Abstract
The c-kit receptor (KIT) and its ligand, stem cell factor (SCF), represent one of the key regulators of testicular formation, development, and function and have been extensively studied in various animal models. The present study was undertaken to characterize the pattern of localization and expression of c-kit in normal adult human testis. Immunohistochemical analysis showed that KIT is expressed in the cytoplasm of spermatogonia, acrosomal granules of spermatids, and Leydig cells. Interestingly, a rather heterogenous pattern of expression of the protein along the basement membrane was observed. Intense protein localization in spermatogonia was detected in stages I–III, whereas low expression was observed in stages IV–VI of the seminiferous epithelium, indicating that the expression of the molecule was stage specific. In situ hybridization studies revealed that the transcripts of the gene were also localized in a similar non-uniform pattern. To the best of our knowledge, such a stage-specific expression of KIT has not been reported previously in the human testis. The results of the present study may expand current knowledge about the c-kit/SCF system in human spermatogenesis.
T
Expression studies indicate that KIT is developmentally regulated in male germ cells during both fetal and postnatal development (Mauduit et al. 1999; Prabhu et al. 2006) and has been implicated in the transduction of extracellular signals that control cell proliferation, survival, and differentiation (Ullrich and Schlessinger 1990; Blume et al. 1991). In the adult mouse testis, KIT is mainly expressed by spermatogonia (Manova et al. 1990; Sorrentino et al. 1991) and Leydig cells (Motro et al. 1991). A 5.5-kb mRNA transcript is highly expressed in spermatogonia and is significantly reduced in meiotic spermatocytes. A truncated transcript of 3.5 and 2.3 kb is found in haploid germ cells (Rossi et al. 1992). Correspondingly, an altered truncated form of protein, tr-kit, is expressed in round spermatids and acrosomal heads of sperm (Albanesi et al. 1996). Expression studies in the mouse indicated that KIT may have a role in regulation of spermatogonial proliferation and maturation of round spermatids (Sorrentino et al. 1991; Albanesi et al. 1996).
The observation that KIT is involved in the development of mouse spermatogonia has led to the speculation that loss of KIT may be a cause of infertility in a subset of infertile men. Indeed, two studies by the same group reported decreased KIT expression in testis of infertile men as compared with fertile (Sandlow et al. 1996; Feng et al. 1999). The pattern of cellular localization of the receptor, though, has not been adequately described. Controversies exist about the cellular localization of the receptor in the human testes. Lammie et al. (1994) reported KIT only in the interstitial testicular cells, and no reactivity to the spermatogonia, in contrast to others who have reported the presence in germ cells and Leydig cells (Strohmeyer et al. 1995; Sandlow et al. 1996,1997).
Thus, the present study was undertaken with the aim of investigating the localization and expression of KIT in the normal adult human testis. Such an exploration is required to provide insights into the role of the receptor and its ligand in the regulation of human spermatogenesis.
Materials and Methods
Subjects
The present study was approved by the Ethics Committee of the National Institute for Research in Reproductive Health and King Edward Memorial Hospital (KEM). Testes were collected from five men (57–69 years old) undergoing therapeutic orchidectomy as part of their management for malignant prostate carcinoma at KEM Hospital, after appropriate written informed consent. None of the men received antiandrogens before surgery.
Tissue Processing
The tissue, upon removal during surgery, was collected and transported to the laboratory in NaCl solution (0.9% w/v) on ice and was immediately preserved in different fixatives including (1) Bouin's, (2) 70% ethanol, (3) neutral buffered formalin (NBF) at room temperature, and (4) NBF at 4C. The samples were processed for paraffin sections following standard protocols. Sections 5 μm thick were cut and processed for hematoxylin and eosin staining, immunostaining, and in situ hybridization. Three out of five testicular tissue samples were selected for further analysis because they exhibited a normal homogeneous histological appearance and the majority of seminiferous tubules and interstitial spaces appeared normal. At least two paraffin blocks from each of the testes were examined histologically.
RT-PCR
Total RNA was extracted from human testicular tissue using TRIZOL (Invitrogen; Carlsbad, CA) according to the manufacturer's instructions. After the RNA was incubated with 5000 units of RNase-free DNase (DNase 1; Amersham Biosciences, Piscataway, NJ) at 37C for 30 min, it was used for RT-PCR. First-strand cDNA was synthesized using the Omniscript RT Kit according to the manufacturer's instructions (Qiagen; GmbH, Germany). Briefly, 1 μM random hexamer primers, 0.5 mM deoxynucleotide triphosphates (dNTPs), 5 units of Moloney murine leukemia virus RT, and 10 units of RNasin in a single-strength reaction buffer were used to synthesize the first strand of cDNA in a total volume of 20 μl. The primers and the RNA were incubated at 70C for 2 min and then at 37C for 1 hr. cDNA mix (2 μl) was amplified using 10 pmol of each primer as described (Table 1), 1 unit of Taq DNA polymerase (Fermentas Life Sciences; Vilnius, Lithuania), 1.5 mm MgCl2, and 10 mM dNTPs in a 25-μl reaction volume in a G STORM thermocycler (Gene Technologies; Braintree, UK). Amplification was carried out for 35 cycles, with each cycle consisting of denaturation at 94C for 1 min, annealing at the specified temperature for each set of primers (Table 1) for 1 min 30 sec, and extension at 72C for 2 min. The products were analyzed on 1.5% agarose gel stained with 0.5 μg/ml ethidium bromide (Bangalore Genei; Bangalore, India) and visualized under ultraviolet transillumination. The product size was approximated using a 100-bp DNA ladder (Bangalore Genei). The negative control did not include cDNA in the reaction mixture. The primers used for amplification of c-kit were commercially synthesized (Numex Chemicals; Mumbai, India), and the sequences are shown in Table 1.
Immunohistochemical Localization
The 5-μm-thick paraffin-embedded sections were fixed on grease-free slides, and immunohistochemical localization of KIT was performed using the Super Sensitive Polymer-HRP Detection System according to the manufacturer's instructions (BioGenex; San Ramon, CA). In brief, the sections were deparaffinized in xylene and rehydrated through a graded ethanol series. Epitope retrieval entailed bathing the hydrated sections in 0.01 M Tris buffer, 0.001 M EDTA, pH 9.0, and heating the sections for 20 min using a water bath set to 94C or treating the sections with sodium citrate buffer (10 mM sodium citrate), pH 6.0, at high power for two cycles of 5 min each in a microwave oven. The sections were allowed to come to room temperature before continuing with the immunostaining procedure. Endogenous peroxidases were inactivated using Peroxide Block (provided in the kit), then blocking was carried out using Power Block (provided in the kit) for 45 min at room temperature, followed by an overnight incubation at 4C with the c-kit polyclonal antibody (1:700 diluted) that recognizes the carboxy cytoplasmic domain of the receptor (catalog no. A4502; DakoCytomation, Glostrup, Denmark). The slides were incubated for 20 min at room temperature in Poly Enhancer (provided in the kit) and then with Poly HRP for 30 min at room temperature. After stringent washings in PBS, the slides were visualized by treatment with 0.05% diaminobenzidine (Sigma-Aldrich; St Louis, MO) in PBS with 0.06% hydrogen peroxide for 10 min. The sections were briefly counterstained with hematoxylin, dehydrated, coverslipped with DPX and examined with a Nikon 90i microscope (Nikon; Tokyo, Japan). For negative controls, the primary antibody was replaced with goat serum. Immunostaining of sections was repeated at least three times.
Sequences of the primers/probes used for detection of kit

In Situ Hybridization
Testicular sections of 5-μm thickness were placed on 3-aminopropyltriethoxysilane-coated glass slides, dried at 37C for 2 hr, and stored at room temperature until use. The sections were hydrated, washed, and incubated in 2X sodium saline citrate (SSC; 1X SSC containing 0.15 M NaCl and 0.015 M sodium citrate, pH 7). Prehybridization was carried out in prehybridization cocktail [50% formamide, 10% dextran sulfate (Sigma-Aldrich), 0.25% yeast tRNA, and 0.25% herring sperm DNA (Roche Diagnostics GmbH; Mannheim, Germany), 4X SSC] at room temperature for 30 min in a moist chamber. c-kit oligoprobe (Table 1), was tail labeled with digoxigenin (dig) as detailed previously (Shah et al. 2005). The probe was diluted at a concentration of 5 pmol/μl in prehybridization cocktail and applied to the sections. Hybridization was carried out at 42C overnight in a moist chamber. The next day, the slides were sequentially washed at room temperature in 4X, 2X, and 1X SSC and equilibrated in buffer 1 (0.01 M Tris/HCl, pH 7.4, with 20 mM NaCl) for 5 min at room temperature. Blocking was performed with 3% normal sheep serum containing 0.3% Triton X-100 for 2 hr at room temperature. Bound probe was visualized by incubation with alkaline phosphatase-conjugated anti-dig antibody (Roche Diagnostics) (diluted 1:500 in blocking solution) and subsequent substrate detection using nitroblue tetrazolium salt/5-bromo-4-chloro-2-indoyl phosphate, according to the manufacturer's instructions (Roche Diagnostics).
Western Blotting
Testicular tissue was homogenized in buffer containing 20 mM phosphate buffer, 150 mM NaCl, 50 mM sodium fluoride, 0.5 mM sodium orthovanadate, 2% v/v Triton X and Complete mini protease inhibitor (Roche Diagnostics). The homogenate was centrifuged at 12,000 × g for 10 min, and the supernatant was collected. Aliquots of the preparation were stored at −20C, and the concentration of the total protein was estimated using Bradford's reagent (Sigma-Aldrich). Electrophoresis was carried out on 10% SDS-PAGE gel under reducing conditions (Laemmli 1970). Briefly, the samples were heated at 95C for 5 min with sample buffer and chilled immediately. Each lane was loaded with 40 μg of protein along with molecular mass marker (Amersham Biosciences). The separated proteins were transferred on a nitrocellulose membrane (Hybond-C Extra; Amersham Biosciences), followed by blocking in 5% non-fat dried milk powder in Tris-buffered saline (TBS) at room temperature for 1 hr. The blots were further incubated at 4C for 18–20 hr with c-kit polyclonal antibody diluted 1:250 in TBS. The blots were washed with TBS three times for 10 min and then incubated for 2 hr at room temperature with horseradish peroxidase-conjugated goat anti-rabbit secondary antibody (Sigma-Aldrich) diluted 1:35,000 in TBS, washed with TBS five times for 10 min each, and detected using the chemiluminescence detection system, ECL-Extra Sensitive Kit (Amersham Biosciences), followed by exposure to X-ray film.
Data Acquisition and Analysis
Staging of the seminiferous epithelium was based on the scheme described by Clermont (1963), but was significantly aided by the stage diagram published by Shah et al. (2005). After ensuring reproducibility of results with the immunostaining protocol, four consecutive sections and four random sections from each of the testes were immunostained. Care was taken to ensure that all the sections were treated in an identical fashion and that they were subjected to identical times in all of the steps during immunostaining. At the completion of the immunostaining protocol, staining pattern was evaluated and scored. To minimize edge artifacts possibly interfering with the scoring of the immunostaining, only tubules at the center of the testes were evaluated. At least ten tubules were counted from each of the testicular sections that met these criteria. Because it was difficult to consistently distinguish between stages I and II and between stages IV and V, data from these stages were pooled. In addition, results from stage VI were pooled with those from stages IV and V. Images were acquired using an Evolution VF CCD camera (MediaCybernetics, Inc.; Bethesda, MD) in a Nikon 90i microscope. The images were assessed using ImagePro software (MediaCybernetics).
Results
Detection of c-kit mRNA by RT-PCR
RT-PCR analysis revealed the presence of a single band of the expected size (205 bp) (Figure 1). No bands were observed when RT was omitted from the reaction mixture. The PCR products were sequenced, and a similarity search revealed complete homology with conventional kit.
Localization of KIT in Human Testis
To rule out nonspecific staining and fixation artifacts, the experiment was repeated using different fixatives and antigen retrieval protocols [Tris EDTA (TE), pH 9.0, at 94C using water bath, and sodium citrate, pH 6, using microwave]. Although good immunostaining was observed in NBF-fixed tissues, nonspecific staining was observed in ethanol-fixed tissues, and no staining was observed in Bouin's-fixed tissues (results not shown). Optimal staining was observed in sections of tissues fixed with NBF at 4C using TE, pH 9.0, for epitope unmasking. Hence, these conditions were followed for the rest of the study. The specificity of the polyclonal antibody was checked using Western blotting. A single band of ∼145 kDa was detected. No bands were visible in the case of negative controls, where the antibody was replaced with an isotype control (Figure 2).
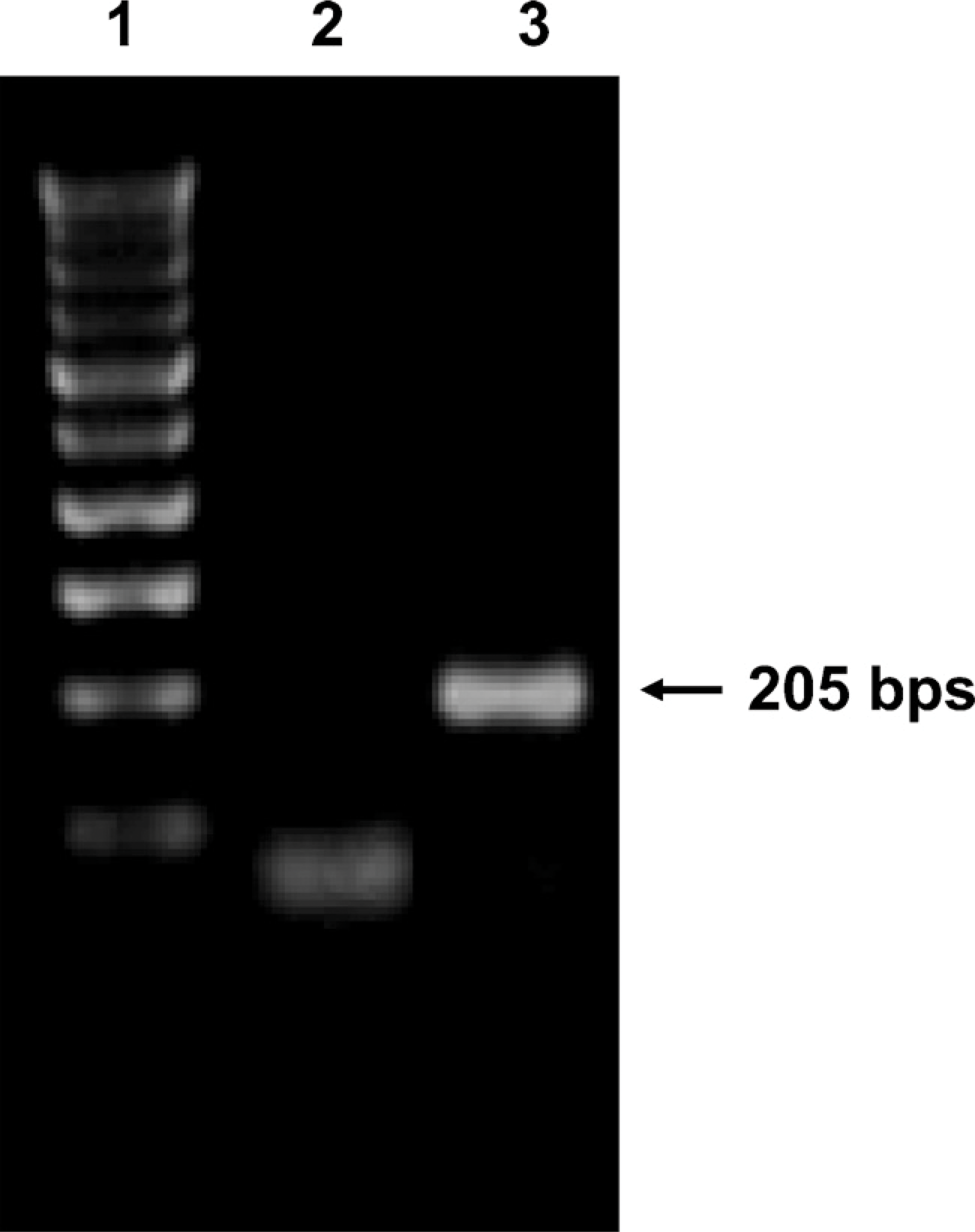
RT-PCR amplification of kit in the adult human testis. Lane 1: 100-bp ladder. Using specific primers for kit (Table 1) a band of 205 bp was detected in Lane 3. Lane 2: negative control.
KIT was localized in the cytoplasmic membrane and the cytoplasm of spermatogonia of the adult testis (Figures 3A and 3B). KIT was also detected in the peritubular Leydig cells and the acrosomal granules of the round spermatids (Figures 3G and 3H). Somatic Sertoli cells and the differentiated germ cells, such as the spermatocytes and elongating spermatids, were consistently negative. No staining was detected in the negative control sections, where the primary antibody was replaced by isotype controls (Figure 3C). However, KIT immunopositivity in the spermatogonia lining the basement membrane was heterogenous. Some areas of the seminiferous tubule displayed weak or no staining, whereas other areas within the same tubule were stained intensely (Figure 3A). This pattern of KIT staining was consistently observed in all testicular specimens examined.
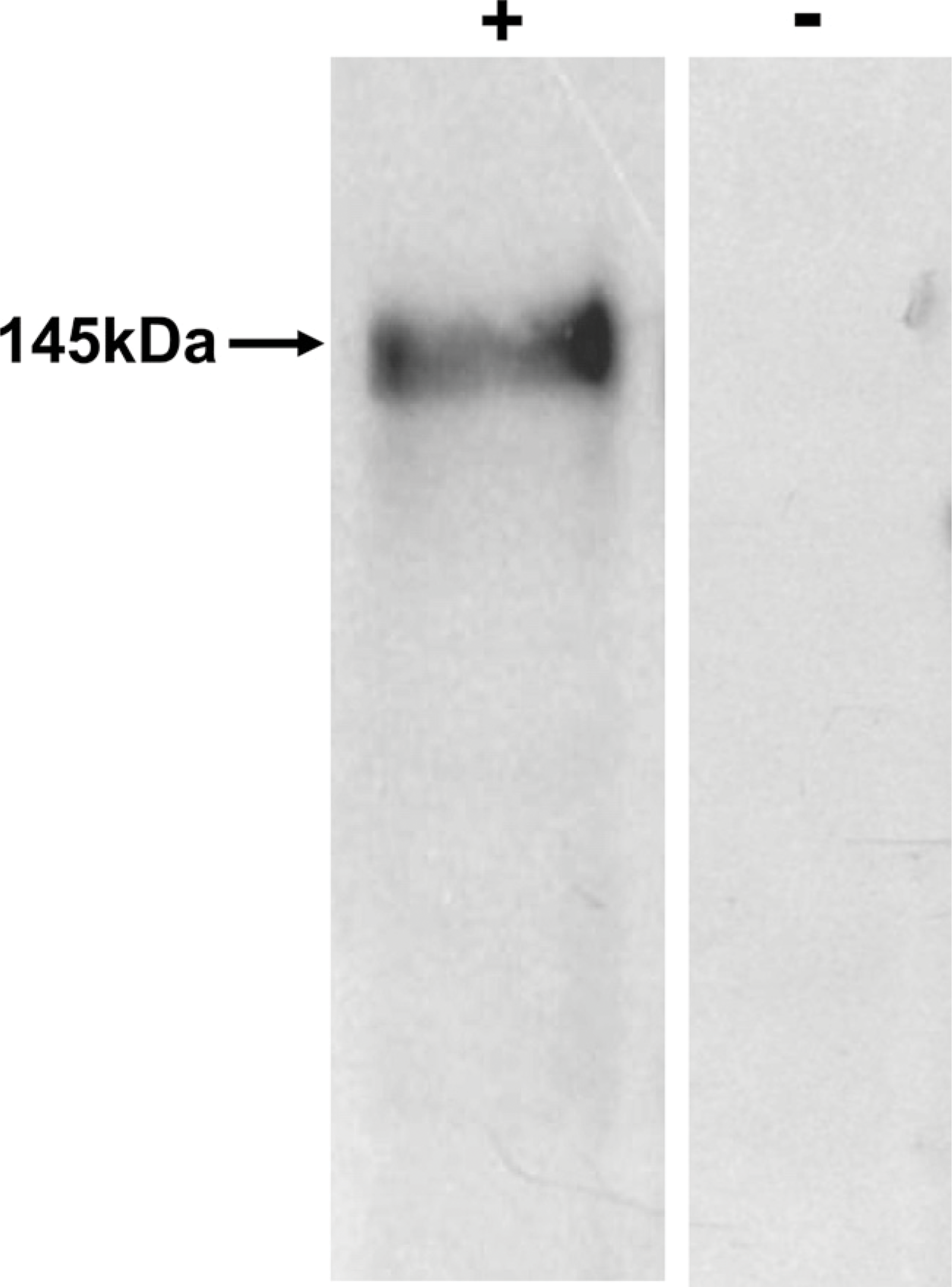
Western blot analysis to detect KIT in the human testis. An expected band of ∼145 kDa was detected, confirming the specificity of the polyclonal primary antibody. No signal was detected in the negative control.
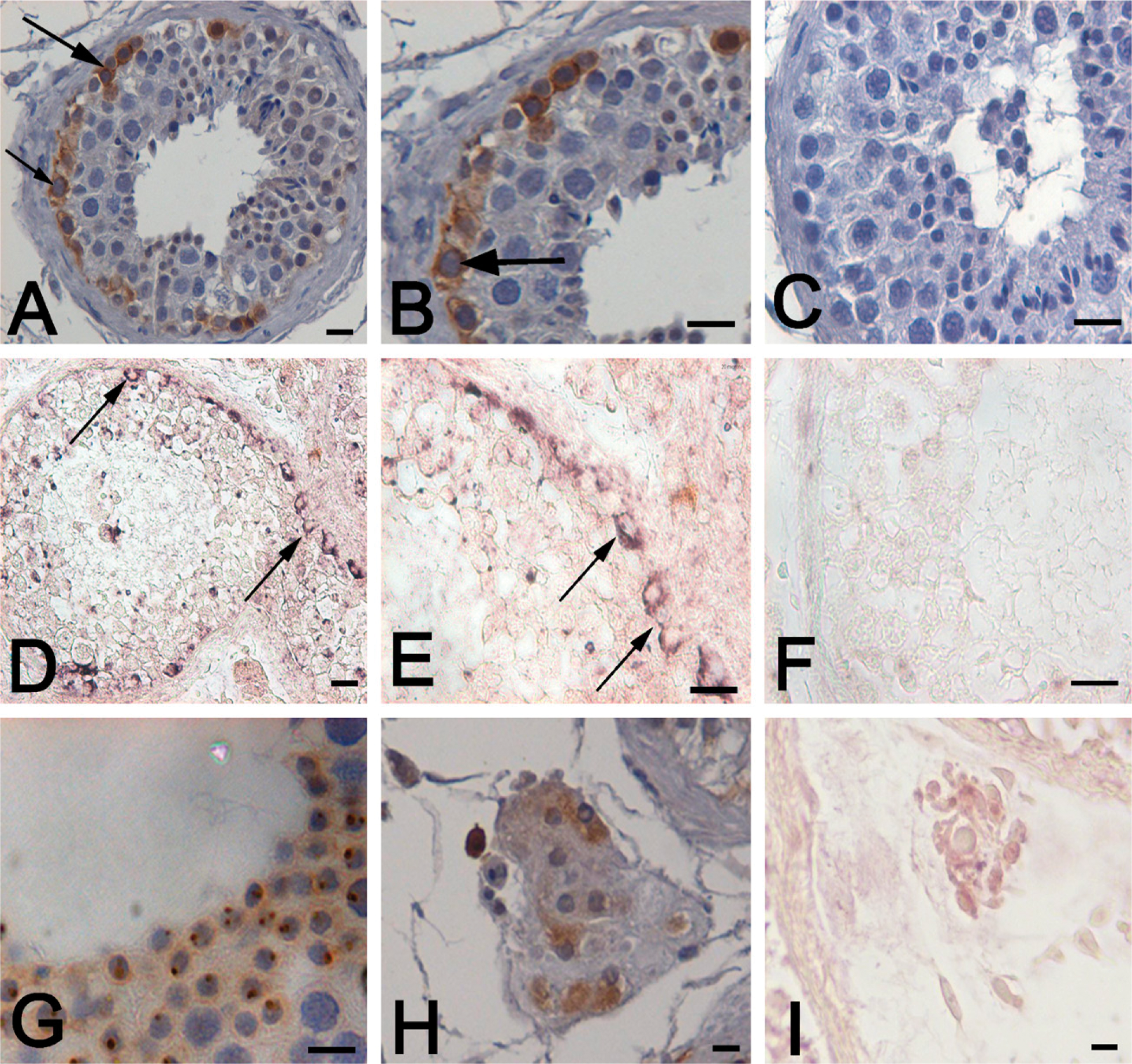
Localization of KIT in the adult human testis. Immunohistochemical localization in human testicular tissue (
Stage-specific Expression of KIT During Spermatogenesis
Within the spermatogonia, maximal expression of c-kit was observed at stages I–III (Figures 4A–4D). A dramatic decline in intensity was observed in stages IV–VI (Figures 4E and 4F). However such dynamic variation in KIT localization was not observed in the Leydig cells and in the acrosomal granules of the round spermatid, where it was constantly expressed. No nuclear staining was observed in any of the stages.
In Situ Localization of kit Transcripts in Human Testis
Non-radioactive in situ hybridization was carried out on testicular sections to confirm the stage-specific localization of kit in the testis. Strong positive signals were detected in the spermatogonia (Figures 3D and 3E). The Leydig cells generated a weaker signal (Figure 3I). In some areas of the seminiferous tubule, intense signals were generated, whereas other areas within the same tubule stained weakly, revealing a heterogenous pattern similar to that seen in the immunolocalization studies (Figure 3D). This pattern of localization was consistently observed in the sections of all the samples analyzed. No signals were detected when a sense probe was used for hybridization (Figure 3F).
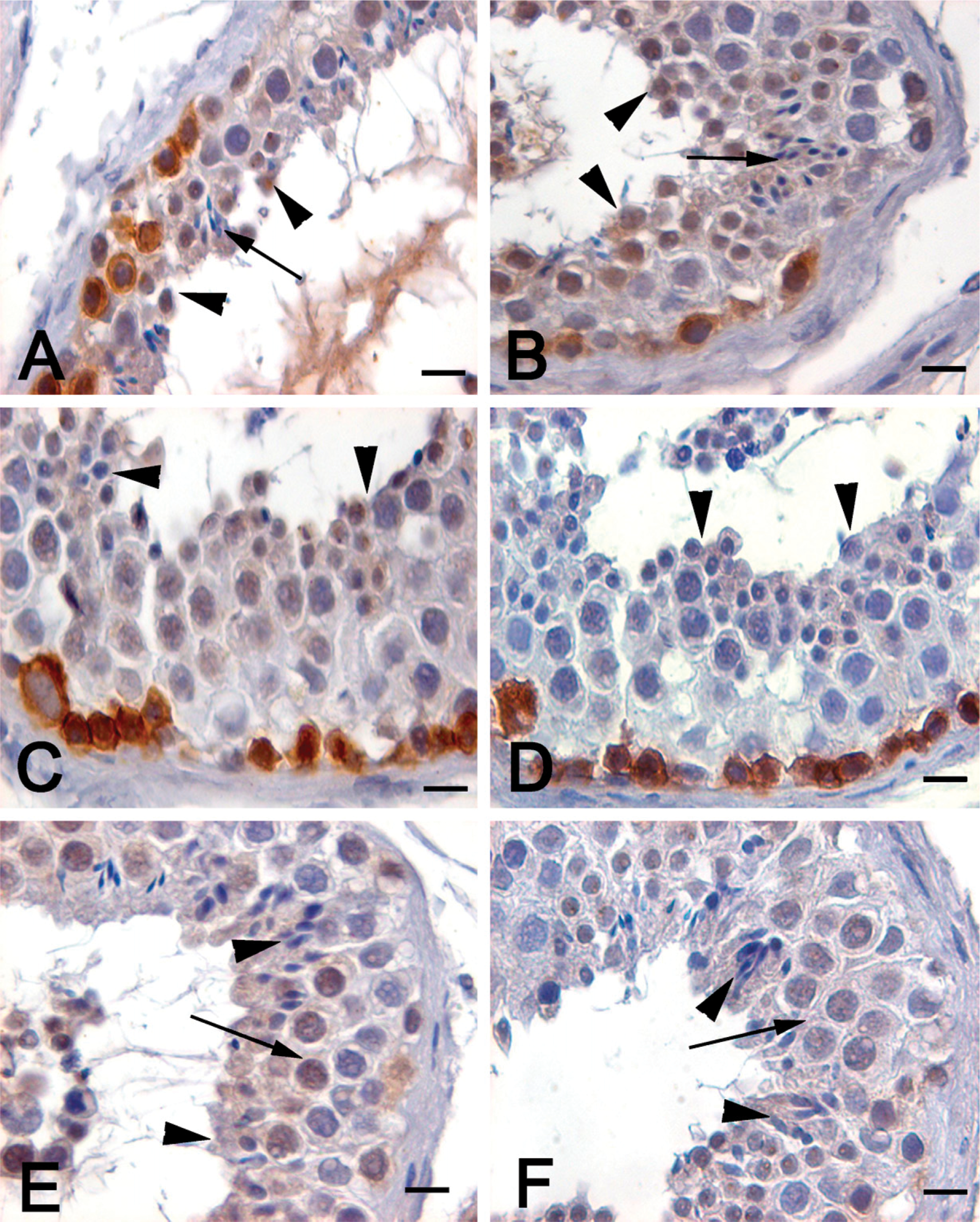
Stage-specific immunolocalization of KIT in the human testis. Intense staining for KIT was observed in stage III of the human seminiferous epithelium, characterized by the presence of only round spermatids (arrowheads) and no elongating/mature forms (
Discussion
The results of the present study demonstrate that c-kit mRNA and protein are expressed in the spermatogonia of the human testis and that this expression is regulated in a stage-specific manner. To the best of our knowledge, such a stage-specific expression of c-kit has not been reported previously in the human testis.
kit is a proto-oncogene and is expressed in the subset of germ cells, melanocytes, and hematopoietic cells of several mammalian species (Rossi et al. 2000; Sette et al. 2000). It has been extensively studied and detected in mouse (Yoshinaga et al. 1991), rat (Dym et al. 1995), and hamster and monkey testis (Von Schonfeldt et al. 1999). In agreement with these observations, we consistently detected the presence of kit transcript (205 bp) in the human testis by RT-PCR. A band of the expected molecular mass (145 kDa) was also detected in the protein lysate isolated from normal human testis, when probed with a specific polyclonal antibody against KIT.
We next tested the cellular localization of the protein in paraffin-embedded testicular sections. A series of fixatives were tested, in combination with two antigen-unmasking protocols (TE, pH 9.0, and sodium citrate, pH 6) to detect the in situ localization of the protein. In our hands, fixing the tissue in NBF at 4C for 48 hr with antigen retrieval in TE at pH 9.0 gave the most consistent and optimal staining. Such fixative dependence of immunogenicity for several proteins has been reported previously (Jacobsen et al. 1980; Berod et al. 1981; Peschke et al. 1986; Saed 2008). Factors such as epitope masking and cross-linking with other proteins may contribute to such observations. Using an antibody directed against the C terminus, intense immunoreactivity of KIT was detected in the membrane and cytoplasm of spermatogonial cells located at the base of the seminiferous tubule and in peritubular Leydig cells. These observations are in contrast to two previous reports, where KIT expression was reported in all the germ cell types, including the spermatocytes and spermatids, along with Sertoli cells (Strohmeyer et al. 1995; Bokemeyer et al. 1996). However, our results confirm the observations, where KIT has been immunolocalized to the spermatogonia and Leydig cells of several mammalian species, including human (Yoshinaga et al. 1991; Dym et al. 1995; Sandlow et al. 1996,1997; Stoop et al. 2008). In addition to spermatogonial cells, strong immunoreactivity was also observed in the acrosomal region of the round spermatids. A postmeiotic staining of KIT in the developing acrosome has been reported in the mouse (Albanesi et al. 1996) and in humans (Sandlow et al. 1996). Earlier, antibodies against the extracellular domain of the KIT receptor stained only spermatogonia, but not spermatids (Yoshinaga et al. 1991). However, a polyclonal antibody raised against the cytoplasmic terminal part of the KIT receptor detected staining of the developing spermatid, in addition to a clear membrane staining in spermatogonia. This form of KIT is believed to be truncated, arising out of an altered haploid cell transcript containing part of the cytoplasmic domain but not the extracellular or transmembrane domains. Our results also imply that the C terminus region of KIT seems to be retained in the human diploid spermatogonia and the haploid cells. The truncated KIT might play a role in sperm cell function during or after fertilization (Albanesi et al. 1996).
Although the presence of KIT in the spermatogonia is a well-accepted observation, in this study, we observed that not all the spermatogonia were uniformly stained for KIT. A rather heterogenous pattern of KIT-positive cells was observed in the immunostained testicular sections, with some areas within the same tubule staining more than others. This peculiar pattern of KIT staining was consistently observed in all testicular sections examined. Unlike in rodents, the human testis has a multistage organization of spermatogenic stages per tubular cross section. There are atypical cellular associations, owing to missing spermatogonia, spermatocytes, or spermatids, and also to intermingling of associations, leading to the chaotic nature of the human seminiferous epithelium (Ehmcke and Schlatt 2006; Amann 2008). Because the expression of KIT also appeared to be chaotic in most sections examined, we hypothesize that KIT expression in the human spermatogonial cell may be stage specific. Indeed, analysis of four consecutive sections and four random sections from each of the testes revealed that most c-kit-positive cells were associated with stages I–III, whereas stages IV–VI were generally devoid of KIT-positive spermatogonial cells. To the best of our knowledge, such stage-specific expression of KIT in the spermatogonial cells has not been reported previously.
To investigate further, if this stage-specific expression of KIT is as a result of differential transcription of the gene or owing to differential utilization of the mature protein, we performed in situ hybridization. kit-positive cells were localized to the spermatogonial cells at the base of the semniferous tubules. These kit-positive cells were also heterogeneously distributed across the tubule, similar to that observed for the protein. Although we were unable to determine the specific stages where kit-positive cells were present (because the sections were not counterstained), the chaotic pattern of kit-positive cells, like that of the protein, prompts us to believe that the transcript of c-kit in the spermatogonial cell seems to be dependent on the stage.
Stage-specific expression of several proteins has been reported in the rodent and human testis and supports our observations (Vincent et al. 1998; Hakovirta et al. 1999; Schrans-Stassen et al. 1999; Zhang et al. 2004; Shah et al. 2005). Similar data in the mouse report maximal staining of KIT in spermatogonia A at stages IX–I (Schrans-Stassen et al. 1999). The mouse seminiferous epithelium exhibits 12 stages of spermatogenesis, of which stages IX through stage I (i.e. stages IX, X, XI, XII, and I) report maximal staining of c-kit in spermatogonia. Because these stages are believed to be analogous to stages I–III in human seminiferous epithelium, it is likely that the regulation of KIT expression in the spermatogonial cells in the mouse and human may be identical. It will be of interest to compare the promoters of the mouse and human KIT, to identify the common regulatory units that may be responsible for such stage-specific expression of this factor.
What could be the possible role of such stage-specific expression of KIT in the germ cells? It is hypothesized that in the normal seminiferous epithelium, the Apale spermatogonia proliferate from epithelial stage I until about stage III, during which time they produce many Apale and B spermatogonia. In stage IV, the B spermatogonia differentiate into early spermatocytes and enter meiosis (Clermont 1963; de Rooij and Russell 2000; Amann 2008). Because KIT-positive cells are observed at stages I–III in human testis, we hypothesize that Apale and B spermatogonia gradually develop from KIT-positive cells in stages I–III into KIT-negative cells near the end of the period of proliferation, around stage IV. The Apale spermatogonia continue to persist, but undergo downregulation of KIT during the period of meiosis in stages IV–VI, at which time they prepare themselves to differentiate. The downregulation of KIT may allow irreversible differentiation of spermatogonial cells into spermatocytes. In rodents, KIT receptor expression was observed in types A1–A4 differentiating spermatogonia during the proliferative stages, and a marked reduction of expression was reported in the spermatogenic cells entering meiosis (Sorrentino et al. 1991; Yoshinaga et al. 1991; Rossi et al. 1992). This pattern of upregulation of KIT during spermatogonial proliferation and its subsequent downregulation at the onset of meiosis appears to be conserved across species and is suggestive that KIT may be associated with regulation of proliferation and differentiation of spermatogonia. Indeed transient silencing of KIT in mouse spermatogonial cells by siRNA led to an induction of cell cycle arrest and increased apoptosis (Sikarwar and Reddy 2008). In humans, too, a decrease in the expression of KIT receptor has been reported to be associated with an increase in apoptosis (Feng et al. 1999).
To summarize the results of our study, we have demonstrated that c-kit mRNA and protein are expressed in the spermatogonia, acrosomal granules of the round spermatids, and Leydig cells in the adult human testis. In the spermatogonia, the expression seems to be stage specific. We believe that the results of the present study will expand current knowledge about the c-kit/SCF system in human spermatogenesis. In vitro studies on the effect of exogenous KIT ligand on spermatogonial cell activity are ongoing in our lab and may provide deeper insights into the role of the c-kit/SCF system in spermatogonial survival, renewal, or differentiation in human spermatogenesis.
Footnotes
Acknowledgements
This work was funded by the National Institute for Research in Reproductive Health Core Funds. S.K.U. is grateful to The Council of Scientific and Industrial Research, New Delhi, for providing a research fellowship supporting her work toward a PhD.
We thank Mr. H. Pawar and Mr. Praveen from the Institute histology department for help in tissue processing and sectioning. Technical assistance provided by Ms. Sandhya, Mr. Dattatray, and Mr. Karekar is acknowledged.
