Abstract
The pituitary gland dynamically changes its hormone output under various pathophysiological conditions. One of the pathways implicated in the regulatory mechanism of this gland is a dopaminergic system that operates the phosphoinositide (PI) cycle to transmit downstream signal through second messengers. We have previously shown that diacylglycerol kinase β (DGKβ) is coexpressed with dopamine D1 and D2 receptors in medium spiny neurons of the striatum, suggesting a plausible implication of DGKβ in dopaminergic transmission. However, it remains elusive whether DGKβ is involved in the dopaminergic system in the pituitary gland. The aim of this study is to investigate the expression and localization of DGK in the pituitary gland, together with the molecular components involved in the PI signaling cascade, including dopamine receptors, phospholipase C (PLC), and a major downstream molecule, protein kinase C (PKC). Here we show that DGKβ and the dopamine D2 receptor are coexpressed in the intermediate lobe and localize to the plasma membrane side by side. In addition, we reveal that PLCβ4 and PKCα are the subtypes expressed in the intermediate lobe among those families. These findings will substantiate and further extend our understanding of the molecular-anatomical pathway of PI signaling and the functional roles of DGK in the pituitary intermediate lobe.
Keywords
T
To date, five different dopamine receptor subtypes have been cloned from different species. It is believed that the dopamine D1- and D2-like receptors are expressed in a different subset of the striatal projection neurons (Gerfen et al. 1990; Gerfen 1992). The dopamine D1-like receptors are expressed preferentially in neurons along the striatonigral pathway, whereas the dopamine D2-like receptors are expressed in the medium spiny neurons of the striatopallidal pathway. In the pituitary gland, the dopaminergic system has been implicated in the inhibitory regulation of the secretion of several hormones. The tuberoinfundibular dopaminergic pathway originates in neurons of the hypothalamus and regulates secretion of prolactin from the anterior pituitary (Fuxe et al. 1969). A pharmacological study showed that the dopamine D2 receptor occurs on cells of the intermediate lobe and mediates inhibition of peptide hormone release from these cells (Cote et al. 1986). Furthermore, PKC, a major downstream DG signal, has been shown to play a role in regulating pituitary hormone synthesis and secretion in the anterior lobe (Garcia-Navarro et al. 1994; Korytko et al. 1998). However, little is known about the morphological aspects of the molecular components involved in the PI cycle of this gland.
In relation to the PI cycle, we have been working on DG kinase (DGK), which catalyzes the phosphorylation of DG. DGK is now revealed to represent a large gene family of isozymes (Topham 2006; Goto et al. 2007; Sakane et al. 2007). Previous studies have reported that DGK isozymes exhibit remarkable heterogeneity in structure, tissue expression, and enzymological properties. We have shown the detailed cellular expression of mRNAs for the isozymes and their functional implications in the central nervous system and heart (Goto et al. 1992, 1994, 2006; Goto and Kondo 1993, 1996; Hozumi et al. 2003; Ito et al. 2004; Takahashi et al. 2005; Arimoto et al. 2006). These observations suggest that each isozyme has its own specific function in various biological processes.
Of the DGK isozymes, we have previously shown that DGKβ is abundantly expressed in the striatum of the brain (Goto and Kondo 1993; Hozumi et al. 2008). Further study revealed that DGKβ is selectively expressed in dopamine D1 receptor– and dopamine D2 receptor–positive medium spiny neurons and exhibits dense accumulation at perisynaptic sites on dendritic spines forming asymmetrical synapses (Hozumi et al. 2008). A similar pattern of the expression of DGKβ and dopamine receptors in the brain led us to further scrutinize the relationship between these molecules in other regions.
In the present study, we investigated the expression and localization of DGK in the pituitary gland, together with the molecular components involved in the PI signaling cascade, including dopamine receptors, PLC, and a major downstream molecule, PKC. Here we show that DGKβ and the dopamine D2 receptor are expressed in the intermediate lobe and localize to the plasma membrane side by side. In addition, we reveal that PLCβ4 and PKCα are the subtypes expressed in the intermediate lobe among those families. Molecular anatomical identification of the DG signaling molecules would provide us clues to further investigate the regulatory mechanism of cells in the pituitary intermediate lobe.
Materials and Methods
Animals
This study was carried out in accordance with the Guide for Animal Experimentation, Yamagata University School of Medicine. Animals were purchased from Japan SLC (Hamamatsu, Japan).
RT-PCR Analysis
Total RNAs were extracted from pituitaries of 9-week-old Wistar rats by TRIzol (Invitrogen; Carlsbad, CA). First-strand cDNA was synthesized from 2 μg of RNA using Moloney murine leukemia virus (M-MLV) reverse transcriptase (Promega; Madison, WI) following the manufacturer's instructions. Polymerase chain reaction (PCR) amplification was performed with KOD-plus polymerase (Toyobo; Tokyo, Japan) using the gene-specific oligonucleotide primers for rat DGK isozymes shown in Table 1 (Ito et al. 2004; Katagiri et al. 2005). PCR conditions were as follows: 94C for 5 min; 33 cycles of 94C for 30 sec, 62C for 30 sec, and 68C for 40 sec, and 68C for 2 min. For control, rat glyceraldehyde-3-phosphate dehydrogenase mRNA was simultaneously amplified using the primers shown in Table 1. PCR products amplified were separated by agarose gel electrophoresis and stained with ethidium bromide. We repeated the RT-PCR twice for one sample derived from one rat (total two rats). Similar expression patterns were obtained from these experiments, and one experiment representative of four experiments is shown.
PCR primer name, sequence, and product size
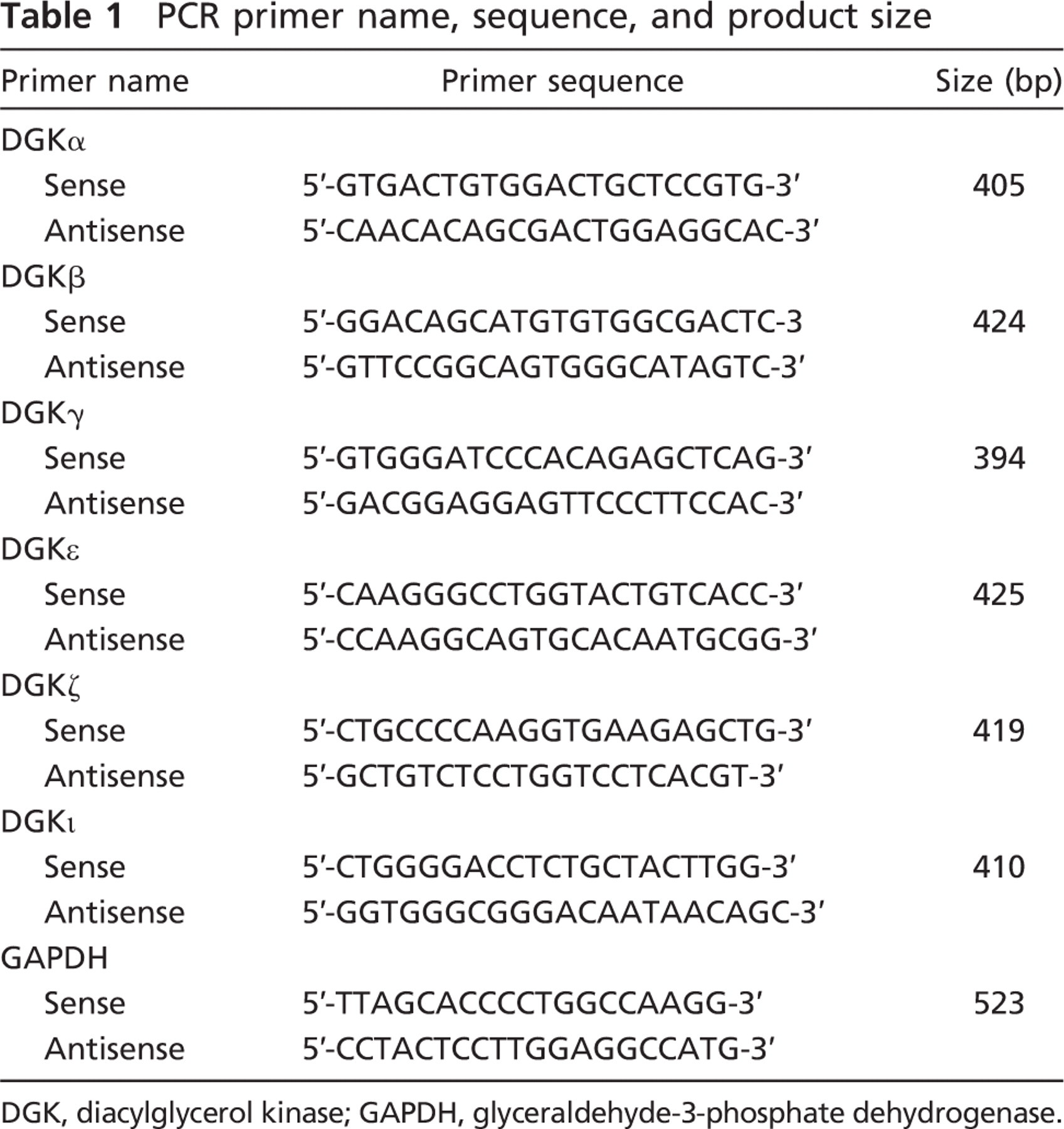
DGK, diacylglycerol kinase; GAPDH, glyceraldehyde-3-phosphate dehydrogenase.
In Situ Hybridization
Cryostat sections of adult rat pituitary gland were hybridized with 0.5–1.0 × 106 cpm per slide of the same cDNA probe labeled with [α-[35S]thio]dATP as described previously (Goto and Kondo 1993). After exposure to Hyperfilm-βmax (GE Healthcare UK Ltd; Buckinghamshire, UK) for 2–3 weeks, the sections were dipped in Kodak NTB2 emulsion and exposed for 3 months.
Immunoblotting
Pituitary gland, brain, and liver of 9-week-old rats were homogenized with 4 vols of a buffer containing 10 mM Tris-HCl (pH 7.4), 20 mM KCl, 0.1 mM EDTA, and 0.25 M sucrose, and centrifuged at 1000 × g for 10 min at 4C to remove debris. Protein concentration was determined using BCA protein assay reagent (Thermo Scientific; Rockford, IL). Values were the means of triplicate determinations. The resulting supernatant (30 μg) was boiled for 5 min in sodium dodecylsulfate (SDS) sample buffer (New England Biolabs, Inc.; Beverly, MA) and subjected to 10% SDS-PAGE. The proteins were then electrophoretically transferred to a polyvinylidene difluoride membrane (NEN Life Science Products, Inc.; Boston, MA). After the nonspecific binding sites were blocked with 5% non-fat dry milk (w/v) in phosphate-buffered saline (PBS) containing 0.02% sodium azide and 0.2% Tween 20, the membrane was incubated for 1 hr at room temperature with 0.5 μg/ml guinea pig anti-DGKβ antibody (Hozumi et al. 2008) or mouse anti-β-actin (1:5000; Sigma-Aldrich; St. Louis, MO) in PBS containing 0.1% Tween 20. Sites of antigen–antibody reaction were visualized using the chemiluminescent ECL Plus Western blotting detection system (GE Healthcare UK, Ltd.; Buckinghamshire, UK). We repeated the Western blot three times and obtained the same results from those experiments.
Tissue and Section Preparation
Adult male Wistar rats at 9 weeks of age were used. For immunohistochemistry, rats anesthetized with ether were fixed transcardially with periodate lysine paraformaldehyde (PLP) for immunofluorescence microscopy (Hozumi et al. 2003). Pituitaries were carefully isolated under the stereomicroscope and immediately fixed for 2 hr with PLP. Pituitaries were embedded in 2% agarose/PBS before sectioning with the Vibratome (VT1200S, Leica; Nussloch, Germany). Pituitaries were carefully positioned to enable cutting 50-μm sections through all lobes and cleft.
Immunohistochemistry
The immunohistochemical procedures employed were previously reported (Krylyshkina et al. 2005). We examined three sections from three rats for each antibody. When using microslicer sections, a brief section pretreatment with methanol greatly enhanced immunoreactivity for DGKβ (Hozumi et al. 2008). Therefore, sections for immunofluorescence were treated by dipping successively in 30, 60, and 100% methanol for 2 min each. All immunohistochemical incubations were performed at room temperature (~20C). Permeabilization of the sections was achieved with 30-min incubations in saponin [0.5% in PBS (PBS-Sap); Wako, Osaka, Japan]. Aspecific binding sites were preadsorbed with 10% normal goat serum.
In double immunofluorescence, guinea pig anti-DGKβ (1.5 μg/ml) or anti-dopamine D1 receptor [(D1R) 1 μg/ml] (Narushima et al. 2006; Hozumi et al. 2008) antibody diluted with PBS-Sap was immunoreacted overnight in a mixture with one of the following rabbit antibodies: anti-dopamine D2 receptor [(D2R) 1 μg/ml] (Narushima et al. 2006, Hozumi et al. 2008), or 1 μg/ml anti-PLCβ4 (Nakamura et al. 2004). After thorough washing in PBS-Sap, sections were incubated for 2 hr with Alexa Fluor 488–conjugated anti-rabbit IgG (1:200; Molecular Probes, Eugene, OR) and biotinylated anti-guinea pig IgG (1:200; Vector Laboratories, Burlingame, CA). Sections were then incubated for 1 hr with streptavidin, Alexa Fluor 546 conjugate (1:100; Molecular Probes) in PBS-Sap.
For single immunofluorescence, 1 μg/ml rabbit anti-mGluR1α (Tanaka et al. 2000), 1 μg/ml goat anti-mGluR5 (Uchigashima et al. 2007; Hozumi et al. 2008), 1 μg/ml rabbit anti-PLCβ1 (Fukaya et al. 2008; Hozumi et al. 2008), 1 μg/ml goat anti-PLCβ2 (sc-31757; Santa Cruz Biotechnology, Santa Cruz, CA), 1 μg/ml rabbit anti-PLCβ3 (Nomura et al. 2007), 1 μg/ml guinea pig anti-DGKβ, 1 μg/ml rabbit anti-DGKε (Nakano et al. 2009), 1 μg/ml rabbit anti-DGKζ (Hozumi et al. 2003), 1 μg/ml rabbit anti-PKCα (Nakano et al. 2006), 1 μg/ml rabbit PKCβII (Hasegawa et al. 2008), or 1 μg/ml rabbit anti-PKCγ antibody (Yoshida et al. 2006) diluted with PBS-Sap was immunoreacted overnight at room temperature. After thorough washing in PBS-Sap, sections were incubated for 2 hr with Alexa Fluor 488–conjugated anti-rabbit, guinea pig, or goat IgG (1:200; Molecular Probes). Sections were scanned using a confocal laser scanning microscope (LSM510META, Carl Zeiss; Göttingen, Germany) in multitrack mode.
Quantification of Labeled Cells
In the present study, we stained the nuclei of cells using TO-PRO-3 (nucleic acid stains) (data not shown). In an area that included 100 cells in total, we counted the number of mGluR1α-, PKCα-, PKCβII-, DGKε-, or DGKζ-positive cells in the anterior, intermediate, and posterior lobe and calculated the expression ratio of those positive cells in each lobe. We counted three independent areas derived from three rats.
Results
Expression of DGK Isozymes in the Pituitary Gland
We first examined whether DGKβ is expressed in the pituitary gland. RT-PCR analysis clearly showed that expression signals for DGKβ are intensely detected in this organ (Figure 1A). In addition, the signals were also detected intensely for DGKζ, moderately for DGKε, and faintly for DGKα. The signals for DGKγ and -ι were below the detection level. The results show that DGKβ is the major isozyme in the pituitary gland.
Expression and Distribution of DGKβ in the Pituitary Gland
To examine regional and cellular localization of DGKβ, we then employed immunohistochemical analysis of the pituitary gland using a specific antibody. The antibody recognized a single band of DGKβ at an estimated molecular mass in lysates of the pituitary gland and brain, but not in that of the liver (Figure 1B). We observed intense labeling for DGKβ solely in the intermediate lobe, and not in the anterior and posterior lobes (Figure 1C). In the intermediate lobe, immunohistochemical signals for DGKβ were seen as tiny puncta on or close to the plasma membrane (Figure 1D). These puncta appeared to outline the boundary between the cells, suggesting that DGKβ is expressed in nearly all of the cells in this lobe. On the other hand, immunolabeling for DGKβ was hardly detected in the cytoplasm. The expression of DGKβ in cells of the intermediate lobe was further confirmed by in situ hybridization analysis that showed that mRNA signals for DGKβ were clearly detected in this lobe (Figure 1E).
DGKβ Is Coexpressed With D2R in Cells of the Intermediate Lobe
Our previous study revealed that DGKβ is selectively expressed in D1 R-positive and D2R-positive medium spiny neurons in the striatum and exhibits dense accumulation at perisynaptic sites on dendritic spines forming asymmetrical synapses (Hozumi et al. 2008). In this regard, it is known that the dopaminergic system is involved in the inhibitory regulation of the secretion of several peptide hormones in the pituitary (Ben-Jonathan et al. 1989). Similar patterns of expression between DGKβ and dopamine receptors in the brain led us to investigate the relationship between these molecules in the pituitary gland. Immunohistochemical staining revealed that immunolabeling for D2R was detected solely in the intermediate lobe and was seen as tiny puncta on or close to the plasma membrane of cells (Figure 2). This pattern of expression and localization is extremely similar to that of DGKβ. When compared by double immunofluorescence, however, D2R-positive puncta were apposed side by side to DGKβ-positive puncta but rarely overlapped with them (Figure 3). On closer examination, we found that D2R immunoreactivity was occasionally enriched in zonal regions 2–3 μm long. In contrast to D2R, immunolabeling for D1R was exclusively detected in the anterior lobe and was seen diffusely in the cytoplasm of scattered cells that occupied 20–30% of the population of this lobe (Figure 2). It remained to be determined which cell types are responsible for the expression of D2R in relation to hormone production. Taken together, it is suggested that DGKβ is involved in the D2R signal cascade in the intermediate lobe and that these molecules are apposed side by side, but not in a stable complex.
DGKβ Is Coexpressed With PLCβ4 in Cells of the Intermediate Lobe
D2R belongs to a family of seven transmembrane domain G protein–coupled receptors, suggesting an involvement of the β type of PLC (PLCβ) in transmitting signals. To determine which subtypes are responsible for this role in the intermediate lobe, we employed immunohistochemical anaysis. Of the PLCβ subfamily, the immunolabeling was solely detected for PLCβ4 in the pituitary gland (Figure 4), but no labeling was recognized for PLCβ1, −2, and −3 (data not shown). Immunohistochemical signals for PLCβ4 were seen as tiny puncta near the plasma membrane, though rather diffusely distributed compared with those for DGKβ (Figure 4). When compared by double immunofluorescence, PLCβ4-positive puncta rarely overlapped with DGKβ-positive puncta (Figure 4). Zonal regions characterized by intense D2R immunoreactivity were not encountered for PLCβ4 (Figure 4). These results suggest that PLCβ4 is the only subtype expressed in the pituitary gland and that it is localized near the plasma membrane in cells of the intermediate lobe.
Expression and Localization of Other PI Signaling Molecules
In addition to dopamine receptors, Group I mGluRs, including mGluR1 and −5, are also known to operate PI signaling via G proteins (Gilman 1987; Masu et al. 1991; Simon et al. 1991; Abe et al. 1992; Neer 1995; Exton 1996). A previous study revealed that PLCβ4 forms an immunoprecipitable complex with mGluR1α in the brain (Nakamura et al. 2004). Furthermore, we showed that DGKβ and mGluR5 localize side by side at perisynaptic sites of medium spiny neurons in the striatum (Hozumi et al. 2008). Therefore we investigated whether type I mGluRs are expressed in the pituitary gland, and if they are, where they localize. The immunohistochemical signals for mGluR1α were solely detected in the posterior lobe (Figure 5A), although the signals for mGluR5 were recognized nowhere in this organ (data not shown), showing that Group I mGluRs are below detection level in the intermediate lobe.
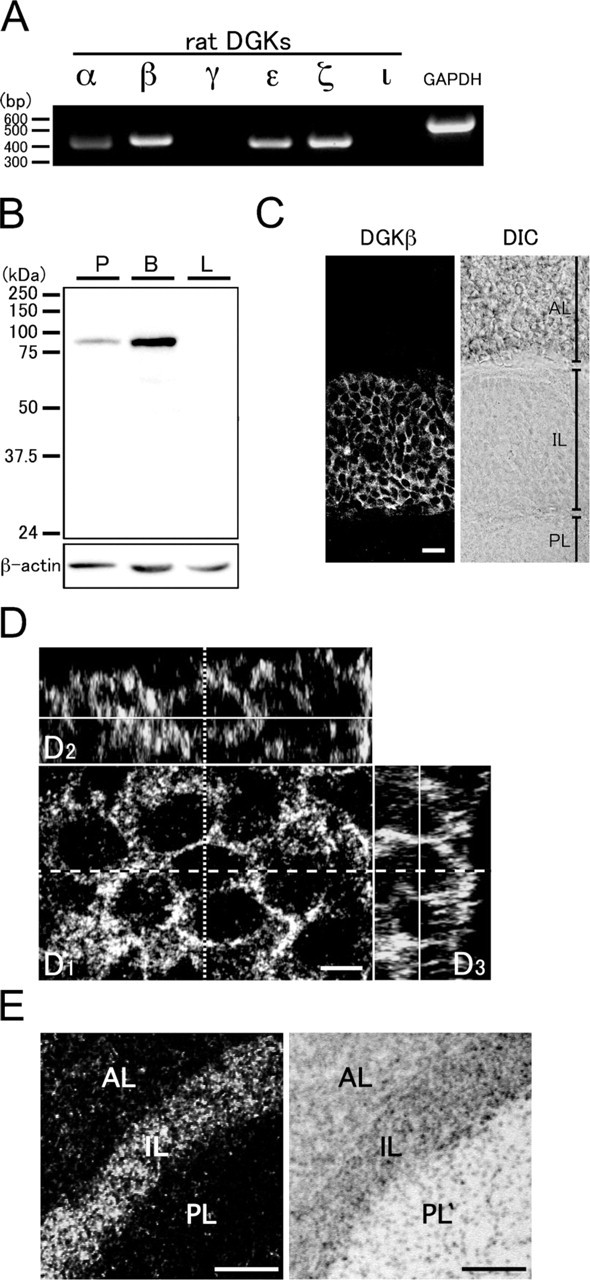
Expression of diacylglycerol kinase (DGK) isozymes in the pituitary gland. (
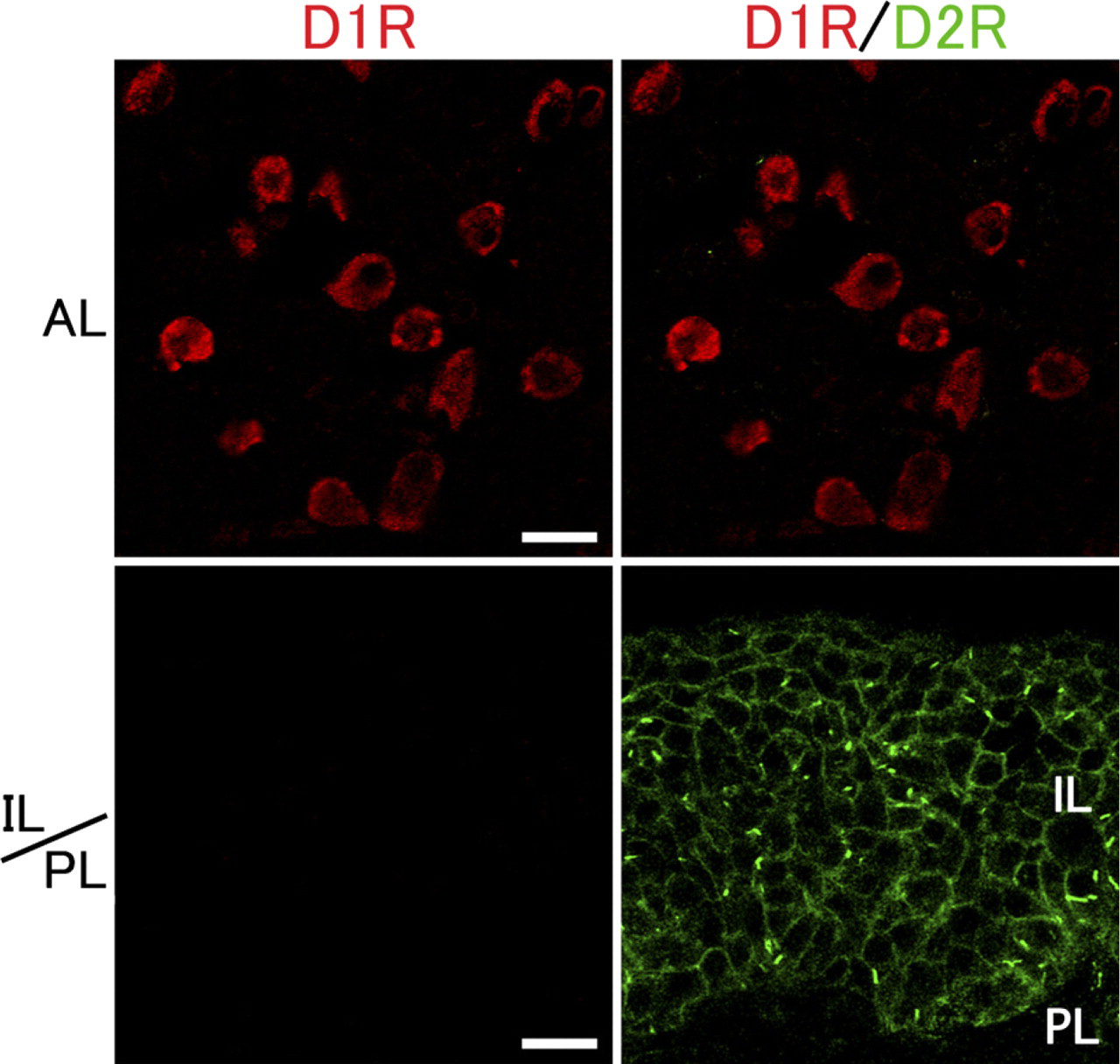
Expression and localization of dopamine D1 receptor (D1R) and D2R in the pituitary gland. Double immunofluorescense for D1R (red) and D2R (green) in the pituitary gland. Immunolabeling for D2R is detected solely in the intermediate lobe and is seen as tiny puncta on or close to the plasma membrane, whereas that for D1R is detected in the anterior lobe, but not the intermediate lobe. AL, anterior lobe; IL, intermediate lobe; PL, posterior lobe. Bar = 20 μm.
With regard to the downstream DG signal, PKC has been shown to play a major role in regulating pituitary hormone synthesis and secretion (Garcia-Navarro et al. 1994; Korytko et al. 1998). Therefore we examined which PKC isoforms are expressed in the pituitary gland by immunohistochemical analysis. Of the classical PKCs, the immunolabeling for PKCα was detected in the anterior, intermediate, and posterior lobes (Figure 5A). The signals for PKCα were seen in the cytoplasm of cells (~30% of population) in the anterior lobe and of almost all of the cells in the intermediate and posterior lobes. On the other hand, the immunolabeling for PKCβII was detected in the anterior and posterior lobes (Figure 5A). The signals for PKCβII were seen in the cytoplasm of cells (~20% of population) in the anterior lobe and of pituicytes (~50% of population) in the posterior lobe. It remained to be determined which cell types are responsible for the expression of PKCα and PKCβII in the anterior lobe in relation to hormone production. No immunolabeling was found for PKCγ in the pituitary gland (data not shown). These results suggest that PKCα is the only isoform of the classical PKCs in the intermediate lobe.
With regard to the other DGK isozymes detected by RT-PCR analysis, DGKζ was observed in the nuclei of almost all of the secretory cells of the anterior and intermediate lobes and the pituicytes of the posterior lobe, whereas DGKε was seen in the cytoplasm of a small number of cells (~20% population) of the anterior lobe but not of the intermediate lobe (Figure 5B). These results suggest that DGKβ and DGKζ are coexpressed and play different roles in each region of cells in the intermediate lobe.
Discussion
The present study reveals that DGKβ is expressed in the pituitary intermediate lobe and localizes to or near the plasma membrane of cells as tiny puncta. This localization pattern is shown to be similar but not identical to that of D2R and PLCβ4, suggesting that the D2R-PLCβ4-DGKβ cascade is likely to operate dopaminergic transmission in the intermediate lobe cells. Previous studies have shown that D2R on cells of the intermediate lobe mediates inhibition of peptide hormone release from these cells (Cote et al. 1986). In addition, pharmacological experiments suggest that dopamine is involved not only in the inhibitory control of the secretion of α-melanocyte–stimulating hormone (α-MSH) from melanotrophs, but also in both the regulation of transcription of propiomelanocortin (POMC) mRNA and the regulation of post-translational processing of the POMC protein (Yamaguchi et al. 1996). In accordance with these findings, D2R-deficient mice show a hypertrophic change in the intermediate lobe of the pituitary gland accompanied by increased expression of POMC, the precursor of α-MSH (Yamaguchi et al. 1996). Therefore it is hypothesized that the D2R-PLCβ4-DGKβ cascade might be involved in these mechanisms, which warrants further investigation.
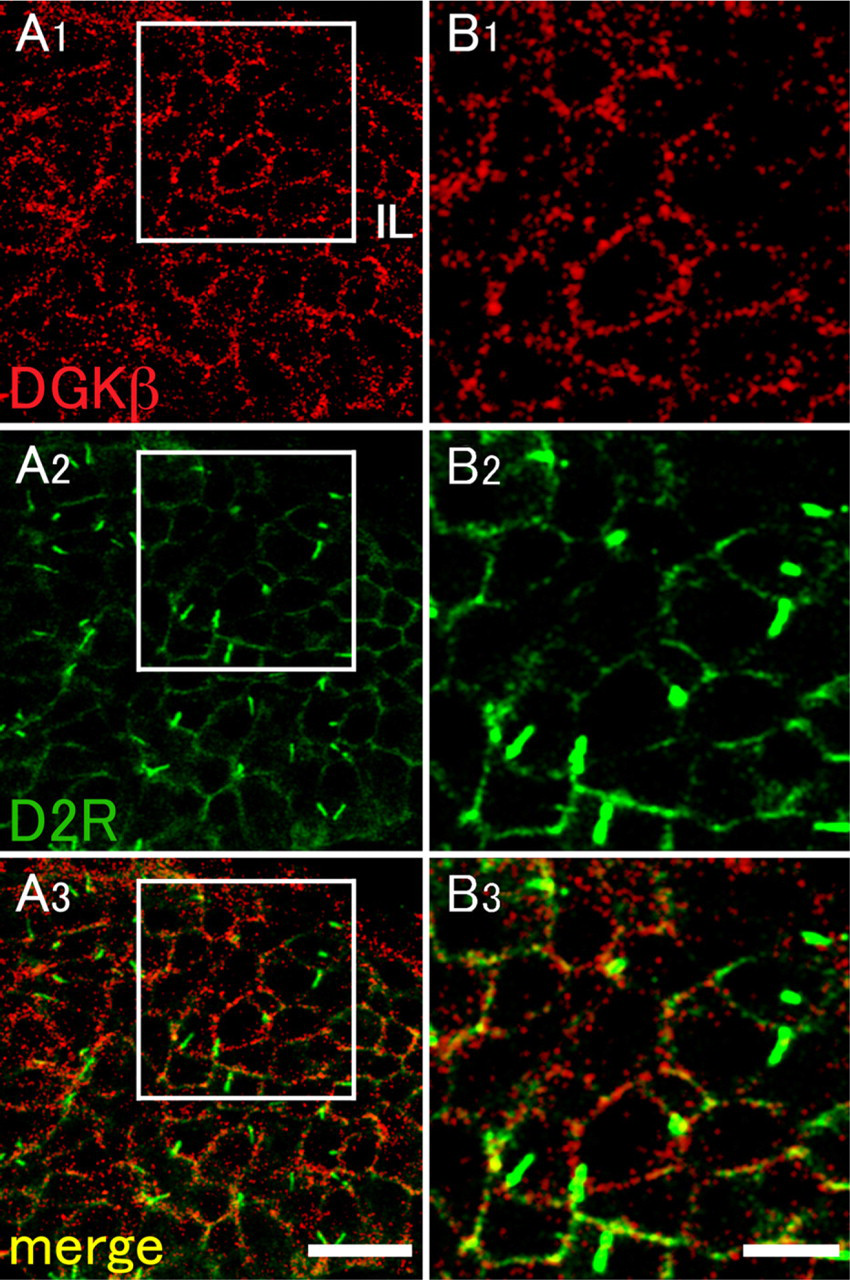
Double-fluorescence immunohistochemistry for DGKβ and D2R in the intermediate lobe. DGKβ as red fluorescence (
What is the functional implication of the compartmentalization of DGKβ from the presumed complex of PLCβ4? Coimmunoprecipitable complex suggests a stable, if not completely so, molecular interaction, which is exemplified by the previous study on PLCβ4 and mGluR1α in the brain (Nakamura et al. 2004). In this case, ligand stimulation of mGluR1α is converted to PLCβ4 activation, leading to the immediate generation of the second messenger signal, DG. Once DG is generated, a fine tuning of this cascade depends on DGK, which controls how long DG stays at a given place, to activate downstream molecules. In this sense, compartmentalization of DGKβ from PLCβ under unstimulated conditions, as shown in the present study, may be reasonable, because in the initial phase of stimulation, compartmentalization certainly delays the access of DGKβ to DG and its attenuation, which results in a transient accumulation of DG on the membrane. Longer segregation of DGKβ from the presumed molecular complex of PLCβ4 contributes to continued activation of the downstream signaling. This speculation is also supported by the results of our previous study, showing that immunolabeled puncta for DGKβ and for mGluR5 or PLCβ1 are not overlapped but rather are positioned side by side in medium spiny neurons of the striatum, showing that DGKβ and mGluR5 or PLCβ1 are compartmentalized and form distinct but closely associated molecular clusters in neurons of the brain (Hozumi et al. 2008).
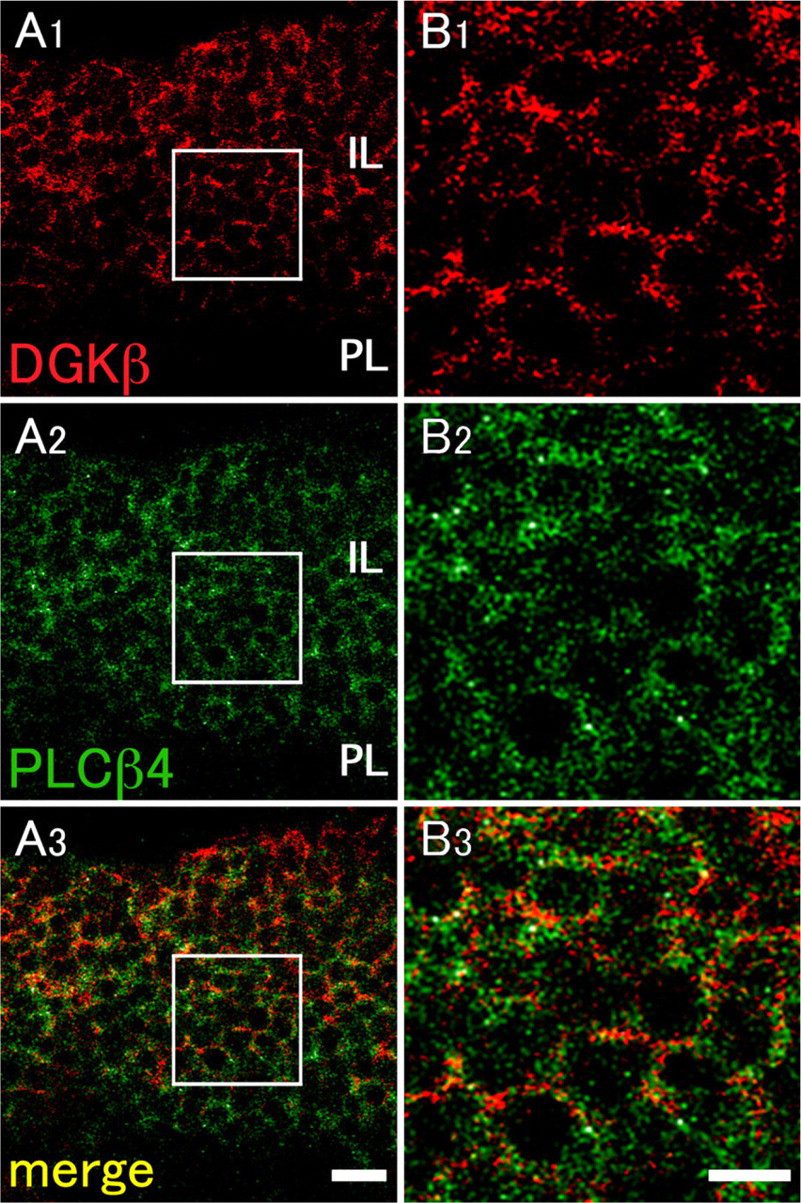
Double-fluorescence immunohistochemistry for DGKβ and phospholipase C β4 (PLCβ4) in the intermediate lobe. DGKβ as red fluorescence (
With regard to the downstream pathway of DG signal, previous studies have shown that PKC activators consistently stimulate secretion of luteinizing hormone (Smith and Vale 1980; Conn et al. 1987; Johnson et al. 1993; Thomson et al. 1993), growth hormone (Smith and Vale 1980; Summers et al. 1985; Johnson et al. 1993; Thomson et al. 1993; Cuttler et al. 1995), prolactin (Summers et al. 1985), thyroid-stimulating hormone (Smith and Vale 1980), and adrenocorticotropic hormone (Abou-Samra et al. 1986) from the anterior pituitary gland, although how PKC is implicated in the intermediate lobe remained unclear. In the present study, we reveal for the first time that PKCα is a molecule that acts, if not exclusively, as a downstream pathway of DG signal in the intermediate lobe, although the functional role of PKCα remains to be elucidated. One possibility is that PKCα mediates the inhibitory role of dopamine in the secretion of α-MSH from melanotrophs. The other possibility might be suggested by previous studies showing that the D2R is one of the substrates for PKC, which confer desensitization of this receptor (Namkung and Sibley 2004; Morris et al. 2007). If this is the case, longer activation of D2R would lead to its desensitization via the DG-PKC pathway, which could be regarded as a feedback mechanism of dopaminergic transmission. Further studies are needed to verify this hypothesis.
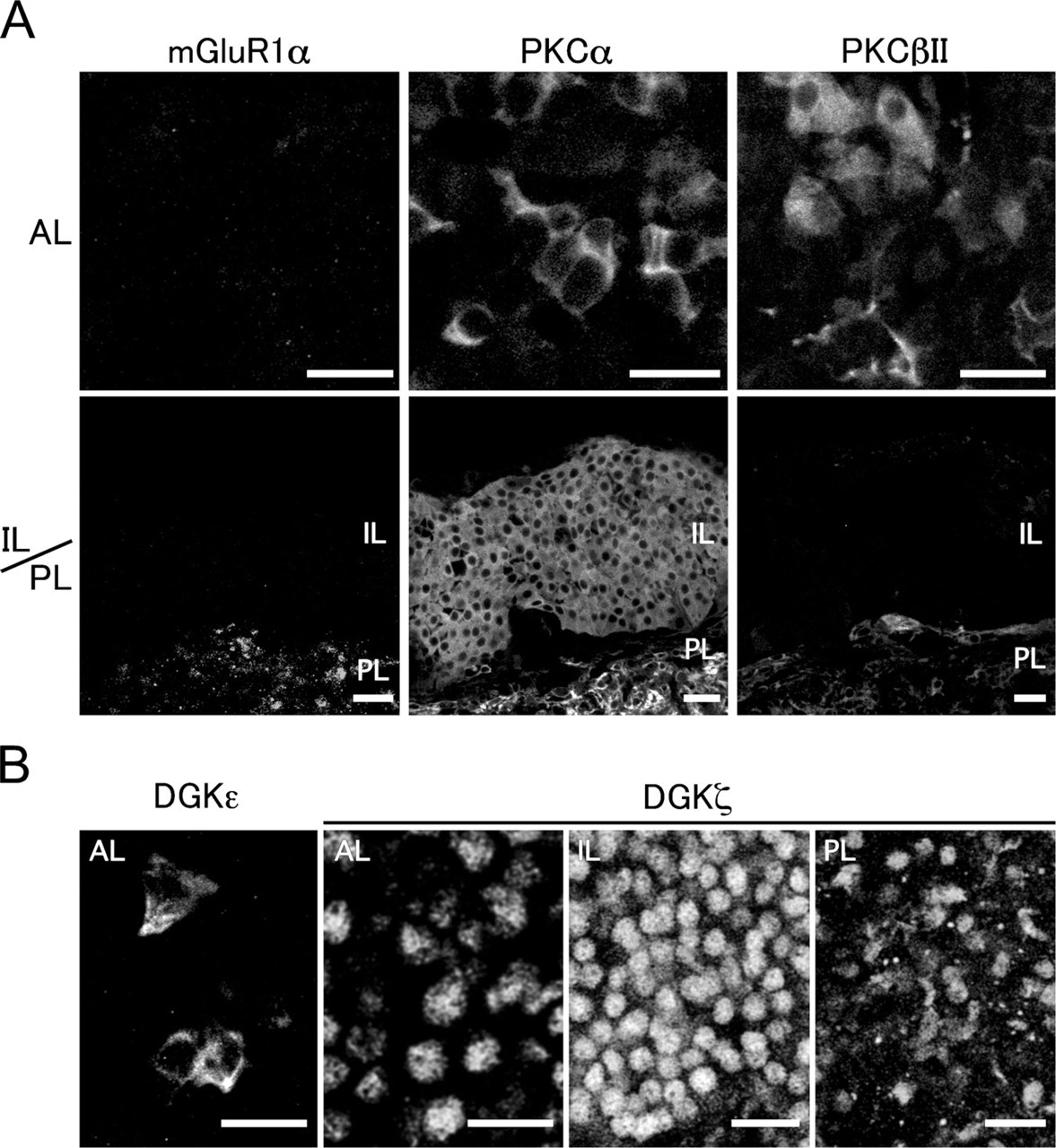
Expression and localization for mGluR1α, PKCα, PKCβII, DGKε, and DGKζ in the pituitary gland. (
Footnotes
Acknowledgements
This work was supported by a grant-in-aid and the Global Centers of Excellence Program from the Ministry of Education, Science, Culture, Sports, Science and Technology of Japan (YH, MW, KG) and from Taisho Pharmaceutical (KG).
