts, a few positive nuclei were seen in the external layer of epithelial cells (Figure 1B). No cytoplasmic staining could be detected. In 4 of 17 cases, we found a few stained stromal cells. The immunostaining for ERβ was detected in the nuclei of ×80% (range, 70–85%) of luminal epithelial cells in acini and interlobular ducts (Figures 1C and 1D). Positive cells were also found in the outer layer of acini and ducts. Those cells were considered myoepithelial cells. We also detected weak to moderate cytoplasmic staining in all positive epithelial cells in acini and ducts. Nuclear staining was also seen in stromal cells, endothelial cells, and lymphocytes.
Localization of AR
About 20% (range, 5–31%) of epithelial cells were immunolabeled, the labeling being restricted to nuclei. Cells were found to be dispersed or formed the internal layer in both acini and intralobular ducts (Figure 1E). In the interlobular ducts, labeled cells were mostly myoepithelial cells (Figure 1F). Some stromal cells also exhibited nuclear staining.
Localization of PRA and PRB
Similar patterns of expression were observed for PRA and PRB, with ×7% (range, 1–27%) of epithelial cells in acini and ducts being labeled. With both antibodies, strong nuclear staining was detected (Figures 2A–2D). Weak to modest staining for PRA was also observed in the cytoplasm of reactive cells (Figures 2A and 2B). In the acini, positive cells were found in the inner layer of epithelial cells (Figures 2A and 2C). In the interlobular ducts, cells staining for either PRA or PRB were located in the outer layer and were then considered myoepithelial cells (Figures 2B and 2D). No stromal cells appeared to be stained, while a few positive lymphocytes were found in 3 of 17 cases.
In all cases, immunolabeling was completely abolished by immunoabsorption of the antibody with the corresponding antigen (data not shown).
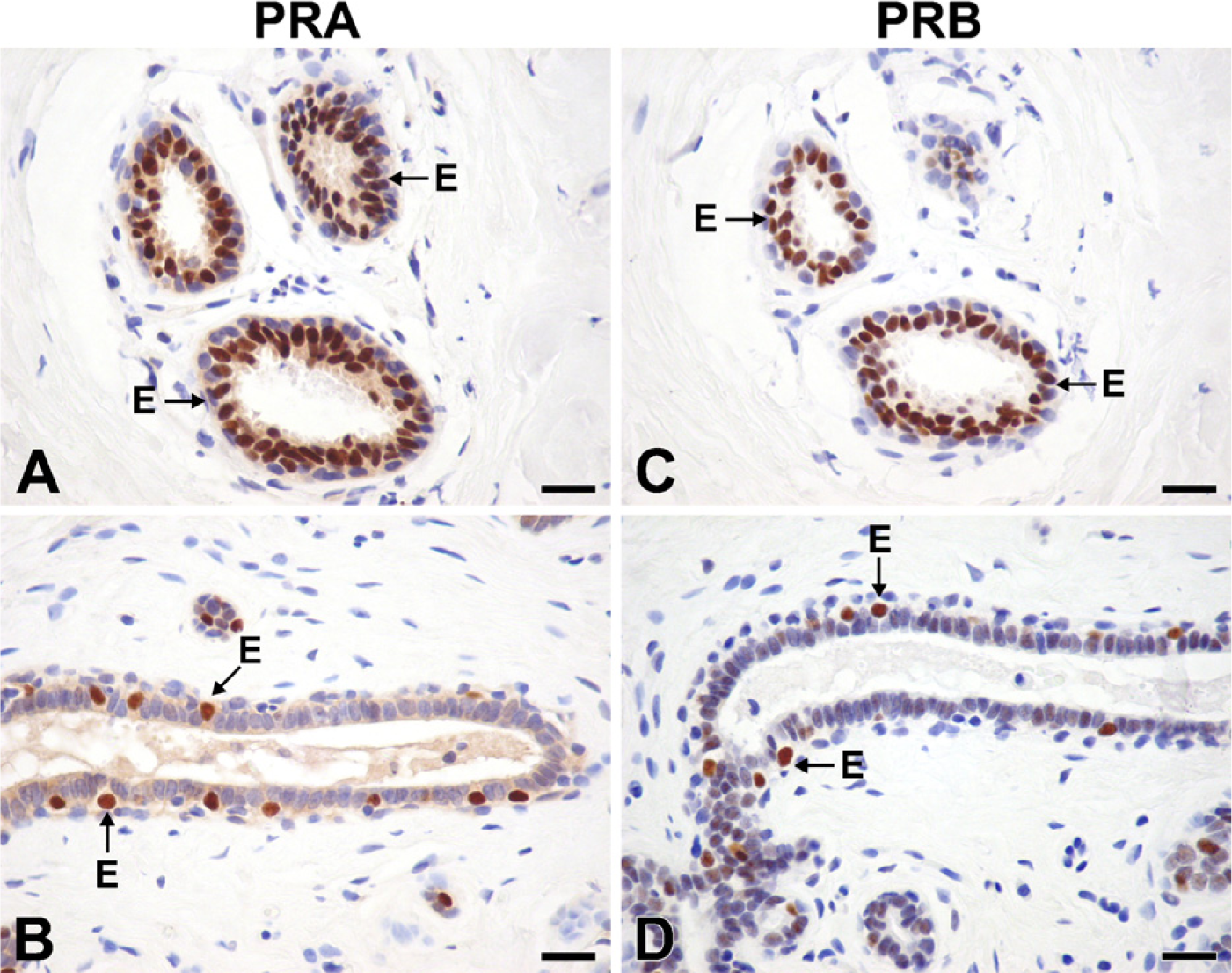
Immunolocalization of PRA (
Discussion
The human adult female breast consists of a branching, tree-like network of ducts and acini lined by a continuous layer of epithelial cells surrounded by a layer of myoepithelial cells (Russo and Russo 1987). In the present study, we demonstrate the cellular localization of the sex steroid receptors in the adult human mammary gland, using specific antibodies to ERα, ERβ, PRA, PRB, and AR, respectively. ERα immunoreactivity was found mostly in epithelial cells of acini and ducts, ×10% of epithelial cells being labeled. It is interesting to note that positive cells were located in the inner layer of epithelial cells in the acini and intra-lobular ducts, while in the interlobular ducts, positive cells were located in the outer layer. We could occasionally find labeled stromal cells. This finding is in contrast with previous results showing that ERα was detectable only in the nuclei of luminal epithelial cells of ducts and lobules and not at all in any of the other cell types in the mammary gland (Petersen et al. 1987; Clarke et al. 1997). Our observations are in agreement with those from a previous report by Shoker et al. (1999) indicating that ERα expression is low (6% to 8%) in mammary gland epithelial cells in premenopausal women. It has also been previously demonstrated that the human mammary gland contains a small but distinct population of ERα-positive cells, consisting of ×7% of the total epithelial cell population (Petersen et al. 1987). Moreover, 87% of the ERα-positive cells were luminal epithelial cells or occupied an intermediate position in the duct wall. Studies of rhesus monkey mammary gland have shown that ERα expression in epithelial cells was decreased and that ERβ expression increased during the menstrual cycle (Cheng et al. 2005). On the other hand, it has been shown that in the human mammary gland, ERα expression was at the lowest level in the luteal phase without any variations in ERβ expression during the menstrual cycle (Shaw et al. 2002). In the present study, all reduction mammoplasty samples were from premenopausal patients whose menstrual status was unknown, so that we did not attempt to correlate ER receptor expression with the hormonal status of the patients.
In comparison with the expression of ERα, the expression of ERβ appears much more widespread, ×70–85% of epithelial cells being positive. Our results with ERβ staining are in accordance with previous results showing that ERβ is detectable in the nuclei of luminal epithelial, myoepithelial, and endothelial cells and in fibroblasts (Shaw et al. 2002; Speirs et al. 2002). In the rat mammary gland, ×60–70% of epithelial cells expressed ERβ at all stages of breast development (Saji et al. 2000). The widespread distribution of ERβ suggests multiple roles for ERβ in the mammary gland. Information for the respective role of ERα and ERβ has been obtained from gene knockout mice. Mammary glands from the ERα knockout (αERKO) mouse were undeveloped, possessing only a rudimentary ductal structure that emanated from the nipple (Korach et al. 1996; Bocchinfuso and Korach 1997; Bocchinfuso et al. 2000). In contrast, ERβ knockout (βERKO) mice appeared to undergo normal mammary development (Krege et al. 1998). These results demonstrate that the ERα receptor alone is both necessary and sufficient to mediate the morphogenic effects of estrogen on the mammary epithelium. However, it seems likely that ERβ plays a dispensable role in the normal mammary gland. In mammary gland hyperplasia, the ratio of ERα to ERβ in cases that progressed to carcinoma was significantly higher than in cases that did not progress, suggesting that ERβ could modulate ERα transcriptional activity (Shaaban et al. 2005).
In the present study, the staining of both PRA and PRB was detected in the nuclei of ×7% epithelial cells of lobules and ducts. Moreover, PRA immunoreactivity was also observed in the cytoplasm of epithelial cells and occasionally in some lymphocytes. Previous reports showed that PRA and PRB were expressed at similar levels in normal human mammary gland without any variations throughout the menstrual cycle (Mote et al. 2002; Branchini et al. 2009). It has also been reported that PRA and PRB were expressed in very similar patterns during the menstrual cycle in the rhesus monkey mammary gland (Cheng et al. 2005). The expression of both PR types was low in the early follicular stage and increased during late follicular and luteal phases (Cheng et al. 2005). In the virgin mouse mammary gland, when ductal development is active, the only PR protein isoform expressed was PRA. PRB was abundantly expressed during alveologenesis observed during pregnancy. PRA and PRB colocalization occurred only in a small percentage of cells (Aupperlee et al. 2005b). It has been demonstrated that unequal and nonoverlapping expression of PRA and PRB may be one mechanism contributing to the different activities of PR protein in the mouse (Mote et al. 2006). With the use of PR-null mutant mice (Lydon et al. 1995), PRAKO mice (Mulac-Jericevic et al. 2000), PRBKO mice (Mulac-Jericevic et al. 2003), PRA transgenic mice (Shyamala et al. 1998), and PRB transgenic mice (Shyamala et al. 2000), it has been shown that PRB may mainly mediate ductal branching and lobuloalveolar growth, while PRA may mediate ductal growth in the mouse. According to in vitro studies of human breast cancer cell lines, the functional outcome of progesterone signaling is determined by the ratio between PRA and PRB expression (Graham et al. 2005). It has also been shown that the PRA:PRB ratio is very high in breast cancer (Graham et al. 1995; Mote et al. 2002). Recently it has been reported that PRA expression in human breast was decreased during pregnancy, while PRB expression was not significantly modified (Taylor et al. 2009). All these results suggest that PRA and PRB play different roles in the mammary gland.
In the present study, AR immunoreactivity was detected in the nuclei of inner epithelial cells of lobules and myoepithelial cells of interlobular ducts. Some stromal cells also exhibited nuclear staining. Our results agree well with previous findings indicating that AR mRNA is detected in mammary epithelium as well as in scattered stromal cells (Zhou et al. 2000). Ruizeveld de Winter et al. (1991) have reported that inner ductal epithelial lining cells showed a moderate staining reaction. In the present study, we observed that ×20% of epithelial cells were immunopositive. In the rhesus monkey mammary gland, AR was expressed in 59–75% of epithelial cells throughout the menstrual cycle (Cheng et al. 2005). In female mice lacking AR, the development of mammary glands is retarded, with reduced ductal branching in prepubertal stages and decreased lobuloalveolar development (Yeh et al. 2003), suggesting involvement of AR in mammary gland maturation. On the other hand, in intact female monkeys, the AR antagonist flutamide induced a 2-fold increase in mammary gland epithelial cell proliferation (Dimitrakakis et al. 2003). These data, although contradictory, strongly suggest that AR might play a role in development and possibly functions of mammary gland. More studies are required to clarify the exact role of androgens in mammary gland function.
In summary, the present findings clearly demonstrate a cell-specific localization of ERα, ERβ, PRA, PRB, and AR in the normal human mammary gland, contributing to the establishment of sites of action of estrogens, progesterone, and androgens. ERα, PRA, PRB, and AR are expressed mainly in the inner layer of epithelial cells in the lobules and in the outer layer of epithelium in the interlobular ducts. ERα and AR are also expressed in stromal cells. The expression of ERβ is more widespread, being detected not only in most of epithelial cells throughout mammary gland but also in stromal cells.
