Abstract
We used immunohistochemistry to examine myosin heavy-chain (MyHC)-based fiber-type profiles of the right and left cricoarytenoideus dorsalis (CAD) and arytenoideus transversus (TrA) muscles of six horses without laryngoscopic evidence of recurrent laryngeal neuropathy (RLN). Results showed that CAD and TrA muscles have the same slow, 2a, and 2x fibers as equine limb muscles, but not the faster contracting fibers expressing extraocular and 2B MyHCs found in laryngeal muscles of small mammals. Muscles from three horses showed fiber-type grouping bilaterally in the TrA muscles, but only in the left CAD. Fiber-type grouping suggests that denervation and reinnervation of fibers had occurred, and that these horses had subclinical RLN. There was a virtual elimination of 2x fibers in these muscles, accompanied by a significant increase in the percentage of 2a and slow fibers, and hypertrophy of these fiber types. The results suggest that multiple pathophysiological mechanisms are at work in early RLN, including selective denervation and reinnervation of 2x muscle fibers, corruption of neural impulse traffic that regulates 2x and slow muscle fiber types, and compensatory hypertrophy of remaining fibers. We conclude that horses afflicted with mild RLN are able to remain subclinical by compensatory hypertrophy of surviving muscle fibers.
Keywords
I
The fiber types and MyHC expression in equine laryngeal muscles are poorly understood. Early analysis using myosin ATPase enzyme histochemistry revealed the presence of two basic fiber types, type 1 (slow) and type 2 (fast) fibers (Gunn 1972; Mascarello and Veggetti 1979; Cahill and Goulden 1986b; Duncan et al. 1991a; Lopez-Plana et al. 1993). Work based on SDS-PAGE suggested that laryngeal muscles affected by equine recurrent laryngeal neuropathy (RLN) expressed only two MyHC isoforms, slow and 2B (Adreani et al. 2006). However, the expression of 2B MyHC in laryngeal fibers of such a large animal is contrary to data from other large animals referred to above. This isoform is also absent in equine limb muscles (Rivero et al. 1999). A definitive study on equine laryngeal muscle fiber types based on MyHC isoform composition is still lacking.
The normal functions of the larynx involve the combined actions of the intrinsic laryngeal muscles. In the equine literature, the posterior cricoarytenoid muscle is referred to as the cricoarytenoideus dorsalis (CAD), whereas the interarytenoid is referred to as the arytenoideus transversus (TrA) (International Committee on Veterinary Gross Anatomical Nomenclature 2005). The CAD serves as the sole abductor of the vocal fold and arytenoid cartilage, and the TrA is an adductor that draws the dorso-medial margins of the arytenoid cartilages together. The cricoarytenoideus lateralis, thyroarytenoideus, and cricothyroideus muscles all serve as adductors. The cranial laryngeal nerve (International Committee on Veterinary Gross Anatomical Nomenclature 2005) innervates the cricothyroid, and the recurrent laryngeal nerve innervates all the other intrinsic laryngeal muscles, including the CAD and TrA. These intrinsic laryngeal muscles perform a diverse repertoire of complex movements that subserve the important functions of airway protection, respiration, and phonation.
Equine laryngeal muscles are of special interest because of the high incidence in this species of RLN, also known as idiopathic laryngeal hemiparesis or hemiplegia. There is now considerable evidence that this condition results from loss of large-diameter myelinated axons that is most profound distally (described as a distal axonopathy) in the left recurrent laryngeal nerve (Duncan et al. 1974; Cahill and Goulden 1986a; Hahn et al. 2008). The subsequent laryngeal muscle denervation and atrophy, which gives rise to the clinical signs of incomplete abduction of the arytenoid cartilage and vocal cord, is therefore typically left-sided. During exercise, these structures collapse into the airway, resulting in abnormal, loud, breathing noises, increased inspiratory resistance, a dramatic reduction in airflow to the lungs, and hypoxaemia (Derksen et al. 1986; Shappell et al. 1988; Dixon 2001). As a result, the most important manifestation clinically is the markedly impaired ability of the horse to perform strenuous exercise. Reinnervation of denervated muscle fibers by neighboring intact nerve terminals often occurs in contiguous muscle fibers by the same nerve fiber, leading to contiguous fibers acquiring the same histochemical properties through the operation of the neural influence, a manifestation referred to as fiber-type grouping, which is a diagnostic sign of early neuropathy (Karpati and Engel 1968).
Fiber-type grouping is known to occur commonly in equine laryngeal muscles (Gunn 1972; Cahill and Goulden 1986b). Up to 30% of horses with no laryngoscopic evidence of laryngeal hemiplegia show fiber-type grouping and histological evidence of neurogenic atrophy in those laryngeal muscles innervated by the left recurrent laryngeal nerve. These apparently normal horses with fiber-type grouping are referred to as “subclinical” cases of RLN (Gunn 1972; Duncan et al. 1974,1977; Cahill and Goulden 1986b; Lopez-Plana et al. 1993).
This study aims to use a battery of highly specific monoclonal antibodies (MAbs) to identify the full range of MyHC isoforms present in equine laryngeal muscles, and to establish a MyHC-based fiber-type classification for these muscles. This classification is important for assessing the contractile characteristics of normal laryngeal muscles and the changes in function as a result of disease, inasmuch as MyHC exerts a profound influence on the speed of contraction of a muscle fiber (Bottinelli et al. 1991). Because it is likely that subclinical cases of RLN would be encountered among the horses studied, it is hoped that these studies will throw some light on pathophysiological mechanisms at work during RLN as well as adaptive mechanisms that enable subclinical horses to remain asymptomatic. This study lays the foundation for future work into the control of equine laryngeal muscle fiber types and changes in laryngeal muscles of horses with manifest laryngeal hemiparesis.
Materials and Methods
Horses and Laryngeal Muscle Preparation
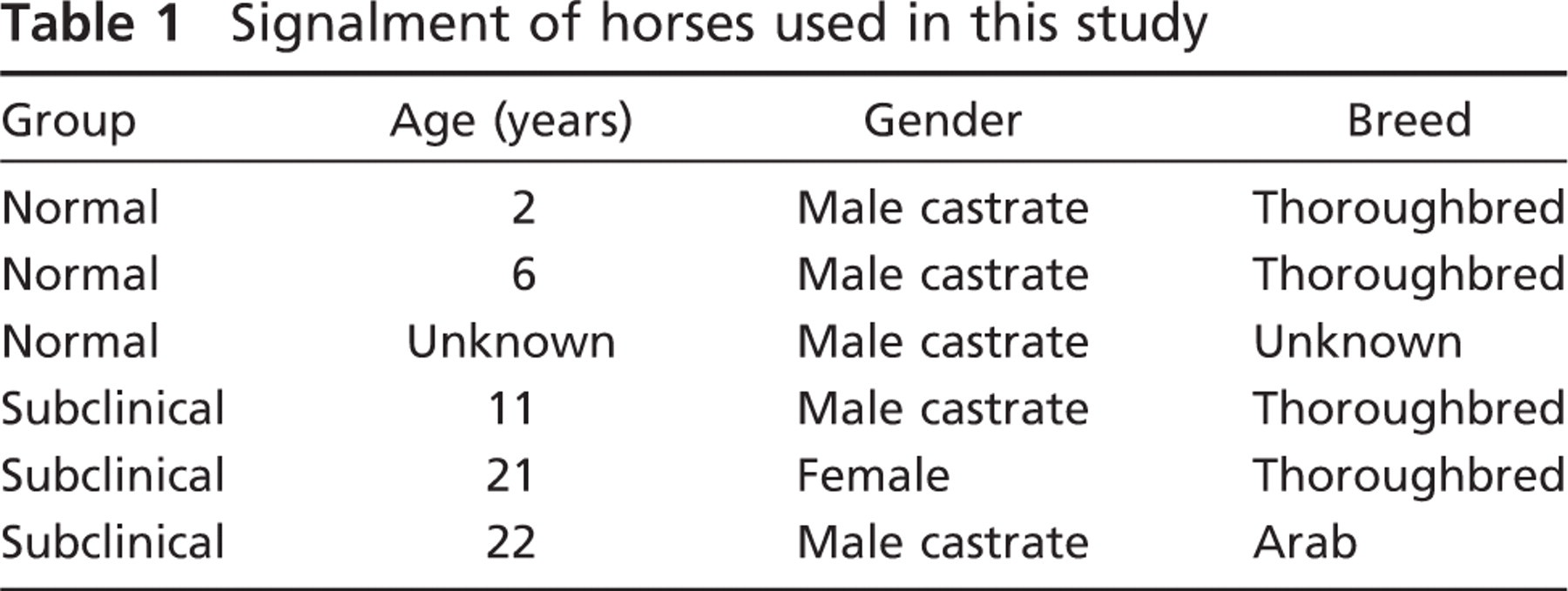
Six horses were used for this investigation. All horses were clinically normal, and on laryngoscopic examination at rest had synchronous and full abduction of the arytenoids cartilages (grade 1 laryngeal function; Rakestraw et al. 1991). They were euthanized in the College of Veterinary Medicine, Michigan State University, for reasons unrelated to any abnormality of the respiratory tract, and institutional guidelines for animal research were followed. The age, breed, and gender of the horses are given in Table 1.
Signalment of horses used in this study
Within 24 hr of the death of the animal, the left and right CAD (L-CAD and R-CAD, respectively) and the left and right TrA (L-TrA and R-TrA, respectively) muscles were removed. Portions of the excised muscles were then mounted on pieces of cork with Tissue-Tek (Miles Scientific; Elkhart, IN), frozen in isopentane cooled in liquid nitrogen, then stored at -80C. The muscles were then packaged in dry ice and air-freighted to Sydney, where they were stored in liquid nitrogen Dewars until used.
Immunohistochemistry and Division of Muscle Into Normal and Subclinical Groups
Serial sections of CAD and TrA muscles from both sides were cut at 10 μm in a cryostat at -20C. Sections from the mid-regions of these muscle blocks were used for immunohistochemistry. Indirect immunoperoxidase histochemistry was carried out as previously described (Hoh et al. 1988) using MAbs against MyHCs as primary antibodies. The MAbs used were 10F5 (anti-2B; Lucas et al. 2000), 6H1 (anti-2X; Lucas et al. 2000), SC-71 (anti-2A; Schiaffino et al. 1989), NOQ7-5–4-D (anti-slow/β-cardiac; Draeger et al. 1987), and 4A6 (anti-EO; Lucas et al. 1995). The MAbs developed in this laboratory (10F5, 6H1, and 4A6) are available from Developmental Studies Hybridoma Bank, University of Iowa. The secondary antibodies used were horseradish peroxidase–labeled rabbit anti-mouse immunoglobulin (Dako Products; Carpinteria, CA) or goat anti-mouse IgM (Sigma-Aldrich; St. Louis, MO) for anti-2X and anti-2B MyHC MAbs.
When immunohistochemically stained sections were viewed microscopically, some muscles showed a normal mosaic pattern of different fiber types, whereas others showed obvious fiber-type grouping, as reported earlier (Gunn 1972). Previous studies had shown that the distal axonopathy affected the left adductors (i.e., the L-TrAs) more severely than the left abductor (L-CAD). Further, the right recurrent laryngeal nerve was sometimes also affected, albeit less severely than the left (Cahill and Goulden 1986a; Duncan et al. 1991a; Lopez-Plana et al. 1993; Hahn et al. 2008). Thus, among the muscles studied, the L-TrA muscle would be the most sensitive for detecting the presence of fiber-type grouping. This muscle was thus selected for sorting the horses into two groups: a “normal” group with a mosaic pattern of fiber staining, and a “subclinical” group in which fiber-type grouping was evident. Sorting was done by four external observers, and the outcome was a complete consensus in their division of the six horses into the two groups, each with three animals, as shown in Table 1. For convenience of description, muscles from “normal” horses are referred to as “normal muscles,” and muscles from “subclinical” horses are referred to as “subclinical muscles,” irrespective of their structural characteristics.
Quantification of Muscle Fiber-type Distribution and Fiber Cross-sectional Areas
The percentages of fibers staining with MAbs against slow, 2A, and 2X MyHCs (i.e., the slow, 2a, and 2x fibers, respectively) present in each muscle were calculated based on counts of all fibers visible in the entire cross sections of the muscle blocks. Counts were done manually using montages of photomicrographs of serial sections. Fibers were classified as hybrid if they were clearly stained by more than one anti-MyHC MAb.
Cross-sectional areas of slow, 2a, and 2x fibers in each muscle were measured from immunohistochemically stained serial sections of the muscle using Image J (National Institutes of Health, version 1.33u). All fibers within a representative microscopic field (≃400–700) were measured.
Statistical comparisons of fiber sizes were performed using the unpaired two-tailed Student's t-test, and were noted as being significant when p<0.05.
Results
MyHC Immunohistochemistry of CAD and TrA Muscles of Horses in the Normal Group
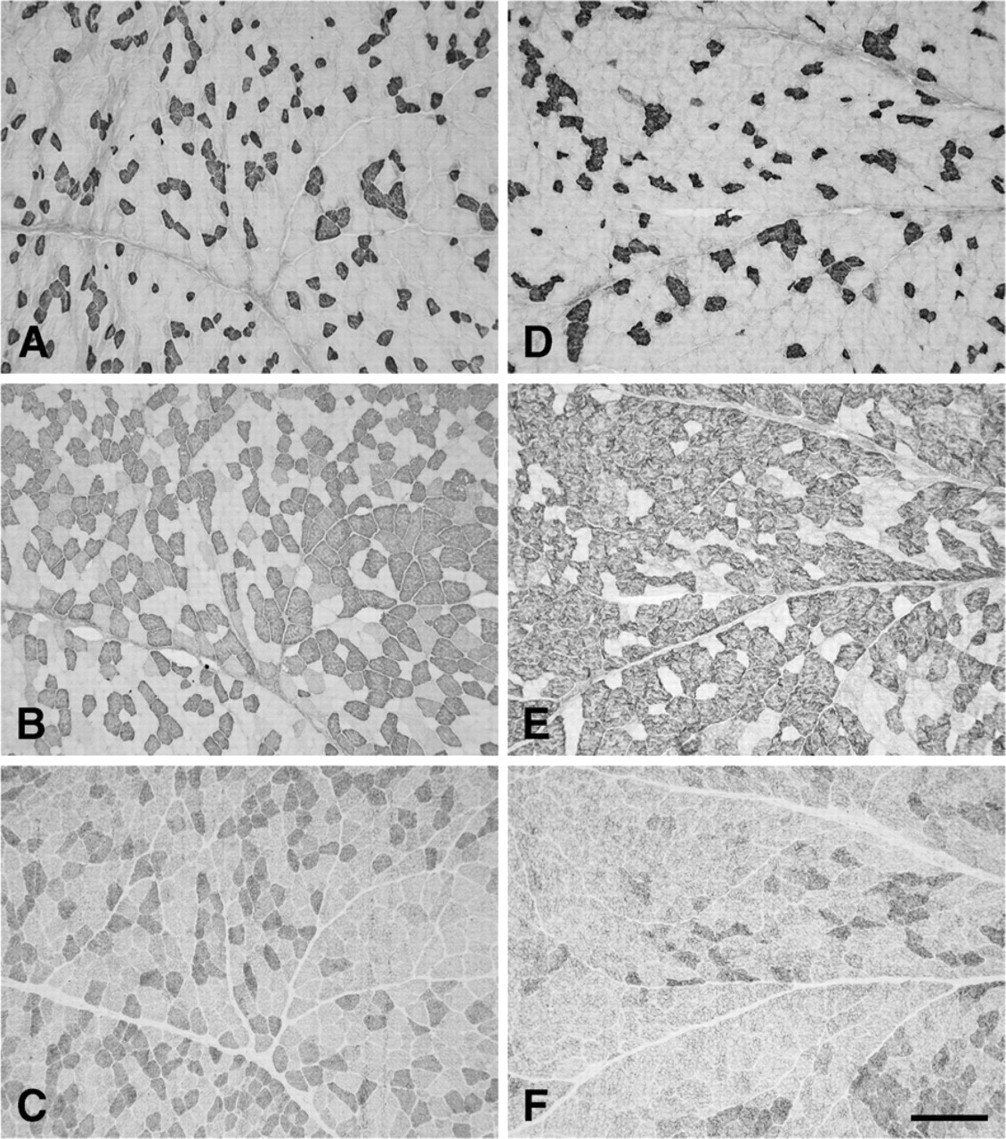
Figure 1 shows low-power photomicrographs of cross sections of the normal R-CAD (Figures 1A–1D) and L-CAD (Figures 1E–1H), from a 6-year-old horse, stained with anti-slow (Figures 1A and 1E), anti-2A (Figures 1B and 1F), anti-2X (Figures 1C and 1G), and anti-2B (Figures 1D and 1H) MAbs. The figure shows that in these muscles, 2a fibers predominate, followed by slow and 2x fibers. Close examination of these photomicrographs revealed that all fibers were stained by one or more of these three MAbs. However, MAb 10F5, which is specific to 2B MyHC in the limb muscles of the rabbit, cat, baboon (Lucas et al. 2000), and seven species of marsupial mammals (Zhong et al. 2001,2008), failed to react with fibers in these muscles (Figures 1D and 1H). Further, the anti-EO MyHC MAb 4A6 also did not stain any fiber (data not shown). The absence of fiber-type grouping is most convincingly seen among the slow fibers, where their low abundance permits easy visualization of a mosaic pattern. High-power photomicrographs of the R-CAD (Figure 2) show clearly that fibers in this muscle comprise only slow, 2a, and 2x fibers, with a small population of hybrid 2a/2x fibers coexpressing 2A and 2X MyHCs.
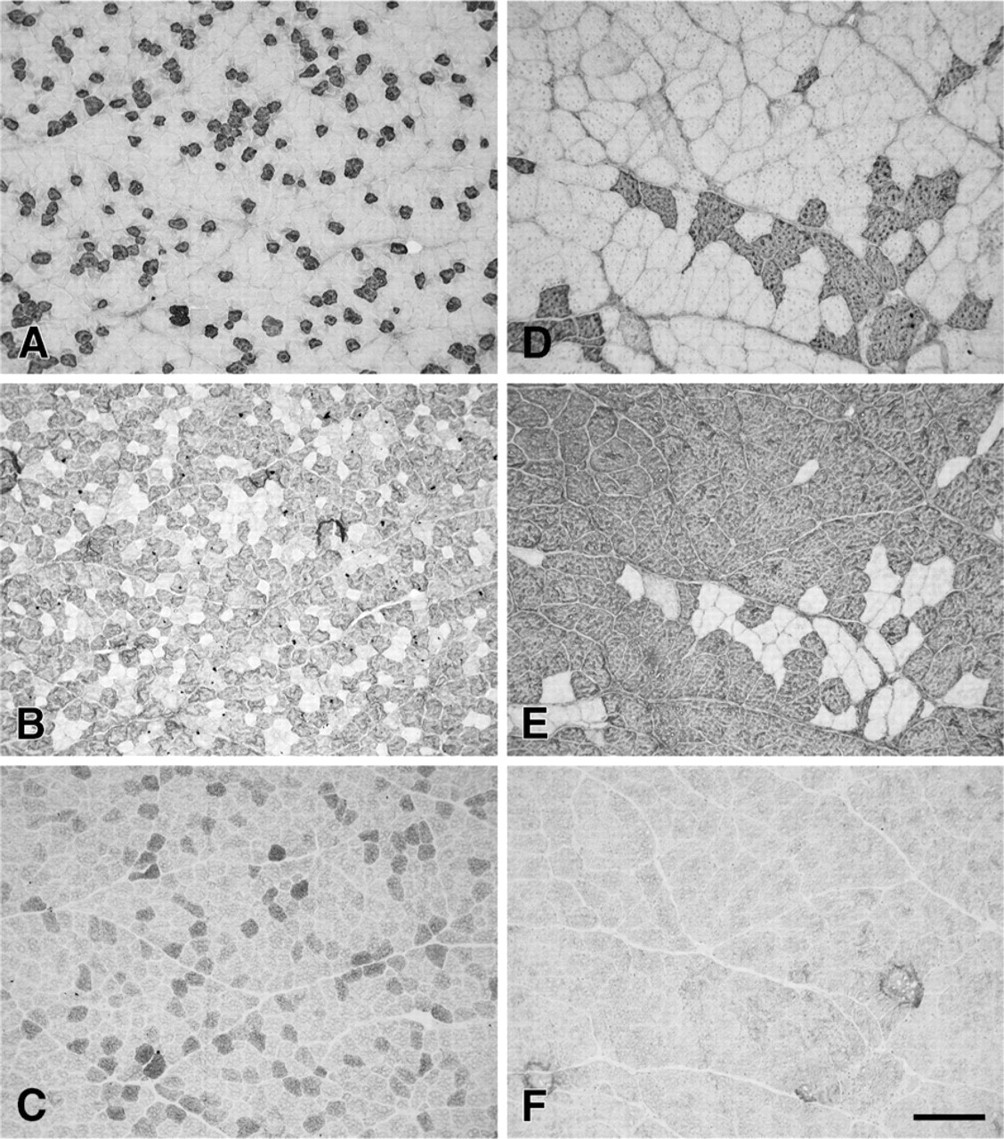
Figure 3 shows photomicrographs of serial cross sections of the normal R-TrA (Figures 3A–3C) and L-TrA (Figures 3D–3F) stained with anti-slow (Figures 3A and 3D), anti-2A (Figures 3B and 3E), and anti-2X (Figures 3C and 3F). The staining patterns of these muscles are very similar to those of normal R-CAD and L-CAD, with a predominance of 2a fibers and absence of fiber-type grouping. These normal R-TrA and L-TrA muscles also did not stain with MAbs 4A6 and 10F5 (data not shown), indicating that they were also devoid of fibers expressing EO and 2B MyHCs.
The mean percentages of various types of fibers in the right and left CAD and TrA muscles from the three normal horses are presented in Table 2. It can be seen that both CAD and TrA muscles are rich in 2a fibers, moderate in slow fibers, but poorer in 2x fibers. Hybrid fibers coexpressing two MyHCs (slow/2a, 2a/2x fibers) are present, but constitute up to little more than 5% of the total fibers. Statistical comparisons between the left and right muscles revealed that the left-sided muscles had significantly fewer slow fibers, compensated for by having more 2a and 2x fibers. This difference in abundance of 2a and 2x fibers between the sides reached statistical significance only for 2x fibers in the TrA. No fibers were found to express 2B and EO MyHCs in any of the normal muscles studied.
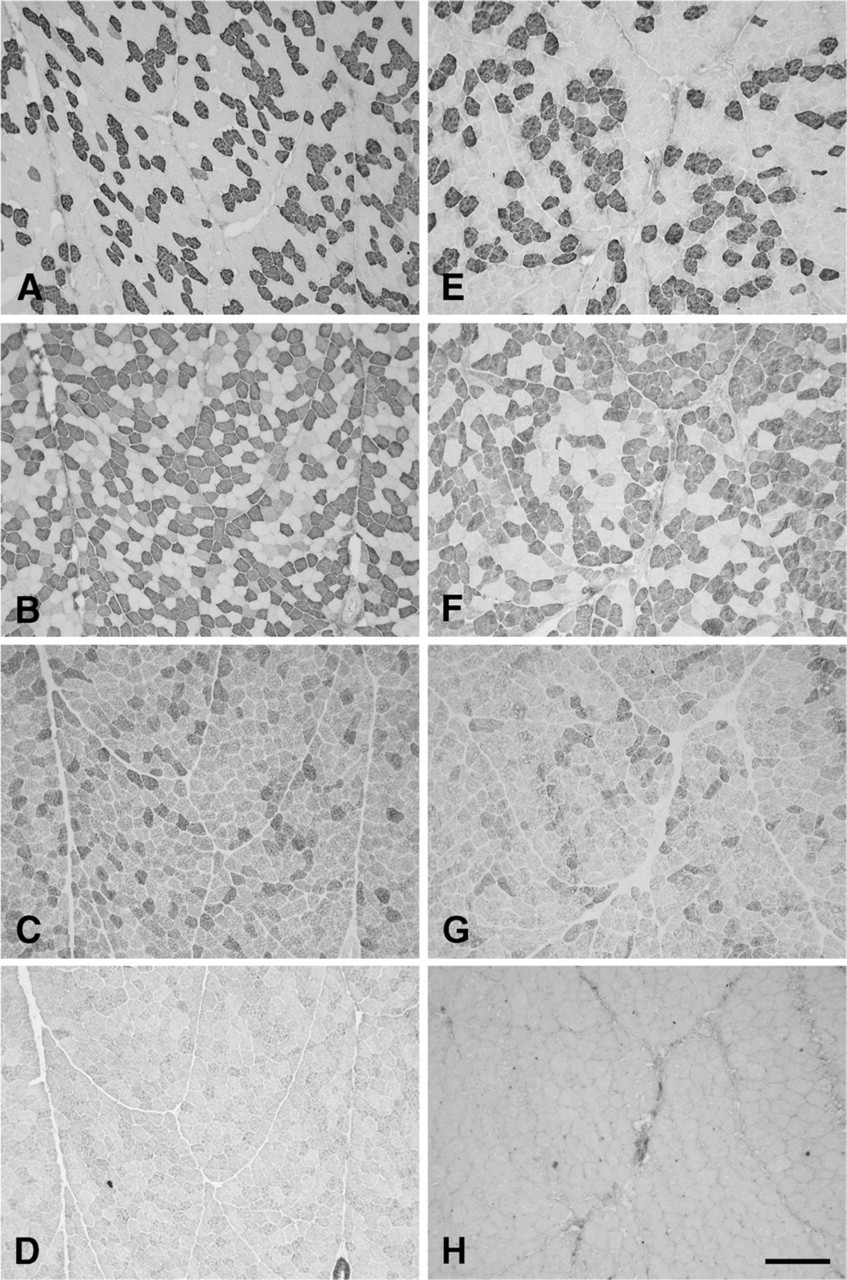
Photomicrographs of immunoperoxidase-stained semi-serial sections of the cricoarytenoideus dorsalis (CAD) muscles from a normal horse aged 6 years old. Panels
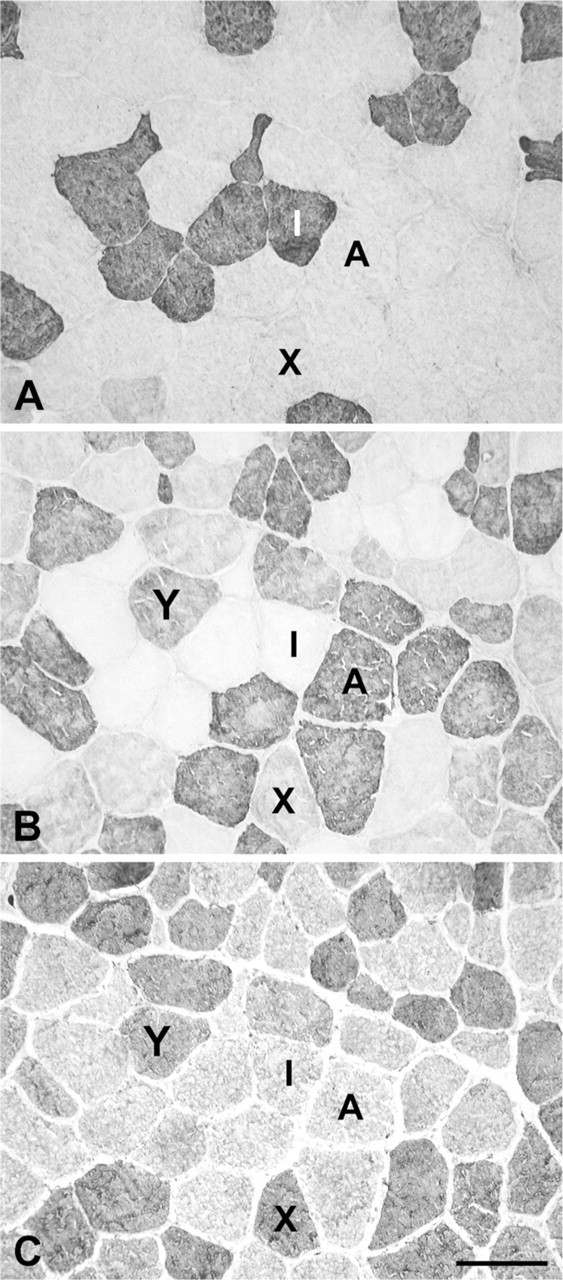
High-power photomicrographs of immunoperoxidase-stained serial sections of the R-CAD muscle shown in Figure 1. The primary antibodies used were anti-slow
MyHC Immunohistochemistry of CAD and TrA Muscles of Horses in the Subclinical Group
Figure 4 shows immunohistochemically stained serial sections of subclinical R-CAD (Figures 4A–4C) and L-CAD (Figures 4D–4F) from a horse aged 22 years. Note that although the immunohistochemical features of this subclinical R-CAD resemble those of normal CAD and TrA muscles, the L-CAD has significantly larger fiber size (see later for quantitative data), shows obvious fiber-type grouping, and, in this field, absence of 2x fibers. These features of the L-CAD are also seen in L-TrA as well as the R-TrA muscles from the same horse (Figure 5). Fiber-type grouping is much more pronounced in the TrA muscles than in the L-CAD, and more pronounced in the L-TrA than in the R-TrA. There were no 2x fibers in L-TrA or R-TrA (Figures 5C and 5F), and no 2b fibers (data not shown).
Quantitative analysis of fiber-type distribution of subclinical CAD and TrA muscles from the three subclinical horses is presented in Table 3. Whereas the profile of fiber types in the subclinical R-CAD resembles those in the normal CAD and TrA muscles (Table 2), the subclinical L-CAD shows a significantly greater abundance of 2a fibers (p<0.005, t-test) and lower abundance of slow fibers (p<0.05, t-test) compared with the subclinical R-CAD. Table 3 shows that subclinical L-TrA also has significantly more 2a fibers (p<0.05, t-test) and correspondingly fewer slow fibers (p<0.005, t-test) than subclinical R-TrA. There were virtually no 2x fibers, as well as no fibers expressing 2B or EO MyHCs, in the subclinical R-TrA and L-TrA.
The proportions of slow, 2a, and 2x fibers in subclinical L-CAD, R-TrA, and L-TrA muscles are compared with corresponding normal muscles in Figure 6. For simplicity, hybrid fibers are not included, inasmuch as they constituted less than 10% of all fibers. Figure 6A shows that the proportion of 2a fibers in subclinical L-CAD muscles is significantly higher, whereas the proportions of 2x and slow fibers were significantly lower than in normal L-CAD. There are very similar changes in the subclinical L-TrA compared with normal L-TrA (Figure 6B): an increase in 2a fibers, but the slight decrease in slow fibers is not statistically significant. However, the most striking change in fiber-type distribution in the subclinical L-TrA is the complete disappearance of pure 2x fibers. The subclinical R-TrA (Figure 6C) resembles subclinical L-CAD and L-TrA in the loss of 2x fibers relative to the corresponding normal muscle (p<0.005, t-test), but this loss is compensated for not by an increase of 2a fibers, but rather by a significant increase in slow fibers (p<0.05, t-test).
Fiber Cross-sectional Areas in Normal Muscles
Table 4 gives the mean values of the cross-sectional areas of slow, 2a, and 2x fibers of CAD and TrA muscles in normal horses and in those with subclinical RLN. In the normal R-CAD, the 2x fibers are the largest in size, followed by 2a fibers; the slow fibers are the smallest, and this order of relative size is the same as in normal mammalian limb muscles (Lucas et al. 2000). This order is disrupted in normal L-CAD, where the order is reversed (slow<2a<2x), and in the normal R-TrA and normal L-TrA, where 2a fibers are larger than 2x fibers. However, comparing the mean cross-sectional area of each fiber type in the normal CAD and TrA muscles on the right with the corresponding value on the left in Table 4, no significant differences were evident.
Distribution of fiber types in equine laryngeal muscles from horses in the normal group
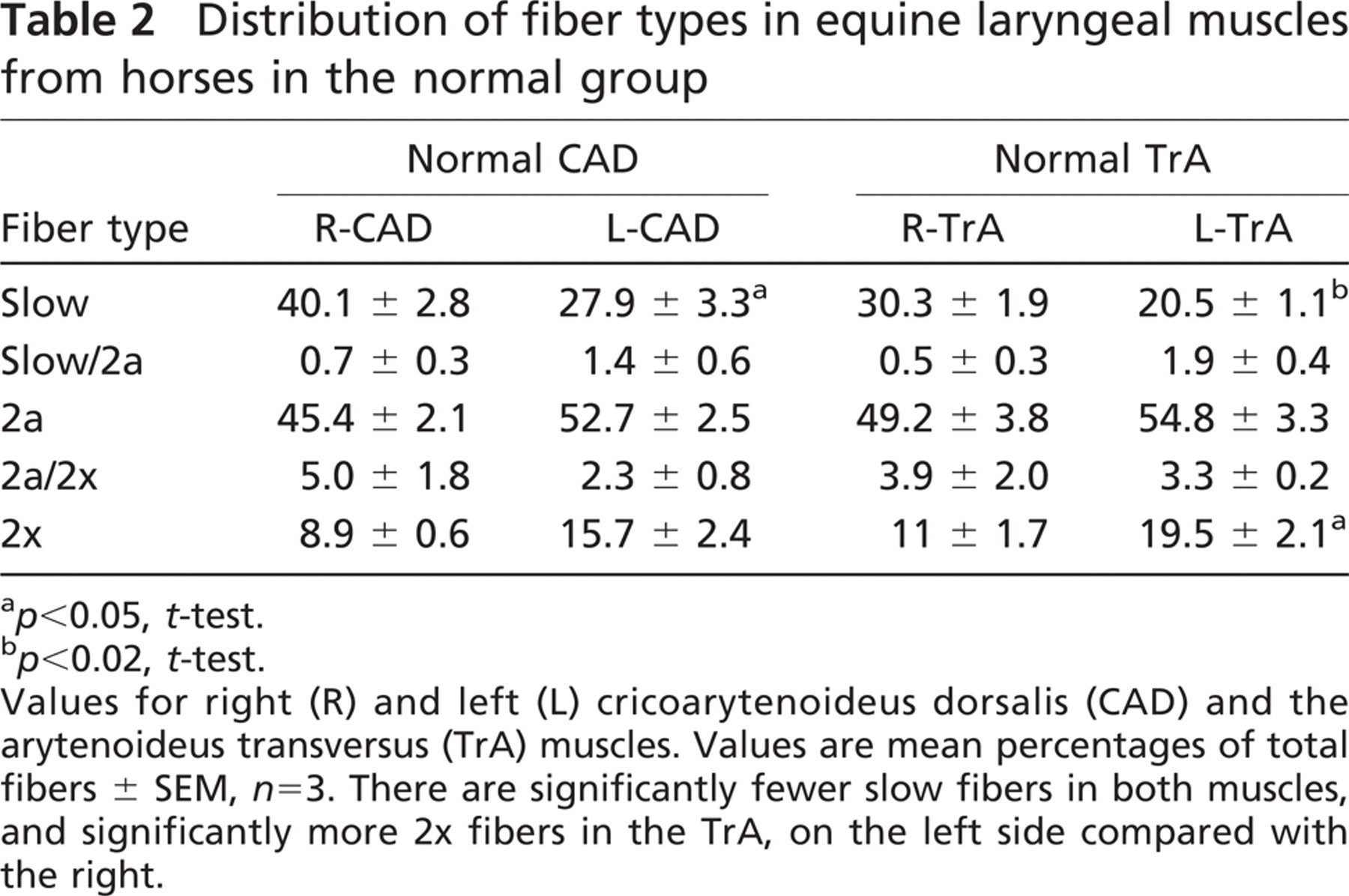
p<0.05, t-test.
p<0.02, t-test.
Values for right (R) and left (L) cricoarytenoideus dorsalis (CAD) and the arytenoideus transversus (TrA) muscles. Values are mean percentages of total fibers ± SEM, n=3. There are significantly fewer slow fibers in both muscles, and significantly more 2x fibers in the TrA, on the left side compared with the right.
Photomicrographs of immunoperoxidase-stained semi-serial sections of the arytenoideus transversus (TrA) muscles from a normal horse aged 6 years old. Panels
Fiber Size Spectra in Subclinical Muscles
Figure 7 compares the fiber size spectra of slow and 2a fibers of R-CAD and L-CAD muscles from a subclinical horse (white columns) with those of corresponding muscles from a normal horse (black columns). Figure 7A shows that the spectra of slow and 2a fibers in subclinical R-CAD are similar to those of corresponding fibers in the normal R-CAD. This is supported by the grouped data on mean cross-sectional areas of these fibers presented in Table 4, which showed no statistically significant differences between these values. Figure 7B shows that the profiles of both slow and 2a fibers in the subclinical L-CAD are shifted toward the right relative to those in normal L-CAD. However, grouped data on mean cross-sectional areas of these fibers shown in Table 4 did not reach a statistically significant difference. Figure 7 also shows that subclinical slow and 2a fibers in L-CAD are shifted to the right relative to corresponding fibers in R-CAD, and this is supported by statistical differences (p<0.05, t-test) between the mean cross-sectional areas of these fibers presented in Table 4.
Photomicrographs of immunoperoxidase-stained semi-serial sections of the CAD from a subclinical horse aged 22 years. Panels
Figure 8 compares the fiber size spectra of slow and 2a fibers of R-TrA and L-TrA muscles from a subclinical horse (white columns) with those of corresponding muscles from a normal horse (black columns). The upper panels of Figure 8 reveal large shifts to the right in size spectra of slow fibers in subclinical R-TrA and L-TrA, compared with respective normal muscles. Data presented in Table 4 show that the mean cross-sectional area of slow fibers in subclinical R-TrA muscles is significantly larger (p<0.05, t-test) than that of normal R-TrA, but the difference for slow fibers between normal and subclinical L-TrA did not reach statistical significance. The lower panels of Figure 8 also show that 2a fibers in subclinical R-TrA, as well as those in normal and subclinical L-TrA, have a moderate shift to the right compared with normal R-TrA. However, Table 4 shows that although the mean cross-sectional areas of 2a fibers in subclinical R-TrA, normal, and subclinical L-TrA muscles are larger than the value for 2a fibers in normal R-TrA, the difference did not reach statistical significance.
Discussion
Isoforms of MyHC Expressed in Equine Laryngeal Muscles
The present work is the first to define the MyHC isoforms expressed in equine laryngeal muscle fibers by using a battery of highly specific MAbs to the five MyHCs found in mammalian laryngeal muscles across various species, i.e., slow, 2A, 2X, 2B, and EO. The MAbs used in this study have been extensively investigated previously, and shown to be MyHC isoform–specific in a wide range of animal species. Our immunohistochemical analyses using these MAbs clearly showed that the equine CAD and TrA muscles express only three of these MyHCs—slow, 2A, and 2X—all of which have been reported to be expressed in equine limb muscles (Rivero et al. 1999). All fibers in these horse laryngeal muscles were stained by one or more of the MAbs against these MyHCs, thus excluding the presence of fibers exclusively expressing other MyHCs.
Distribution of fiber types in the right and left CAD and the TrA muscles of horses with subclinical recurrent laryngeal neuropathy (RLN)
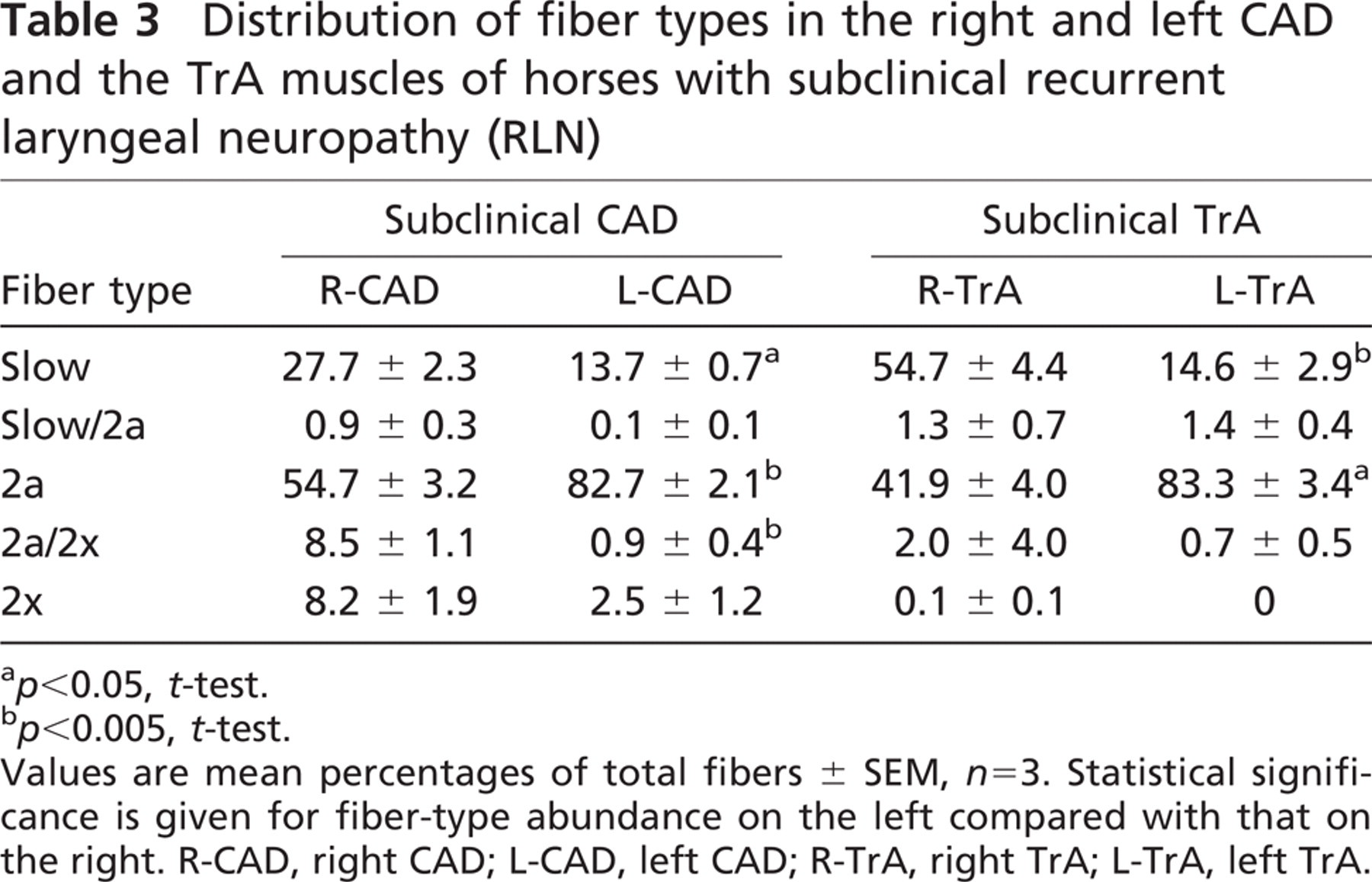
p<0.05, t-test.
p<0.005, t-test.
Values are mean percentages of total fibers ± SEM, n=3. Statistical significance is given for fiber-type abundance on the left compared with that on the right. R-CAD, right CAD; L-CAD, left CAD; R-TrA, right TrA; L-TrA, left TrA.
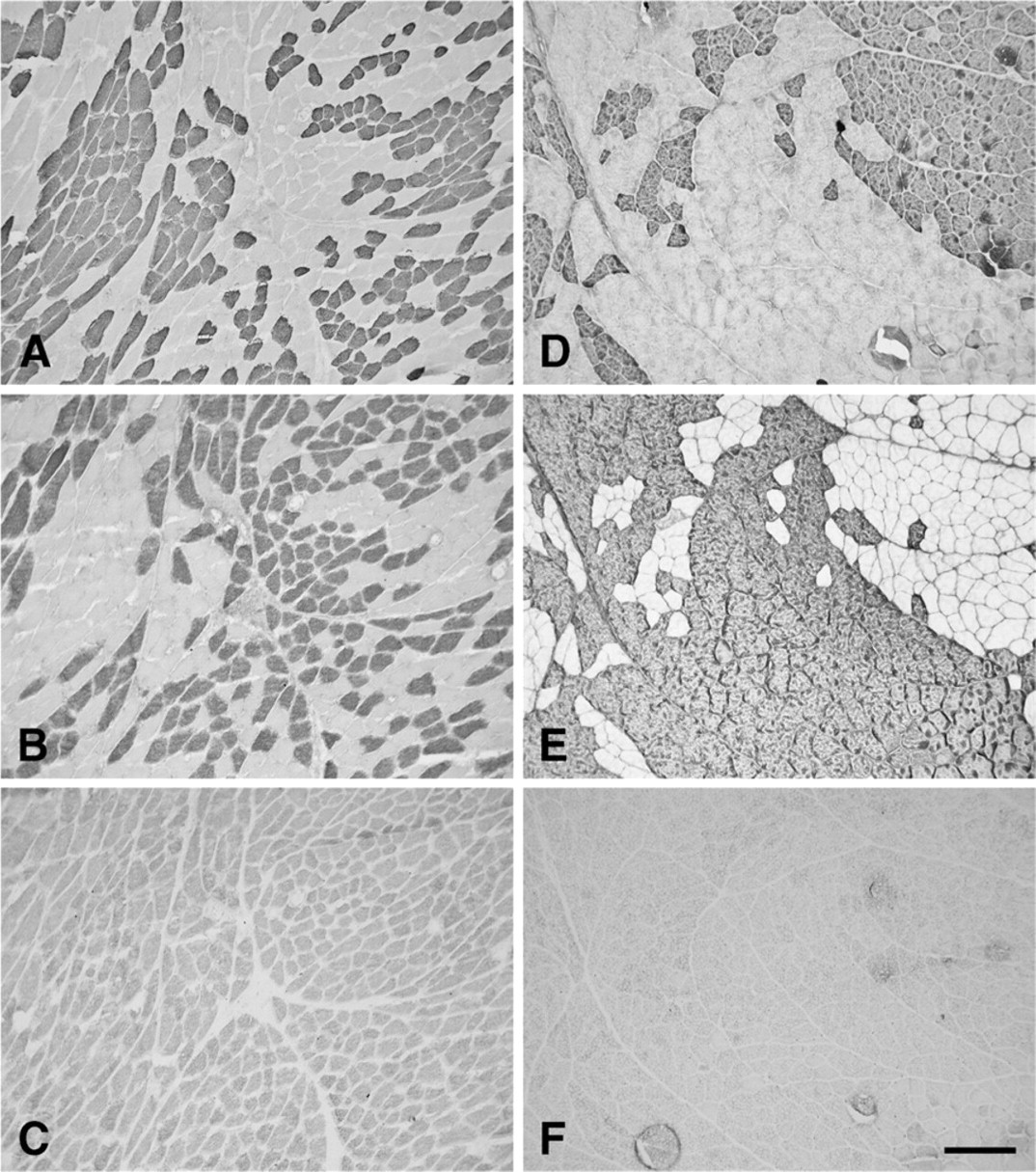
Photomicrographs of immunoperoxidase-stained semi-serial sections of the TrA from the horse with subclinical recurrent laryngeal neuropathy shown in Figure 4. Panels
We note the failure of MAbs against EO and 2B MyHCs, which are specific across a wide range of species, to stain fibers in equine CAD and TrA. The anti-EO MAb 4A6 was shown to be EO MyHC specific in rabbits (Lucas et al. 1995), humans (Pedrosa-Domellof et al. 2000), rats (Rubinstein and Hoh 2000), and cats (Lucas CA and Hoh JFY, unpublished data). Anti-2B MyHC MAb 10F5 has been shown to be specific for this isoform in the mouse (Nair-Shalliker et al. 2004), rat, rabbit, cat, baboon (Lucas et al. 2000), and even marsupial species (Zhong et al. 2008), using both immunohistochemistry and Western blots. Our failure to find 2B MyHC expression in horse laryngeal muscles is in contrast to recent work suggesting that laryngeal muscles from horses with RLN express 2B and slow MyHCs (Adreani et al. 2006). The identification of MyHC isoforms in Adreani et al. (2006) relied on a comparison of the electrophoretic mobilities of equine and murine MyHCs in SDS-PAGE gels, under the assumption that the mobility of each MyHC isoform is independent of species, which clearly cannot be relied upon (Galler et al. 1997; Reiser and Kline 1998; Zhong et al. 2001).
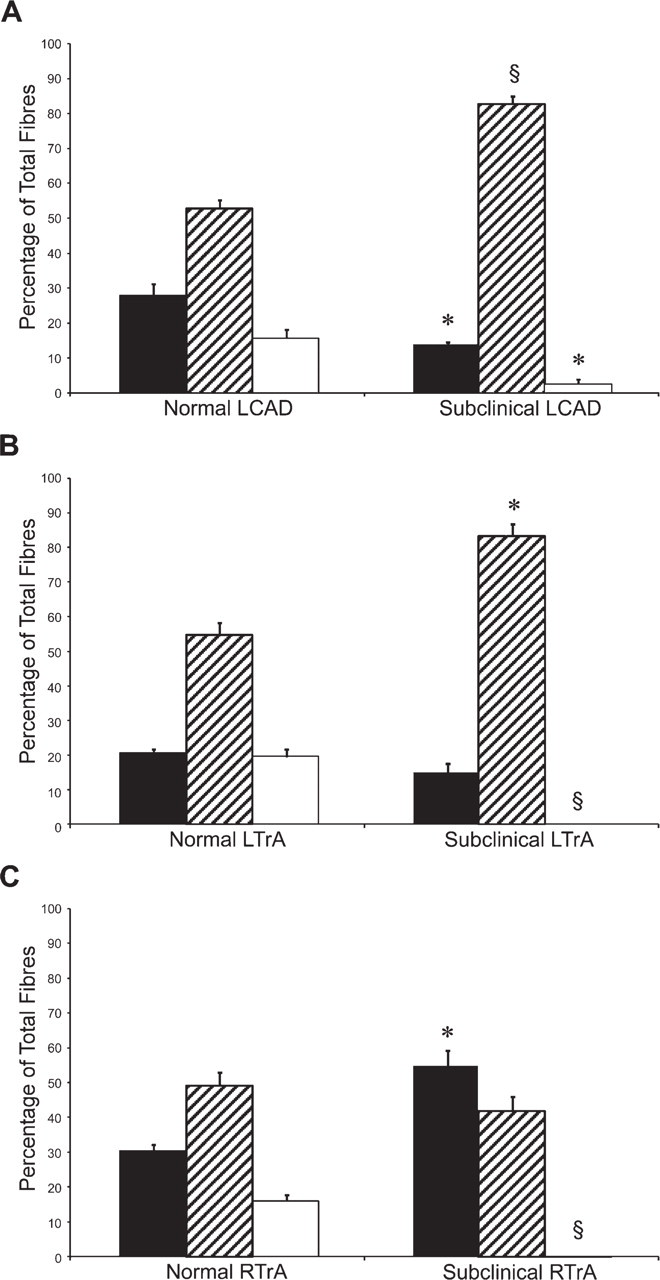
Histograms of fiber-type distribution in normal and subclinical laryngeal muscles. Values show percentages (mean ± SEM, n=3) of fibers expressing pure slow (black), 2A (striped), and 2X (white) MyHCs in L-CAD
The absence of 2B and EO MyHCs in horse laryngeal muscles is consistent with findings in other large animals—humans (Li et al. 2004), baboons (Rhee and Hoh 2008), and cattle (Toniolo et al. 2005). These results reflect the notion that variations in fiber types in laryngeal muscles among species represent an adaptation to optimize the control of airway resistance in relation to the respiratory frequency and metabolic rate of the animal (Rhee and Hoh 2008). 2B and EO MyHCs are found only in small mammals such as rats and rabbits, in which respiratory rates are high, and fast abductor and adductors are needed to regulate airflow during inspiration and expiration, respectively. In animals of intermediate size, such as cats (Rhee and Hoh 2008) and dogs (Wu et al. 2000b), 2B but not the faster EO MyHC is expressed. In larger animals, where respiratory rate is low, even 2B MyHC is no longer expressed.
Cross-sectional areas of the various types of fibers of the R-CAD and L-CAD and the R-TrA and L-TrA muscles in normal horses and those with subclinical RLN
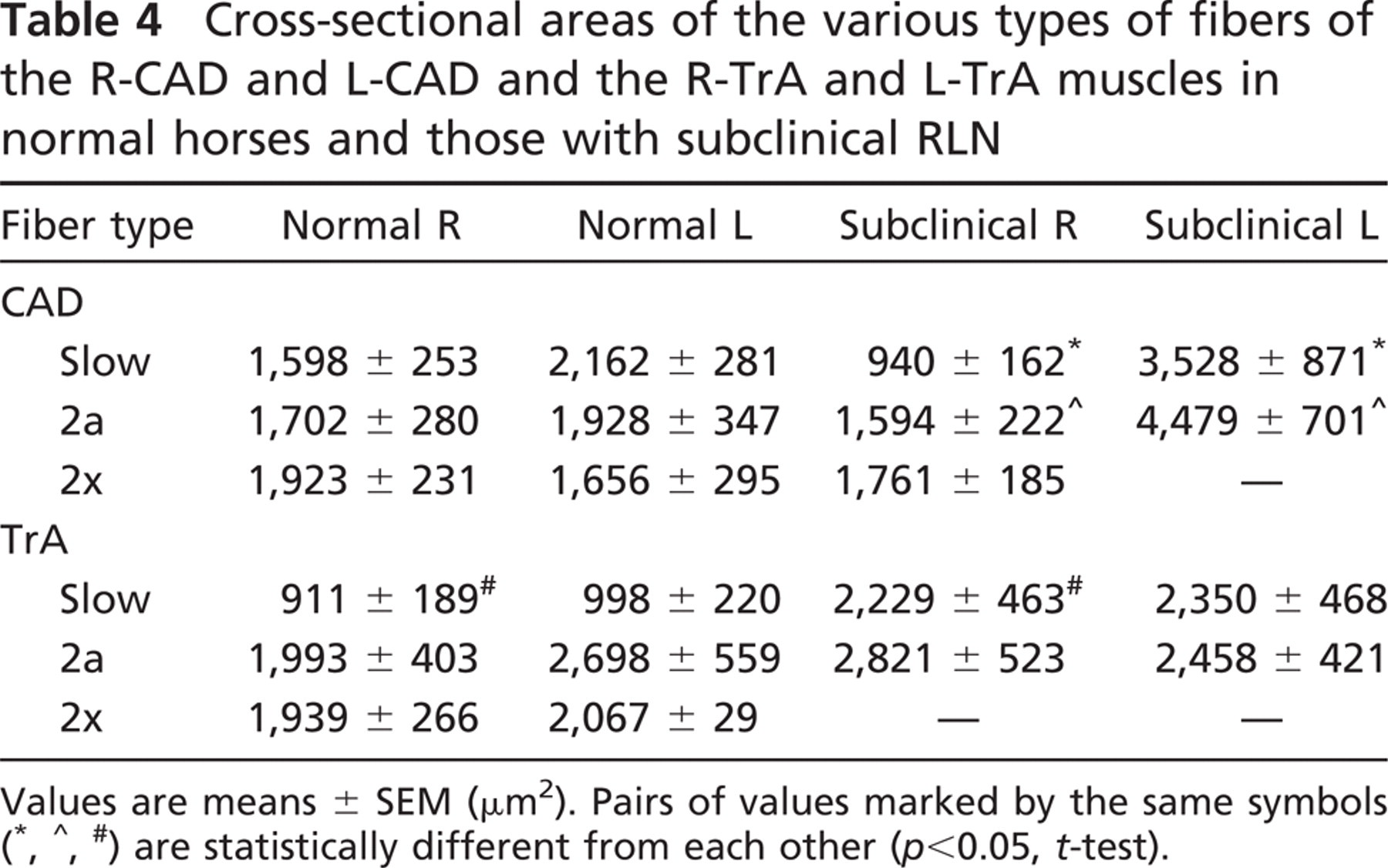
Values are means ± SEM (μm2). Pairs of values marked by the same symbols (∗, ∧, #) are statistically different from each other (p<0.05, t-test).
Muscle Fiber Types in the CAD and TrA of Horses in the Normal Group
Previous works on muscle fiber types in equine laryngeal muscles were based on histochemical analysis, and were only able to demonstrate the existence of two basic fiber types, fast (type 2) and slow (type 1). A minor, hybrid fiber type with histochemical characteristics of both fast and slow fibers was also present (Gunn 1972; Cahill and Goulden 1986b; Duncan et al. 1991a; Lopez-Plana et al. 1993; Adreani et al. 2006). The present work on apparently normal laryngeal muscles, using MyHCs as markers, revealed that the fast fibers could be subdivided into two distinct types, 2a and 2x, each with a distinct motor protein and hence speed of contraction. The three MyHCs expressed in equine laryngeal muscles are distributed among three pure fiber types (slow, 2a, 2x) and two hybrid fiber types (slow/2a and 2a/2x. Mechanical analysis of horse limb muscles shows that the three major fiber types provide a 10-fold range of contraction speed (Rome et al. 1990). Because laryngeal muscle fibers containing the same MyHCs as limb fibers contract at virtually the same speeds (D'Antona et al. 2002), motor units of horse laryngeal muscles are expected to have an equally wide dynamic range. In view of the importance of laryngeal muscles in respiratory control, the wide range of contraction speeds available to horse laryngeal muscles is probably very important functionally in dealing with the wide metabolic scope between rest and maximal activity in this athletic animal.
In both the CAD and TrA, the predominant fibers are the pure 2a and slow fibers, with a moderate proportion of the fastest 2x fibers and up to 10% of hybrid fibers. This profile of fiber types cannot be regarded as static throughout life, inasmuch as laryngeal muscles are subject to neural and hormonal control. Variations in fiber-type distribution can be expected in laryngeal muscles of normal horses, depending upon physiological factors such as the level of physical training and hormonal states.
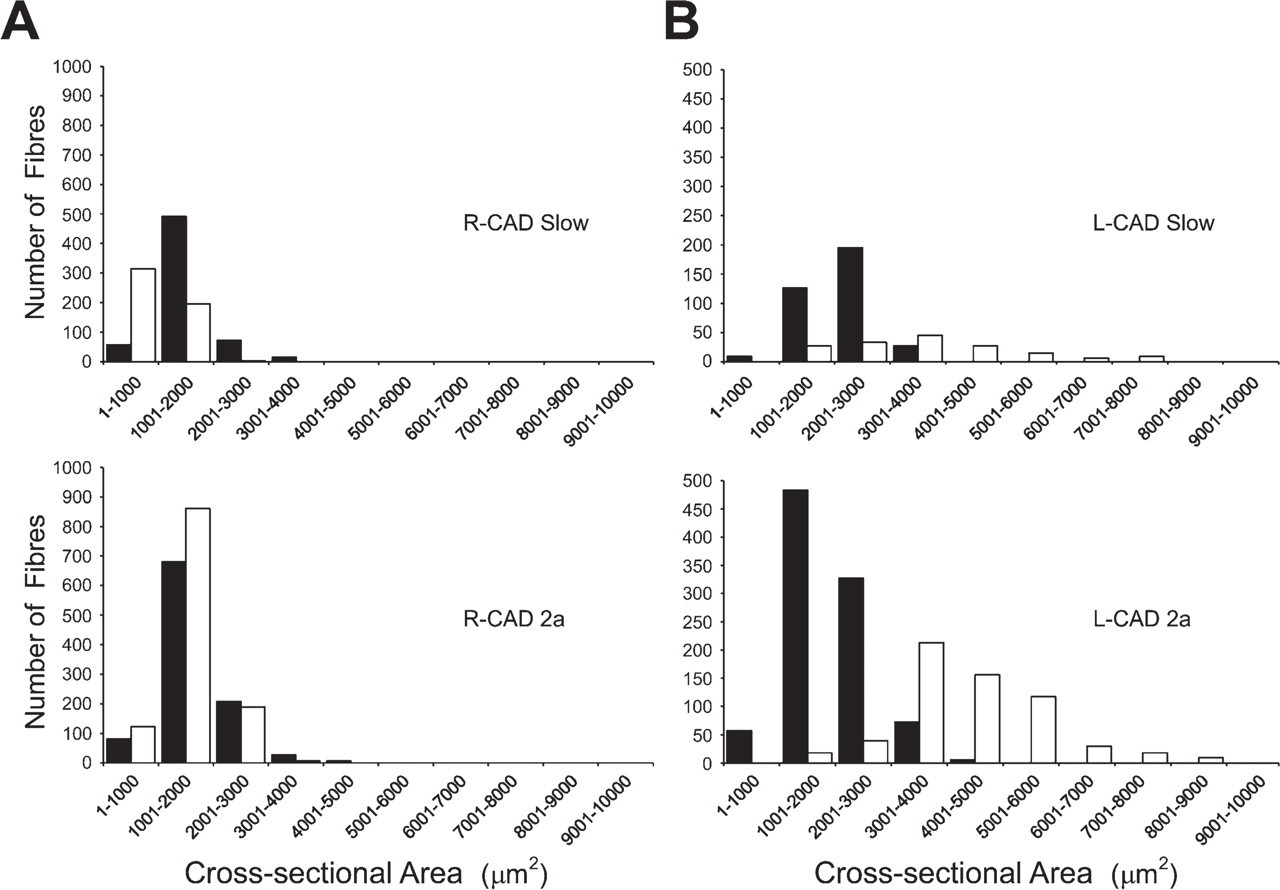
Size spectra of slow and 2a fibers in normal and subclinical R-CAD
Fiber-type Grouping in CAD and TrA Muscles in Horses With Subclinical RLN
In the subclinical group of horses, fiber-type grouping was more pronounced in the L-TrA than in the R-TrA, relatively mild in the L-CAD, but absent in the R-CAD. These results are consistent with earlier reports indicating that the left-sided laryngeal muscles are more severely affected in RLN than the right-sided muscles (Cahill and Goulden 1986b; Duncan et al. 1991a; Griffiths 1991), and that the adductors (including TrA) are more affected than the abductor (CAD) (Duncan et al. 1991a). This view is further supported by the virtual absence of 2x fibers in L-TrA and R-TrA muscles and the reduced abundance of these fibers in L-CAD compared with R-CAD (Table 3). The reduced proportion of slow fibers and the increase in 2a fibers in left-sided muscles relative to right-sided muscles (Table 3) further underscores the greater susceptibility of left-sided muscles to RLN. This fact may be due to the longer axons of left laryngeal motoneurons compared with the right. The function and integrity of motoneurons depend on axoplasmic flow, the bi-directional flow of organelles, signaling proteins, and mRNAs (Cosker et al. 2008; Goldstein et al. 2008). Disruption of axoplasmic transport of mitochondria in long axons has been implicated as a pathogenic factor in peripheral neuropathy (Baloh 2008). The very long axons of laryngeal motoneurons may make them susceptible to axoplasmic transport problems, and thus to RLN.
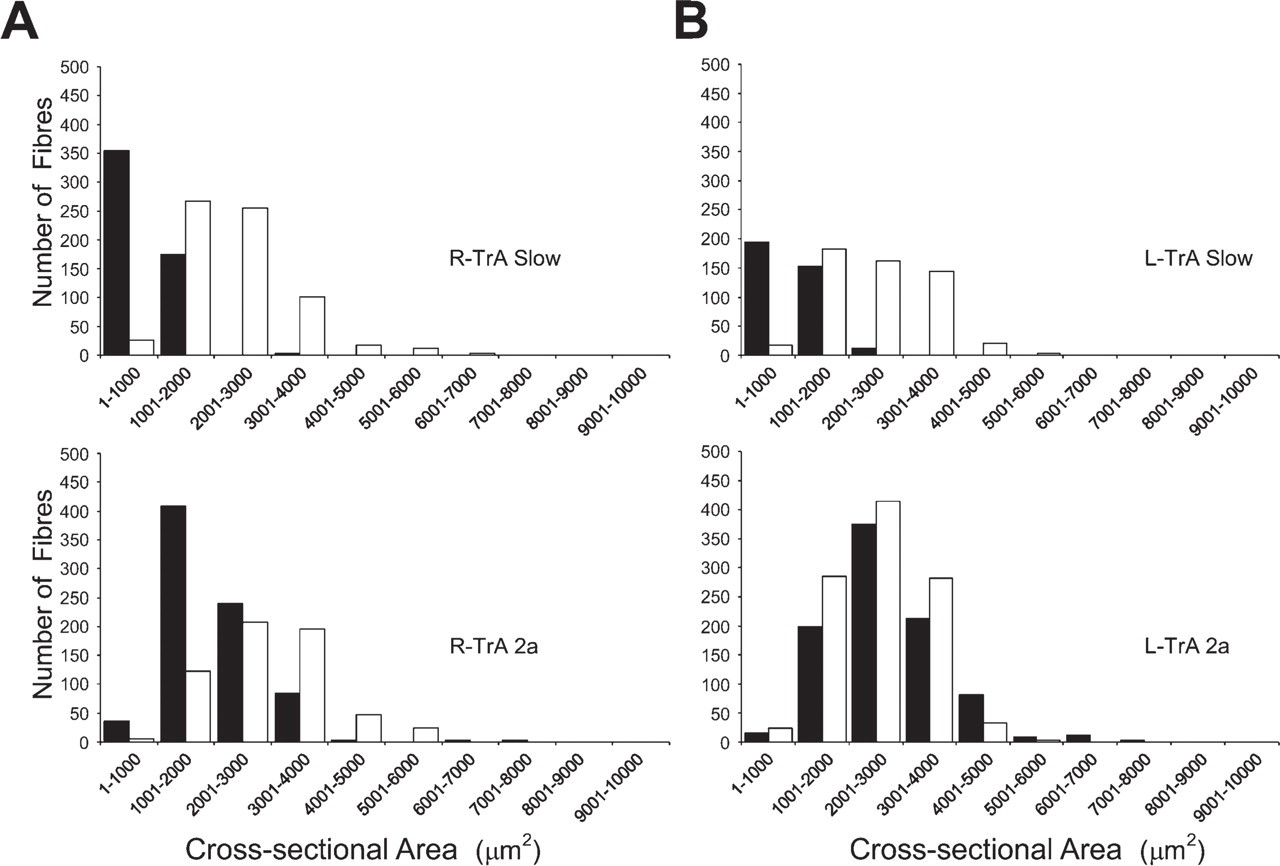
Size spectra of slow and 2a fibers in normal and subclinical R-TrA
In this study, muscles without fiber-type grouping are from horses that are 2 and 6 years old (and from a third horse of unknown age), whereas older (>10 years) horses showed fiber-type grouping (Table 1). Because denervation and reinnervation of muscle fibers have been shown to occur in rats and humans late in life (Luff 1998), the question arises as to whether the observed changes are merely a consequence of normal ageing. Although we cannot exclude this possibility, in view of the small sample size, it is unlikely, inasmuch as age-related fiber-type grouping only becomes significant rather late in life. Further, our finding of the correlation of fiber-type grouping with age is expected from what is known about RLN. RLN is known to affect even young horses, those from 2 to 3 years of age often showing clinical signs of the disease (Cahill and Goulden 1987; Cook 1988). Because RLN is progressive, at least in some cases (Dixon et al. 2002; Davidson et al. 2007), and fiber-type grouping is irreversible as long as motoneuron integrity remains, the probability of finding fiber-type grouping in laryngeal muscles from apparently normal horses necessarily increases with age. A study on a larger number of horses than used in the present work would be necessary to confirm that fiber-type grouping does occur in laryngeal muscles of young, asymptomatic horses.
Changes in Properties of CAD and TrA Fibers in Horses With Subclinical RLN
A novel and most prominent finding in equine laryngeal muscles showing fiber-type grouping (subclinical L-CAD, L-TrA, and R-TrA) was a marked decrease or even total elimination of 2x fibers, compared with their 11–20% abundance in corresponding normal muscles (Tables 2 and 3). Earlier works simply did not identify 2x fibers (Adreani et al. 2006). Loss of these fastest fibers would have reduced the range of shortening speeds of motor units in these muscles from 10- to only 4-fold (Rome et al. 1990). Although all horses in the current study were able to achieve synchronous and full abduction of the arytenoid cartilages during laryngoscopic examination at rest, it seems possible that loss of the fastest, 2x fibers would impact on airway protection and respiratory function during maximal exertion. However, laryngoscopic examination during strenuous exercise on a treadmill of horses that have grade 1 laryngeal function at rest does not typically reveal any compromise in laryngeal function (for a combined analysis of the four major studies, see Ducharme 2004; available at www.havemeyerfoundation.org/monograph.htm). Because adductors are more severely affected than the abductor (the CAD), it is also possible that a reduced range of contraction speeds of adductor muscles is present in subclinical horses. However, unlike humans, adductor deficit is not recognized to be of clinical significance in the horse.
Other significant changes in subclinical L-CAD and L-TrA, relative to muscles on the right side, were a decrease in the proportion of slow fibers, an increase in the proportion of 2a fibers (Table 3), and hypertrophy of these fibers. An almost total loss of slow fibers identified by low ATPase activity has been reported in laryngeal muscles of horses with gross atrophy of the left laryngeal muscles (Adreani et al. 2006), but this finding was not supported by immunohistochemical analysis of L-CAD muscle of horses with laryngoscopic evidence of laryngeal hemiplegia (Steel CM, unpublished data). However, if there is indeed a loss of slow fibers following on the heels of the loss of 2x fibers in subclinical RLN, as the current study suggests, this would drastically reduce the dynamic range of contraction speed of laryngeal motor units. Further, loss of the slow, fatigue-resistant fibers would reduce the capacity of laryngeal muscles to endure sustained periods of function.
The subclinical R-TrA was exceptional in that the loss of 2x fibers was associated with an increase in slow fibers relative to normal R-TrA, whereas the 2a fiber population did not significantly change (Figure 6C). However, these fibers were also hypertrophic relative to normal R-TrA.
Possible Mechanisms for Changing Laryngeal Muscle Fiber Properties in Subclinical RLN
The drastic loss of 2x fibers in muscles with subclinical RLN is an important novel finding in this work. A possible mechanism for this loss is that within the recurrent laryngeal nerve bundle, fibers innervating 2x muscle fibers have a greater susceptibility to damage in RLN. This results in the denervation of 2x fibers, their reinnervation by neighboring nerve fibers innervating 2a or slow fibers, and their consequent conversion into 2a or slow fibers by the neural influence (Pette and Vrbova 1985; Hoh 2005). This mechanism would account for fiber-type grouping but not for the shift of slow fibers toward 2a fibers. The selective loss of medium/large—diameter myelinated nerve fibers reported in the recurrent laryngeal nerves in RLN (Duncan et al. 1974,1978,1991b; Cahill and Goulden 1986a; Hahn et al. 2008) may be cited in support of this hypothesis. In the cat, large spinal motoneurons with large-diameter axons innervate large motor units with fast, fatigable 2x motor units, whereas small motoneurons with small-diameter axons innervate small, fatigue-resistant, slow motor units (Burke 1994). The large myelinated fibers in the recurrent laryngeal nerve of the horse may also innervate 2x fibers in laryngeal muscles. Large axons are more susceptible to damage by pressure (Burke et al. 1992). Their loss in RLN would thus explain the selective loss of 2x fibers. Future work on horse laryngeal motor units correlating nerve fiber diameter, conduction velocity, and innervated muscle fiber type, as defined by MyHC isoform expression, will contribute greatly to the understanding of RLN by testing this hypothesis.
Another possible hypothesis for the loss of 2x fibers may be that early axonopathy of nerve fibers prevented the transmission of high-frequency neural signals necessary to support the 2x fiber type. It is well known that limb muscle fiber types are under the control of neural impulse patterns (Pette and Vrbova 1985). There is evidence that laryngeal muscle fibers are also controlled by neural impulses in a frequency-dependent way (Carraro et al. 1988; Paniello et al. 2001; Rhee et al. 2004). In RLN, chronic demyelination of nerve fibers occurs. In experimental animals, it is well known that demyelination leads to prolongation of the refractory period, which impairs the capacity of the nerve fibers to transmit high-frequency impulses (Low and McLeod 1977). Frequency-dependent conduction block has also been demonstrated in humans with a nerve compression neuropathy, the carpal tunnel syndrome (Watson et al. 2006). It is thus possible that in subclinical RLN, there is impairment in the transmission of high-frequency bursts of impulses that support 2x muscle fibers, while allowing lower frequency bursts to pass through. This could lead to the transformation of 2x fibers into 2a fibers. Such a mechanism could account for an enhanced proportion of 2a fibers relative to slow fibers, but will not per se explain fiber-type grouping, which requires denervation and reinnervation.
Loss of slow fibers may arise from either their selective denervation or from slow-to-fast fiber transformation. There is no evidence in RLN for a selective loss of small myelinated fibers of the recurrent laryngeal nerve (Duncan et al. 1991b) that are likely to innervate slow muscle fibers. Slow-to-fast muscle fiber transformation is well known to occur in limb muscles under conditions of altered impulse activity to slow muscles, such as spinal cord transection (Hoh et al. 1980; Lieber et al. 1986), zero gravity (Baldwin et al. 1994; Talmadge et al. 1996), and hindlimb suspension (Talmadge et al. 1996; Stevens et al. 2000). A similar fiber-type transformation can occur in RLN if mild axonopathy of nerve fibers innervating slow muscle fibers impairs their ability to sustain the transmission of low-frequency impulses for the prolonged periods needed to support slow fibers, while not affecting the short bursts of activity needed to support 2a fibers. In the experimental situations of slow-to-fast muscle fiber transformations cited above, transformed fast fibers undergo atrophy rather than the observed hypertrophy of laryngeal muscles in subclinical RLN (Figures 4, 5, 7, and 8; Table 4). In limb muscles, it is well known that an increase in functional load leads to the hypertrophy of their fibers (Swynghedauw 1986; Goldspink 2005). The presence of hypertrophy in laryngeal muscle fibers with subclinical RLN suggests that there has been an increase in functional load, which may be due to an increase in tissue stiffness or the loss of some muscle fibers from failure of reinnervation. Hypertrophy is a useful compensatory adaptation, and no doubt helps the horse to remain subclinical in spite of the presence of neuromuscular pathology.
In summary, the observed changes in laryngeal muscles showing fiber-type grouping are presumably due to subclinical RLN, and are likely to be due to multiple pathophysiological mechanisms, including selective denervation of 2x muscle fibers and their reinnervation and conversion into 2a or slow fibers, corruption of specific neural impulse patterns that regulate specific muscle fiber types, loss of muscle fibers, and compensatory hypertrophy. From the changes in properties of muscle fibers we observed in laryngeal muscles of horses with subclinical RLN, one could use basic principles to predict the fiber-type characteristics in equine laryngeal muscles with frank RLN. With further denervation, 2x fiber loss and conversion to 2a or slow fibers will be complete, and both types of surviving fibers will hypertrophy even further. When denervation progresses to the extent that surviving nerve fibers can no longer maintain a functionally adequate population of muscle fibers, regenerative myogenesis will be stimulated (Shinners et al. 2006), and many muscle fibers will become chronically denervated and will undergo drastic atrophy. Denervated fibers are likely to express multiple MyHCs, including embryonic and fetal/developmental isoforms (Jakubiec-Puka et al. 1990; Janmot and d'Albis 1994; Shinners et al. 2006). Some of the predicted characteristics—hypertrophy of fast and slow fibers and presence of atrophic fibers—have already been observed in muscles with overt RLN using standard histochemistry (Adreani et al. 2006). These authors identified the surviving fast fibers as type 2b. Our results suggest that they should be type 2a.
