Abstract
The enzyme-labeled antigen method is a histochemical technique that visualizes antigen-specific antibody-producing cells in tissue sections, originally documented in 1968. In this study, we attempted to reemerge this hidden but potentially useful method in rat models immunized with horseradish peroxidase (HRP), ovalbumin (OA), or keyhole limpet hemocyanin (KLH). After repeated immunization in footpads, popliteal, groin, and axillary lymph nodes and spleen were sampled. Paraformaldehyde-prefixed frozen sections were incubated with HRP, biotinylated OA, or biotinylated KLH. Proteinase K pretreatment and the secondary use of HPR-labeled streptavidin were applied in the latter two situations. Plasma cells producing antigen-specific antibodies were visualized. Proportions of antigen-specific antibody-producing cells in total plasma cells shown with the immunoperoxidase method for rat immunoglobulins were evaluated. The percentage of antigen-specific plasma cells reached ∼50% of total plasma cells in the regional lymph nodes. The specificity was confirmed by (a) negativity in non-immune rat tissue, (b) negativity with indifferent antigen probes, and (c) abolishment of the reactivity with the corresponding rat serum. In buffered formalin-fixed, paraffin-embedded tissues, fewer plasma cells were labeled for HRP and KLH antibody reactivity after strong proteolysis and prolonged incubation. Expectedly, this method allows us to observe antigen-specific antibody-producing cells under varied pathological conditions.
Keywords
T
The enzyme-labeled antigen method is potentially useful for analyzing pathological conditions from a novel point of view that enables us to observe the distribution of antigen-specific antibody-producing cells in such lesions accompanying dense infiltration of plasma cells as infectious diseases, malignant tumors, and autoimmune or allergic disorders. In many cases, the nature of the corresponding antigens recognized by antibodies secreted from plasma cells that infiltrate within the lesions remains unclear. The plasma cells should locally produce disease-specific antibodies, simply because the immunocytes are distributed within the lesions. Expectedly, the enzyme-labeled antigen method can be an intriguing technique for localizing the pathogenic antibodies that provoke certain pathological conditions.
Plasma cells infiltrating in infectious lesions may produce antibodies against pathogenic microbes. In tissue sections of infectious diseases, labeled microbial antigens may thus visualize plasma cells locally secreting antimicrobial antibodies. In malignant lesions, plasma cells may produce antitumor cell antibodies. When the candidate antigen is once available, the pathobiological significance of plasma cells infiltrating within the neoplastic lesion may be elucidated. Similarly, autoimmune or allergic processes can be examined by the enzyme-labeled antigen method.
In this report, we attempted to re-emerge the enzyme-labeled antigen method by using paraformaldehyde (PFA)-prefixed frozen sections of the lymph nodes and spleen of rats immunized with HRP, ovalbumin (OA), or keyhole limpet hemocyanin (KLH). We further evaluated the proportion of antigen-specific antibody-producing cells in total plasma cells in tissue sections sampled from these experimental models. The technique was further applied to buffered formalin-fixed, paraffin-embedded specimens of rat lymphoid tissues.
Materials and Methods
Experimental Animals
Specific pathogen-free male Sprague-Dawley rats (Chubu Kagaku Shizai; Nagoya, Japan), 5 weeks old, weighing 150 g, were kept in the animal laboratory of Fujita Health University, Toyoake, Japan, with 50% humidity and under 12:12-hr light and dark cycles, and fed freely with a standard pellet diet (CE-2; CLEA Japan, Tokyo, Japan) and tap water ad libitum. The animal experiments were conducted in accordance with the Guidelines for the Management of Laboratory Animals in Fujita Health University.
Immunization
One mg HRP (Wako Pure Chemical Industries; Osaka, Japan), OA (Sigma; St. Louis, MO), or KLH (Sigma) were dissolved in 1 ml saline and emulsified with an equal quantity of Freund's complete adjuvant (Difco Laboratories; Detroit, MI). The emulsion (0.15 ml each) was injected three times into the footpad of all four legs of rats. At the second and third boosts, 1 and 5 weeks after the initial challenge, Freund's complete adjuvant was replaced by Freund's incomplete adjuvant (Difco Laboratories). Three or four rats were used for immunization of the respective antigen (n = 4 in case of HRP and OA and n = 3 in case of KLH). In each experiment, two rats were immunized with an emulsion of saline and the adjuvant as control “non-immune” animals.
Tissue Sampling
Two weeks after the third antigen injection, the rats were euthanized under inhalation of diethyl ether. Pieces of bilateral popliteal, groin, and axillary lymph nodes and spleen were sampled, rinsed with 0.01 M PBS, pH 7.2, and immersion-fixed in 4% PFA-containing phosphate buffer, pH 7.4, for 4 hr at 4C. The fixed tissues were rinsed in 10% sucrose-containing PBS overnight at 4C and additionally soaked in 15% and 20% sucrose-containing PBS for 4 hr each. The rinsed lymph nodes and spleen were embedded in an embedding medium (Tissue Mount; Chiba Medical, Saitama, Japan), quickly frozen with dry ice-acetone, sectioned on a cryostat (Leica Microsystems; Wetzlar, Germany) at 3 μm thickness, and mounted on 3-aminopropyl-triethoxysilane–coated glass slides. The frozen sections were dried for 30 min with a drier at room temperature and stored at 220C until staining. Part of the tissues was fixed in 0.1 M phosphate-buffered 10% formalin, pH 7.2, overnight at room temperature, and routinely embedded in paraffin. Hematoxylin and eosin–stained sections were prepared to confirm the histological features.
Blood samples were collected from renal veins of the rats. Sera were separated by centrifugation for 10 min at 3000 rpm, collected in microtubes, and stored at −80C until assay.
Titration of Antigen-specific Antibodies in the Serum
The titration assay was carried out by the ELISA method. Microtiter plates (Costar 3590; Corning Incorporated, Corning, NY) were coated with 100 μg/ml HRP, OA, or KLH (100 μl/well) dissolved in 0.1 M carbonate buffer, pH 9.6, overnight at 4C. After removing the carbonate buffer, 100 μl of 3% BSA in PBS was added to the well and incubated for 1 hr at 37C. The plates were rinsed with PBS containing 0.05% Tween-20 (PBS-Tween) three times after each step of the subsequent incubations. Two-fold serial dilutions of the rat sera were examined in a range from 1:100 to 1:205,600 dilutions. An aliquot (100 μl) of the diluted serum was added to the well. After incubation for 1 hr at 37C and subsequent rinsing in PBS-Tween, alkaline phosphatase–conjugated rabbit anti-rat IgG antibodies (Rockland Immunochemicals; Gilbertsville, PA) diluted at 1:1000 were added to the well (100 μl/well). After incubation for 1 hr at 37C and rinsing in PBS-Tween, the color reaction was developed with 100 μl/well 0.01 M diethanolamine buffer, pH 9.8, containing p-nitrophenyl phosphate (Sigma) for 20 min at room temperature. The reaction was stopped by adding 3 N sodium hydroxide (50 μl/well). Finally, the absorbance at 405 nm, representing IgG antibody titers in the serum, was measured.
Biotinylation of HRP, OA, and KLH
For preparing labeled antigens, HRP, OA, and KLH were biotinylated with a sulfo-NHS-LC biotinylation kit (Pierce; Rockford, IL). Biotinylation was performed according to the manufacturer's instructions. Biotinylated HRP was used for cross-checking the specificity of the reactivity.
Enzyme-labeled Antigen Method Using HRP, Biotinylated OA, and Biotinylated KLH as Probes
After rinsing sections in running water for 5 min, endogenous peroxidase activity was inactivated in methanol containing 0.3% hydrogen peroxide for 30 min at room temperature. After brief immersion in PBS, the sections were incubated with HRP, biotinylated OA, or biotinylated KLH solution at the concentration of 1, 10, or 100 μg/ml for 1 hr at room temperature. In the latter two cases using biotinylated probes, the sections were further rinsed three times in PBS for 5 min and incubated with HRP-labeled streptavidin ready-to-use solution (Nichirei Bioscience; Tokyo, Japan) for 1 hr at room temperature. After a final rinse in PBS for 5 min three times, the HRP activity bound to plasma cells was visualized by incubating with 0.05 M Tris-HCl buffer, pH 7.6, containing 0.02% diaminobenzidine (DAB) and 0.006% hydrogen peroxide. The sections, counterstained with Mayer's hematoxylin, were dehydrated through graded ethanol, penetrated with xylene, and mounted in hydrophobic mounting medium (Entellan New; Merck, Germany).
Retrieval of Antigen-binding Activity by Proteinase K Treatment
When necessary, antigen-binding activity was retrieved through digestion with proteinase K (Roche Diagnostics; Mannheim, Germany). Briefly, the sections were treated, after endogenous peroxidase blockage, with 0.05 M Tris-buffered saline, pH 7.6, containing 0.625-20 μg/ml proteinase K for 15 min at room temperature and incubated with HRP, biotinylated OA, or biotinylated KLH.
Use of Buffered Formalin-fixed, Paraffin-embedded Sections for the Enzyme-labeled Antigen Method
Buffered formalin-fixed, paraffin-embedded sections of the axillary lymph nodes were further used for the enzyme-labeled antigen method. Higher concentrations of proteinase K (20, 80, and 320 μg/ml) were evaluated for pretreatment for 15 min at room temperature. Appropriate concentrations of the probe antigen solutions were also examined: 1–100 μg/ml for HRP, 10–100 μg/ml for biotinylated OA, and 100–500 μg/ml for biotinylated KLH. Heat-assisted retrieval through pressure pan cooking in 0.01 M citrate buffer, pH 6.0 or pH 7.0, and in 1 mM EDTA solution, pH 8.0, was also tried (Tsutsumi and Kamoshida 2003). The other procedures were fundamentally the same as in the case of frozen sections. Overnight incubation with the antigen solutions was tested when negative for 1-hr incubation.
Direct Immunoperoxidase Staining With a Cocktail of HRP-labeled Polyvalent Antibodies Against Rat IgG, IgA, and IgM
After a brief dip in running water and endogenous per-oxidase blockage, the frozen sections were treated with Tris-buffered saline containing 5 μg/ml proteinase K for 15 min at room temperature, rinsed in PBS, and incubated with a cocktail of HRP-conjugated goat anti-rat IgG, IgA, and IgM polyvalent antibodies (MP Biomedicals; Solon, OH), diluted at 1:1000, for 1 hr at room temperature. Finally, DAB coloring reaction for 5 min and brief hematoxylin counterstaining followed.
Analysis of the Proportion of Specific Antibody-producing Cells in Total Plasma Cells
The proportions of antigen-specific antibody-producing cells in total plasma cells were calculated as follows. In consecutive frozen sections of the lymph nodes and spleen, both antigen-specific antibody-producing cells and plasma cells immunostained with polyvalent anti-rat immunoglobulin antibodies were comparatively counted. We evaluated a total of four high-power (x400) microscopic fields where plasma cells were accumulated (the medullary cord of the lymph nodes and the red pulp of the spleen).
Specificity Cross-checking With Indifferent Antigen Probes
The specificity of the enzyme-labeled antigen method was cross-checked by incubating with indifferent antigen solutions on fixed frozen sections of the axillary lymph nodes. Proteinase K pretreatment was performed in all frozen sections. For staining in HRP-immunized rat tissues with biotinylated HRP, OA, or KLH as probes, signals were detected in red with alkaline phosphatase–labeled streptavidin and the following new fuchsin color reaction (New fuchsin substrate kit Histofine; Nichirei Bioscience, Tokyo, Japan), to avoid confounding positivity through HRP-labeled streptavidin. To inhibit endogenous alkaline phosphatase activity, 5 mM levamisole (Sigma) was added to the reaction mixture. The relevance of the alkaline phosphatase–mediated reaction was checked by obtaining positivity in the corresponding frozen sections of the lymph nodes immunized with HRP, OA, or KLH.
Specificity cross-checking was similarly done for the paraffin sections using higher concentrations of antigen solutions.
Antibody Absorption Test
Aliquots (90 μl) of the antigen solutions (HRP: 1 μg/ml for frozen sections or 10 μg/ml for paraffin sections, biotinylated OA: 10 μg/ml, and biotinylated KLH: 100 μg/ml in PBS) were preincubated with 10 μl of PBS-diluted serum (1-, 10-, 100-fold) of the antigen-immunized rats or PBS for 1 hr at 37C (final dilutions of the immune serum were 1:10, 1:100, or 1:1000). After centrifugation at 8000 rpm for 1 hr at 20C, the supernatant of the absorbed solution was applied to the enzyme-labeled antigen method sequence.
Antibody titration of immunized or non-immune rat sera determined by the ELISA method

Antigen-specific IgG antibody titers are shown as the optical density at 405 nm (mean ± SE). The rat serum was diluted at 1:12,800. HRP, horseradish peroxidase; OA, ovalbumin; KLH, keyhole limpet hemocyanin.
Statistical Analysis
For evaluating correlations between the proportion of antigen-specific antibody-producing cells in the regional lymph nodes and the titer of antigen-specific IgG antibodies, the ANCOVA was performed where the immunized antigen was regarded as a covariate, the antibody titer as a dependent variable, and the proportion as an independent variable.
The proportions of antigen-specific antibody-producing cells in total plasma cells were statistically compared between the regional lymph nodes (as a group) and the spleen. The nested ANOVA was performed where the individual animals immunized with the respective antigen were nested as a random effect, whereas tissue type (lymph nodes or spleen), antigen type (HRP, OA, or KLH), and the interaction between the tissue type and antigen type were used as fixed effects.
Statistical analysis was performed with JMP software (5.0.1a; SAS Institute, Cary, NC); p<0.05 was regarded as statistically significant.
Results
Serum Titers of Antigen-specific Antibodies
In the ELISA assay for detecting antigen-specific serum IgG antibodies in rats immunized with HRP, OA, or KLH, optical density values of the immunized rat sera were significantly higher than those of the control (non-immune) rat sera, indicating that antigen-specific antibodies were raised in the serum of rats immunized with the corresponding antigen. The serum antibody titers in the ELISA assay are shown in Table 1.
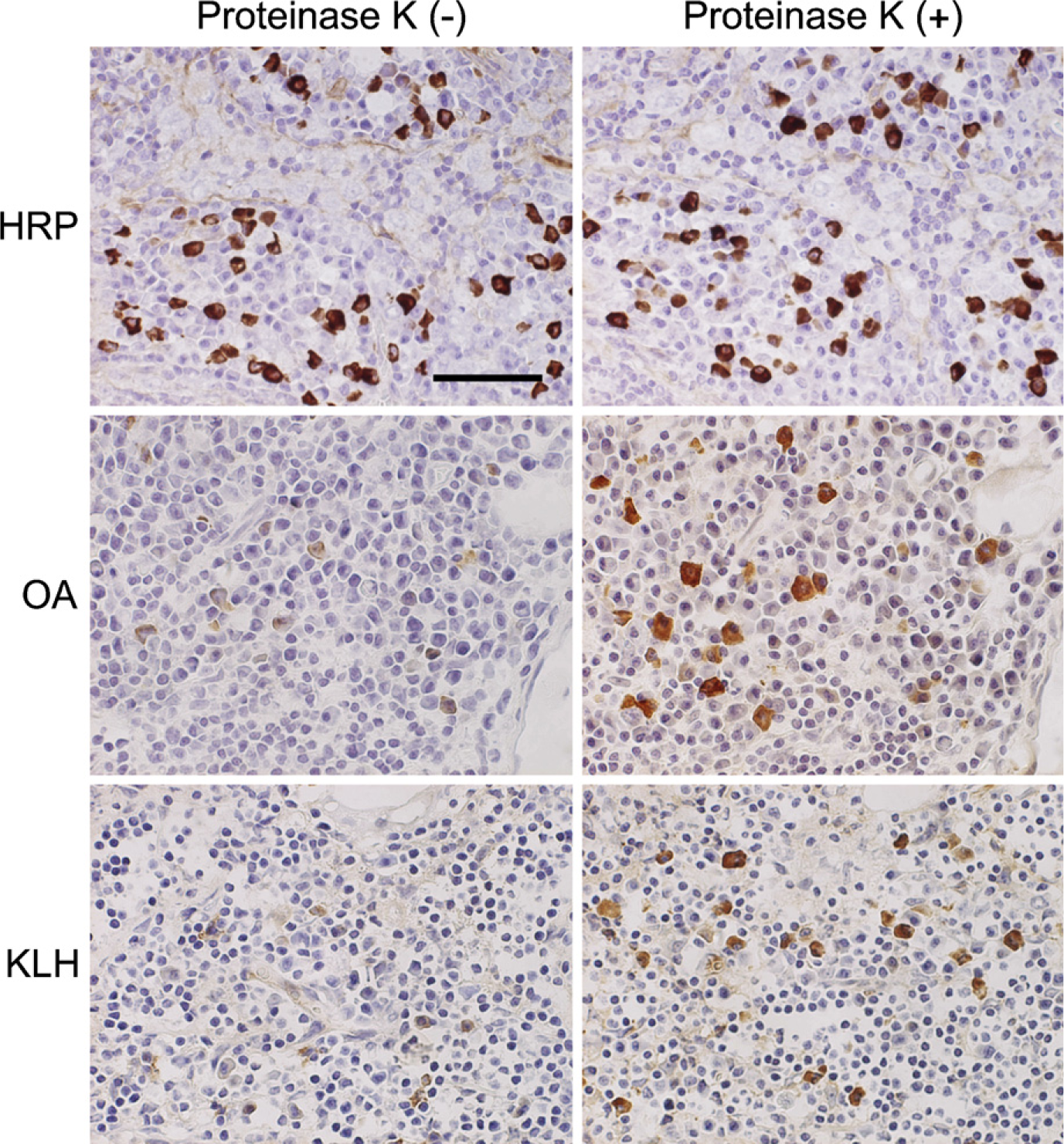
Paraformaldehyde (PFA)-prefixed frozen sections of the popliteal lymph nodes of horseradish peroxidase (HRP)-, ovalbumin (OA)-, or keyhole limpet hemocyanin (KLH)-immunized rats stained with the corresponding antigen probes before (left panels) and after (right panels) proteinase K pretreatment. Antigen-binding activity is evidently retrieved by proteolysis in cases of OA and KLH. The cytoplasm of antigen-specific antibody-producing cells is labeled brown. Bar = 50 μm.
Enzyme-labeled Antigen Method in Fixed Frozen Sections Using HRP, Biotinylated OA, and Biotinylated KLH as Probes and Retrieval of Antigen-binding Activity With Proteinase K Pretreatment
In 4% PFA-prefixed frozen sections of the lymph nodes and spleen, antigen-specific antibody-producing cells were visualized clearly in case of HRP and weakly in case of OA. The cytoplasm of plasma cells distributed in the medullary cord of the lymph nodes and the red pulp of the spleen was stained in brown color (Figure 1, left panels). In sections stained for KLH, specific immunocytes were scarcely identified without proteolytic pretreatment.
Proteinase K pretreatment evidently improved the stainability of antigen-specific antibody-producing cells in the case of OA or KLH (Figure 1, right panels). The cytoplasm of the immunocytes producing anti-OA and anti-KLH antibodies became clearly positive after the proteinase K pretreatment. The optimal concentration of proteinase K was proven to be 5–10 μg/ml, and higher concentrations (20 μg/ml) decreased the number and density of the positive cells and caused higher background staining. In the case of HRP-immunized rats, the pretreatment minimally influenced the staining pattern.
The optimal concentrations of the antigen solutions were 1 μg/ml for HRP, 10 μg/ml for OA, and 100 μg/ml for KLH.
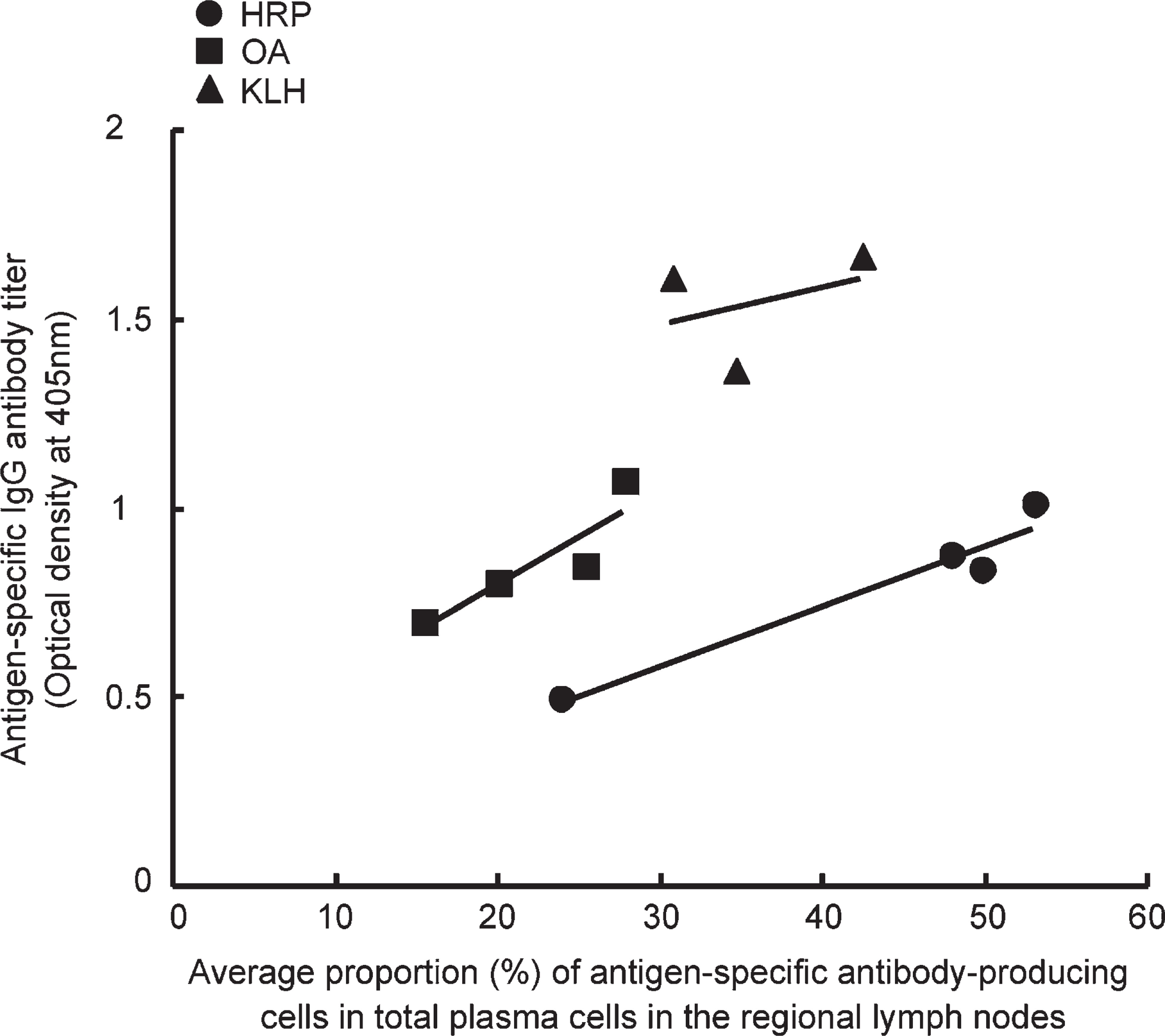
No labeled immunocytes were shown in the lymph nodes and spleen of the control (non-immune) rats (Figure 2, top row). When the tissue sections were incubated with indifferent antigens, no positivity was obtained (Figure 2, second to bottom row). When the antigen solution was preabsorbed with the corresponding rat serum, the signal of positive cells decreased or disappeared in accordance with the serum dilution (Figure 3). The sera of rats with indifferent antigenic challenge never absorbed the signals. These findings were seen in any combination of the three antigens, confirming the specificity of the enzyme-labeled antigen method.
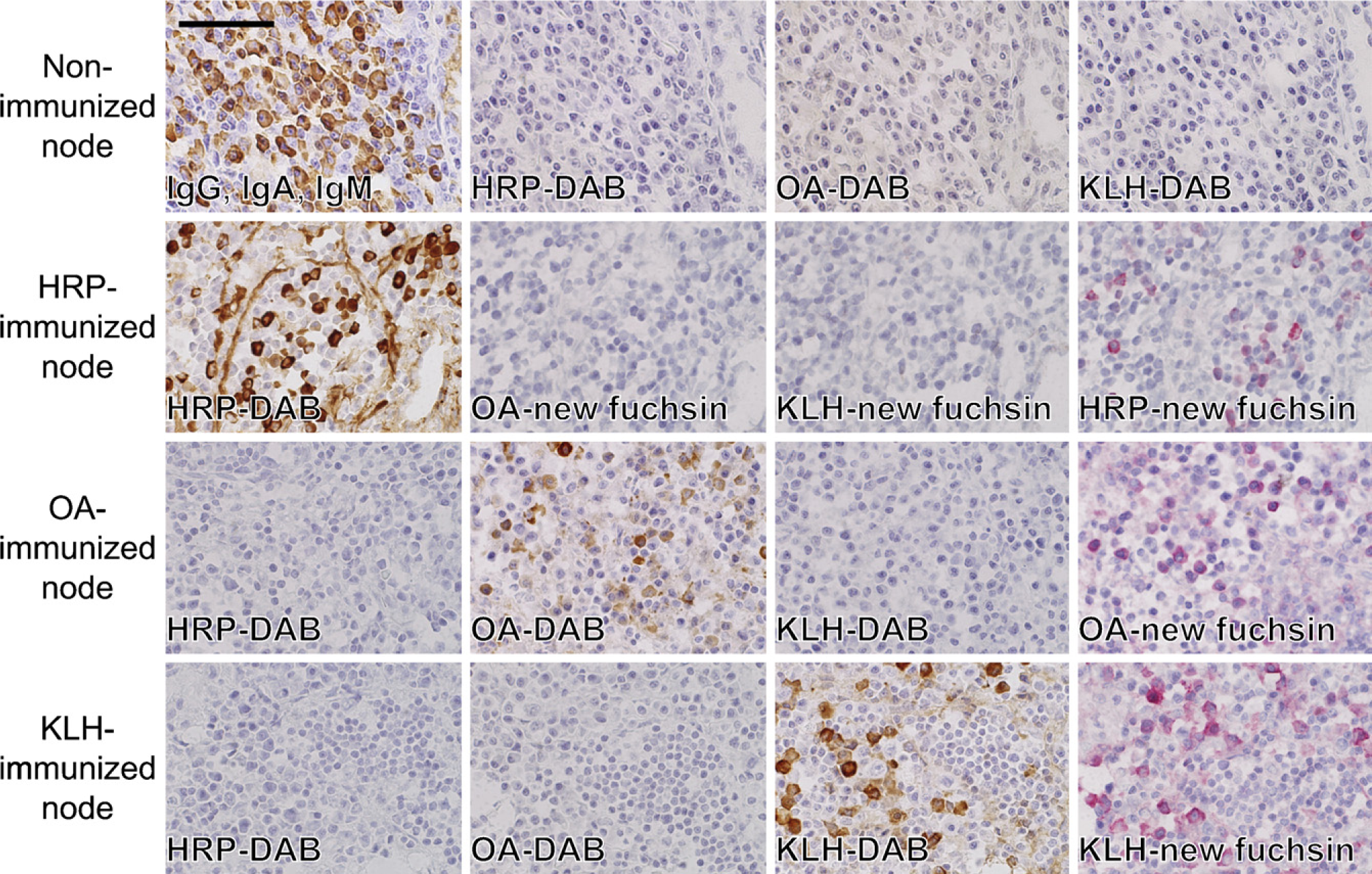
(Top row) PFA-prefixed frozen sections of the popliteal lymph node of non-immunized rats stained, after mild proteolysis, with anti-rat IgG, IgA, IgM polyvalent antibodies, HRP, biotinylated OA, or biotinylated KLH. A good number of immunoglobulin-producing plasma cells are shown by the direct immunoperoxidase method, whereas there are no cells showing antigen-specific antibody production. (Second, third, and fourth row) PFA-prefixed frozen sections of the axillary lymph node stained with the corresponding and indifferent antigens as probes. Proteinase K pretreatment was performed in all sections. The immunocytes are unreactive with indifferent antigen probes. When the sections of HRP-immunized rat tissues were stained with biotinylated OA or KLH, signals were detected in red with alkaline phosphatase–labeled streptavidin and the following new fuchsin color reaction. The rightmost column of the second through bottom rows shows positive controls for the respective antibody activity demonstrated with the alkaline phosphatase reaction. Bar = 50 μm.
Proportions of the Antigen-specific Antibody-producing Cells in Total Plasma Cells
Antigen-specific antibody-producing cells were counted on sections stained with the enzyme-labeled antigen method. Proteinase K-pretreated sections were used for analysis in the case of OA- or KLH-immunized rats. The proportions of the antigen-specific antibody-producing cells in total plasma cells were higher in the lymph nodes than in the spleen. The proportions in the lymph nodes and spleen (mean ± SE) were as follows: 43.5 ± 6.7% and 21.1 ± 5.6% in HRP-immunized rats, 22.0 ± 2.8% and 13.7 ± 3.0% in OA-immunized rats, and 35.8 ± 3.4% and 28.2 ± 5.3% in KLH-immunized rats, respectively. The individual data are summarized in Table 2. In the axillary nodes of the HRP-immunized rats, the average proportion of anti-HRP antibody-producing cells reached 46.3 ± 7.1% (range, 25.7- 68.9%). In the nested ANOVA analysis (Table 3), the proportion of antigen-specific antibody-producing cells in total plasma cells in all the regional lymph nodes (33.6 ± 3.9%) was significantly higher than the spleen (20.4 ± 3.0%). The p value was <0.0001. Representative photomicrographs of the axillary lymph node and spleen are shown in Figure 4.
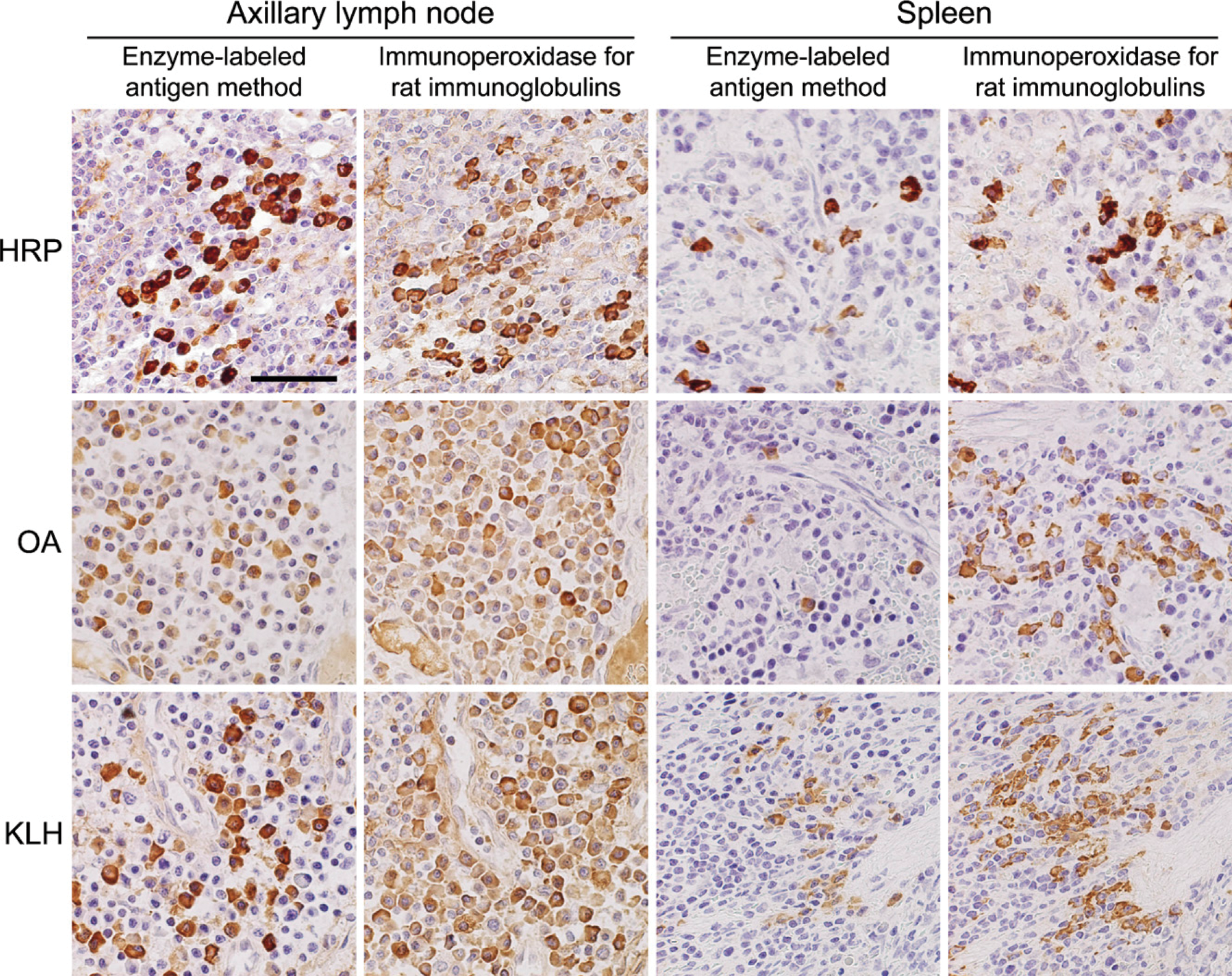
The greater the average proportions of antigen-specific antibody-producing cells in sections of regional lymph nodes, the higher the serum IgG titers (the optical density) in the ELISA analysis (Figure 5). The ANCOVA analysis showed statistically significant correlations between the average proportions of antigen-specific antibody-producing cells in the regional lymph nodes and antigen-specific IgG antibody titers. The p value was 0.0021, where the regression coefficient (β) was 0.0160 and the t(10) value was 4.73.
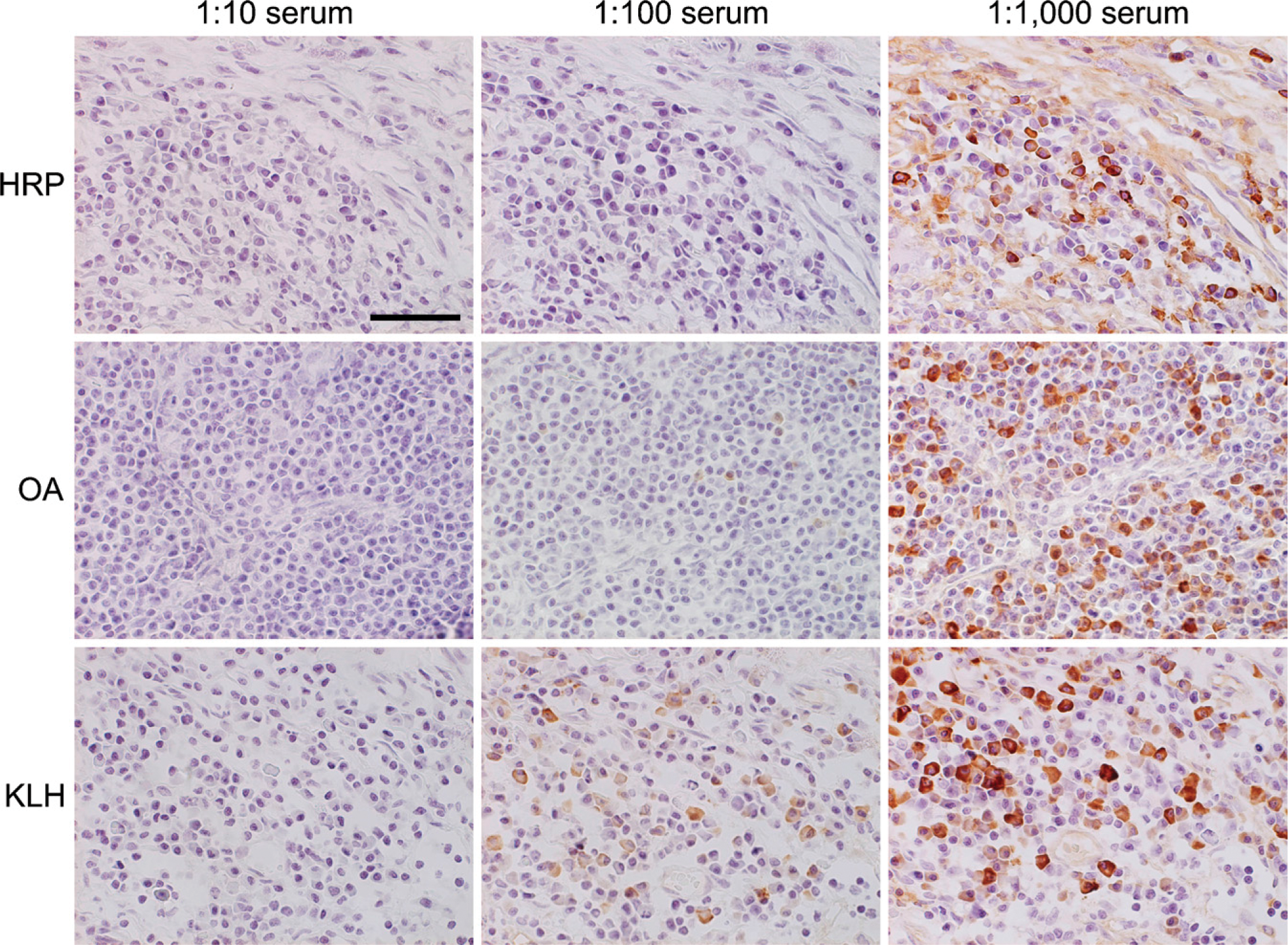
Antibody absorption tests using PFA-prefixed frozen sections of the axillary lymph nodes of the HRP-, OA-, or KLH-immunized rats. The probe antigen solutions were preabsorbed with immunized rat sera (10-, 100-, or 1000-fold diluted). The antigen-binding activity was absorbed only with the corresponding serum in a dilution-dependent manner. Bar = 50 μm.
Proportion of antigen-specific antibody-producing cells in total plasma cells in the popliteal, groin, and axillary lymph nodes and spleen of immunized rats
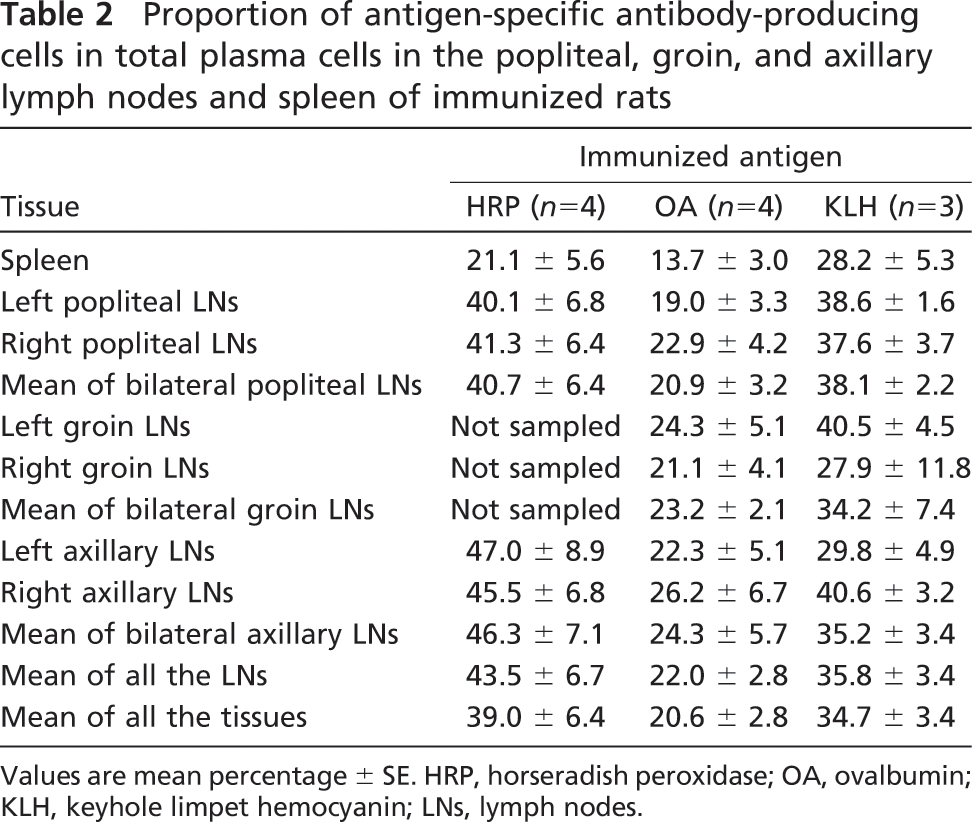
Values are mean percentage ± SE. HRP, horseradish peroxidase; OA, ovalbumin; KLH, keyhole limpet hemocyanin; LNs, lymph nodes.
Enzyme-labeled Antigen Method Using Paraffin Sections
Without proteinase K pretreatment, no positive cells were seen in buffered formalin-fixed and paraffin-embedded sections of the axillary lymph nodes. Heating treatment in citrate buffers or EDTA solution was also ineffective. After pretreatment with proteinase K in a high concentration (80 μg/ml), positive anti-HRP signals were observed in the cytoplasm of plasma cells in paraffin-embedded nodal tissues of HRP-immunized rats (Figure 6A, left). In KLH-immunized nodal sections pretreated with proteinase K, no signal was seen after 1-hr incubation with biotinylated KLH, but a positive reaction appeared after overnight incubation (Figure 6A, right). The signal density was fairly high for anti-HRP activity but weaker for anti-KLH activity. Positive cells in paraffin sections were definitely fewer than those in PFA-fixed frozen sections. No distinct signals were discerned for anti-OA activity, and a relatively high background was observed. In the case of HRP, signals were stably observed in a range of antigen concentrations: 10-100 μg/ml. The concentration of 100 μg/ml was appropriate for identifying anti-KLH activity in paraffin sections, and higher concentrations (250 and 500 μg/ml) gave increased background staining.
The specificity of the positive signals in paraffin sections was confirmed by the cross-check using indifferent antigens and preabsorption tests with diluted sera of the immunized animals. The immunized rat sera absorbed the antigen-specific antibody activity in a dilution-dependent manner (Figure 6B). No signal was identified in non-immune rat tissues.
Discussions
The enzyme-labeled antigen method was first reported by Leduc et al. (1968) and Straus (1968) to histochemically visualize antibody-producing cells in frozen sections of the lymph node. However, this method was never applied thereafter. In this study using HRP, biotinylated OA, and biotinylated KLH as probes, antigen-specific antibody-producing cells were clearly visualized in protease-pretreated or untreated 4% PFA-prefixed frozen sections of the lymph nodes and spleen in HRP-, OA-, or KLH-immunized rats. The specificity of the staining was confirmed by (a) negativity in non-immune rat tissues, (b) negativity with indifferent antigen solutions, and (c) absorption of the reactivity only with the corresponding serum of the immunized rat. Hence, the applicability of the enzyme-labeled antigen method was reconfirmed.
In the analysis on the proportion of antigen-specific antibody-producing cells in total plasma cells, the proportions were significantly higher in the regional lymph nodes than in the spleen. Particularly in the HRP-immunized rats, the proportion reached nearly 50% in the axillary lymph nodes, representing a direct drainage site from the footpad (the antigen-injected site), with the maximal proportion reaching 68.9% in a single node, whereas the proportion was ∼20% in the spleen. It was surprising that an overwhelming large number of antigen-specific antibody-producing cells had locally been provoked in the regional lymph nodes. To our knowledge, there has been no report describing the proportion of antigen-specific antibody-producing cells in total plasma cells in the lymphoid tissue. Kishiro et al. (1995) reported that the establishment of anti-OA antibody-producing hybridomas was ∼10 times more efficient with the use of medial iliac lymph nodes than with the use of the spleen, when OA was immunized in the footpad of the rat. Sado et al. (2006) also demonstrated that the efficiency in the production of monoclonal antibodies against OA was higher in the iliac lymph nodes than in the spleen, when the antigen was challenged into the tail base. The present study showed that a huge number of antigen-specific antibody-producing immunocytes are accumulated in regional lymph nodes sensitized with the antigens.
Nested ANOVA analysis for comparing the proportion of antigen-specific antibody-producing cells in total plasma cells in the regional lymph nodes with that in the spleen
HRP, horseradish peroxidase; OA, ovalbumin; KLH, keyhole limpet hemocyanin.
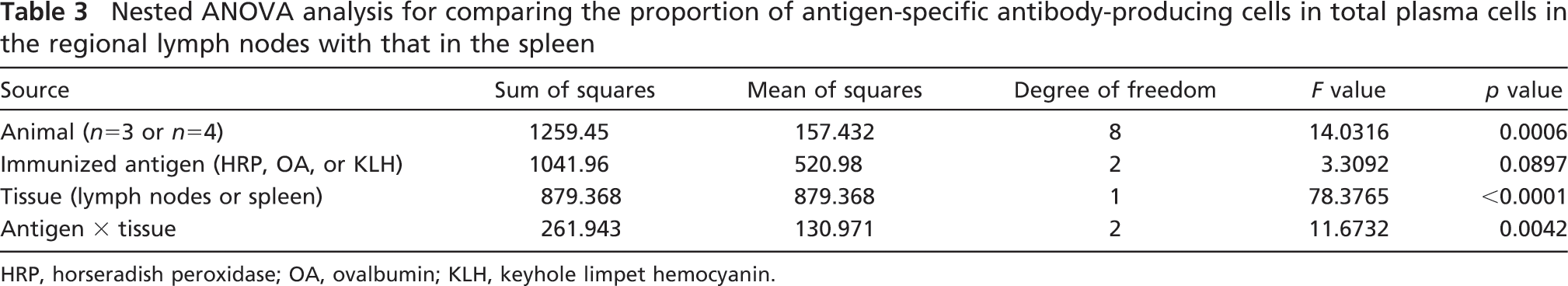
Comparison between the enzyme-labeled antigen method and the direct immunoperoxidase method for rat immunoglobulins. PFA-prefixed frozen sections of the axillary lymph nodes and spleens of HRP-, OA-, or KLH-immunized rats were reacted with the corresponding antigen probes (first and third columns, respectively). Proteolysis was added for localizing anti-OA and anti-KLH activity. The consecutive sections were stained with anti-rat IgG, IgA, and IgM polyvalent antibodies (second column for the node and fourth column for the spleen). A large percentage of plasma cells distributed in the medullary cord of the node show positivity for specific antibody production. Some plasma cells in the splenic red pulp show positivity. Bar = 50 μm.
Proteinase K pretreatment retrieved antigen-binding activity of the plasma cells in 4% PFA-prefixed frozen section of lymph nodes of OA- or KLH-immunized rats. Cross-linkage of amino residues through formaldehyde fixation may cause alteration of three-dimensional structures of the antigen-binding site of immunoglobulin molecules. Subsequently, the antigen-antibody reaction may sterically be hampered (Ramos-Vara 2005). Proteolytic enzyme cleavage of the protein cross-linkage by proteinase K retrieved the antigen-binding activity of immunoglobulins in fixed tissues, as is so in antigen retrieval (unmasking) in the enzyme-labeled antibody method or IHC (Battifora and Kopinski 1986). Proteinase K is a representative of the proteolytic enzymes (Privat et al. 2000). Proteolytic digestion was actually unnecessary for localizing positive signals of anti-HRP antibody activity in prefixed frozen sections. In the case of HRP, the antigen itself carries enzyme activity and it does not need to be labeled with another enzyme or biotin. HRP is a highly sensitive and stable enzyme for color development in the DAB reaction, and the molecular mass is relatively low, ∼44 kDa (Welinder 1979). The penetration of the antigen molecules to plasma cells in frozen sections may thus be relatively prompted.
Regression lines shown between the antigen-specific IgG antibody titer in the rat serum and the average proportion of antigen-specific antibody-producing plasma cells in the regional lymph nodes. Significant correlation was observed between the serum antibody titer and the number of plasma cells producing specific antibodies. The ANCOVA analysis shows statistical significance (p<0.0021).
OA is also a low-molecular-mass protein, ∼44 kDa, but is not an enzyme by itself (Tai et al. 1977; Nisbet et al. 1981). As a result, it is plausible that streptavidin-labeled HRP molecules bound through biotinylated OA onto the immunocytes in fixed frozen sections were fewer than HRP molecules in the anti-HRP series. Biotinylation may cause alteration of the antigenicity of OA. In addition, HRP carries a higher amount of carbohydrate chains on the molecule than OA and KLH: HRP, 20.1%; OA, 3.2%; KLH, 3.3% (Tai et al. 1977; Welinder 1979; Kurokawa et al. 2002). The carbohydrate residues may be antigenic, and the carbohydrate antigen may be more stable in antigenicity than the protein antigen. They may give molecular stability to the core protein structures. Expectedly, the antigenicity of HRP was thus more stably exposed onto immunoglobulin molecules in prefixed frozen sections than that of OA.
In contrast, KLH is composed of macromolecules with a wide range of molecular mass from 345 kDa to 8.3 MDa (Söhngen et al. 1997). Therefore, it seems to be difficult for KLH molecules to penetrate frozen sections to reach the target antibodies. Proteolytic pretreatment may assist the probe molecules by penetrating the prefixed tissue sections. Because proteinase K is a nonspecific serine protease, the antigen-binding activity of immunoglobulins may be inactivated by cleaving their antigen recognition site. In this situation, other proteases such as trypsin, pepsin, pronase, ficin, and actinase (Hazelbag et al. 1995; Liapis and Hutton 1997) can be tried for “antibody retrieval.”
In a variety of pathological conditions, including inflammatory bowel diseases, Epstein-Barr virus-related carcinomas such as lymphoepithelioma of the nasopharynx and gastric carcinoma with lymphoid stroma, autoimmune disorders, and infectious diseases, a large number of lymphocytes and plasma cells infiltrate within the lesions (Fukayama et al. 1998; Rowley et al. 2000; Hoang et al. 2004; Rahman et al. 2007). However, it has never been clarified what kind of antigens are recognized by immunoglobulins locally secreted from the plasma cells. It is reasonable to consider that the immunoglobulins are closely correlated with the pathological process of the disorder in question, simply because they are produced locally within the lesion. It is expected that a large proportion of plasma cells may produce antibodies against limited sorts of pathogenic antigen molecules. Therefore, once the probe antigen is available, disease-specific antibodies may be identified in tissue sections by the enzyme-labeled antigen method. In a variety of infectious diseases, specific antibody titers against pathogens raise in the patients' sera (Tsutsumi 2007). The antibodies against pathogens should be produced at the local site of infection or in the regional lymphoid tissues. Topological relationship between the pathogen and antipathogen immune reaction will be visualized with this old-fashioned but novel methodology. Similarly, the site of production of autoantibodies can be specified. In many examples of infectious diseases and autoimmune disorders, the antigenic features have been well documented, and thus the antigen probes can be prepared and used.
There is no doubt that the fixation condition is important for properly applying the enzyme-labeled antigen method. In this model study, 4% PFA fixation was shown to be appropriate for reproducibly detecting antibody activity against HRP, OA, or KLH. When sections of the lymph node of HRP-immunized rat were preliminarily stained with HRP as a probe, there was no clear difference in stainability of anti-HRP antibody-producing plasma cells between PFA-prefixed frozen sections and fresh frozen sections postfixed in PFA (data not shown). The morphological preservation was much better in the prefixed sections than that in the postfixed sections. At this point, we judged PFA prefixation as an appropriate procedure for this methodology. In the case of OA and KLH, we did not compare the stainability between the prefixed and postfixed frozen sections. However, other fixatives, including ethanol-based ones, should further be evaluated in accordance with the property of the target antigens.
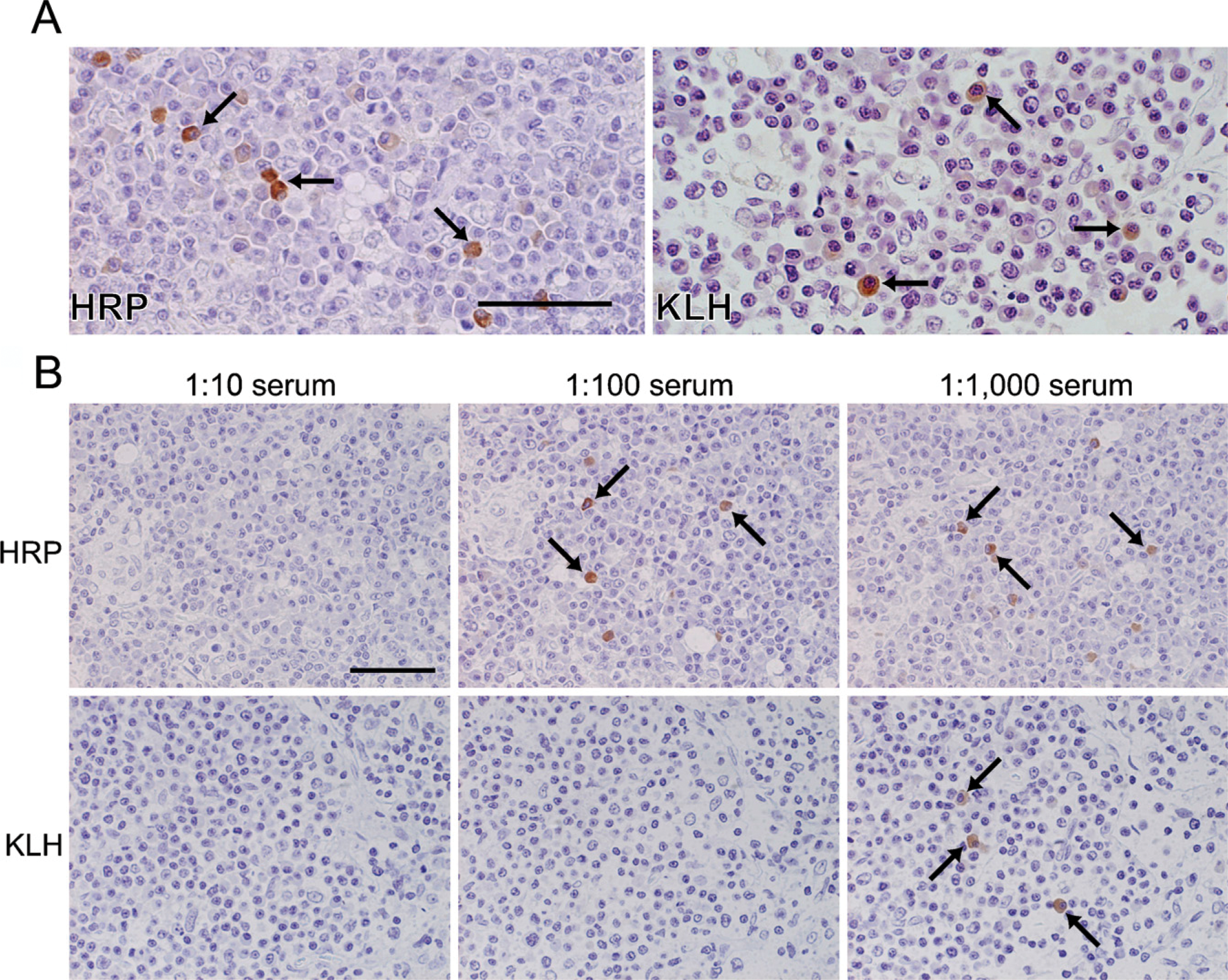
(
It is potentially important, particularly for pathologists, that the specific antibodies against HRP are detectable in buffered formalin-fixed, paraffin-embedded sections after strong proteolytic pretreatment. Anti-KLH antibody activity was also shown in protease-pretreated paraffin sections after prolonged (overnight) incubation with the antigen probe. However, positive immunocytes in paraffin sections were fewer than those in frozen sections. The specificity of the anti-HRP and anti-KLH reactivities was confirmed. In contrast, anti-OA activity was undetectable. As in the immunoperoxidase method, improved and relevant retrieval of the antigen-binding activity may lead to the wide use of archival paraffin sections for the enzyme-labeled antigen method in the near future.
Footnotes
Acknowledgements
This work was supported by Grants-in-Aid 17390106 and 18659103 from the Ministry of Education, Research, Technology and Labor, Japan.
The authors thank Prof. Shuji Hashimoto, PhD, Department of Hygiene, Fujita Health University School of Medicine, Toyoake, for valuable advice and suggestions in the statistical analysis.
