Abstract
Immunogold cytochemistry was applied to reveal the intracellular location of AMP-activated protein kinase (AMPK) subunits in liver tissue of normal rats fed ad libitum. AMPK α and β subunits were located both in the cytosol and in close association with rosettes of glycogen particles (α particles). To reveal their true in situ association with glycogen, particular tissue processing conditions that retain glycogen in the cells were required. These included fixation with a combination of glutaraldehyde and paraformaldehyde, followed by postfixation with osmium tetroxide and lead citrate and embedding in Epon. Processing by less-stringent fixation conditions and embedding in Lowicryl led to the extraction of the glycogen deposits, which in turn resulted in the absence of any labeling. This indicates that the loss of glycogen deposits leads to the loss of closely associated proteins. Labeling for the α1 and α2 subunits of AMPK was found to be about 2-fold greater over glycogen than over cytosol, whereas labeling for β1 was 8-fold higher over the glycogen particles than over the cytosol. Immunogold combined with morphometric analysis demonstrated that the β1 subunits are located at the periphery of the glycogen rosettes, consistent with a recent hypothesis developed via biochemical approaches.
G
AMP-activated protein kinase (AMPK) is a multi-substrate kinase that acts as a sensor of cellular energy status and is activated by a large variety of stresses that increase cellular AMP and decrease ATP levels (Hardie and Carling 1997; Hardie 2007). AMPK is also regulated by hormones such as leptin and adiponectin that control whole-body energy balance (Minokoshi et al. 2002; Yamauchi et al. 2002; Kahn et al. 2005). Once activated, AMPK switches off anabolic processes that consume ATP, such as lipid, glucose, and protein synthesis, while switching on catabolic processes that generate ATP, including glucose uptake, glycolysis, fatty acid oxidation, and mitochondrial biogenesis (Kahn et al. 2005; Hardie 2007). It achieves these effects both by direct phosphorylation of metabolic enzymes and via effects on transcription (Leclerc et al. 2002). In this manner, AMPK matches the supply of ATP to demand and maintains energy balances at both the cellular and whole-body levels.
AMPK exists as α-β-γ heterotrimers with multiple subunit isoforms encoded by seven genes giving rise to up to 12 possible enzyme combinations, each with varying tissue and subcellular locations (Stapleton et al. 1996, 1997; Thornton et al. 1998; Cheung et al. 2000; Kemp et al. 2003). AMPK requires the presence of all three subunits for activity (Dyck et al. 1996). The α sub-unit, which appears to be unstable unless coexpressed with the β and γ subunits (Dyck et al. 1996; Woods et al. 1996), contains a serine/threonine-specific kinase domain followed by an auto-inhibitory domain (Pang et al. 2007) and a C-terminal domain required for association with the β subunit (Iseli et al. 2005; Xiao et al. 2007). The α subunit is only active after phosphorylation at Thr-172 within the activation loop (Hawley et al. 1996) by upstream kinases, of which the most important is the tumor suppressor kinase LKB1 (Hawley et al. 2003; Woods et al. 2003). The C-terminal domain of the β subunit acts as a molecular scaffold that binds the α and γ subunits (Iseli et al. 2005; Xiao et al. 2007). The γ subunit contains four tandem repeats of structures known as CBS motifs that form the regulatory nucleotide-binding domains (Adams et al. 2004; Scott et al. 2004). These act in pairs to reversibly bind two molecules of AMP or ATP in an antagonistic manner, plus a third molecule of AMP that is tightly bound and non-exchangeable (Xiao et al. 2007). Binding of AMP (but not ATP) causes activation by inhibiting dephosphorylation of the α subunit at Thr-172 (Davies et al. 1995; Sanders et al. 2007), and by further allosterically activating the phosphorylated enzyme.
The β subunit in all eukaryotic species also contains a central conserved domain that is a member of the CBM20 family of carbohydrate-binding modules (Machovic and Janecek 2006) and causes the AMPK complex to bind to glycogen both in cell-free assays and in intact cells (Hudson et al. 2003; Polekhina et al. 2003). The structure of this domain has been determined in the presence of the cyclic oligosaccharide β-cyclodextrin, and several residues involved in binding the carbohydrate have been identified (Polekhina et al. 2005). Many interesting links between glycogen content and AMPK activity have been reported in physiological studies, although there are also some intriguing paradoxes. In cell-free assays, AMPK inactivates glycogen synthase via phosphorylation at site 2 (Carling and Hardie 1989), and this site becomes phosphorylated when muscle is perfused with the AMPK activator AICAR in wild-type but not in AMPK α2 knockout mice (Jorgensen et al. 2004). However, although AMPK is activated by muscle contraction (Winder and Hardie 1996), following contraction, glycogen synthase is paradoxically found to be dephosphorylated and activated (Aschenbach et al. 2001). High muscle glycogen content has been found to repress AMPK activation in response to AICAR in rat muscle (Wojtaszewski et al. 2002) or contraction in human muscle (Wojtaszewski et al. 2003), although in McArdle's disease, AMPK is found to be hyper-activated following exercise, despite the high muscle glycogen content (Nielsen et al. 2002). Some of these paradoxes may be explained by recent findings that the glycogen-binding domain (GBD) on the β subunit of AMPK is a regulatory domain that both allosterically inhibits the kinase and inhibits its phosphorylation and activation by upstream kinases (McBride et al. 2009). Although the GBD also binds unbranched oligosaccharides (Koay et al. 2007), marked inhibition only occurs with oligosaccharides containing an α1:6 linkage (McBride et al. 2009). These findings led to the hypothesis that AMPK binds to the surface of full-size glycogen particles but is not inhibited, leading to phosphorylation of glycogen synthase at site 2 and providing a feedback regulation of glycogen synthesis. Conversely, when glycogen is depleted (following exercise in muscle or fasting in liver) the glycogenbound AMPK would become inhibited by the exposed branch points, leading to dephosphorylation of glycogen synthase by glycogen-bound protein phosphatase-1 (Aschenbach et al. 2001), which is thus primed for rapid glycogen resynthesis. If this hypothesis is correct, AMPK acts as a glycogen sensor that ensures that glycogen stores are rapidly replenished when depleted (McBride et al. 2009).
To further examine this hypothesis, a key question concerns the proportion of AMPK in the cell that is bound to glycogen. Although the GBD clearly causes AMPK to bind to glycogen in intact cells, to date this has only been examined in cultured cells in which AMPK has been overexpressed and where glycogen deposits of abnormal size are formed. Indeed, it has been reported that following purification of α particles of glycogen from liver under gentle extraction conditions, no endogenous AMPK could be detected (Parker et al. 2007).
We have, herein, examined the association of AMPK α and β subunits with glycogen α rosettes in liver cells of normal fed rats, using in situ molecular morphology. This has allowed us to demonstrate that both types of subunits are associated with glycogen particles in situ. However, in keeping with the presence of the GBD domain on the β subunit, its association with glycogen is stronger and/or more stable. Furthermore, by using the quantitative morphometrical approach, we were also able to establish that the binding of the β subunit takes place at the edge of the glycogen rosette, a result consistent with the hypothesis that a significant proportion of AMPK may be bound to the non-reducing ends at the surface of glycogen particles (McBride et al. 2009).
Materials and Methods
Tissue Processing
One-month-old male Sprague Dawley rats weighing 100 g were obtained from Charles River Co. (St-Constant, QC, Canada). Animals were fed ad libitum and handled following the guidelines of the Canadian Council of Animal Care. Small pieces of liver tissue were sampled from the anesthetized animals (urethane, 1 g/kg body weight) and processed for electron microscopy. Tissue samples were fixed by immersion in 0.1 M phosphatebuffered (pH 7.2) 1% glutaraldehyde or in a fixative solution derived from Karnovsky (1965), as reported by Simionescu et al. (1972), composed of 5% glutaraldehyde plus 3% paraformaldehyde in 0.1 M phosphate buffer (pH 7.2) at 4C for 2 hr, followed by a 1 hr postfixation with a solution of 1% osmium tetroxide and a saturated solution of lead citrate in 0.1 M phosphate buffer. This fixation protocol was reported to be particularly useful for preserving polysaccharide particles in tissues with an enhanced contrast to all cellular structures (Simionescu et al. 1972). Some samples of the glutaraldehyde-fixed tissues were dehydrated in methanol and embedded in Lowicryl at −30C according to techniques particularly appropriate for immunocytochemistry (Bendayan 1995). All other tissues were dehydrated in graded ethanol and embedded in Epon (Luft 1961) according to standard procedures.
Tissue thin sections were cut, mounted on nickel grids, and counterstained with uranyl acetate and lead citrate or processed for immunocytochemistry.
Immunocytochemistry
For the immunocytochemical detection of AMPK subunits, the protein A-gold technique (Bendayan 1995) was applied. Tissue thin sections were first treated with a saturated solution of sodium metaperiodate for 10 min at room temperature, followed by 15 min incubation with 1% ovalbumin, and then transferred to one of the specific anti-AMPK subunit antibodies for an overnight incubation at 4C. Upon thorough washing with PBS, the grids were incubated with 1% ovalbumin and then for 30 min at room temperature with the protein A-gold complex (10-nm gold particles) prepared as described previously (Bendayan 1995). Upon thorough washing with PBS and rinsing with water, the grids were dried and counterstained with uranyl acetate and lead citrate. Examination was completed with a Philips 410 LS electron microscope.
Control experiments were carried out by omitting the antibody step; the labeling protocol was performed with the protein A-gold alone.
For morphometrical evaluation, a large number of images were recorded from several labeling experiments at the original magnifications of ×16,900. These were then enlarged (200%), printed, and processed for the morphometrical evaluation of the intensity of labeling. Cytoplasmic areas comprising large glycogen deposits and areas of cytosol rich in rough endoplasmic reticulum and devoid of glycogen deposits were measured by planimetry, and the number of gold particles located in these areas was counted using an image processing system (Videoplan 2, Carl Zeiss; Toronto, Canada). Results are expressed as number of gold particles per μm2 (mean values ± SEM). Statistical analyses were performed applying Student's t-test.
To further evaluate the exact topographical location of the immunogold particles in reference to the glycogen rosettes, we recorded images at higher magnification (×31,000), and upon printing them (200%), we measured the distance of each gold particle to the center of the rosette as well as the radius of the particular rosette. The ratio of these values reflects the location of the gold particle to the edge of the glycogen rosette. A distribution curve was drawn.
Antibodies
The anti-AMPK α1 and α2 were from Bethyl Laboratories, Inc. (Montgomery, TX) and were applied at a 1:10 dilution in PBS. Their specificity was previously well demonstrated (Zang et al. 2004; Doi et al. 2005; Cammisotto et al. 2008). By Western blot, they generate the characteristic doublet bands at the appropriate molecular mass (64 kDa) (Zang et al. 2004; Fraser et al. 2005; Rubin et al. 2005; Cammisotto et al. 2008). Three anti-AMPK β1 antibodies were used: one was from Santa Cruz Biotechnologies, Inc. (Santa Cruz, CA); the second was from Epitomics, Inc. (Burlingame, CA); and the third was raised using a synthetic peptide. This third antibody is a rabbit polyclonal against the N-myristoylated synthetic peptide, AMPK β1 (2-24) N-myristoyl-Gly-2, Cys-24 (myr-GNTSSERAALER-QAGHKTPRRDC-amide) coupled to keyhole limpet hemocyanin (Sigma, H-2133; Oakville, ON, Canada) via the C-terminal cysteine residue of the peptide using the heterobifunctional reagent N-succinimidyl-3-(2-pyridyldithio) propionate (Pharmacia; Uppsala, Sweden). Anti-AMPK β1 antibody was purified by peptide affinity chromatography using the immunizing peptide coupled to Sulfo Link coupling gel (Pierce P/N 20,402; Pierce Biotechnology, Inc., Rockford, IL). All antibodies were used at 1:10 dilution in PBS.
Specificity of the three anti-β1 antibodies was assessed by Western blots using liver tissue extracts from normal and AMPK β1 knockout mice as well as from normal rat. Details on the AMPK β1 knockout mice have been published previously (Scott et al. 2008). The three antibodies yield a single band at the mobility expected from the predicted molecular mass, which was absent in extracts from knockout mice (Figure 1). These immunochemical tests support the high specificity of the probes used. The study focused on the AMPK β1 subunit because it is highly expressed in liver with low expression in skeletal muscle, whereas the opposite pattern is observed for the β2 isoform (Thornton et al. 1998).
Results
Processing tissues for electron microscopy, particularly without osmium postfixation, has the tendency to extract most of the glycogen, leaving empty spaces within the cell cytoplasm. Liver cells prepared under these conditions showed large empty areas corresponding to the location of the glycogen deposits (Figure 2). In contrast, when we processed the same tissue with a protocol that includes postfixation with osmium tetroxide and lead citrate, large deposits of electron-dense particles characteristic of glycogen were found in the cytosol. The deposits are formed by rosettes of dense conglomerated particles (Figures 3 and 4).
For the immunocytochemistry of AMPK subunits, the two fixation protocols yielded very different results. Immunogold with any of the AMPK subunit antibodies on glutaraldehyde-fixed, Lowicryl-embedded material resulted in a labeling by gold particles only over the cytosol (Figure 2). Clear areas presumed to have been filled with glycogen deposits prior to fixation were completely devoid of labeling. Indeed, extraction of the glycogen deposits appears to have led to the loss of the AMPK, inasmuch as little immunolabeling was found for any of the subunits tested (Figure 2). The remaining AMPK subunits revealed by the gold particles were restricted to areas of the cytosol associated with the rough endoplasmic reticulum (Figure 2). On the other hand, immunocytochemistry performed on sections of tissues processed using postfixation in osmium tetroxide and lead citrate and embedded in Epon, which retains the glycogen (Figures 3 and 4), yielded labelings of the AMPK subunits at the level of the cytosol-rough endoplasmic reticulum, but more significantly, at the level of the glycogen rosettes (Figures 3 and 4). Labeling for the three AMPK subunits (α1, α2, β1) was found closely associated with the glycogen particles. Other cellular structures such as mitochondria and lysosomes exhibited very few gold particles (Figures 3 and 4). The three antibodies directed against the AMPK β1 subunit yielded identical qualitative results, demonstrating strong association with the glycogen rosettes.
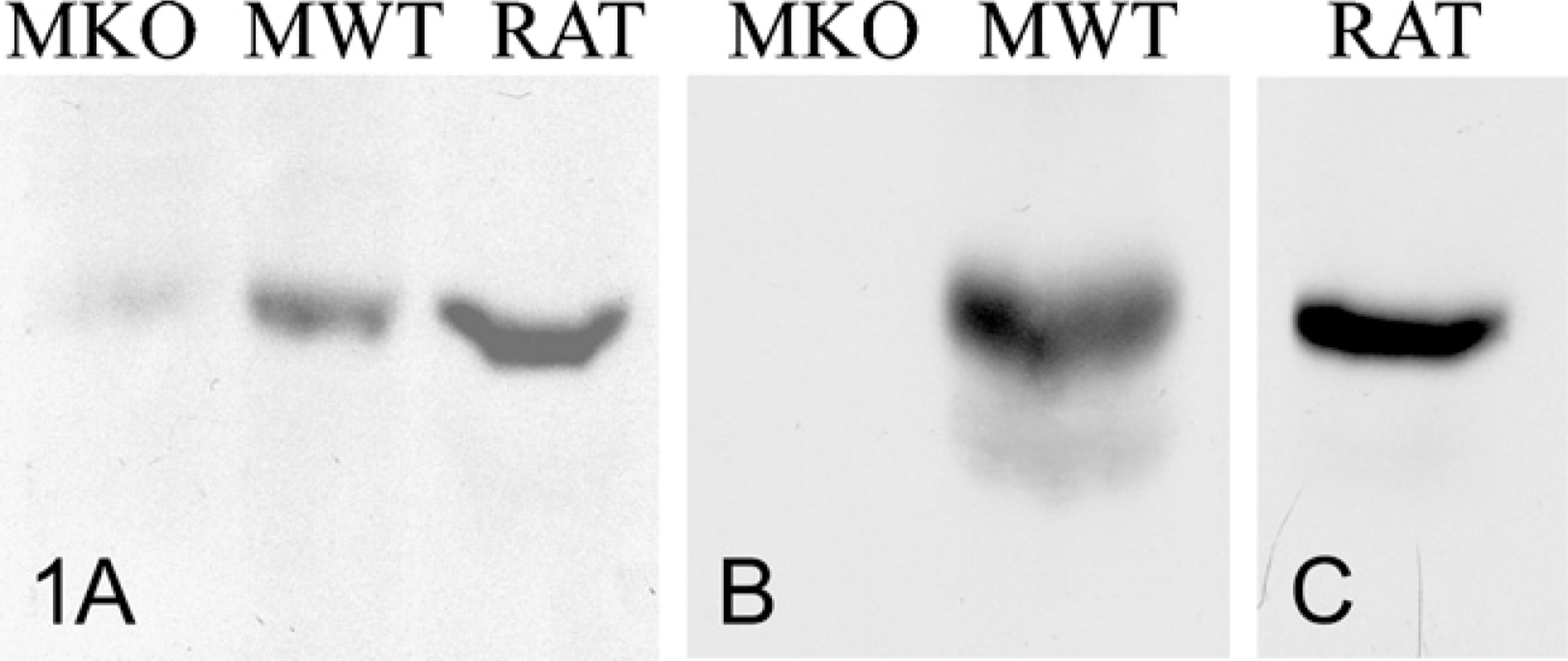
Results from Western blot analyses using two of the antibodies against the AMP-activated protein kinase (AMPK) β1 subunit. The antibody from Santa Cruz (
In all the control experiments, we observed major reductions in labeling, with few gold particles scattered throughout the cell cytoplasm (results not shown), supporting the high specificity of the labelings.
Quantitative evaluations of the intensity of labeling in areas of cytosol and glycogen revealed significant differences between the α and β AMPK subunits (Table 1). Labelings for AMPK α1 and α2 were approximately 2-fold greater over glycogen than over the cytosol (Table 1). On the other hand, the AMPK β1 subunit showed a preferential association with glycogen, with a labeling eight times as much over the glycogen particles than over the cytosol (Table 1). Quantitation of the labeling obtained under the control experiments confirms the high specificity of these labelings, with very low background values (Table 1).
To assess the location of the AMPK β1 subunit within the glycogen rosette, we used highly magnified images of immunogold labeling (Figure 5) and measured the distance of the gold particle from the center of the rosette as well as the radius of the rosette itself. The ratio of these values gives a good indication, with statistical support, of the location of the gold particles within the glycogen rosette. The distribution curve is shown in Figure 6. The mean ratio was found to be 0.96 ± 0.02 (value of 1.00 being the exact edge of the rosette), indicating that the gold particles are located at the surface of the glycogen rosette. To better interpret this distribution curve and the presence of some gold particles slightly detached from the glycogen, it should be noted that the gold particles have a diameter of 10 nm and each is coated with protein A molecules, which in turn, are bound to IgG molecules present on the antigenic site at the surface of the tissue section. This superposition of proteins adds significantly to the final size of the probe (Bendayan 1995) and can explain why some gold particles appear to be located a short distance away from the glycogen rosette.
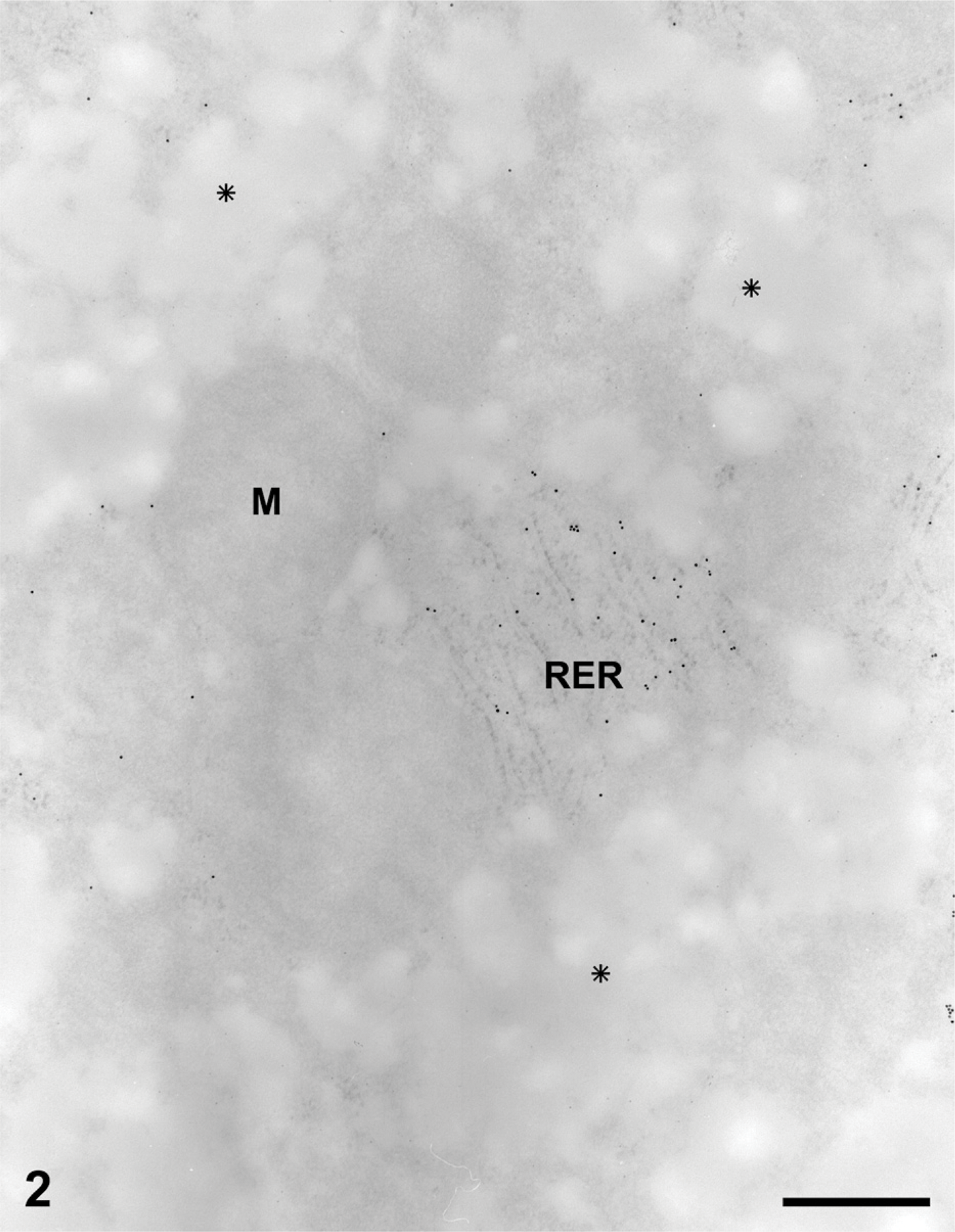
Liver tissue from normal rats fed at libitum. Protein A-gold immuno-cytochemistry for the AMPK subunits. Tissue sample fixed with glutaraldehyde and embedded in Lowicryl. The glycogen deposits have been extracted, leaving empty spaces within the cytoplasm (asterisks). Other structures such as mitochondria (
Discussion
It has been well established that for successful immunocytochemistry, retention of tissue and cell protein antigenicity, as well as ultrastructural preservation, is crucial. Successful immunocytochemistry implies assigning particular and specific cellular locations to antigens with the highest resolution possible. Such resolution can only be achieved on tissues and cells displaying excellent ultrastructural preservation, which is solely obtained by efficient fixation protocols. However, fixation of tissues and retention of antigenicity are two opposing criteria (Bendayan 1995, 2001). Indeed, strong fixatives have the tendency to alter protein structure by cross-linking amino acids, leading to significant alteration of antigenic epitopes. Similar and additional alterations in protein antigenicity can further take place during dehydration of the tissues in solvents and embedding in resins. Various fixation and embedding protocols for successful immunocytochemistry have been introduced over the years (Bendayan 1995). A general consensus on the best approach leads to a compromise between retention of sufficient amounts of antigenicity to allow immunolabeling, and satisfactory ultrastructural preservation to permit adequate identification of cellular organelles (Bendayan 1995, 2001).
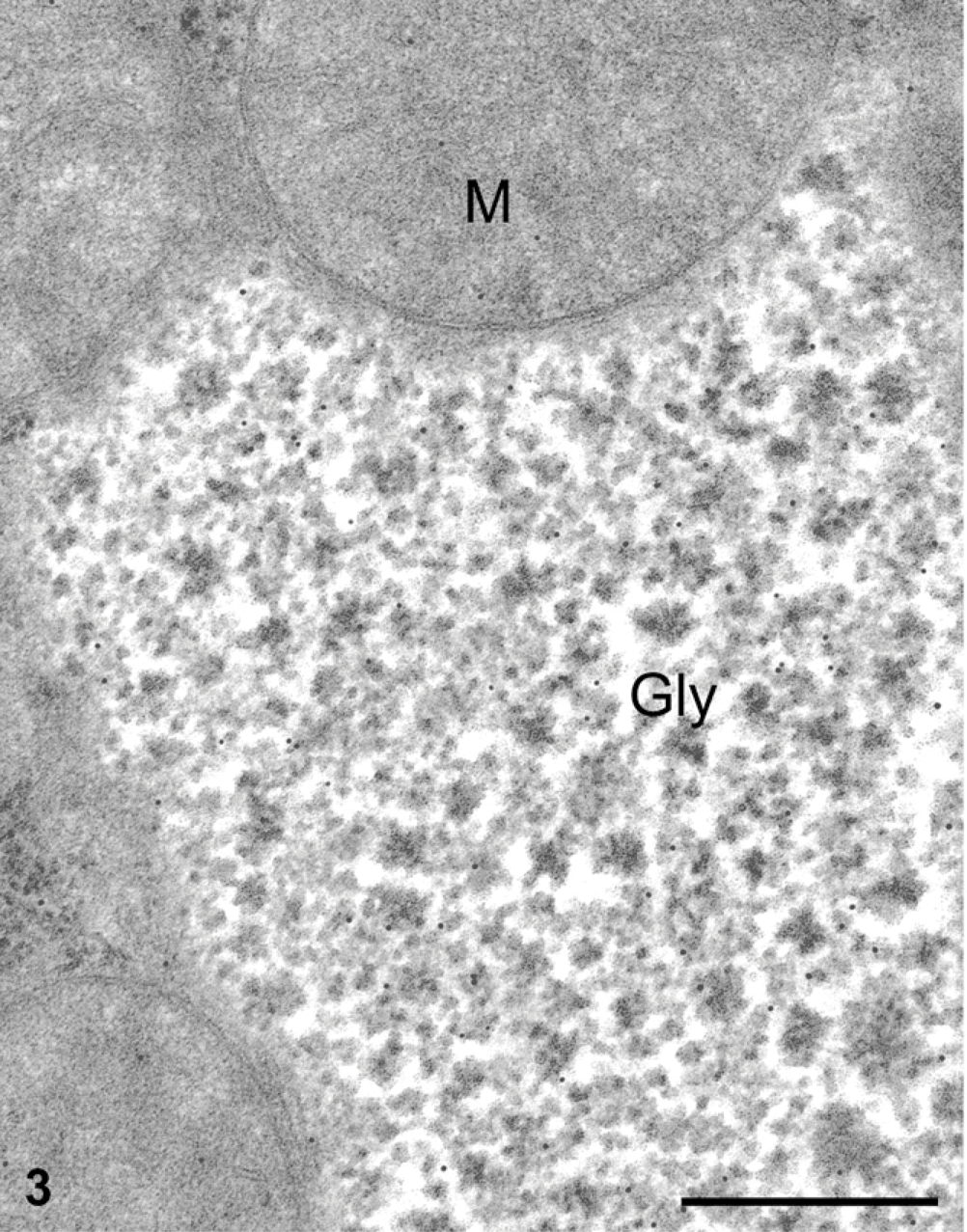
Liver tissue from normal rats fed at libitum. Protein A-gold immunocytochemistry for the AMPK subunits. Liver sample fixed with the paraformaldehyde-glutaraldehyde followed by postfixation with the osmium tetroxide-lead citrate combination and embedded in Epon. The glycogen deposits (Gly) are retained as electron-dense particles arranged in rosettes. They are labeled for the AMPK subunit α1. The gold particles are preferentially associated with glycogen deposits. Mitochondria (
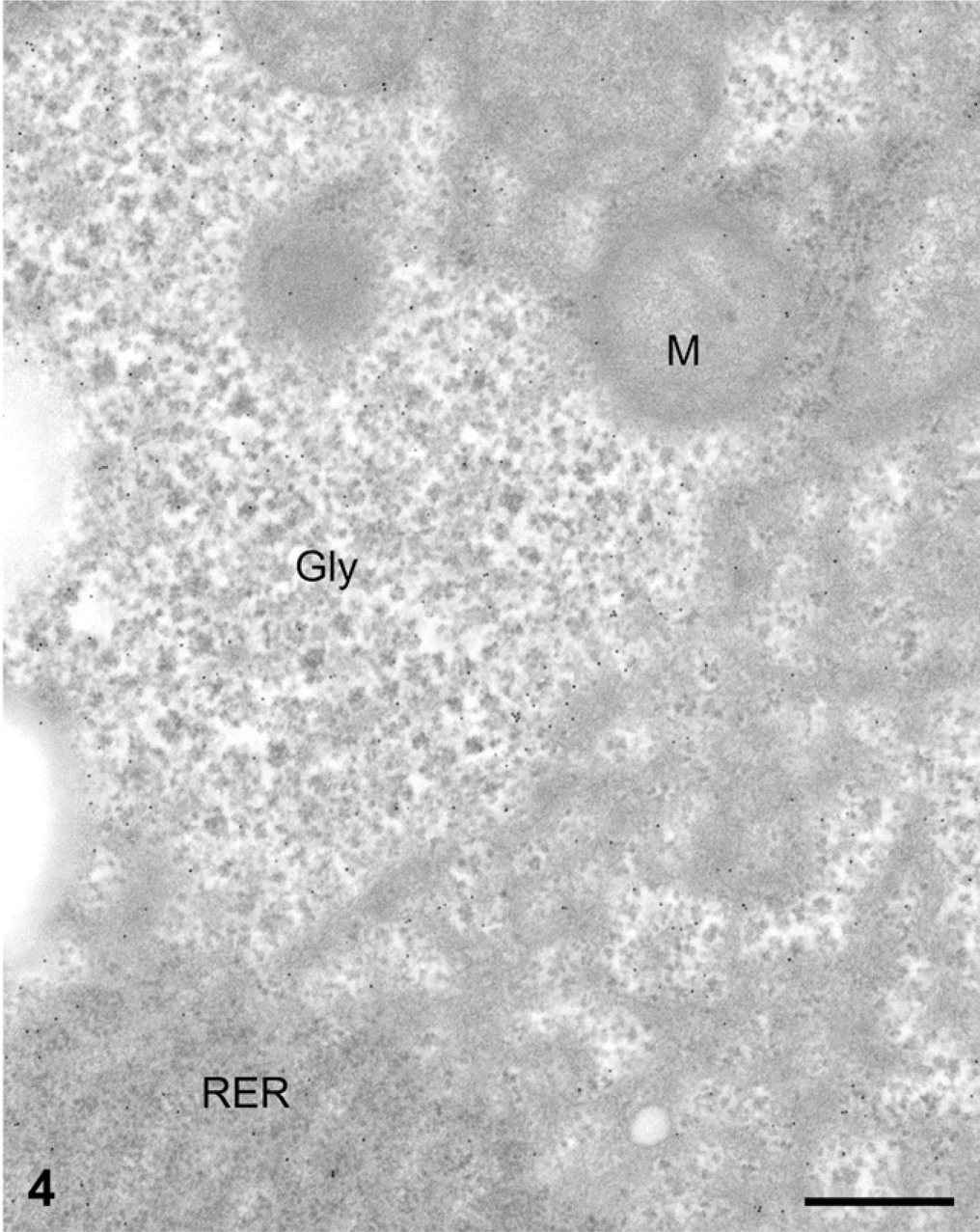
Liver tissue from normal rats fed at libitum. Protein A-gold immunocytochemistry for the AMPK subunits. Liver sample fixed with the paraformaldehyde-glutaraldehyde followed by postfixation with the osmium tetroxide-lead citrate combination and embedded in Epon. The glycogen deposits (Gly) are retained as electron-dense particles arranged in rosettes. They are labeled for the AMPK subunit β1. The gold particles are preferentially associated with glycogen deposits. Some particles are also present over the cytosol associated with the rough endoplasmic reticulum (RER) whereas mitochondria (
Glycogen, probably because of its polysaccharide nature, is one of the cellular components that is most difficult to retain in tissue sections. Strong fixation protocols are required to immobilize glycogen, and this is most likely to be achieved by fixing proteins linked to it or present in its close vicinity (Simionescu and Palade 1971). We have shown that weak fixation protocols extract the glycogen deposits from the cell cytoplasm. We have further found, in the present study, that when glycogen is extracted from the tissue, the immunolabelings for AMPK subunits, as well as that for other glycogen-associated proteins such as glycogen synthase (unpublished results), are also depleted. This indicates that extraction of glycogen, due to poor fixation, also removes the associated proteins bound to it. Thus, any morpho-cytochemical study of proteins associated with glycogen metabolism and therefore associated with glycogen storage requires retention of the glycogen within the cells. When glycogen is retained in the tissue, so are the proteins that are bound to it, and our present results with the AMPK subunits constitute an excellent demonstration of this.
Density of immunolabelings for AMP kinase α1, α2, and β1 in normal rat liver cells

Significantly different from the corresponding value over the cytosol; p<0.001.
Gold particles/μm2; mean values ± SEM. Values within parentheses correspond to the total cellular compartment area evaluated.
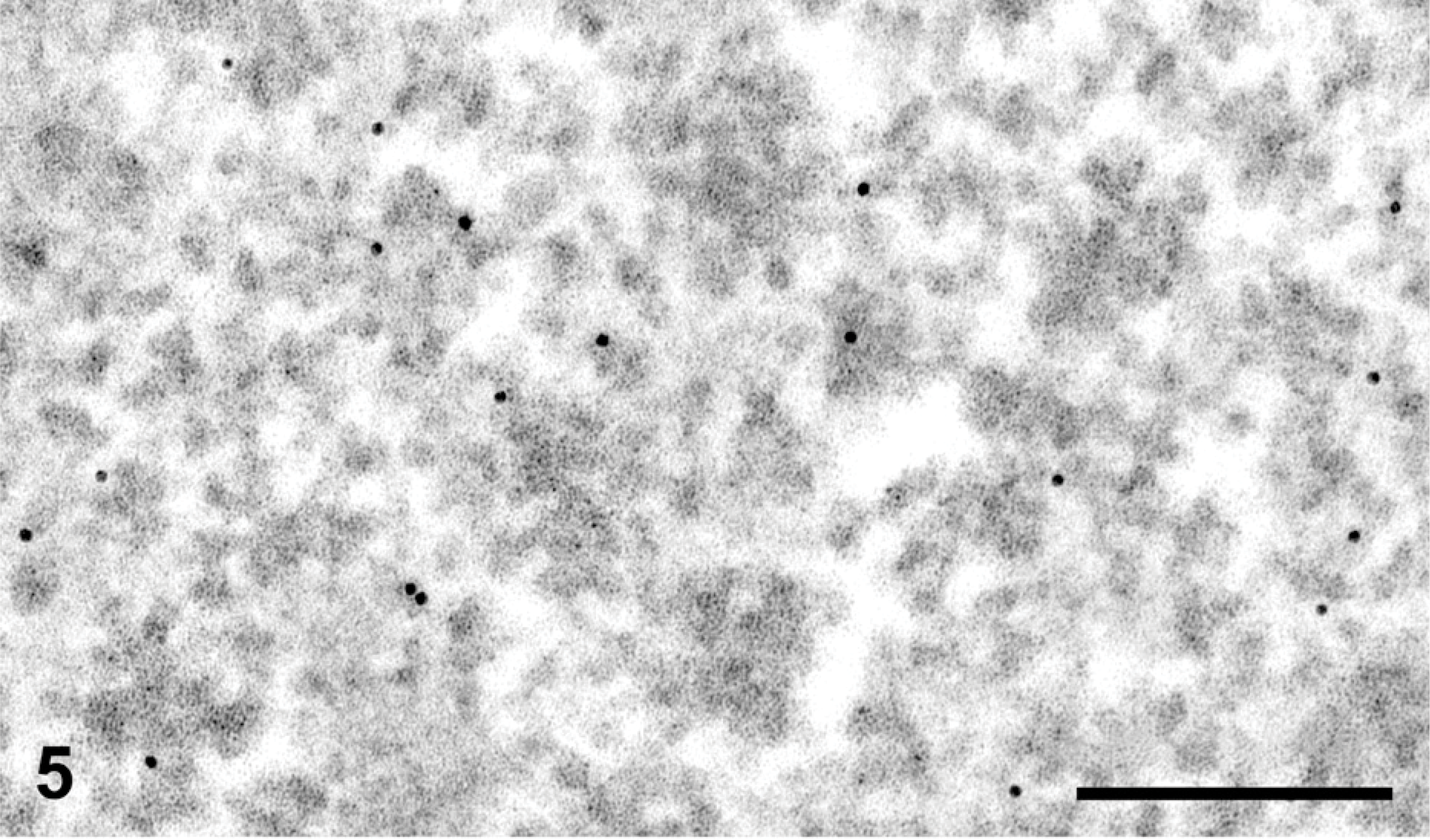
Liver tissue from normal rat fed at libitum. Protein A-gold immunocytochemistry for AMPK-β1 subunit. At very high magnification, we can assess the position of the gold particles (AMPK β1) over the glycogen rosettes. The gold particles are mostly at a peripheral location on the glycogen rosettes. Bar = 0.1 μm.
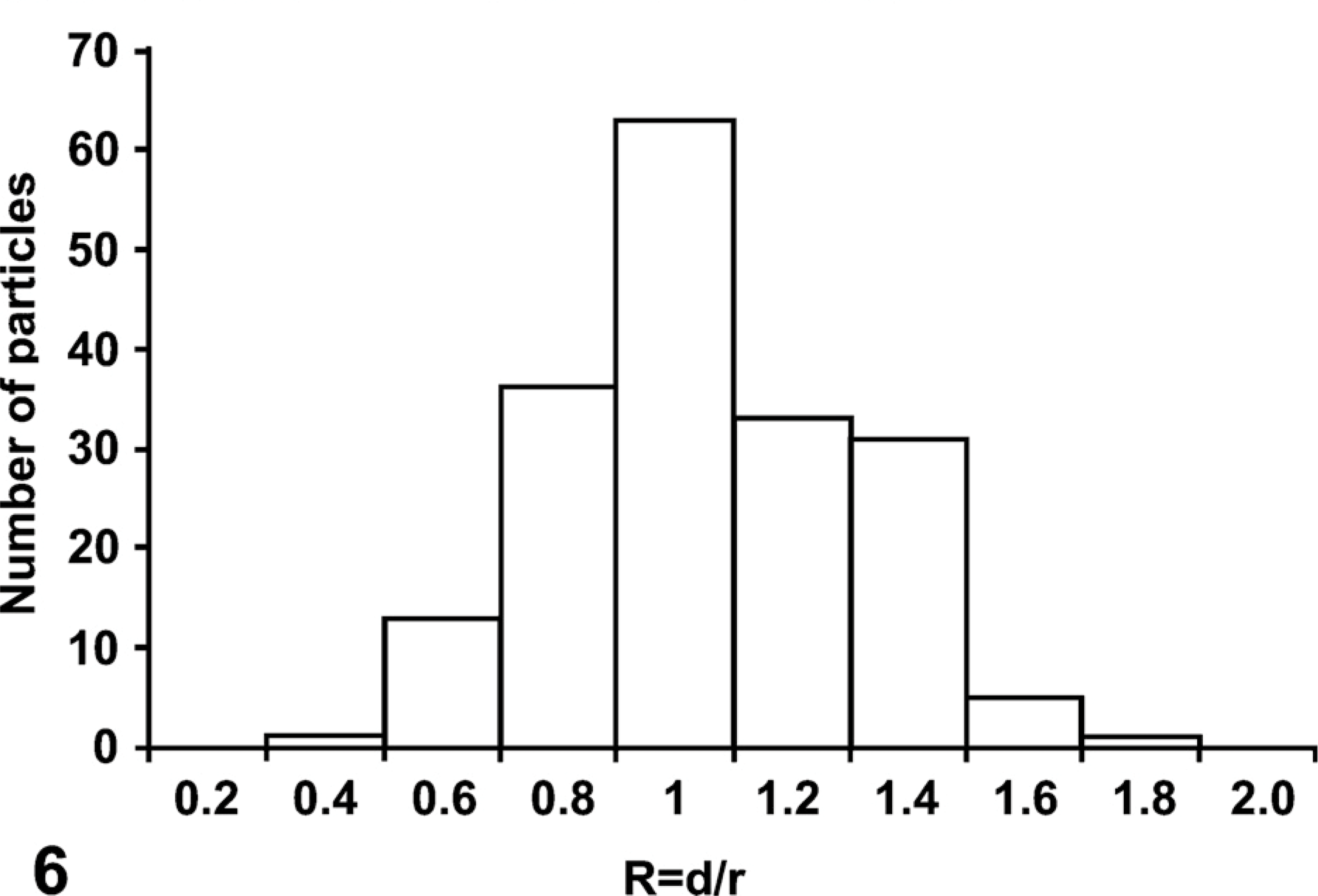
Liver tissue from normal rat fed at libitum. Protein A-gold immunocytochemistry for AMPK-β1 subunit. Histogram revealing the distribution of the gold particles (AMPK β1) over the glycogen rosettes. The evaluation was carried out in the following way: the distance (d) between the gold particle and the center of the corresponding rosette was measured and divided by the radius (r) of the same glycogen rosette. The ratio (
Although it has previously been shown that the presence of the GBD on the AMPK β subunits causes the complex to associate with glycogen particles, this was only achieved by overexpression of the β1-GFP (Polekhina et al. 2003) or various combinations of GFP α1 or α2 with β1/β2 and γ1/γ2/γ3 (Hudson et al. 2003) in cultured cells. In these studies, glycogen particles were visualized by fluorescence microscopy using costaining with antibodies against glycogen or glycogen synthase (Hudson et al. 2003), or by coexpression with RFP-labeled glycogen phosphorylase (Polekhina et al. 2003). However, these are not physiological situations, because normal glycogen particles of diameter up to 200 nm are too small to be resolved by light microscopy. It also appears that overexpression of β1 or of AMPK complexes may lead to aggregation of glycogen particles into clusters of abnormal sizes (Polekhina et al. 2003). In this report, we show for the first time that endogenous rat liver AMPK, and particularly the β1 subunit, is associated with the α rosettes of glycogen of normal size in liver from normal fed rats.
By combining optimal ultrastructural preservation of liver cells and protein A-gold immunocytochemistry, we were able to demonstrate that the AMPK β1 subunit is topographically associated with the glycogen molecules and that among the subunits, it has a stronger association with glycogen particles than do the the α1 or α2 subunits. Although direct comparative evaluations of intensities of labeling among antigens cannot be drawn because antigen-antibody affinity varies among antibodies (Bendayan 1995), our quantitative evaluations of the labelings revealed that the proportion of the α1 and α2 subunits associated with glycogen particles is broadly similar, whereas that of β1-associated is much higher. This is consistent with previous results indicating that the binding of the AMPK complex to glycogen is mediated by the GBD on the β subunit (Hudson et al. 2003; Polekhina et al. 2003). Our results may also indicate that there is a glycogen-bound pool of β1 subunit in rat liver not bound to any α subunit, although further work will be required to confirm this.
Our stereological study has further demonstrated that the binding of AMPK to glycogen rosettes takes place at the periphery of the glycogen molecule. Previous work has suggested that glycogen binding is mediated by the carbohydrate-binding module on the β subunits (Hudson et al. 2003; Polekhina et al. 2003). Crystallographic analysis of a partial complex of the budding yeast ortholog of AMPK (the SNF1 complex) has suggested that the GBD is located on the periphery with the carbohydrate-binding site on the outside of the complex (Amodeo et al. 2007). The results shown in Figures 5 and 6 are consistent with the proposal (McBride et al. 2009) that the AMPK complex can only bind to the surface of a full-size glycogen particle, and may be unable to gain access to internal branch points due to the tight packing of the outer layer of non-reducing ends.
In conclusion, in the present study, we have demonstrated that weak fixation protocols used for electron microscopy tissue preparation lead to extraction of glycogen from the cells. This extraction is followed by the loss of proteins bound to glycogen. On the other hand, preservation of glycogen in cells also retains associated proteins and allows revealing of glycogen-associated proteins such as the AMPK subunits. Our results suggest that a significant proportion of AMPK in normal rat liver cells is bound to glycogen. Our finding that AMPK complexes or β1 subunits are associated with the periphery of α rosettes of glycogen is also consistent with the recent proposal that AMPK binds to the surface of glycogen via the GBD on the β subunit, allowing the kinase to act as a sensor of glycogen structure (McBride et al. 2009).
Footnotes
Acknowledgements
This work was supported by grants from the Canadian Institutes of Health Research (MB, MP), Diabete Quebec (MB) and the National Institutes of Health (MB, NR, MP).
The authors acknowledge the technical assistance of D. Gingras.
