Abstract
The kidneys and liver are the major routes for organic anion elimination. We have recently shown that acute obstructive jaundice is associated with increased systemic and renal elimination of two organic anions, p-aminohippurate and furosemide, principally excreted through urine. This study examined probable adaptive mechanisms involved in renal elimination of bromosulfophthalein (BSP), a prototypical organic anion principally excreted in bile, in rats with acute obstructive jaundice. Male Wistar rats underwent bile duct ligation (BDL rats). Pair-fed sham-operated rats served as controls. BSP renal clearance was performed by conventional techniques. Renal organic anion-transporting polypeptide 1 (Oatp1) expression was evaluated by immunoblotting and IHC. Excreted, filtered, and secreted loads of BSP were all higher in BDL rats compared with Sham rats. The higher BSP filtered load resulted from the increase in plasma BSP concentration in BDL rats, because glomerular filtration rate showed no difference with the Sham group. The increase in the secreted load might be explained by the higher expression of Oatp1 observed in apical membranes from kidneys of BDL animals. This likely adaptation to hepatic injury, specifically in biliary components elimination, might explain, at least in part, the huge increase in BSP renal excretion observed in this experimental model.
Keywords
A large and diverse number of organic or weak organic acids that exist as anions at physiological pH are secreted by mammalian renal tubules. Although this system secretes a number of endogenous compounds, it is generally accepted that it is particularly important in secreting numerous exogenous compounds, including pharmacologically active substances, industrial and environmental toxins, and plant and animal toxins (Burckhardt and Pritchard 2000).
Several members of multispecific organic anion transporters have been identified, which mediate renal elimination of organic anions, such as organic anion transporter (OAT), organic anion-transporting poly-peptide (Oatp), sodium-phosphate transporter (NPT), multidrug resistance-associated protein (MRP), and peptide transporter (PEPT) families (Sekine et al. 2006).
We have recently shown (Brandoni et al. 2003, 2006) that acute obstructive jaundice is associated with increased systemic and renal elimination of two organic anions, p-aminohippurate and furosemide, principally excreted through urine. The upregulation of OAT1 that we observed in cholestatic animals is one of the mechanisms involved in this phenomenon.
It would be of great importance to know if the renal elimination of an organic anion preferentially excreted by the liver would be altered in this experimental model. If so, adaptive mechanisms involved in organic anions elimination would be relevant in the presence of obstructive cholestasís to compensate for the impairment in biliary excretion.
Bromosulfophthalein (BSP), an organic anion principally excreted in bile, is secreted negligibly by the kidney in non-pathological conditions (Fleck and Bräunlich 1991, 1995).
In this study, we evaluated the physiological and molecular mechanisms involved in renal BSP elimination in rats with extrahepatic cholestasis.
Materials and Methods
Experimental Animals
Male Wistar rats from 110 to 130 days old were used throughout the study (380–410 g body weight). For surgical procedures, the animals were anesthetized with sulfuric ether. After an upper abdominal incision performed under sterile technique, the common bile duct was isolated and double-ligated close to the liver hilus immediately below the bifurcation and cut between the ligatures (BDL group). Controls underwent a sham operation that consisted of exposure, but not ligation, of the common bile duct (Sham group). The abdominal incision was closed by single sutures. All the studies were performed 21 hr after surgery. Animals were allowed free access to standard laboratory chow and tap water and housed in a constant temperature and humidity environment with regular light cycles (12 h) during the experiment. Animals were cared for in accordance with the principles and guidelines for the care and use of laboratory animals, recommended by the National Academy of Sciences and published by the National Institute of Health (NIH publication 86–23 revised 1985).
Biochemical Determinations
The day of the experiment, blood was withdrawn from the femoral artery of Sham and BDL animals. Serum was separated by centrifugation (3000 rpm, 3 min). These samples were used to measure total and direct bilirubin as parameters indicative of hepatic function and urea serum levels as a parameter indicative of global renal function. The mentioned biochemical analyses were performed with optimized spectrophotometric techniques, using commercial kits (Wiener Laboratory; Rosario, Argentina).
Urinary Excretion of BSP
As a preliminary study, we evaluated BSP renal excretion after administering a single bolus of BSP (10 mg/kg body weight, aqueous solution) through a femoral venous catheter. Urine was collected 40 min after administration in proper vials to measure the quantity of BSP. Concentration of BSP in urine was measured spectrophotometrically by alkalinization with 0.1 M NaOH (Quaglia et al. 2002).
Renal Clearance Studies
These studies were performed as previously described (Brandoni et al. 2004; Villar et al. 2004, 2005). A priming dose of inulin (30 mg/kg body weight) and BSP (12 mg/kg body weight) in 1.5 ml saline solution was administered through the venous catheter. A solution containing inulin (12 g/liter), BSP (0.05 g/liter), and saline solution (9 g/liter) was infused through the venous catheter using a constant infusion pump (KDS210; KD Scientific, New Hope, PA) at a rate of 1 ml/hr/100 g body weight. The glomerular filtration rate (GFR) was calculated from the clearance of inulin to determine filtered load of BSP. The excreted, secreted, and filtered loads of BSP and fractional excretion of BSP were calculated by conventional formulae for each animal. BSP concentrations in serum and urine were determined spectrophotometrically by alkalinization with 0.1 M NaOH (Quaglia et al. 2002), and inulin concentrations were assayed by the procedure of Roe et al. (1979). The volume of urine was determined by gravimetry.
Binding of BSP to plasma proteins was determined by ultrafiltration as previously described by Cerrutti et al. (2001).
Preparation of Liver Plasma Membranes (LPMs)
LPMs from Sham and BDL rats were obtained as described previously by van Amelsvoort et al. (1978) with some modifications. Briefly, the liver was immediately dissected out, rinsed in saline solution, and homogenized in 20 ml of 0.2 mM CaCl2/0.25 M sucrose/0.1 mM PMSF/10 mM HEPES-Tris (pH 7.40). EDTA was added until reaching a final concentration of 1 mM. Afterward, the homogenate was diluted with homogenization buffer (+1 mM EDTA) to a final volume of 100 ml. All operations were performed at 0–4C. After centrifugation for 10 min at 1000 × g, the supernatant and upper fluffy layer of the pellet were collected and centrifuged later for 30 min at 20,000 × g. The resulting pellet, which represents crude membranes, was resuspended gently in 0.3 M mannitol/0.1 mM PMSF/10 mM HEPES-Tris (pH 7.50). Each preparation represents the liver of one rat. For each experimental group, four different preparations were made. Protein quantification of samples was performed using the method of Sedmak and Grossberg (1977).
Preparation of Brush Border Membranes (BBMs) From Kidney
BBMs from Sham and BDL rats were isolated from renal tissue by Mg/EGTA precipitation as previously described (Ohoka et al. 1993), with some modifications (Torres et al. 2003). Each preparation represents renal tissue from four animals. For each experimental group, four different preparations were made. Protein quantification of samples was performed using the method of Sedmak and Grossberg (1977).
Body weight pre- and postsurgery, variation of body weight, and hematocrit of Sham and BDL rats
p<0.05.
Results are expressed as means ± SEM. BDL, bile duct ligation.
Fluorescence Polarization Measurements
The fluidity of BBMs was assessed by measuring the steady-state fluorescence anisotropy of 1,6-diphenyl-1,3,5-hexatriene (DPH) as previously described (Montagna et al. 1998; Cerrutti et al. 2002). BBMs (270 μg protein) were incubated for 1 hr at 25C with DPH (final concentration 1 μM). The DPH was excited at 366 nm, and emission was observed at 433 nm at 25C. The fluorescence anisotropy was calculated using the equation: r = (I|| − GI∗∗∗/I|| + 2GI∗∗∗) where I|| and I∗∗∗ are the fluorescence intensities observed with the polarized light emitted parallel and perpendicular, respectively, to the excitation polarizer. G is the correction factor for the optical system, given by the ratio of vertically to the horizontally polarized emission components when the excitation light is polarized in the horizontal direction.
Electrophoresis and Immunoblotting
Immunoblotting and subsequent densitometry for Oatp1 were performed in LPM, renal homogenates, and BBM as previously described (Cerrutti et al. 2002; Brandoni et al. 2003, 2004, 2006; Villar et al. 2004, 2005) using a commercial rabbit polyclonal antibody against rat Oatp1 (Alpha Diagnostic International; San Antonio, TX). Blots were processed for detection using enhanced chemiluminescence system (ECL; Amersham, Buckinghamshire, UK). For comparison between groups of animals, staining with ponceau red showed that equal quantities of proteins were deposited (Cerrutti et al. 2002; Brandoni et al. 2003, 2004, 2006; Villar et al. 2004, 2005).
IHC Microscopy
Kidneys from Sham (n = 4) and BDL (n = 4) rats were processed as previously described (Villar et al. 2005; Brandoni et al. 2006). Kidney sections were incubated with commercial polyclonal antibodies against Oatp1 (10 μg/ml) overnight at 4C. The sections were incubated with biotinylated secondary antibody against rabbit immunoglobulin for 1 hr (biotinylated Ig; Multi-Link Biogenex, San Ramon, CA). After being rinsed with PBST, the sections were incubated for 30 min with horseradish peroxidase (HRP)-conjugated streptavidin solution (Streptavidin/HRP complex; Multi-Link Biogenex). To detect HRP labeling, a peroxidase substrate solution with diaminobenzidine (0.05% diaminobenzidine in PBST with 0.05% H2O2) was used. The sections were counterstained with hematoxylin before being examined under a light microscope.
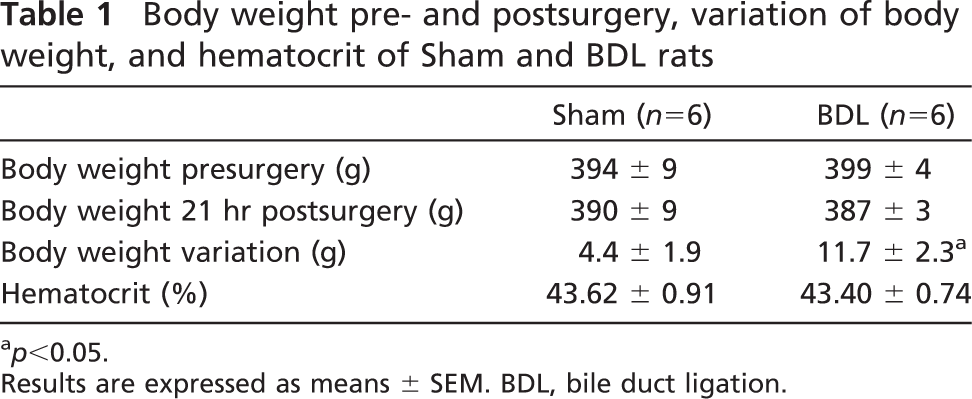
Liver histology from Sham (
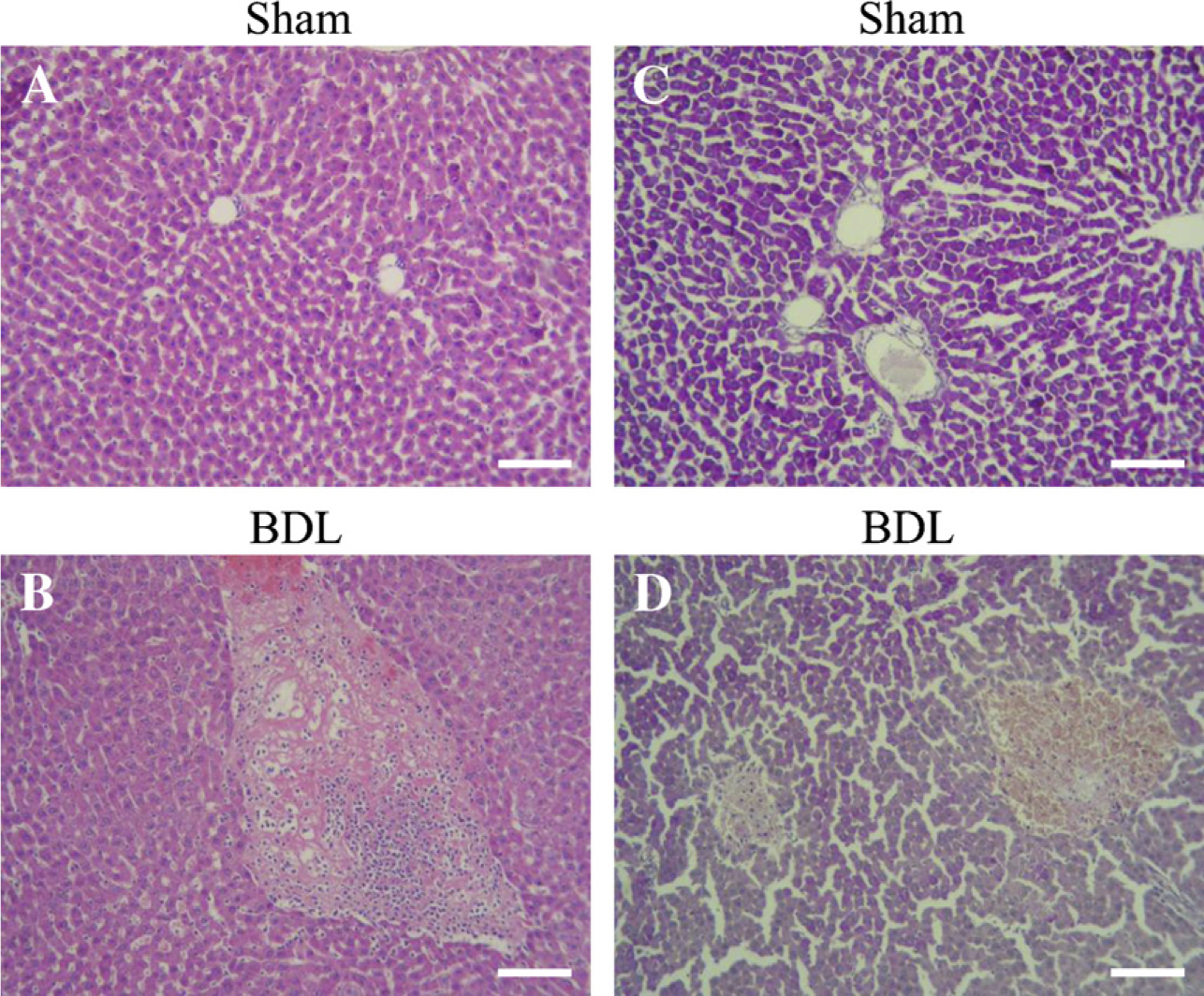
Kidney histology from Sham (
Controls using antibody solution absorbed with excess synthetic peptide or omission of primary or secondary antibody showed no labeling.
Histopathological Studies
Histopathology of livers and kidneys was performed after fixing in 10% neutral-buffered formaldehyde solution for 4 hr and embedding in paraffin; 4-μm-thick sections were processed for routine staining with hematoxylin—eosin or with hematoxylin and periodic acid-Schiff (PAS).
Materials
Chemicals were purchased from Sigma (St. Louis, MO) and were analytical grade pure.
Statistical Analysis
Statistical analysis was performed using an unpaired t-test. When variances were not homogeneous, a Welch's correction was used. p<0.05 was considered significant. Values are expressed as means ± SEM. For these analyses, GraphPad (San Diego, CA) software was used.
Results
Body weight of animals was measured before surgery and at the day of the experiments (21 hr after the corresponding surgery). Rats with extrahepatic cholestasis lost weight in comparison with sham-operated animals (Table 1). These results are similar to those previously described (Wòjcicki et al. 2000; Leung et al. 2001). No difference between groups was seen in the hematocrit.
Bilirubin levels, both total and direct, were significantly higher in BDL than in sham-operated animals, attesting to the adequacy of biliary obstruction (total bilirubin, mg/liter; Sham, n = 6: 5.1 ± 0.3, BDL, n = 6: 44.1 ± 2.8, p<0.05; direct bilirubin, mg/liter; Sham, n = 6: 1.9 ± 0.1, BDL, n = 6: 34.5 ± 3.4, p<0.05). There was no significant difference between groups in serum urea levels (Sham, n = 6: 0.40 ± 0.08 vs. BDL, n = 6: 0.43 ± 0.06 g/liter).
Kidneys weight, kidney/body weight ratio, GFR, PBSP, BSP unbound to proteins fraction, ELBSP, FLBSP, and SLBSP in Sham and BDL rats
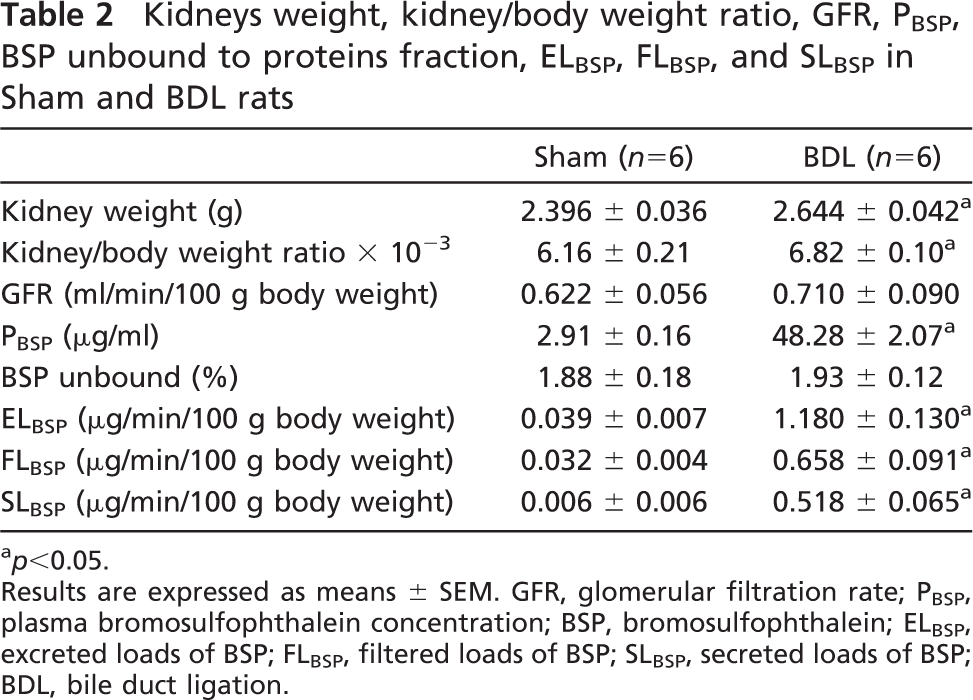
p<0.05.
Results are expressed as means ± SEM. GFR, glomerular filtration rate; PBSP, plasma bromosulfophthalein concentration; BSP, bromosulfophthalein; ELBSP, excreted loads of BSP; FLBSP, filtered loads of BSP; SLBSP, secreted loads of BSP; BDL, bile duct ligation.
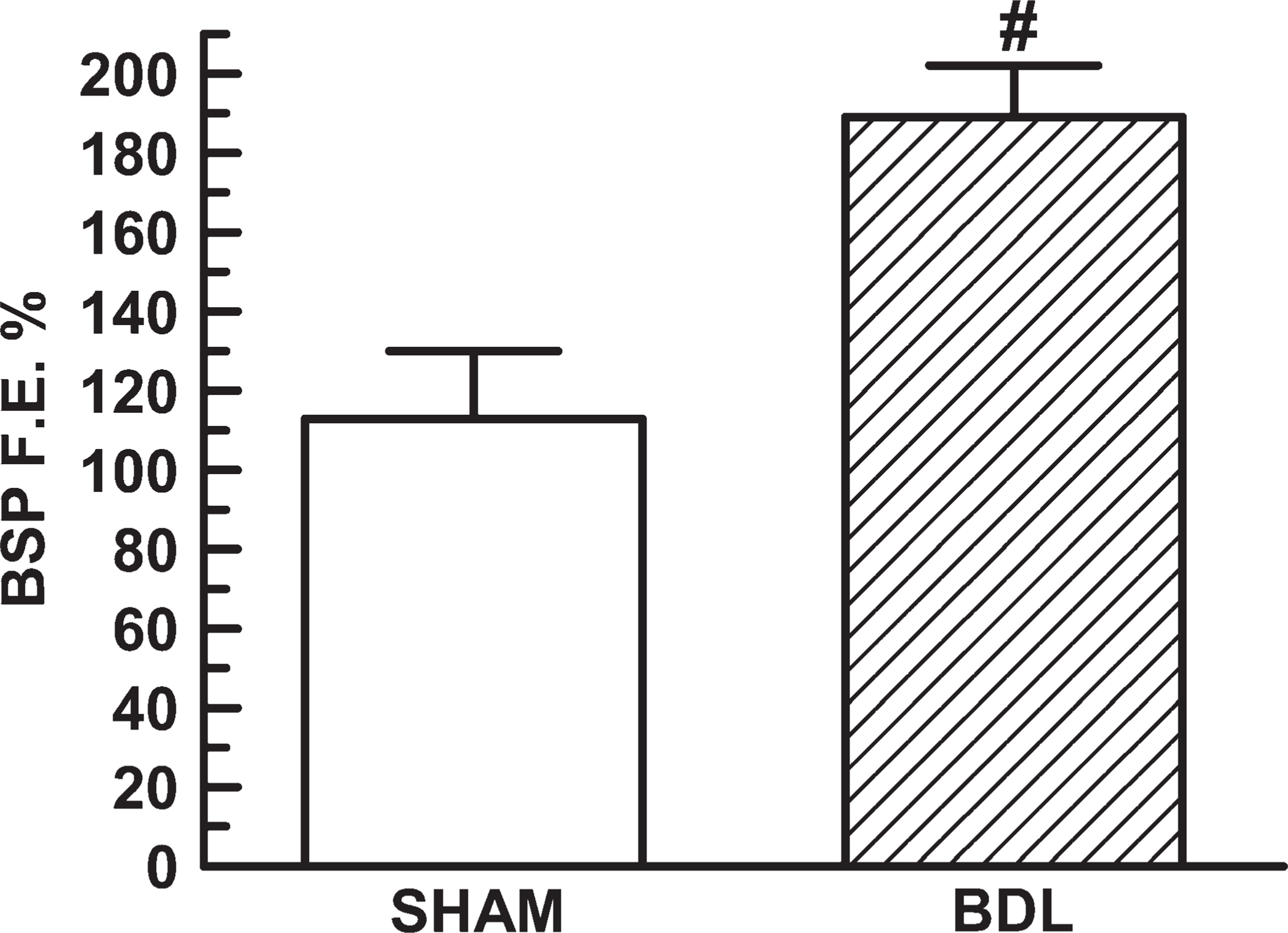
Bromosulfophthalein (BSP) fractional excretion (BSP F.E. %) in Sham (n = 6) and BDL (n = 6) rats. Results are expressed as means ± SEM. #p<0.05.
Histological studies showed that livers from the Sham group were of normal morphology, whereas in rats with extrahepatic cholestasis, some abnormalities were seen (Figure 1). An enlargement of portal spaces was seen. Some mononuclear cell infiltrations in the widened portal areas were also noted. Furthermore, numerous and dispersed zones of necrosis were observed. Furthermore, PAS staining showed the normal disposition of the liver in Sham rats. In animals with mechanical cholestasis, an evident diminished PAS staining was observed. This observation is related to the presence of recent necrosis areas. These observations corroborated the morphological liver damage that underwent animals with extrahepatic cholestasis (Wòjcicki et al. 2000).
Light microscopy only showed significant renal morphological alterations in PAS-stained kidneys (Figure 2). In BDL rats, renal tubules showed cells with PAS cytoplasmic granules. The height of the epithelial cells and their apical folds were reduced. A few tubules resembled thyroid follicles because of the presence of PAS acidophilic material in their lumen. These results are similar to those described by Wòjicki et al. (2000).
The urinary excretion of BSP in the preliminary studies turned out to be hugely increased in BDL rats (μg BSP; Sham, n = 7: 1.92 ± 0.34; BDL, n = 8: 32.4 ± 12.93, p<0.05).
Kidney and the respective kidney/body weight ratio were always greater in BDL rats (Table 2). No difference between groups was observed in GFR. Plasma BSP concentration was markedly greater in BDL rats. There was no difference between both experimental groups in BSP unbound to proteins fraction. The excreted, filtered, and secreted loads of BSP were all higher in BDL rats compared with Sham ones. In addition, Figure 3 shows a significant increase in BSP fractional excretion of the BDL group.
Renal expression of Oatp1 was examined in homogenates and BBMs from Sham and BDL animals by immunoblotting analysis. Figure 4 shows no difference between Sham and BDL rats in the homogenates Oatp1 expression. Alternatively, a higher abundance of Oatp1 was observed in BBMs from BDL rats in comparison to Sham rats.
On the other hand, as reported earlier (Dumont et al. 1997), Oatp1 hepatic expression was lower in liver plasma membranes from BDL rats (%; Sham, n = 4: 100 ± 3; BDL, n = 4: 83 ± 5; p<0.05).
The IHC study showed staining for Oatp1 at the apical plasma membrane of the S3 segment of the proximal tubule cells (Figure 5). Increasing labeling was seen at the apical membranes of the tubules from kidneys of BDL rats (Figure 5B). This pattern parallels the data from the immunoblotting study.
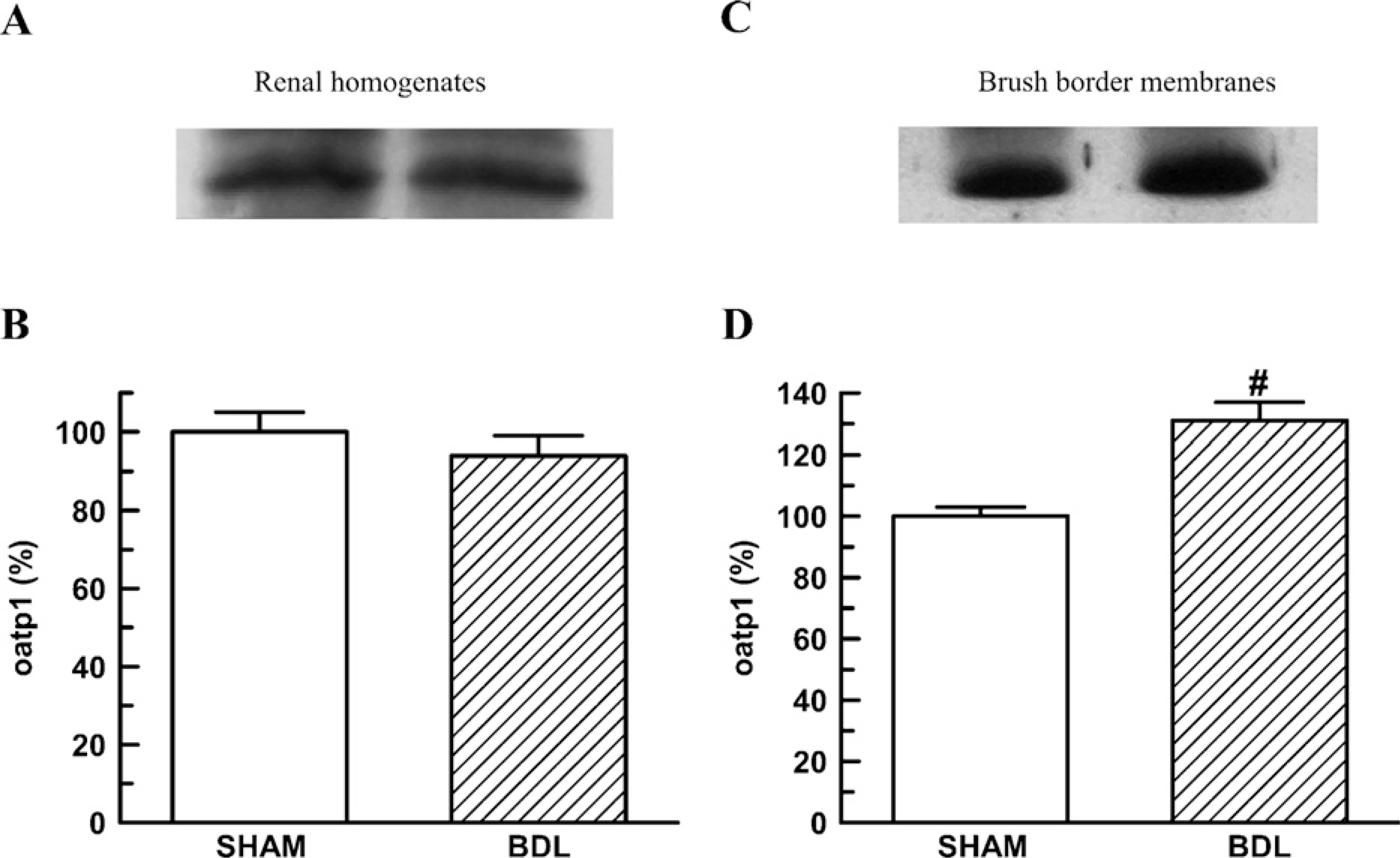
Renal homogenates (50 μg proteins) (
Membrane fluidity of BBM was similar in both experimental groups [anisotropy values (r); Sham, n = 4: 0.1876 ± 0.0008; BDL, n = 4: 0.1838 ± 0.0025].
Discussion
The kidneys and liver are the major routes for organic anion elimination (Fleck and Bräunlich 1991; Burckhardt and Pritchard 2000). Numerous compounds, such as drugs, environmental substances, plant and animal toxins, and metabolites of both foreign and endogenous origins, are classified as organic anions.
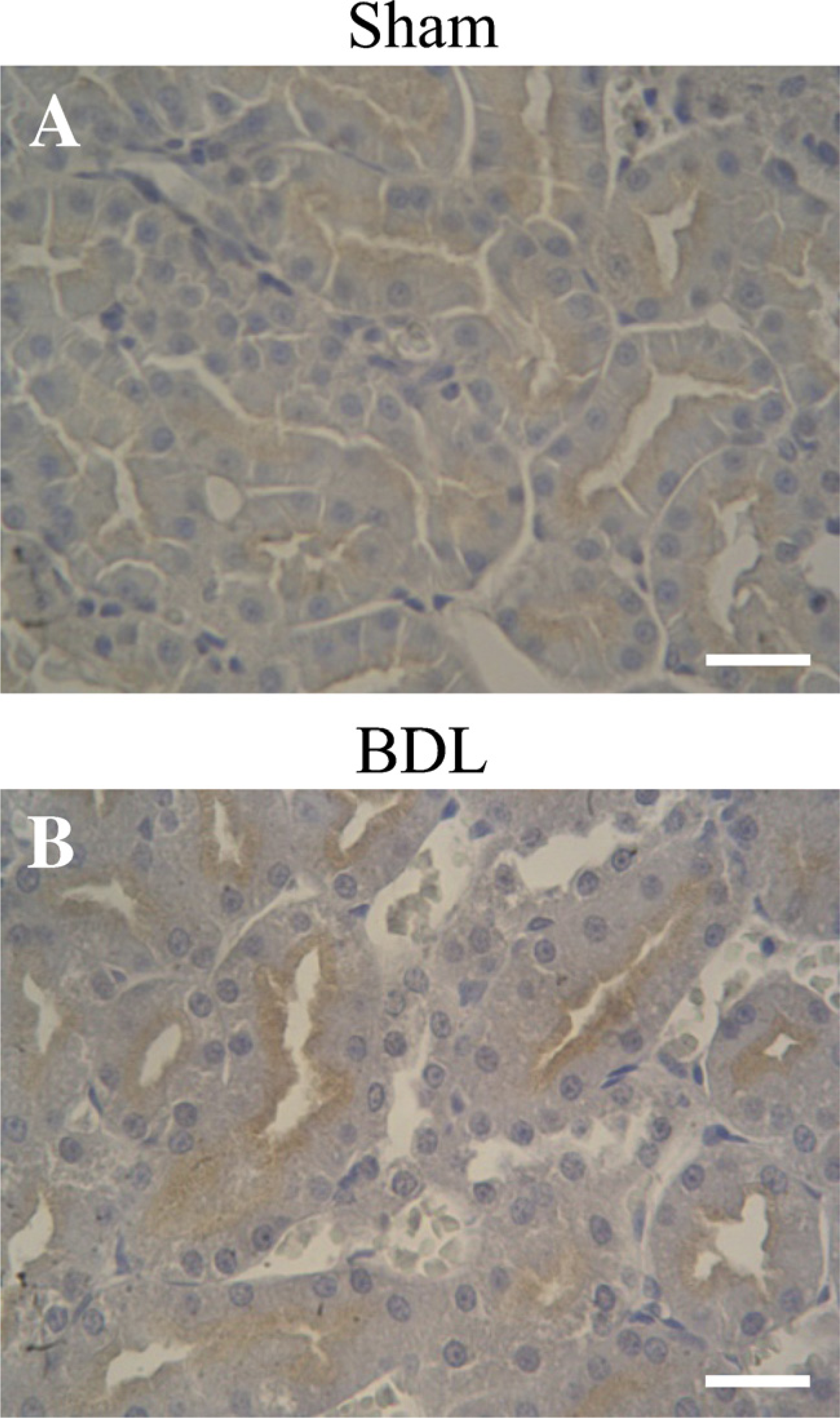
Immunoperoxidase IHC for Oatp1 in kidneys of Sham (
Cholestasis has been shown to alter the transport of the bile salts and of other organic anions (Reichen and Simon 1988; Lee et al. 2001; Denk et al. 2004). Extrahepatic cholestasis refers to obstruction of large bile ducts outside the liver, for instance, caused by gallstones. Renal function in the course of obstructive jaundice has been the subject of great interest.
As a preliminary study, we evaluated the renal excretion of BSP after a single intravenously dose. In this study, we found an imperative increase in BSP urinary excretion in rats with extrahepatic cholestasis. Therefore, we decided to evaluate possible renal mechanisms involved in this higher renal elimination performing more specific studies.
The increase in BSP excreted load, seen in renal clearance studies, would be explained by the following mechanisms: on one hand, by the increase in the filtered load that resulted from the huge increase in plasma BSP concentration in BDL rats, because GFR showed no difference with Sham group, on the other hand, by the increase in the secreted load.
Renal secretion of organic anions, which occurs principally along the proximal portion of the nephron, plays a critical role in regulating their plasma concentrations and in clearing the body of potentially toxic xenobiotics agents (Miyazaki et al. 2004; Sekine et al. 2006). The transepithelial transport involves separate entry and exit steps at the basolateral and luminal aspects of renal tubular cells. Several carrier proteins have been cloned and are functionally characterized from both membrane domains of rats kidneys (Sekine et al. 2006). Defining the modifications on the expression of these transporters is important, both to understand the cholestatic process and to identify potential therapeutic targets.
In a previous study, we showed (Brandoni et al. 2006) that OAT3 expression in renal basolateral membranes was unchanged in rats at 21 hr after BDL. BSP is a substrate for this transporter. In addition, MRP2 mediates the transport of BSP-conjugated derivates, and MRP2 upregulation in BDL rats was described in recent studies (Tanaka et al. 2002; Villanueva et al. 2006).
Proteins other than MRP2 may be involved in active transport of compounds, such as BSP, across the apical membrane of renal tubular cells. Therefore, we decided to evaluate Oatp1 renal expression as an additional responsible mechanism for the increased BSP secretion observed in BDL animals.
The first member of the Oatp gene family of membrane transporters, Oatp1, has been isolated from rat liver and shown to mediate Na+-independent saturable transport of BSP (Bergwerk et al. 1996; Eckhardt et al. 1999; Hagenbuch and Meier 2003). Oatp1 is also localized, in addition to the basolateral plasma membrane of hepatocytes, at the apical membranes of the kidney proximal tubule (S3 segment). Thus, this transporter could be responsible for reabsorption of organic compounds that are freely filtered, such as estradiol-17β-glucuronide, or important for the secretion of certain organic compounds that are taken up into proximal tubular cells across the basolateral membrane (Gotoh et al. 2002).
In rats with cholestasis caused by BDL, the hepatic expression of Oatp1 is downregulated (Dumont et al. 1997; Donner et al. 2007; Geier et al. 2007). This transporter mediates hepatic uptake of numerous organic anions from the systemic circulation. This observation may also be an adaptive change because BDL rats need to restrict excess uptake of various organic anions into hepatocytes. Renal Oatp1 mediates a bidirectional transport of organic anions at the apical (brush border) membrane (Bergwerk et al. 1996; Dubuisson et al. 1996; Gotoh et al. 2002; Lash 2005). It is worth noting that differential processing and trafficking of this transporter in liver and kidney may have important functional and regulatory consequences.
The function of Oatp1 in renal cells under physiological and pathological conditions has not yet been fully elucidated.
In this study, our data showed that BDL rats have a higher renal expression of Oatp1 protein at apical membranes despite no change in Oatp1 abundance in kidney homogenates. In addition to immunoblotting, IHC study shows increased Oatp1 labeling associated with apical plasma membrane domains. These observations suggest an alteration in Oatp1 trafficking that might be caused by an increased recruitment of preformed transporters into the membranes or an inhibition in the internalization of membrane transporters.
The results of this study have shown that the kidneys are able to adapt rapidly to obstructive cholestasis because BSP renal elimination had increased nearly 30-fold during the first day after induction of bile duct obstruction. This study shows evidence that this renal adaptation to cholestasis involves an upregulation of the transport capacity of the proximal tubular organic anion-transporting polypeptide 1, Oatp1. This increase in Oatp1 protein units at the apical membrane of renal cells may be a compensatory mechanism for reducing injury to hepatocytes or renal epithelia from cytotoxic materials that may occur in rats with obstructive cholestasis.
Membrane fluidity has been shown to affect numerous membrane functions including passive permeability enzymatic activities and transport systems (Montagna et al. 1998; Torres et al. 2003). This effect can result from a direct action on the conformational changes required for the function of a given protein or may involve the ease with which proteins associate or dissociate. In this cholestatic model, we found no difference in BBM fluidity between groups. Because fluidity measures the bulk physical properties of membrane lipids, which are important determinants of transport properties, the observed difference in BSP transport in BDL groups would not be related to changes in this parameter.
Extrahepatic cholestasis induces a complex series of hormonal changes in kidneys (Holt et al. 1999; Leung et al. 2001), which might influence the regulation of Oatp1. Likely, several local and systemic factors are produced at the same time, and the role of such factors in the regulation of channels and transporters in the presence of BDL is unknown.
The accumulation of bilirubin, bile acids, and other potential toxins existing in this cholestatic model may affect transcriptional regulatory mechanisms (e.g., fetal transcription factor, pregnene × receptor) and post-transcriptional regulations (Zollner et al. 2003; Donner et al. 2007; Geier et al. 2007). Accordingly, Tanaka et al. (2002) found that bilirubin, sulfate-conjugated bile acid, and human bile upregulate the expression of MRP2 in renal tubular cells but not in hepatocytes. Some experiments are currently being performed in our laboratory to elucidate whether these regulatory mechanisms are involved in Oatp1 modulation in renal tubular cells in this cholestatic model in rats.
In summary, we present evidence that cholestasis induced by common BDL in the rat induces a redistribution in renal Oatp1 protein units into apical membranes from renal cells. Moreover, this likely adaptation to hepatic injury, specifically in biliary components elimination, present in this model might explain, at least in part, the huge increase in BSP renal excretion observed in this experimental model.
Footnotes
Acknowledgements
This study was supported by Fondo para la Investigaciòn Científica y Tecnològica (PICT 05-20201) and Consejo Nacional de Investigaciones Científicas y Técnicas (PIP 5592).
We thank Prof. Juan C. Picena (Cátedra de Anatomía y Fisiología Patològicas, Facultad de Ciencias Médicas, U.N.R.) and Alejandra Martínez (Area Morfología, Facultad de Ciencias Bioquímicas y Farmacéuticas, U.N.R.) for their contribution to the histological studies and Wiener Lab Argentina for analytical kits.
