Abstract
To test the hypothesis that a perturbation of endoplasmic reticulum (ER) function is involved in the pathogenesis of osteoarthritis (OA), articular cartilage was isolated from non-OA patients secondary to resection of osteo- or chondrosarcomas. Intra-joint samples of minimal and advanced osteoarthritic cartilage were isolated from patients undergoing total knee arthroplasty and scored for disease severity. Glucose-regulated protein-78 (grp78) and bcl-2-associated athanogene-1 (bag-1) were detected via immunofluorescence as markers of non-homeostatic ER function. Additionally, the expression of type VI collagen and its integrin receptor, NG2, was determined to examine cartilage matrix health and turnover. There was an upregulation of grp78 in advanced OA, and variable expression in minimal OA. Non-OA cartilage was consistently grp78 negative. The downstream regulator bag-1 was also upregulated in OA compared with normal cartilage. Collagen VI was mainly cell-associated in non-OA cartilage, with a more widespread distribution observed in OA cartilage along with increased intracellular staining intensity. The collagen VI integral membrane proteoglycan receptor NG2 was downregulated in advanced OA compared with its patient-matched minimally involved cartilage sample. These results suggest that chondrocytes exhibit ER stress during OA, in association with upregulation of a large secreted molecule, type VI collagen.
O
Chondrocytes are subjected to a variety of stressors, such as oxidative, osmotic, and biomechanical stress (Lee et al. 2002; Pfander and Gelse 2007; Wuertz et al. 2007), and mechanisms must exist to maintain homeostasis in spite of these stressors. It has recently been reported in our lab that chondrocytes also undergo ER stress in vitro in response to physiological and pharmacological agents (Yang et al. 2005). To probe the function of ER activity in OA, we examined the expression and distribution of two proteins known to be involved in ER stress: glucose-regulated protein-78 (grp78) and bcl-2-associated athanogene-1 (bag-1). Grp78 [also known as immunoglobulin-binding protein (BiP)] is a member of the heat shock protein-70 (hsp70) family, which performs numerous functions within the ER, including translocation of newly synthesized proteins across the ER membrane, maintaining proteins in a form that prevents improper aggregation, and allowing proper protein folding and oligerimization, along with the targeting of misfolded proteins for proteasomal degradation (Hendershot 2004; Li and Lee 2006; Lai et al. 2007). Grp78 is a target gene of the unfolded protein response (UPR) signaling pathway, and its upregulation makes it an accepted marker for ER stress (Lee 2001; Banhegyi et al. 2007).
Bag-1 has recently been shown to be a downstream regulator of ER stress in chondrocytes. Our lab has demonstrated in vitro that overexpression of bag-1 protects chondrocytes from ER stress-induced downregulation of collagen type II and apoptosis, and that exogenously induced ER stress causes downregulation of bag-1 (Yang et al. 2007). From these data, we hypothesized that if chondrocytes do, in fact, undergo ER stress during the pathogenesis of OA, an upregulation of grp78 will be observed, which may or may not be associated with a downregulation of bag-1.
To qualitatively assess the health and turnover of the ECM in non-OA and OA cartilage, we examined the expression of type VI collagen and its integral membrane proteoglycan receptor, NG2. A number of ECM molecules have been reported to be localized to the pericellular matrix, including the network-forming collagen type VI, which is a vital component of both ECM signaling and cell-matrix structural stability (Poole 1997; Alexopoulos et al. 2005; Guilak et al. 2006). Collagen VI has been reported to increase expression in osteoarthritic cartilage, and to lose its peri-cellular localization (Hambach et al. 1998; Pullig et al. 1999; Söder et al. 2002). As with other secreted collagens, type VI is modified and assembled within the ER (Sipila et al. 2007). Here, we reexamine the expression of collagen VI and NG2 (Burg et al. 1996) in minimal and advanced osteoarthritic cartilage, speculating that upregulation of a large secreted molecule such as a collagen, in coordination with the many other alterations in gene expression and ECM secretion that have been shown to occur in OA, might result in ER stress.
Within an osteoarthritic knee joint, the separate medial and lateral joint compartments experience dissimilar loading forces that play a role in the rate of advancement of disease, even as both compartments are presumably undergoing disease processes at the cellular and molecular level (Shelburne et al. 2005; Sandell 2007). Using our previously published model system utilizing intra-joint comparisons to represent minimal and advanced stages in the morphological progression of OA (Yagi et al. 2005), we examined specific cellular and matrix changes associated with human OA. Our results demonstrate an upregulation of grp78 associated with human OA, suggesting that chondrocytes are experiencing altered ER function. Further, increased expression of bag-1 suggests an overall perturbation of chondrocyte homeostasis. These alterations in cellular markers are associated with an upregulation of collagen VI, and downregulation of its integral membrane proteoglycan receptor, NG2.
Materials and Methods
Obtaining Cartilage Samples
Cartilage samples were isolated from medial and lateral femoral condyles from patients undergoing total knee arthroplasty within 3 hr of surgery. Initial characterization was performed by macroscopic observation, and tissue was defined as minimal or advanced based on gross morphological characteristics. Full-thickness cartilage samples of advanced OA were obtained within 1 cm of a macroscopic defect on the medial condyle, and minimal OA samples were obtained from areas with no obvious surface defect from the lateral femoral condyle (patient n = 18; average age, 63.6 years). In most cases, only one minimally involved and one advanced OA sample were taken from each patient. However, if selection criteria granted more than one minimal or advanced sample to be retrieved, then multiple samples were taken. Healthy, non-OA knee articular cartilage from patients diagnosed with chondro- and osteosarcomas (n = 3), along with hip (n = 4) and ankle (n = 2) cartilage, were obtained as examples of normal, homeostatic cartilage (average age 42.4 years). There were no significant differences in immunostaining between these three locations, and non-OA figures show the most representative sample without taking location into consideration. Tissue samples were fixed in 10% neutral buffered formalin for 24 hr for histological analysis. Age range of selected OA patients was 51–83 years, and the study was approved by the Northeastern Ohio Universities College of Medicine (NEOUCOM) institutional review board.
Scoring of Cartilage Samples
The cartilage samples were scored as: fibrillation (1, even surface; 2, uneven surface; 3, fibrillation on only superficial zone; 4, fissures in mid-zone; 5, fissures in deep zone), chondrocyte cloning (1, no clusters; 2, chondrocyte clusters in superficial zone, of no more than four cells; 3, chondrocyte clusters beyond superficial zone, of no more than four cells; 4, chondrocyte clusters of more than four cells), and matrix depletion (ratio of non-metachromatic area to entire area).
A derived total score was calculated as the fibrillation score + cloning score + (matrix depletion score/10), with a highest possible score of 19. Scoring criteria were based on Yagi et al. (2005). Statistics were calculated using the Mann-Whitney independent samples test, with p value less than 0.05 considered significant.
Immunofluorescence
Tissue samples were fixed in 10% neutral buffered formalin (Fisher Scientific; Fair Lawn, NJ), processed, and paraffin embedded according to standard laboratory procedures. Paraffin blocks were sectioned at 6 μm and stained with 0.1% thionin for basic histological analysis and scoring. Primary antibodies for immunofluorescence were as follows: all reactive against human epitopes and applied at 1:100 concentration; grp78 goat polyclonal (Santa Cruz Biotechnology; Santa Cruz, CA, sc-1050); bag-1 rabbit polyclonal (Santa Cruz Biotechnology; sc-939); collagen VI rabbit polyclonal (Fitzgerald; Concord, MA, RDI-600401108); and NG2 rabbit polyclonal, a generous gift from Dr. William Stallcup (Atlanta Veterans Affairs Medical Center, The Burnham Institute for Medical Research, Cancer Research Center; La Jolla, CA) via Dr. Kathleen Doane (NEOUCOM). Secondary antibodies were purchased from Molecular Probes (Invitrogen; Carlsbad, CA) and used at 1:200 concentration: goat anti-rabbit Alexa Fluor 568 (A11011), and rabbit anti-goat Alexa Fluor 488 (A11078).
In general, unmasking was achieved using chondroitinase ABC (Sigma-Aldrich; St. Louis, MO) for 20 min at 37C. Primary antibodies were incubated overnight at 37C, and secondary antibodies were applied for 1 hr at room temperature. Slides were counterstained with 4', 6-diamidino-2-phenylindole (DAPI) (Vectashield Hard Set Mounting Medium with DAPI; Vector Laboratories, Burlingame, CA) and coverslipped. Control slides were also prepared for each protein examined, performing every step of the procedure except the addition of primary antibody, for which blocking serum remained overnight. Images were obtained using Bioquant Osteo II (Bioquant Image Analysis; Nashville, TN), and background exposure was normalized to the associated minus-primary control slide, with any additional staining considered positive.
Results
Characterization of Osteoarthritic Cartilage
Figure 1 illustrates characteristics of the different types of cartilage samples that were analyzed in this study, including degrees of metachromasia, surface fibrillation, and chondrocyte cloning. Cartilage sections were stained with thionin to demonstrate metachromasia, which provides an indirect estimation of proteoglycan content. A progressive loss of metachromasia is observed between non-OA, minimally involved, and advanced OA cartilage samples (Figures 1A-1C). Additionally, normal cartilage retains a smooth, intact articular surface, whereas deep surface fibrillations are found in advanced OA (Figures 1A and 1C). Minimal-OA cartilage often displays minor fibrillations, predicting further damage from loading and changes in ECM composition that will lead to advanced disease (Figure 1B). Although chondrocytes are distributed uniformly throughout the depth and breadth of the cartilage matrix in normal tissue and minimal OA (Figures 1A and 1B), large isogenous groups of four or more cells are found in the superficial to mid zones of advanced OA cartilage (Figure 1C, arrows).
The cartilage samples were scored according to a previously described method (Yagi et al. 2005). A derived total score was calculated as the fibrillation score + cloning score + (matrix depletion score/10). Patient-matched minimal- and advanced-OA total scores are graphically visualized in Figure 2. There is a statistically significant difference in total scores between minimal and advanced cartilage (p<0.001), although some patients show similar total scores.
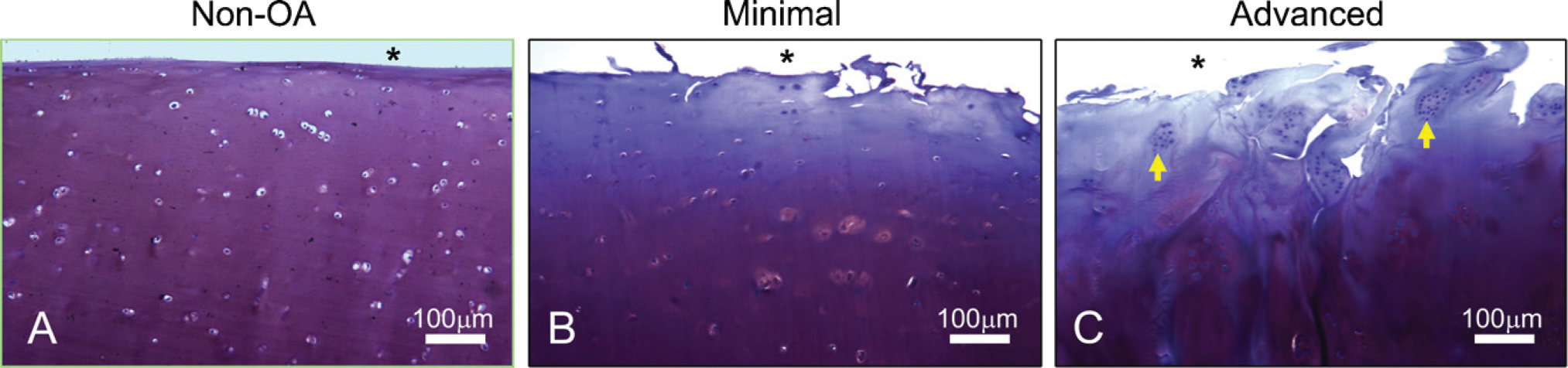
Non-osteoarthritis (OA) (
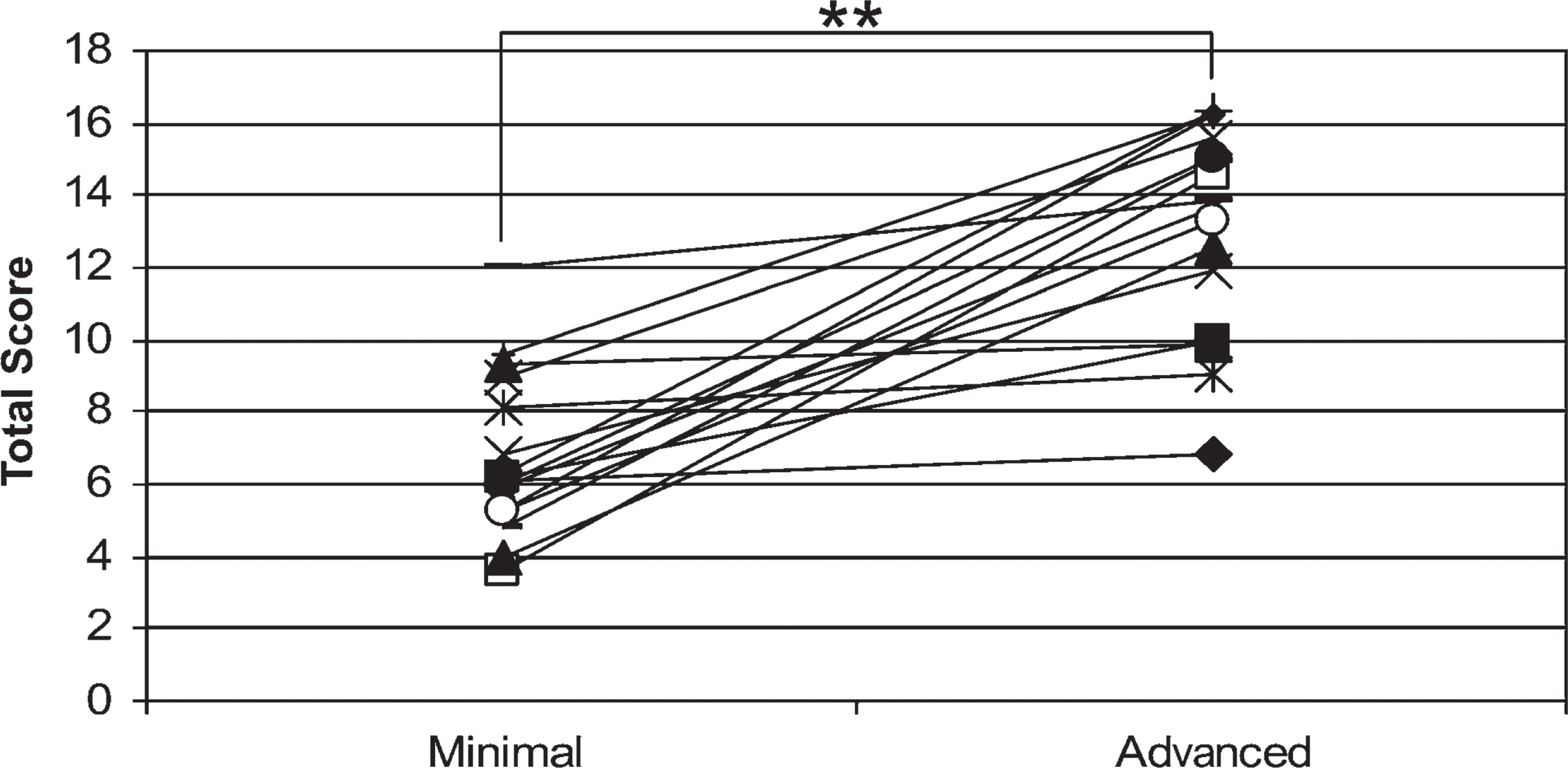
Total scores of patient-matched minimal and advanced osteoarthritic cartilage, calculated as the fibrillation score + cloning score + (matrix depletion score/10). Mann-Whitney test returns p value < 0.001∗∗.
Expression Patterns of ER Stress-associated Proteins
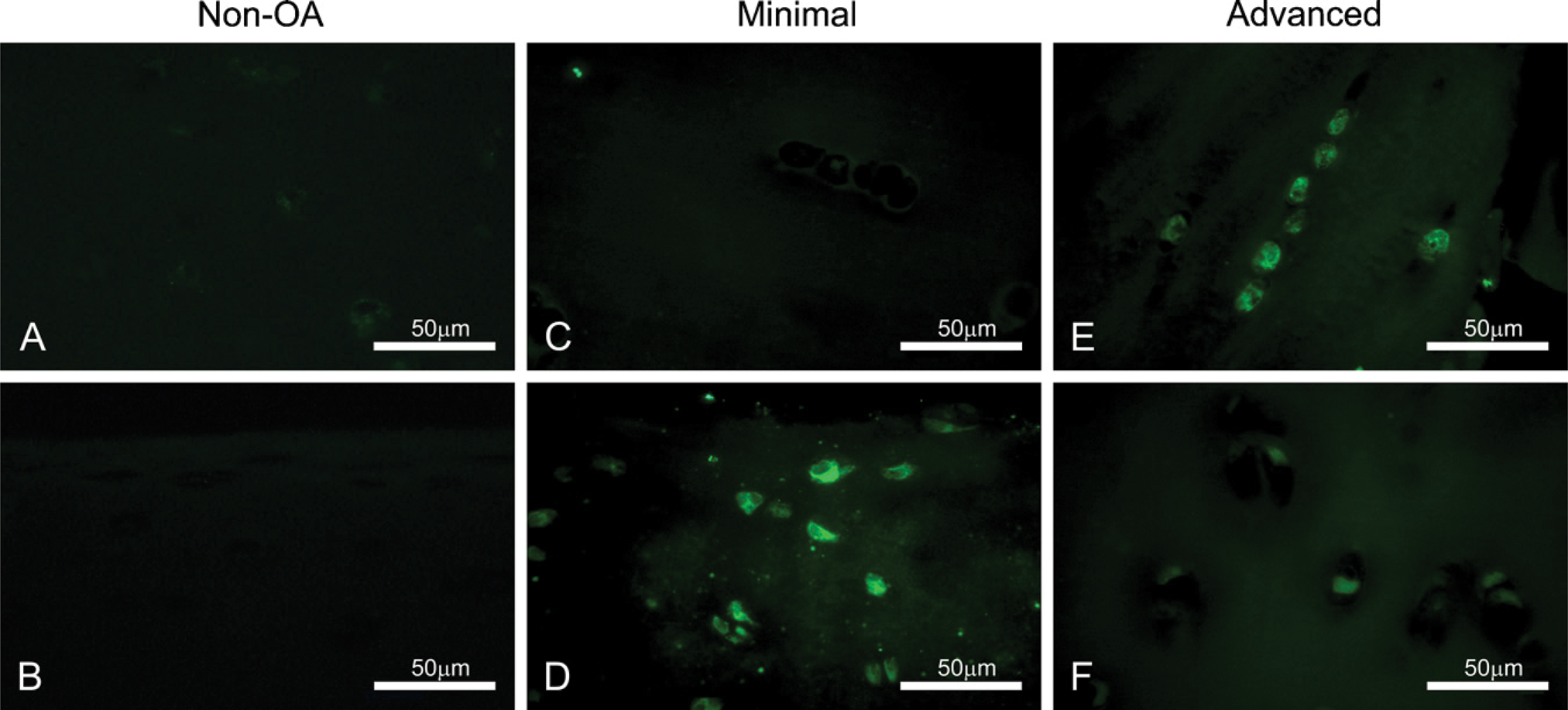
Grp78 was expressed to varying degrees in both minimally involved and advanced OA cartilage (Figure 3). Two representative patients are shown, demonstrating the range in staining degree and intensity that was observed. Normal, non-OA cartilage showed consistently low staining for grp78, and although OA cartilage showed consistently positive staining, staining was more prevalent in advanced samples vs minimally involved cartilage.
Bag-1 expression is present at low levels in healthy articular cartilage (Figure 4A). However, minimally involved OA cartilage exhibits an upregulation of bag-1, especially in the superficial zone (Figure 4B). Moreover, advanced OA chondrocytes upregulate bag-1 throughout the depth of the cartilage (Figure 4C). Although not every cell in advanced OA was positive for bag-1, there was a consistent upregulation across the depth of cartilage for every patient examined. Figures 4E and 4F show high-magnification images of bag-1-positive cells in normal and advanced-OA cartilage staining in the peri-nuclear region and cytoplasm.
Expression of Collagen VI and Its Receptor, NG2
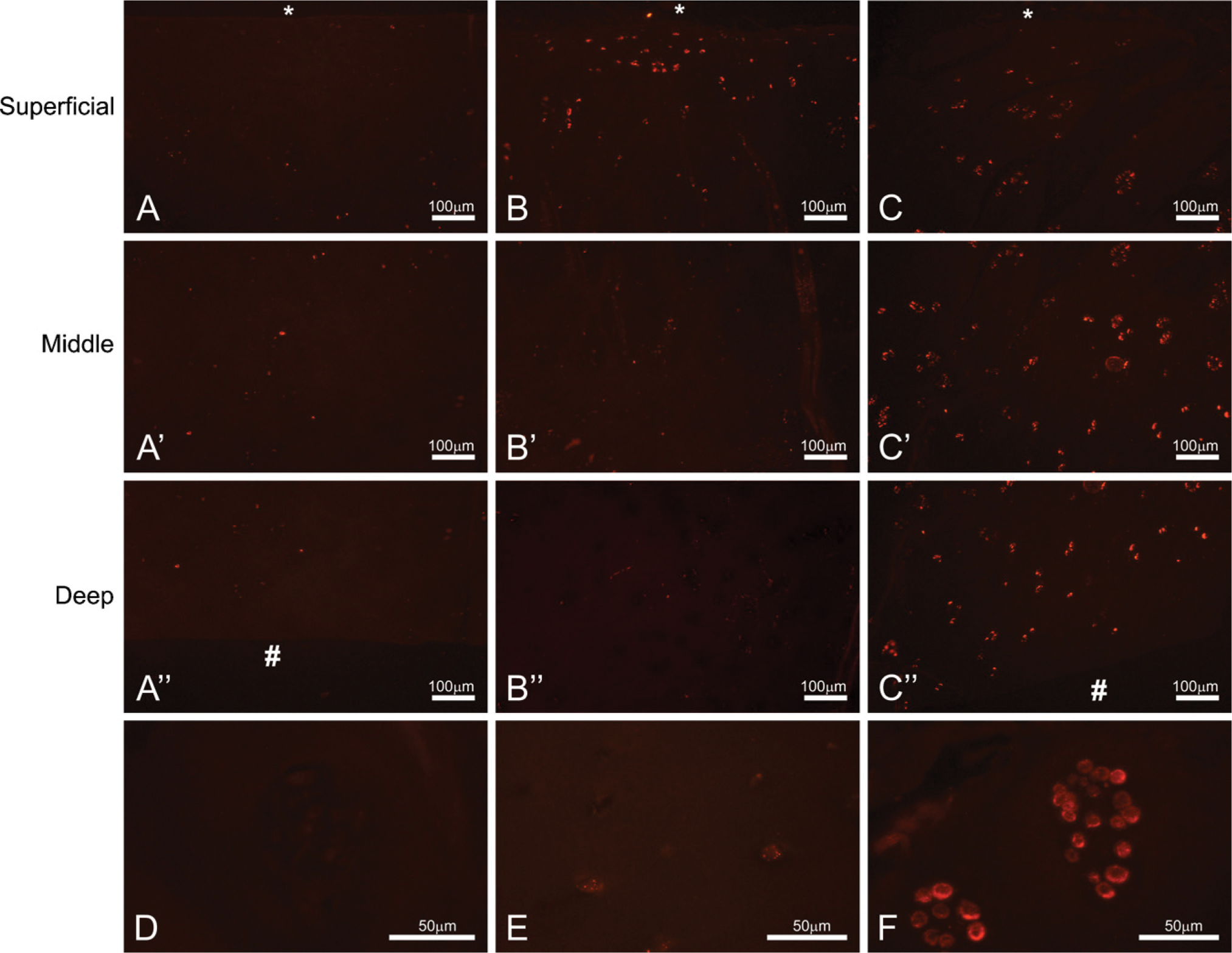
In healthy articular cartilage, collagen VI is found localized to the peri-cellular region known as the chondron (Poole 1997; Figure 5A). Through the progressive stages of OA, a change in distribution of collagen VI occurs, from the peri-cellular chondron to the interstitial matrix (Figures 5B and 5C). In addition to a change in distribution, collagen VI is highly upregulated in both minimal- and advanced-OA cartilage (Figures 5B and 5C), demonstrated by the increased staining intensity in these samples (Figures 5B”’ and 5C”, arrows). Through DAPI staining (not shown), the authors have determined that the majority of staining is outside the cell, and not intracellular.
Non-OA cartilage showed consistently positive cellular staining for NG2 (Figure 6). In every patient examined (n = 8), NG2 was downregulated in advanced-OA cartilage compared with its minimal-OA patient-matched cartilage sample, for which three representative patients are shown in Figure 6.
Discussion
Utilization of an intra-joint model system for describing the varying degrees of cartilage involvement during OA controls for genetic and age-related variances in gene and protein expression (Yagi et al. 2005). Minimal and advanced osteoarthritic cartilage samples were obtained and characterized according to defined end points, from which it was determined that advanced-OA cartilage differed significantly from minimal-OA cartilage, based on the means of their derived total scores. Quantifying these differences allowed us to draw conclusions from protein expression data based on the degree of similarity or difference between minimal and advanced patient-matched samples. It is also important to emphasize the differences in protein expression between normal cartilage and minimal-OA cartilage observed in this study, which demonstrates that disease-associated changes are occurring at the cellular and ECM level prior to macroscopic structural deformities.
Non-OA (
Distribution of bcl-2-associated athanogene-1 (bag-1) in non-OA (
Recent work has demonstrated that ER stress-inducing agents, such as glucose withdrawal, and the pharmacological agents tunicamycin and thapsigargin upregulate ER stress markers such as cleaved caspase-12 and gadd153 in isolated chondrocytes (Yang et al. 2005). Proteomic analysis of normal and OA chondrocytes has revealed changes in stress-related proteins (Ruiz-Romero et al. 2008). Yamasaki et al. (2006) reported resistance to ER stress-induced apoptosis in rheumatoid arthritic synovial cells. Additionally, ER stress is implicated in skeletal dysplasias such as pseudoachondroplasia and multiple epiphyseal dysplasia, in which mutated cartilage oligomeric matrix protein and collagen type IX are retained within the ER (Hecht et al. 1998). ER stress can also be induced by hypoxia and ischemic preconditioning, which reduce neuronal cell death by upregulating chaperones such as grp78 (Hayashi et al. 2003). Grp78 upregulation in coordination with a cellular ER stress response can also occur via photic injury (Yang et al. 2008). Here we have examined the in vivo expression of the ER stress marker grp78, and show that ER stress does not occur in normal, healthy articular cartilage, but does occur in advanced-OA cartilage and to varying degrees in minimal OA. The range of staining in minimally involved cartilage exemplifies the complexity of the disease process occurring at the cellular and molecular level, with changes in protein expression occurring at non-distinct intervals through the progression of disease. Examining chondrocyte mRNA levels for grp78 would be valuable in determining the timing of the ER stress response in coordination with other markers of OA disease progression. Grp78 is a critical ER chaperone protein that performs multiple functions, and is upregulated under stress conditions in an attempt to restore order to ER function and processing (Hendershot 2004; Li and Lee 2006; Lai et al. 2007). These data represent the first evidence of ER stress-associated proteins being expressed in osteoarthritic chondrocytes in vivo, and future studies will focus on the role of ER stress in the pathogenic mechanisms of OA.
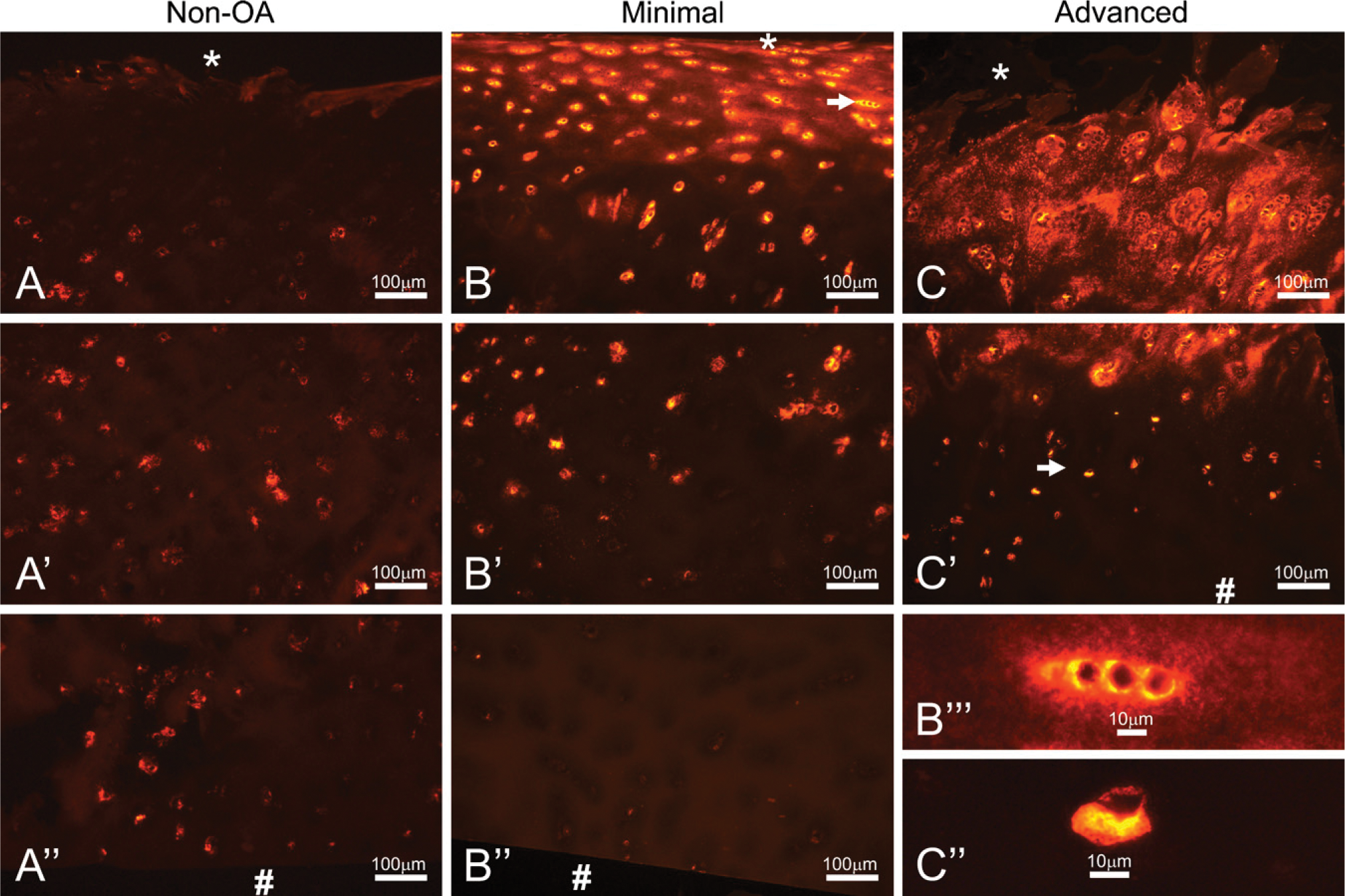
Non-OA (
Chondrocytes synthesize and secrete several types of collagens under homeostatic conditions, including types II, VI, IX, and XI, and the ER is the site of synthesis and folding of these molecules. Additionally, chondrocytes in pathological OA conditions disrupt homeostasis by up- and downregulating many secreted molecules, including these collagens. The peri-cellular matrix in articular cartilage is a unique region composed of specific ECM and transmembrane molecules, of which collagen VI is a vital component. The specificity of collagen VI to this region makes it an excellent marker for determining the relative health and turnover of cartilage matrix by chondrocytes in normal and osteoarthritic tissue. In previous studies, collagen VI has been shown to be upregulated in OA cartilage, but only in the mid to deep zones (Hambach et al. 1998; Pullig et al. 1999). Here we show that collagen VI changes its peri-cellular localization to an increasing degree with OA disease severity, especially in the superficial to mid zones. In addition to this change in distribution, there is an overall upregulation of collagen VI, as evidenced by increased staining intensity with the progression of disease. Although it is possible that partial degradation and fragmentation of collagen VI could also lead to increased immunohistochemical staining intensity, it is well characterized in the literature that collagen VI is upregulated in OA, which is corroborated here. We hypothesize that as chondrocytes lose the ability to interact with their surrounding matrix via collagen VI, they upregulate transcription of the gene to replenish the peri-cellular matrix and restore signaling pathways. Moreover, the loss of cell-matrix structural stability provided by the interaction of collagen VI with both the chondrocyte and other ECM molecules may contribute to the ECM degradation and fibrillation observed in advanced OA. However, it is also possible that increased expression is forcing the distribution interstitially, and that more collagen VI is being secreted than can be retained peri-cellularly.
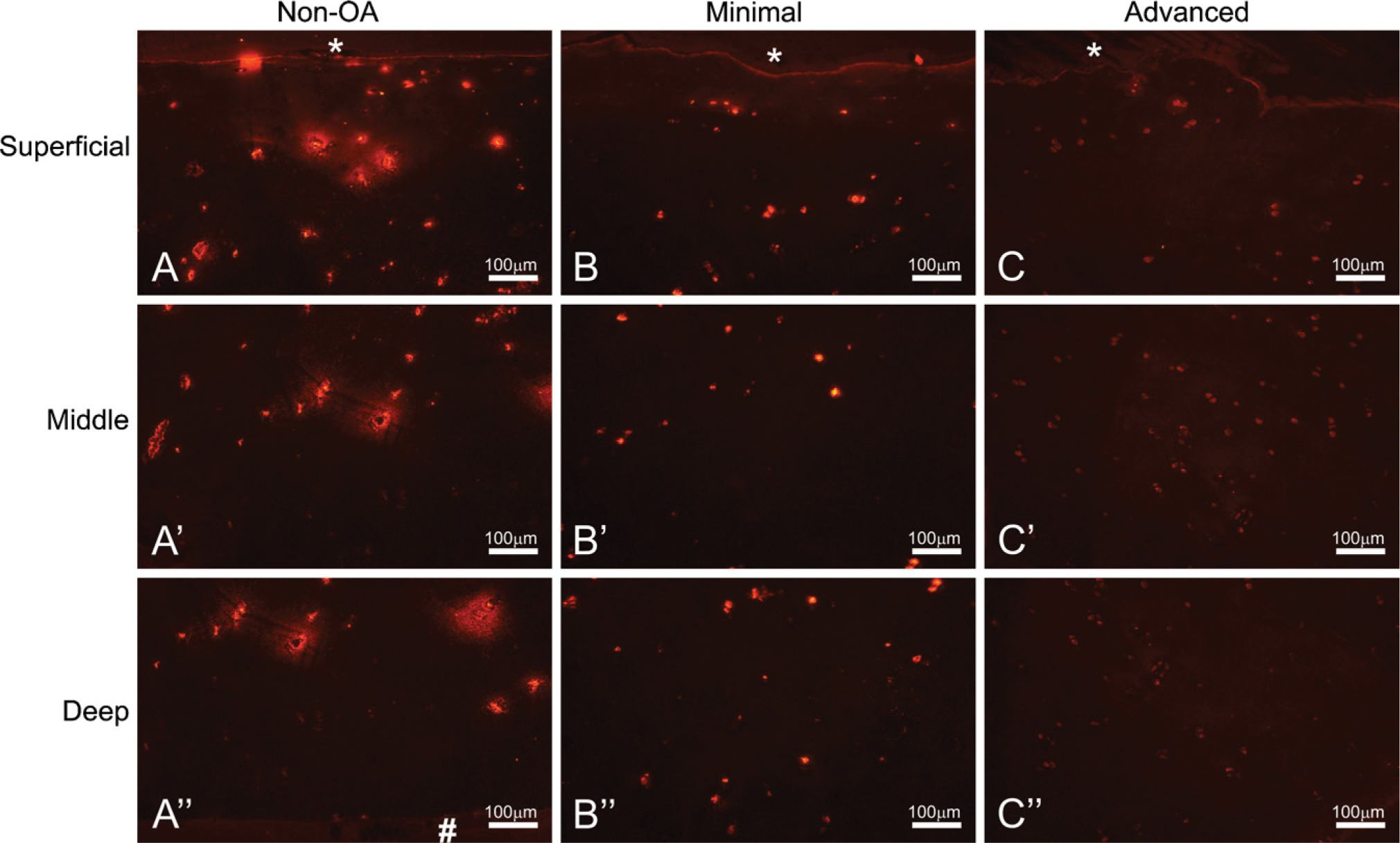
Representative samples of non-OA cartilage (
Tsang et al. (2007) report delayed terminal differentiation in hypertrophic chondrocytes of the growth plate in response to ER stress, brought on by the retention of collagen type X in the ER. Additionally, it is reported that grp78 is upregulated during early mouse development in the heart, somites, and other tissues, and that its expression decreases during later phases of development (Barnes and Smoak 2000). It has also been suggested that the GRP family of proteins serve as detectors for the level of activity of cellular secretion (Kozutsumi et al. 1988). From this work and the multitude of reports in the literature regarding upregulation of secreted molecules in OA, it is reasonable to speculate that significant upregulation of collagen VI and other secreted proteins could be a contributing factor to the occurrence of ER stress in an already diseased chondrocyte. However, future studies must be performed to link the upregulation of secreted proteins to ER stress in OA.
The combination of the change in collagen VI distribution to the interstitial matrix and its up-regulation suggests that the osteoarthritic chondrocyte is attempting to replenish its immediate environment without success. For this reason, we chose to examine the expression of NG2, the integral membrane proteoglycan to which collagen VI binds and which serves as a pathway for signaling activity and cell-matrix structural stabilization (Burg et al. 1996; Iyengar et al. 2005). Even though overall staining intensities were different within minimal and advanced groups, comparisons of patient-matched minimal- and advanced-OA pairs revealed a relative downregulation of NG2 in eight out of eight patients examined. Petrini et al. (2005) have shown that NG2 protein is downregulated in the skeletal muscle of patients with Ullrich scleroatonic muscular dystrophy, an inherited disorder characterized by a collagen VI mutation; however, the mRNA for NG2 is upregulated. An interesting continuation of our study would be to perform in situ hybridization on the normal and OA cartilage to determine whether this is also the case here.
Bag-1 is a member of the BAG family of proteins that bind and enhance the anti-apoptotic function of bcl-2 (Takayama et al. 1995). However, bag-1 serves a variety of other functions within the cell, including the mediation of proliferation via raf-1 activation and acting as a cochaperone in the ER through interaction with hsp70 (Wang et al. 1996; Takayama et al. 1997, 1999). As cells undergo ER stress, hsp70 is upregulated and competes with raf-1 for binding to bag-1 (Wang et al. 1996). It has been shown in our lab that bag-1 is downregulated in chondrocytes in monolayer culture in response to ER stress-inducing agents (Yang et al. 2007). We have also shown that bag-1 expression declines in mouse articular cartilage with age (Kinkel et al. 2004). From these data, we hypothesized that bag-1 expression would decline in osteoarthritic cartilage as a result of ER stress, disease, age, or a combination of these. However, we clearly show that bag-1 expression is increased in chondrocytes residing in more-morphologically involved cartilage, and this upregulation is observed throughout all zones of the cartilage. This is suggestive of a diseased or abnormal phenotype in chondrocytes, even in the mid to deep zones, where macroscopic defects have yet to develop. Bag-1 has been shown to localize to the mitochondria and ER during ER stress (Yang et al. 2007), indicative of its anti-apoptotic and ER stress-associated functions, respectively. These findings could suggest a number of different possibilities for the role of increased bag-1 expression in OA. Chondrocytes may upregulate bag-1 in a role protective against the increased apoptosis that occurs during OA. Apoptosis has been shown to occur at increased rates in OA compared with non-diseased cartilage, and across the depth of cartilage (Hashimoto et al. 1998; Héraud et al. 2000). Bag-1 is known to bind to and enhance the anti-apoptotic properties of bcl-2 (Takayama et al. 1995); therefore, its upregulation may be an attempt to antagonize apoptotic signals. Up-regulation of bag-1 may also occur to stimulate a proliferative signal to replenish cellularity in a degrading matrix, inasmuch as bag-1 binds to and activates the raf-1 MAP-kinase proliferation pathway (Wang et al. 1996). These anti-apoptotic and proliferative functions may also be working in coordination. Finally, increased bag-1 may aid in the UPR that occurs in response to ER stress, under which new protein translation is halted, misfolded proteins are shuttled for degradation, and molecular chaperones are upregulated, including the bag-1 binding proteins hsp70 and grp78 (Lee 2001; Banhegyi et al. 2007). The ubiquitin domain of bag-1 serves as an intermediary between hsp70 and the proteasome (Lüders et al. 2000), and its upregulation here may be in conjunction with the UPR characteristic of cells undergoing ER stress.
To summarize, we have shown that osteoarthritic cartilage expresses ER stress-associated proteins in vivo and mis-expresses proteins that may contribute to or respond to this stress. These data open the door to a new and exciting area of research bridging the OA and ER stress fields.
Footnotes
Acknowledgements
This work was supported by grants from the Arthritis Foundation and the National Institutes of Health. The authors thank Dr. William Stallcup for the generous donation of NG2 antibody.
