Abstract
Angiopoietin-1 (Angpt1; previously Ang-1) participates in vascular maintenance and remodeling. In the current study, we investigated the distribution of Angpt1 protein in rat brain. We detected Angpt1 immunoreactivity (IR) in cerebral blood vessels, cuboidal ependyma, and tanycytes, which are specialized hypothalamic bipolar ependymal cells. We also evaluated patterns of IR of endothelium-specific receptor tyrosine kinase 2 (Tie2, the receptor for Angpt1). Tie2 IR was present in Angpt1-immunoreactive cuboidal ependyma in a membranous pattern, suggesting an autocrine or paracrine role for Angpt1- Tie2. Tie2 IR was also associated with peri-ependymal blood vessels, some of which were contacted by tips of Angpt1-immunoreactive tanycyte processes, implying a potential functional ligand2receptor interaction mediating communication between the cerebrospinal fluid and vascular compartments. Because we previously found that cerebral Angpt1 expression was modulated by 17β-estradiol (E2), and because some tanycyte functions are modulated by E2, we tested the hypothesis that E2 affects ependymal and tanycyte Angpt1 expression in vivo. No gross E2 effect on the ependymal pattern of Angpt1 IR or cerebral Angpt1 protein content was observed.
T
Tanycytes are specialized bipolar ependymal cells located in the mediobasal hypothalamus and are implicated in neuroendocrine modulation (reviewed in Bruni 1998; Wittkowski 1998; and Rodriguez et al. 2005). Tanycytes are thought to be modified radial glia and are classified as α1, α2, β1, and β2 based on exact location, connectivity, morphology, as well as ultrastructural and molecular features (reviewed in Rodriguez et al. 2005). Most tanycytes produce glial fibrillary acidic protein (GFAP), which makes GFAP immunoreactivity (IR) a useful general tanycyte marker. In addition to GFAP, more than 35 different molecules, from structural proteins to growth factors and growth factor receptors, have been identified in tanycytes (Rodriguez et al. 2005).
The unique location and structure of tanycytes, i.e., the apical tip of the bipolar cell in contact with the cerebrospinal fluid (CSF) and the tip of the long, radially oriented basal process in contact with hypothalamic blood vessels, neurons, or glia, suggests that tanycytes are the anatomical substrate of communication between the CSF compartment and the hypothalamic-pituitary axis. The observation that tanycytes endocytose substances from the CSF and transport them down their basal processes supports this hypothesis (Peruzzo et al. 2004).
Tanycyte functions may be under hormonal control (Fernandez-Galaz et al. 1996; Rodriguez et al. 2005; Garcia-Segura et al. 2008). Subpopulations of tanycytes express estrogen receptors, and it has been hypothesized that E2 modulates hypothalamic neuronal activity during the estrous cycle by altering tanycyte function (reviewed in Rodriguez et al. 2005). An observation that lends credence to this hypothesis is the dynamic interaction of tanycytes and gonadotrophin-releasing hormone (GnRH)-secreting neurons. GnRH is released into portal blood, in a pulsatile manner, from nerve endings that contact portal capillaries but are separated from the perivascular space by a barrier composed of the tips of tanycyte basal processes (reviewed in Rodriguez et al. 2005). The structure of the tanycyte barrier was shown to vary with experimental manipulation of gonadal hormones, leading to the proposal that estrous cycle- mediated changes in the tanycyte barrier alter access of GnRH-containing nerves to capillaries, thus modulating GnRH release (King and Rubin 1994,1995).
In the current study, we investigated Angpt1 protein localization in brains of adult rats and observed Angpt1 IR in third-ventricular and aqueductal cuboidal ependyma and tanycytes. We then evaluated Tie2 IR in relation to Angpt1. Finally, because we have previously found that E2 augments cerebral Angpt1 mRNA (Ardelt et al. 2005), we hypothesized that E2 modulates Angpt1 in tanycytes. We investigated cerebral Angpt1 protein expression in normal adult male and female rats, as well as ovariectomized adult female rats treated with exogenous E2 to achieve different serum E2 levels (Brown-Grant et al. 1970).
Materials and Methods
All procedures performed on rats were approved by the University of Alabama Institutional Animal Care and Use Committee and were in accordance with National Institutes of Health guidelines for animal research.
Animal Care
Rats were maintained in cages of three on a normal light-dark cycle (6 AM-6 PM) and were provided ad libitum with water and diet (Teklad 2018SX; Harlan Breeders, Indianapolis, IN). Female and male Wistar rats, 200–225 g, were obtained from Harlan.
Ovariectomy and Pellet Implantation
Rats were anesthetized with isoflurane (5% for induction; 2% for maintenance) in an oxygen–-room air carrier (O2 at 0.05 l/min; room air at 1.0 l/min). Rats were prepped and draped in a sterile manner; the ovaries were removed through bilateral abdominal incisions; and placebo or E2-releasing pellets (25 μg or 100 μg, 21-day release; Innovative Research of America, Sarasota, FL) were implanted in a subcutaneous pocket in the neck.
Tissue Preparation
Animals were euthanized 10 to 14 days after ovariectomy and pellet implantation. Blood for E2 radioimmunoassay (RIA) was collected via cardiac puncture, and animals were perfused with 80 ml of phosphate-buffered saline (PBS) followed by 100 ml of 4% paraformaldehyde (PF). Brains were removed and postfixed overnight in 4% PF at 4C, followed by embedding in paraffin. Eight-μm-thick sections were cut on a microtome and collected on glass slides.
E2 RIA
E2 RIA was performed on serum obtained at euthanasia using a double-antibody kit (Diagnostic Systems Laboratories; Webster, TX).
Immunolabeling
Three Angpt1 antibodies were used for immunolabeling in tissue sections (Table 1). The Angpt1/4 (C-19) antibody is directed against the carboxyl terminus and recognizes both Angpt1 and Angpt4 (which is not found in rodents). The Angpt1/4 (C-19) antibody was utilized in the majority of immunolabeling experiments, and the two additional Angpt1 antibodies (both directed against the N terminus) were used to corroborate observations made with the Angpt1/4 (C-19) antibody.
For immunolabeling using amplification and visualization with tyramide-FITC (Perkin Elmer; Waltham, MA), tissue sections were first deparaffinized and incubated for 10 min in 0.5% pepsin (Sigma-Aldrich Chemical Company; St. Louis, MO) in 0.01 M HCl at 37C. Tissue sections were then sequentially blocked with 3% H2O2, 1% avidin (Vector Laboratories; Burlingame, CA) in PBS, 1% biotin (Vector) in PBS, and 1% horse serum (Sigma) in PBS. Primary antibodies were incubated on tissue sections for 2 hr at room temperature or overnight at 4C. Biotin-labeled secondary antibodies (Antibodies Incorporated; Davis, CA) were applied for 1 hr at room temperature. Signal was amplified with streptavidin-HRP and tyramide-FITC according to the kit protocol (Perkin Elmer), except that tyramide-FITC was used at a dilution of 1:500.
Primary antibodies and lectin used in tissue section labeling, immunoprecipitation, and Western blotting
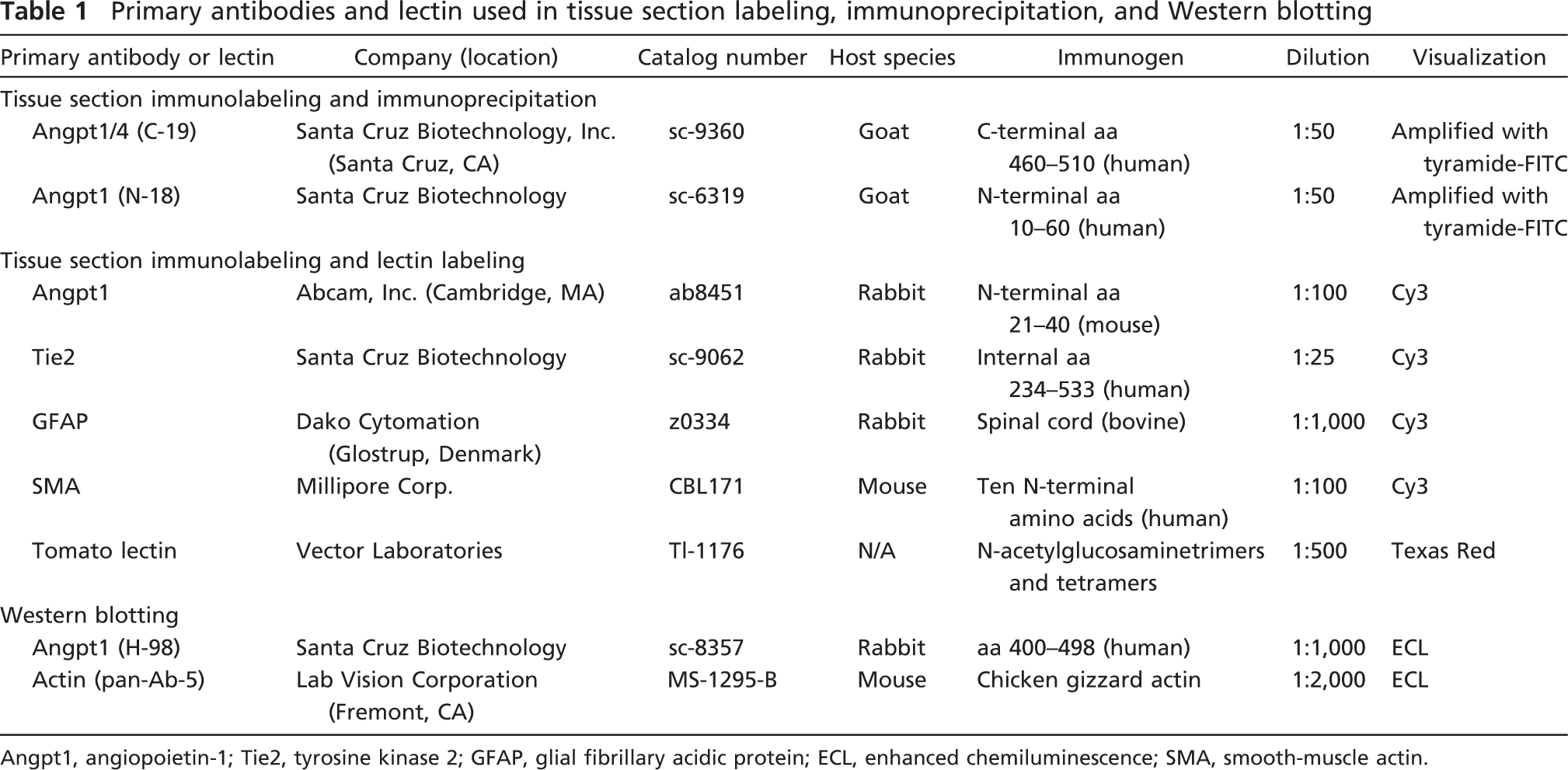
Angpt1, angiopoietin-1; Tie2, tyrosine kinase 2; GFAP, glial fibrillary acidic protein; ECL, enhanced chemiluminescence; SMA, smooth-muscle actin.
For visualization without amplification, tissues were deparaffinized and blocked in 1% horse serum (Sigma) in PBS, and primary antibodies were incubated on sections for 2 hr at room temperature or overnight at 4C. Cy3-coupled secondary antibodies (Millipore; Billerica, MA) at a dilution of 1:100 were then incubated on tissue sections for 1 hr at room temperature.
In double-label characterization of vascular Angpt1 IR, tomato lectin (Lycopersicon esculentum) and antibodies against GFAP and α-smooth muscle actin (SMA) were utilized to detect endothelium (Porter et al. 1990), astrocytes, and pericytes (Bandopadhyay et al. 2001), respectively (Table 1). In double-label characterization of ligand-receptor localization, antibodies against Angpt1 generated in goats were mixed with antibodies against the Angpt1 receptor, Tie2, generated in rabbits (Table 1). After immunolabeling, tissue sections were dipped in bisBenzimide H 33258 (Hoechst; Sigma-Aldrich) to label cell nuclei.
Immunizing peptides used in blocking experiments with the Angpt1/4 (C-19) antibody and the antibody directed against Angpt1 N-terminus aa 21–40 were purchased from the respective parent companies (Table 1).
Angpt1 Immunolabeling Specificity
Angpt1 antibodies unexpectedly labeled a proportion of nuclei in tissue sections, although with variable intensity from experiment to experiment. Angpt1 antibodies immunoprecipitated a protein, which migrated at 70 kDa from rat brain (see below) and labeled purified recombinant Angpt1 protein in Western blots, suggesting good specificity (data not shown). The significance of nuclear Angpt1 labeling in tissue sections is unknown.
Angpt1 Immunoprecipitation
Protein lysates were generated from rat brains 10 to 14 days after ovariectomy/pellet implantation, n=3 per treatment group (placebo pellet; 25-μg or 100-μg E2 pellet). A 1:1 mixture of the Angpt1/4 (C-19) and Angpt1 (N-18) antibodies (Table 1) was used for immunoprecipitation in 0.5 mg of protein per brain lysate. Immunoprecipitates were subjected to gel electrophoresis and blotted. Blots were labeled with an antibody that recognizes several angiopoietins, including Angpt1 (H-98; Table 1) and an antibody against actin (Table 1) as a control. Bands were visualized with the enhanced chemiluminescence technique (Amersham Biosciences Corporation; Piscataway, NJ), and optical density was determined using Metamorph software (Offline version 4.6r4, Molecular Devices Corporation; Sunnyvale, CA).
Microscopy and Photography
Coronal sections from bregma −2.12 through −3.30 were photographed. Digital photography was performed by an investigator blinded to the hormonal status of tissues, either with a conventional microscope (Olympus BX41 microscope and DP71 camera; Olympus America, Center Valley, PA) or a confocal microscope (Zeiss LSM410 microscope and camera; Carl Zeiss Micro-imaging, Inc., Thornwood, NY). Care was taken to photograph control slides from which primary antibodies were omitted and control slides in which primary antibodies were mixed with immunizing peptides at the same exposure as slides labeled with primary antibodies. Confocal images used in the figures were reconstructed from source images using Metamorph software (Molecular Devices). Digital postprocessing with Adobe Photoshop 5.0 (Adobe Systems, Inc.; San Jose, CA) was performed on two panels in one of the figures (indicated in the figure legend).
Semi-quantitative Hypothalamic Angpt1 IR Assessment
An investigator blinded to the hormonal status of the source animals examined at least five tissue sections containing the ependyma per animal. In GFAP- and Angpt1-labeled slides, the investigator made a visual assessment of Angpt1/GFAP double-labeled processes as either <50% or ≥50% of the total number of GFAP-positive processes.
Results
Brain tissue from the following groups of animals was evaluated with immunolabeling and lectin labeling: (a) adult male rats, n=2; (b) normal adult cycling female rats, n=2; (c) adult female rats, ovariectomized and implanted with a placebo pellet, n=3, serum E2 level = 3 ± 2 pg/ml; (d) adult female rats, ovariectomized and implanted with a 25-μg E2 pellet, n=4, serum E2 level = 29 ± 20 pg/ml; and (e) adult female rats, ovariectomized and implanted with a 100-μg E2 pellet, n=2, serum E2 level = 125 ± 30 pg/ml.
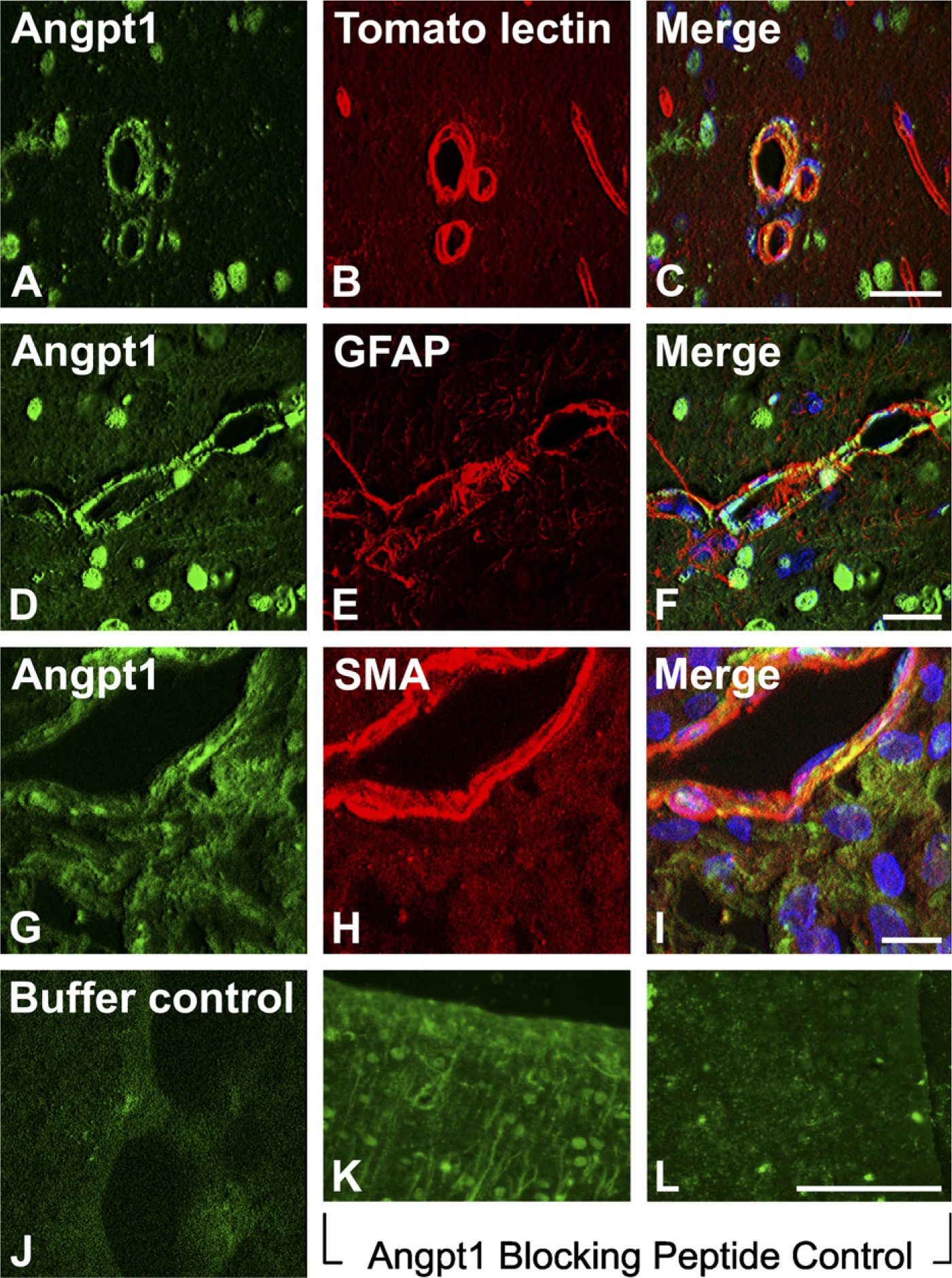
Vascular localization of angiopoietin-1 (Angpt1) immunore-activity (IR) in normal adult female rat brain (confocal microscopy) and labeling controls (confocal and conventional microscopy). (
Patterns of Angpt1 and Tie2 immunolabeling were similar in all rats regardless of sex, hormonal status (treatment group), or plasma E2 level. Immunolabeling was most robust with the Angpt1/4 (C-19) antibody, but all three Angpt1 antibodies resulted in a similar pattern of IR. Angpt1 IR was found in vascular structures (Figures 1A-1I), as well as cuboidal ependyma and tanycytes (Figures 1K, 2, and 3). IR was not present when the primary antibody was omitted and was abrogated by incubation of Angpt1 antibodies with immunizing peptides (Figures 1J and 1L).
Vascular and Ependymal Angpt1
Within vascular structures, Angpt1 IR was associated with pericytes and astrocytes. Specifically, Angpt1 IR was located external to the endothelium and internal to astrocytic processes, with some areas of overlap (Figures 1C, 1F, and 1I).
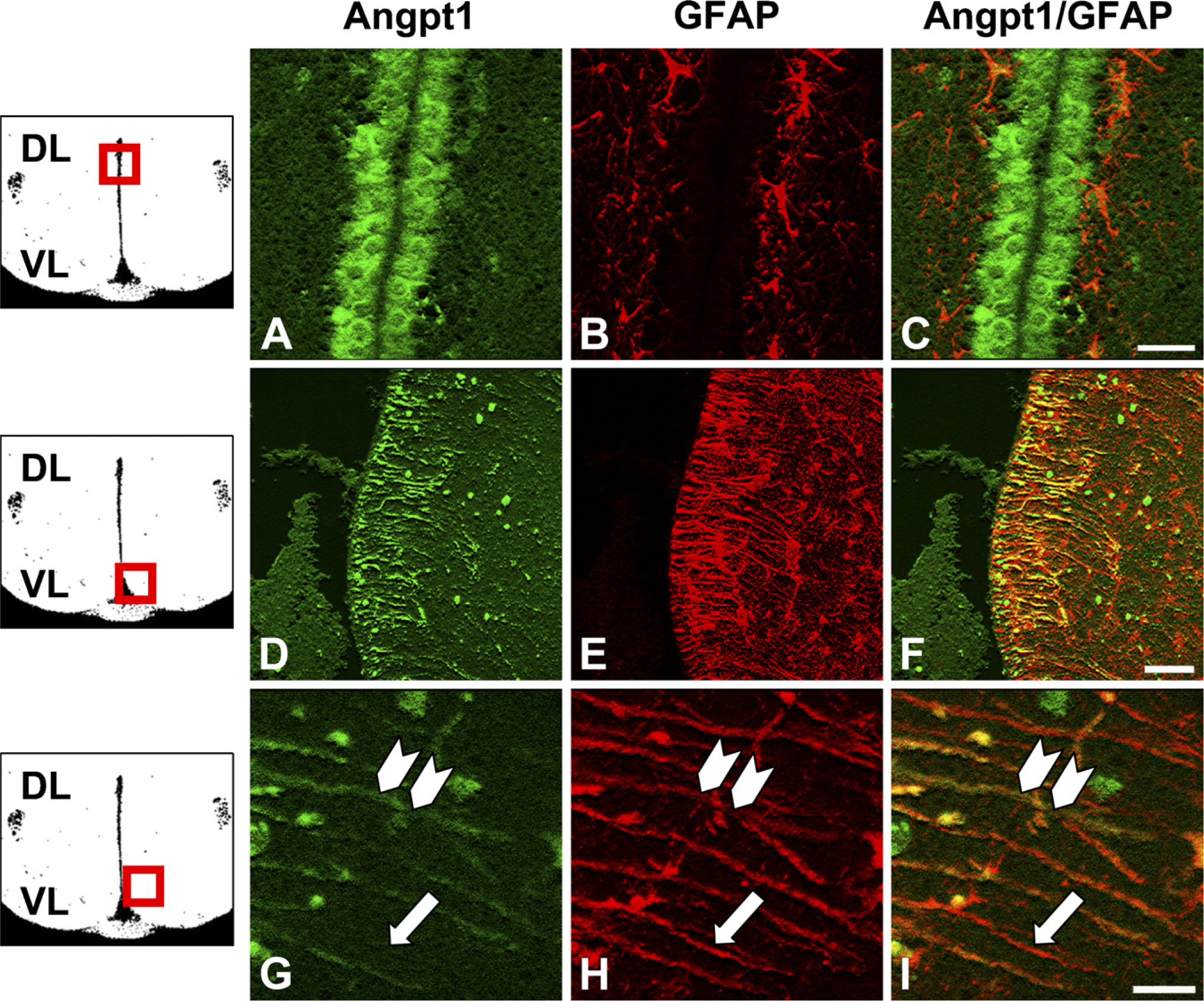
Angpt1 IR and GFAP IR relationships in hypothalamic third-ventricular ependyma in normal adult female rats (confocal microscopy). Diagrams show the orientation and approximate location of the images in each row (boxed area; not to scale). DL, dorsal; VL, ventral. (
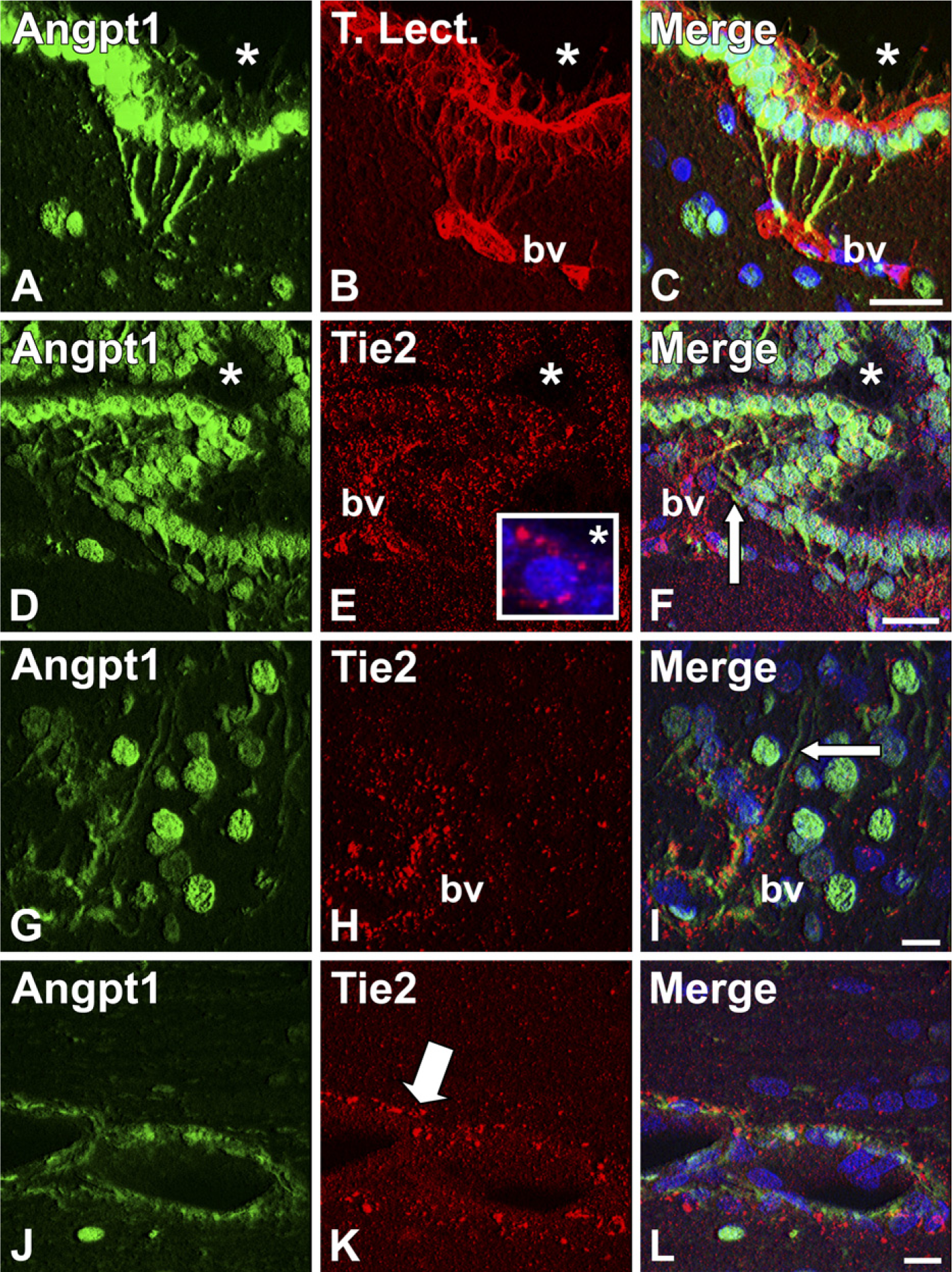
Relationships of ependymal and vascular Angpt1 IR and tyrosine kinase 2 (Tie2) IR in normal adult female rats (confocal microscopy). (
Within the third-ventricular ependyma, Angpt1 IR was present in the cytoplasm of cuboidal ependymal cells in the dorsal aspect of the third ventricle. In this region, GFAP-immunoreactive cells with the morphology of astrocytes were located outside of the Angpt1-immunoreactive cuboidal ependymal cell layer (Figures 2A-2C). In contradistinction, within the ventral regions of the third-ventricular ependyma, Angpt1 IR was localized in GFAP-immunoreactive processes originating within the ependymal layer and coursing radially from the ventricle (Figures 2D-2F). Based on the location and morphology of the processes, the GFAP-immunoreactive/Angpt1-immunoreactive bipolar cells in the ventral hypothalamic third-ventricular ependyma were identified as tanycytes (Rodriguez et al. 2005). In all rats sampled, ≥50% of GFAP-immunoreactive tanycytes were also Angpt1 immunoreactive (Figures 2G-2I). Angpt1 IR was present in α1, α2, β1, and β2 tanycytes, based on location within the ependyma.
Cuboidal ependymal cells of the Sylvian aqueduct exhibited Angpt1 IR in the cytoplasm, as well as in projections that were in contact with nearby vascular structures (Figures 3A-3C).
Angpt1-Tie2 Relationships
To assess anatomical distribution of Angpt1 and its receptor, Tie2, we evaluated Angpt1 IR relative to Tie2 IR in double-labeled tissue sections. Tie2 IR was enriched in the ependyma and adjacent vascular structures (Figures 3D-3L). In Angpt1-immunoreactive cuboidal ependyma, Tie2 IR was distributed in a pattern suggesting membrane localization that was especially prominent in regions of contact between adjacent ependymal cells and on the lumenal surface (Figures 3D-3F). Tips of some Angpt1-immunoreactive tanycyte processes were found in close proximity to Tie2-immunoreactive vascular structures coursing parallel to the ependymal surface (Figures 3D-3I). Vascular Angpt1 IR and Tie2 IR were also closely associated (Figures 3J-3L).
Effect of Hormonal Status on Angpt1 Expression
Based on previous observations that cerebral Angpt1 mRNA expression is modulated by E2 (Ardelt et al. 2005), Angpt1 protein expression was evaluated in brains of rats with different hormonal milieus. Similar amounts of Angpt1 protein were immunoprecipitated from brains of ovariectomized rats treated with three different doses of E2 (Figure 4A). The pattern of ependymal Angpt1 IR was very similar in all rats tested regardless of sex or hormonal status (Figures 4B and 4C).
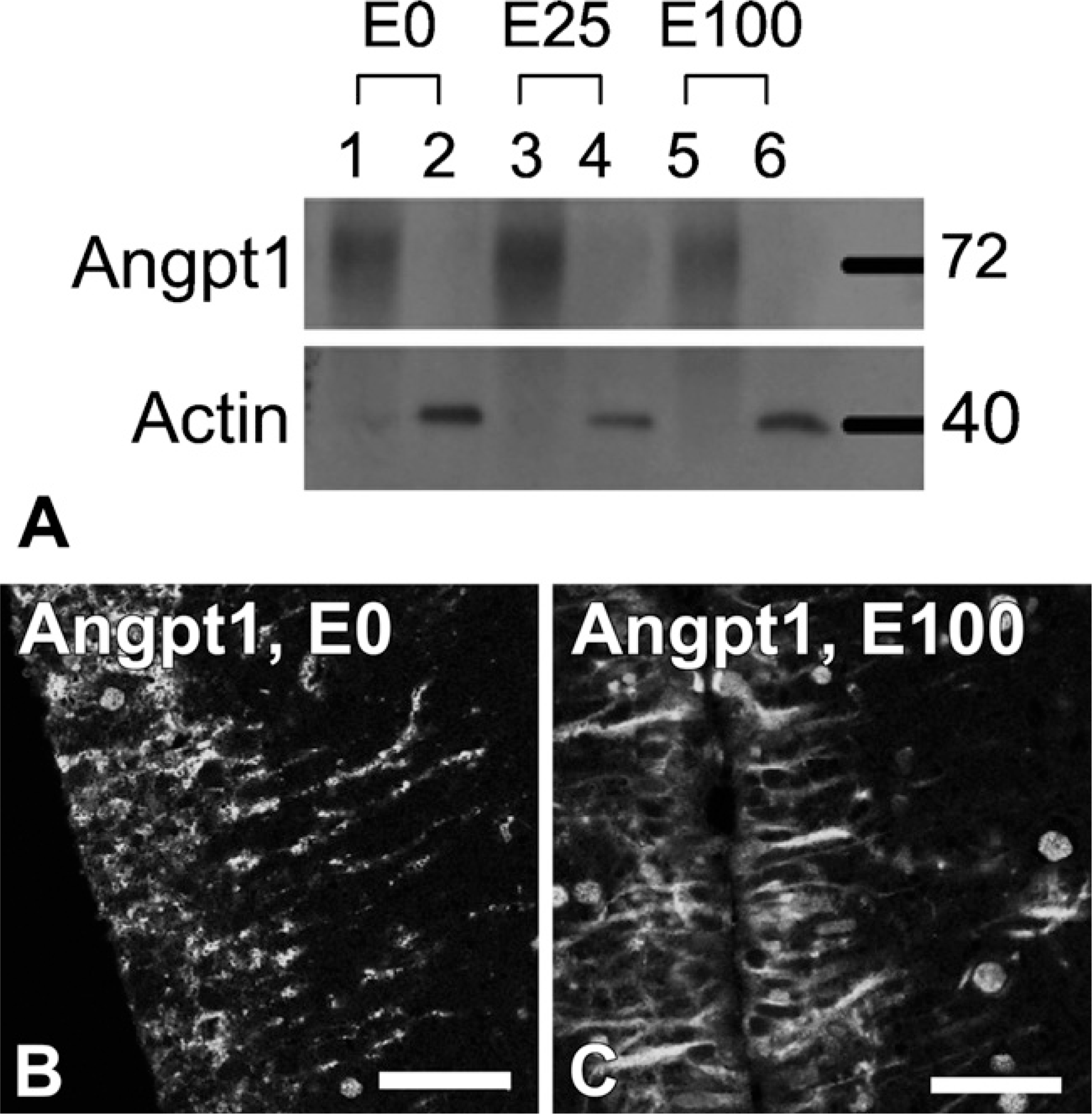
Effect of hormonal status on cerebral Angpt1 expression (immunoprecipitation and confocal microscopy). (
Discussion
In this investigation, we characterized the patterns of Angpt1 and Tie2 expression in rat brain. We found that antibodies against Angpt1 labeled cuboidal ependyma and tanycytes. Antibodies against the Angpt1 receptor, Tie2, labeled the membranes of Angpt1-immunoreactive cuboidal ependymal cells, and Angpt1-immunoreactive tanycyte processes were in close association with Tie2-immunoreactive blood vessels.
Tanycytes synthesize a variety of proteins, as well as transport molecules from the ventricular CSF (Rodriguez et al. 2005). Although it is not known whether Angpt1 protein is synthesized in tanycytes or taken up from the ventricle, the finding of Tie2 IR on the ventricular surface of ependymal cells suggests the possibility of receptor-mediated Angpt1 endocytosis. It is tempting to speculate that tanycytes, by virtue of their ability to sample the CSF, may use CSF-derived Angpt1 as a sentinel of physiological or pathological conditions.
The finding of close anatomical relationships between Angpt1-immunoreactive/Tie2-immunoreactive cuboidal ependymal cells, and between Angpt1-immunoreactive tanycyte processes and Tie2-immunoreactive blood vessels, implies functional coupling. One possible functional endpoint of ependymal Angpt1–-vascular Tie2 interaction is modulation of occludin expression (Iizasa et al. 2002; Wakui et al. 2006). Angpt1 secreted from tanycyte basal processes into the perivascular space may influence properties of the neurovascular unit; for example, tanycyte-derived Angpt1 may locally augment the blood–-brain barrier by upregulating occludin.
Tanycyte functions may be under hormonal control (reviewed in Rodriguez et al. 2005; Garcia-Segura et al. 2008). We previously observed that cerebral Angpt1 mRNA content was augmented by E2 (Ardelt et al. 2005), and, therefore, we hypothesized that altering the hormonal milieu will affect cerebral Angpt1 protein expression. Grossly, we did not find evidence that E2 modulates hypothalamic Angpt1; however, our in vivo experiments were not designed to detect small quantitative effects. Further testing of this hypothesis may be better suited to controlled in vitro systems such as hypothalamic organotypic slices (Levy et al. 1996).
In conclusion, we observed Angpt1 IR in cuboidal ependyma and tanycytes. We found no E2-mediated effects on cerebral Angpt1 expression. We demonstrated Tie2 IR in Angpt1-immunoreactive cuboidal ependyma, as well as in vascular targets of Angpt1-immunoreactive tanycyte basal processes. These anatomical relationships, coupled with previously published data on the function of tanycytes, suggest ligand-receptor interactions that may affect cerebral vascular and ependymal function.
Footnotes
Acknowledgements
This work was supported by National Institutes of Health Grant K-08 NS-050167 to Dr. Ardelt and National Institutes of Health Neuroscience Blueprint Core Grant NS-57098 to the University of Alabama at Birmingham.
