Abstract
Periostin is a unique extracellular matrix protein, deposition of which is enhanced by mechanical stress and the tissue repair process. Its significance in normal and neoplastic colon has not been fully clarified yet. Using immunohistochemistry and immunoelectron microscopy with a highly specific monoclonal antibody, periostin deposition was observed in close proximity to pericryptal fibroblasts of colonic crypts. The pericryptal pattern of periostin deposition was decreased in adenoma and adenocarcinoma, preceding the decrease of the number of pericryptal fibroblasts. Periostin immunoreactivity appeared again at the invasive front of the carcinoma and increased along the appearance of cancer-associated fibroblasts. ISH showed periostin signals in cancer-associated fibroblasts but not in cancer cells. Ki-67–positive epithelial cells were significantly decreased in the colonic crypts of periostin−/- mice (∼0.6-fold) compared with periostin+/+ mice. In three-dimensional co-culture within type I collagen gel, both colony size and number of human colon cancer cell line HCT116 cells were significantly larger (∼1.5-fold) when cultured with fibroblasts derived from periostin+/+ mice or periostin-transfected NIH3T3 cells than with those from periostin−/- mice or periostin–non-producing NIH3T3 cells, respectively. Periostin is secreted by pericryptal and cancer-associated fibroblasts in the colon, both of which support the growth of epithelial components.
P
Periostin has also been shown in various neoplastic tissues, including cancers of the breast, pancreas, and colon (Sasaki et al. 2003; Bao et al. 2004; Shao et al. 2004; Tai et al. 2005; Grigoriadis et al. 2006; Baril et al. 2007), but the tissue distribution has been controversial. Periostin, when transfected to cancer cells, promotes tumor angiogenesis, metastatic growth, cancer cell motility, and adhesion (Gillan et al. 2002; Bao et al. 2004; Shao et al. 2004; Kudo et al. 2006; Yan and Shao 2006; Baril et al. 2007). Although most of these studies postulate cancer cells as a source of periostin, there have been unexpectedly few cancer cell lines that expressed significant amounts of periostin mRNA in vitro (Gillan et al. 2002; Bao et al. 2004; Yan and Shao, 2006) (unpublished data). It is also controversial which types of cells produce periostin in the neoplastic tissue by morphological studies. Baril et al. (2007) showed mRNA of periostin in cancer cells by ISH in pancreatic cancer. In contrast, strong expression of periostin mRNA was observed in cancer stromal cells in lung cancers and breast cancers (Sasaki et al. 2001, 2003). Erkan et al. (2007) showed that stellate cells produced periostin in normal and neoplastic pancreas.
In this study, we investigated localization and function of periostin in non-neoplastic and neoplastic colon, with special attention to the PCF and the other type of myofibroblast, the cancer-associated fibroblast (CAF). Furthermore, functional studies, using periostin knockout mice, also showed growth-promoting function of periostin molecules.
Materials and Methods
Tissue Specimens
Tissue specimens consisted of non-neoplastic tissues of human colon including the biopsy specimens of ulcerative colitis (3 cases) and the neoplastic tissues; carcinoma in adenoma (16 cases), carcinoma with early invasion (8 cases) and advanced invasion (24 cases); and metastatic colon cancer of the liver (13 cases). All specimens were retrieved from the files of the Department of Diagnostic Pathology, University of Tokyo Hospital. According to the criteria of the Japanese Research Society for Cancers of the Colon and Rectum (Japanese Society for Cancer of the Colon and Rectum 2006), we regarded early carcinomas as those showing invasion to the submucosal layer and advanced invasive as colon cancers beyond the proper muscular layer. Tissue samples had been formalin fixed and embedded in paraffin for histopathological examination.
For the assessment of RT-PCR, frozen tissue samples of the carcinoma and non-neoplastic mucosa were obtained from the surgically resected colons for the treatment of the carcinomas at Jichi Medical School Hospital. These tissue specimens were immediately frozen and stored at −80C. Informed consent was obtained from each patient to allow the use of portions of tissue for research purposes, and the study was approved by the ethical committee of the institutions.
For the histological analysis of the colon of periostin−/- mice, we used three 12-week-old mice homozygous for the disrupted periostin gene (Kii et al. 2006; Shimazaki et al. 2008) and the same number of wild-type littermates.
Mice were treated in accordance with the policies of the Animal Ethics Committee of the University of Tokyo.
Cell and Culture
The colon cancer cell line HCT116, mouse fibroblast cell line NIH3T3, and periostin+/+ or periostin−/- mouse lung primary-cultured fibroblasts were used for proliferation assay and/or colony formation assay. They were routinely grown in DMEM (Sigma; St. Louis, MO) with 10% FBS, 100 U/ml penicillin, and 100 μg/ml streptomycin, at 37C saturated with 5% CO2 in a humid atmosphere. Primary-cultured lung fibroblasts derived from periostin+/+ and periostin−/- mice were obtained by the digestion method with DISPASE I (GODO SHUSEI Co.; Tokyo, Japan) according to the manufacturer's instructions. These primary-cultured fibroblasts were used in experiments before the fifth passage.
Transfection
Expression vectors for mouse periostin with HA tag and enhanced green fluorescent protein (EGFP) were previously constructed in pCAGIPuro (Li et al. 1999) (kindly provided by Dr. H. Niwa, RIKEN Center for Developmental Biology, Kobe, Japan) (Kii I, et al., unpublished data). Cells were transfected by FuGene 6 Transfection Reagent (Roche Diagnostics; Indianapolis, IN) according to the manufacturer's instructions. Selection and cloning were performed using puromycin (Sigma).
Immunohistochemistry and Double Immunofluorescence
The primary antibodies used in this study were a rabbit polyclonal anti-human periostin antibody (Shimazaki et al. 2008), a mouse monoclonal anti-α smooth muscle actin (αSMA) (1A4, 1:50; DakoCytomation, Glostrup, Denmark), a rabbit polyclonal anti-single-strand DNA antibody (ssDNA, 1:100; DakoCytomation), and a rat monoclonal anti-mouse Ki-67 antibody (TEC-3, 1:50; DakoCytomation), FITC-conjugated monoclonal anti-αSMA (mouse monoclonal 1A4-FITC, 1:300; Sigma). For immunohistochemistry, antibody binding was visualized by the avidin-biotin complex technique using 3,3-diaminobenzidine (DAB) as the chromogen. For double immunofluorescence, binding of the periostin antibody was visualized by goat anti-rabbit IgG labeled with Alexa fluor 594 (Molecular Probes; Eugene, OR), and sections were mounted with 4′,6-diamidine-2′-phenylindole dihydrochloride (DAPI) for nuclear staining (Vectashield Hardset Mounting Medium with DAPI; Vector Labs, Burlingame, CA).
For the assessment of pericryptal immunoreactivity of the colonic crypts or tumor glands with semiquantification, we defined positive crypts or glands when all circumferences were surrounded by immunoreactive material. The population of positive crypts or glands was counted and scored in four groups according to prevalence: score 0, <10%; score 1, 10-49%; score 2, 50-80%; score 3, >80%.
Immunoelectron Microscopy
Normal mouse colon tissues of Jcl: MCH (ICR) mice, 10 weeks old (CLEA Japan; Tokyo, Japan), were fixed in 4% paraformaldehyde-phosphate buffer (pH 7.0) overnight at 4C and washed in a series of ascending concentrations from 10% to 15%, 20% sucrose in PBS each for 4 hr, and then immersed in 20% sucrose and 5% glycerol in PBS for 1 hr. The tissues were embedded in OCT compound and frozen. After frozen sections (8 μm thick) were cut in a cryostat, primary antibody and secondary antibody were applied, and antibody binding was visualized by DAB as described above. They were then embedded in Epon 812. For light microscopic analysis, semithin sections (1 μm) were stained with toluidine blue. For immunoelectron microscopy, ultrathin sections were contrasted only with uranyl acetate omitting the staining of lead citrate and examined with an electron microscope (Model 1200EX; JEOL, Tokyo, Japan).
ISH for Periostin mRNA
Antisense and sense cRNA probes were prepared by in vitro transcription of the EcoRI-XbaI fragment of human periostin cDNA (Accession number D13665) (Takeshita et al. 1993), using a DIG Labeling MIX (Roche; Indianapolis, IN). The mRNA ISH was conducted according to the manufacturer's instructions (Roche).
Quantitative Periostin mRNA Real-time RT-PCR
Total RNA was isolated by the acid guanidine-thiocyanate/phenol/chloroform method or RNeasy Mini Kit (Qiagen; Valencia, CA). Reverse transcription was performed using Ready To Go 1st strand cDNA kit (GE Healthcare Bio-Sciences; Uppsala, Sweden).
For the quantitative periostin real-time RT-PCR, the housekeeping gene, β-actin, was used as an internal reference. The sequences of the primer sets were as follows: human periostin, 5′-GACTCAAGATGATTCCCTTT-3′ (forward) and 5′-GGTGCAAAGTAAGTGAAGGA-3′ (reverse); β-actin, 5′-GGCCACG-GCTGCTTC-3′ (forward), 5′GTTGGCGTACAGGTCTTTGC-3′ (reverse). Real-time RT-PCR for periostin and β-actin was performed in parallel and in triplicate per sample, using an iCycler iQ Multi-Color Real Time PCR Detection System (Bio-Rad Laboratories; Hercules, CA) and the following protocol: 50C for 2 min, 95C for 15 min, and 45 cycles at 94C for 30 sec, 60C for 30 sec, and 72C for 30 sec. PCR was performed in a 50-μl reaction mixture containing 25 μl of Platinum SYBR Green qPCR SuperMix-UDG (Invitrogen; Carlsbad, CA), 2.5 μl of forward and reverse primer, 1 μl of each cDNA sample, and 19 μl of ddH2O. The relative ratio of expression of periostin and β-actin are shown as mean ± SEM.
Proliferation Assay
Proliferation assay was performed using Cell Counting Kit-8 (CCK-8; Dojindo Laboratories, Kumamoto, Japan), a variant MTT assay kit, as the technical manual described with the supplementation of a final concentration of 0.1% BSA and either recombinant human periostin (Biovendor; Heidelberg, Germany) dissolved in PBS or equal amounts of PBS without periostin.
Three-dimensional Co-culture With Cancer Cells and Fibroblasts for the Colony Formation Assay
Collagen gel matrix was prepared using a type I collagen culture kit (Cellmatrix; Nitta Gelatin, Osaka, Japan). HCT116 and either primary-cultured lung fibroblasts derived from periostin+/+ and periostin−/- mice or NIH3T3 with transfection of periostin or EGFP at each final concentration of 1 × 106/ml were mixed with chilled reconstituted collagen, and 1 ml of the mixture was pored into a 12-well culture plate and allowed to gel at 37C for 30 min. The next day, the matrix was detached from the bottom of the dish and floated below the surface of the medium. Floating collagen gel culture was conducted for 2 weeks. After that, 10% phosphate-buffered formalin-fixed and paraffin-embedded sections were studied with hematoxylin and eosin stain. Photographs of >36 focuses of high-power fields (HPFs) at random were taken, and each colony area was squared using Multi Gauge ver.2.2 software (FUJIFILM; Tokyo, Japan).
Statistical Analysis
All numerical results are shown as the mean ± SEM. The data were evaluated by Mann-Whitney U test or Student's t-test for statistical comparisons (cut-off for statistical significance, p<0.05) using Statview software (SAS Institute Inc.; Cary, NC).
Results
Immunoreactive Periostin in PCFs of Human and Mouse Colon
In normal colorectal mucosa of both humans and mice, periostin immunoreactivity was observed in the stroma surrounding the crypts (Figure 1A), making up the “pericryptal pattern.” There was no immunoreactivity in the epithelial cells of the colonic crypts or other mesenchymal cells, except for vascular smooth muscle cells. Immunofluorescent double staining for periostin and αSMA (Figure 1B) showed a close approximation of periostin immunoreactivity to αSMA-positive cells, which represent the PCFs. Importantly, periostin deposition was present outside of the basement membrane and frequently faced the αSMA-positive PCFs (Figure 1B).
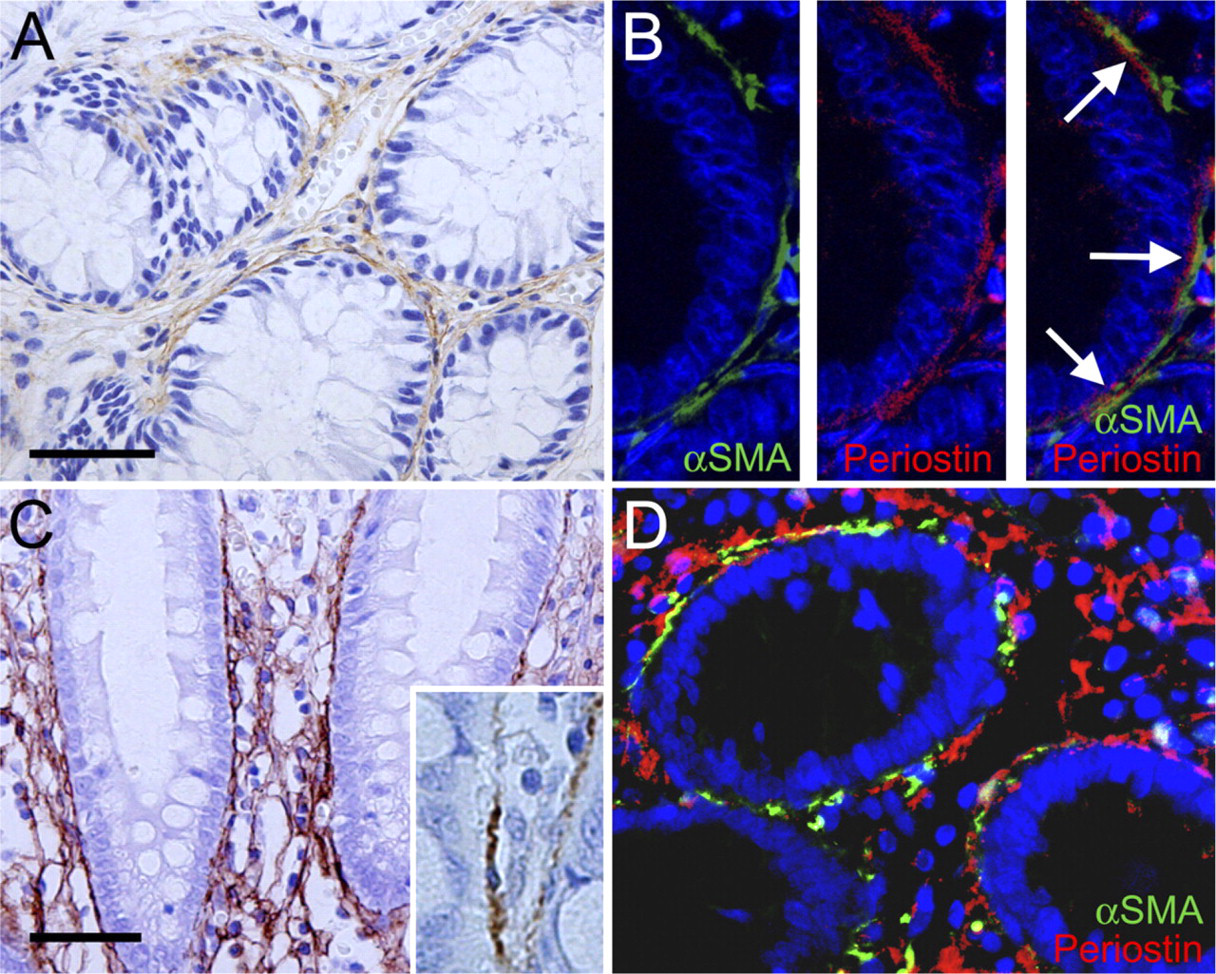
Periostin in non-neoplastic colorectal mucosa. (
In the inflammatory condition such as ulcerative colitis, the intensity of the pericryptal pattern of periostin immunoreactivity became stronger; besides the linear immunoreactive material, a mesh pattern was observed throughout the lamina propria (Figures 1C and 1D).
Immunoelectron microscopy of the mouse colonic mucosa (Figure 2) showed the localization of immunoreactivity for periostin. Immunoreactive material was predominantly identified in the narrow space between the PCF and basement membrane, and it mainly delineated the surface of the cytoplasm of PCFs (Figure 2B). There was a small amount of immunoreactive material within the cytoplasm of the PCF, but it was difficult to localize it precisely at the subcellular organella. On the other hand, there was no immunoreactivity identified within the cytoplasm of the epithelial cells. Thus, these findings indicate that PCFs primarily secrete periostin outside to the interstitium, resulting in a pericryptal pattern in the non-neoplastic colorectal mucosa at the light microscopic level.
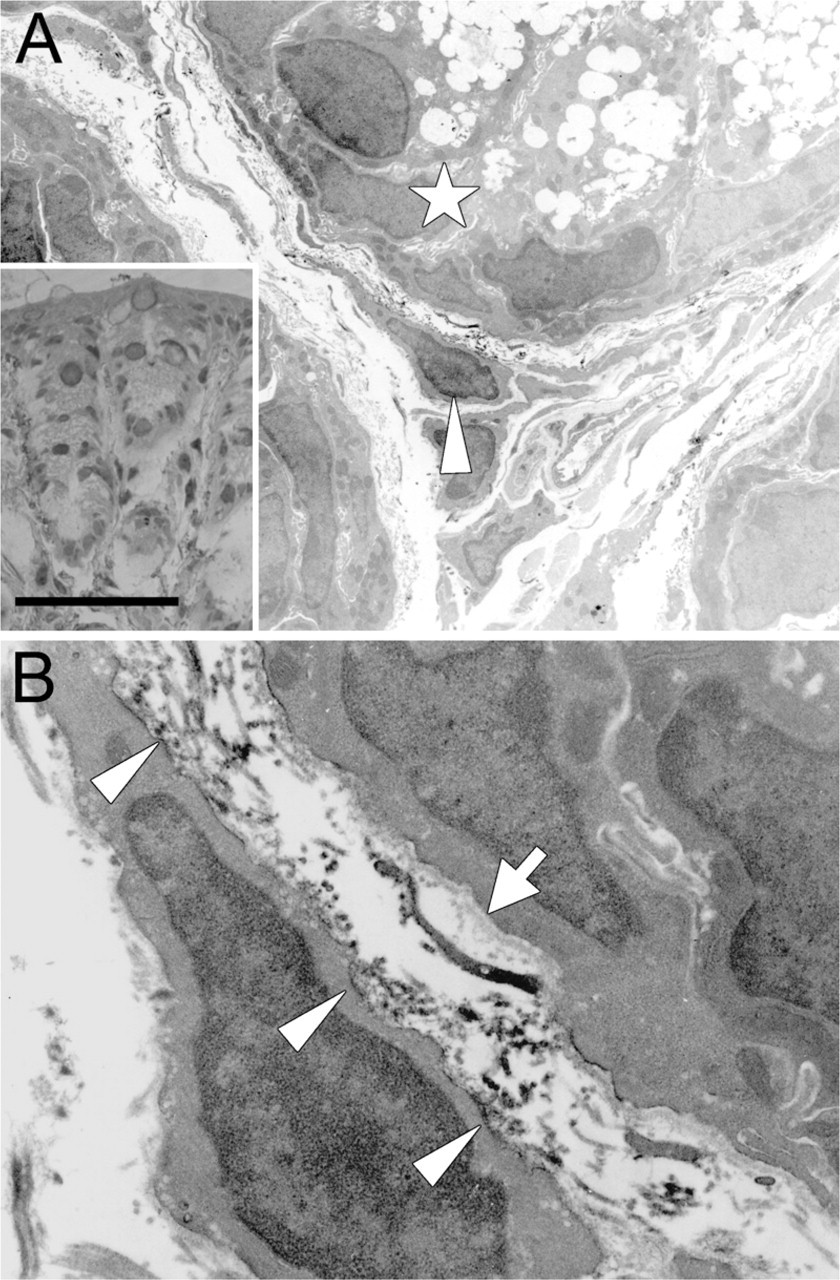
Immunoelectron microscopy of periostin in normal colonic mucosa of the mouse. (
Dynamics of Periostin Deposition in Relation to the PCF in Pathological Conditions
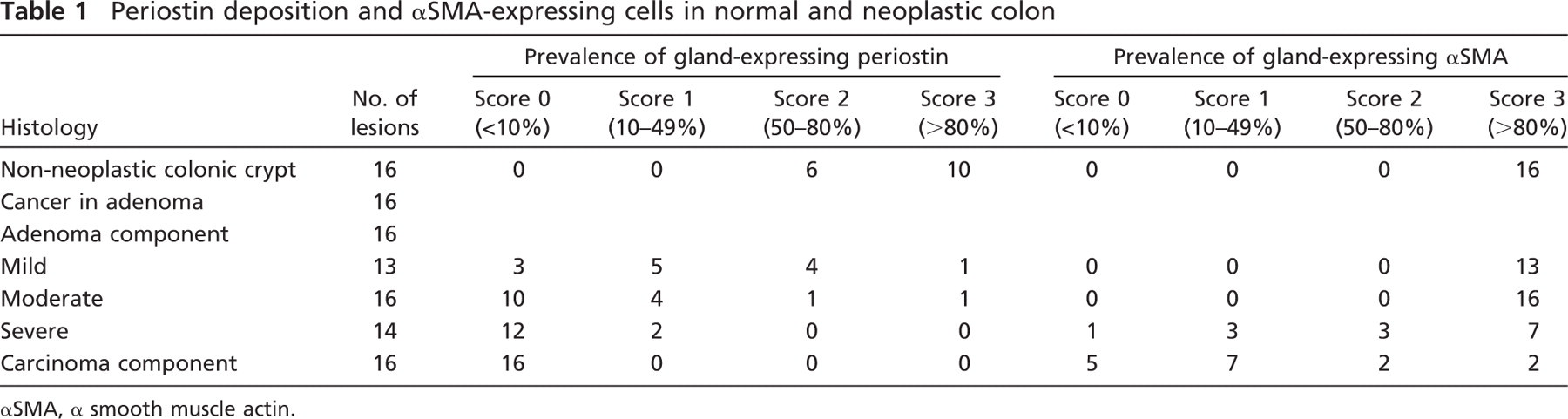
It is known that the number of PCFs is decreased along the adenoma-carcinoma sequence (Yao and Tsuneyoshi 1993; Higaki et al. 1999; Li et al. 1999). Immunoreactive periostin in the stroma, pericryptal pattern was weakened (Figures 3A and 3B) or disappeared in the adenoma component and completely disappeared in the adenocarcinoma component (Figures 3C and 3D). On immunofluorescence double staining, the amount of periostin immunoreactivity was decreased, and its intensity became weak in the adenoma, even when the number of αSMA-positive cells seemed to be similar to that in normal or hyperplastic mucosa (Figure 3E). Thus, semiquantitative comparative evaluation of periostin and αSMA immunoreactivity showed that the decrease of pericryptal immunoreactivity of periostin precedes the decrease of PCFs along the adenoma-carcinoma sequence (Table 1; Figure 3F).
Immunoreactive Periostin in Cancer-associated Fibroblasts
Periostin immunoreactivity was observed again at the stroma of the early invasive cancer (Figure 3G). The immunoreactive material showed a fibrillar shape, but the intensity was relatively weak and confined to the invasive front. When the carcinoma further invaded beyond the proper muscles, the stroma integrated within the carcinoma exhibited strong periostin immunoreactivity (Figures 3H and 3I). It became strong and dense compared with immunoreactivity observed in early invasive carcinoma. There were many fibroblastic cells with comparatively plump nuclei within the area showing periostin immunoreactivity (Figure 3J). No staining was observed in carcinoma cells. These findings were highly reproducible; the intensity and localization of immunoreactive periostin did not change by size, grade, or type of carcinoma.
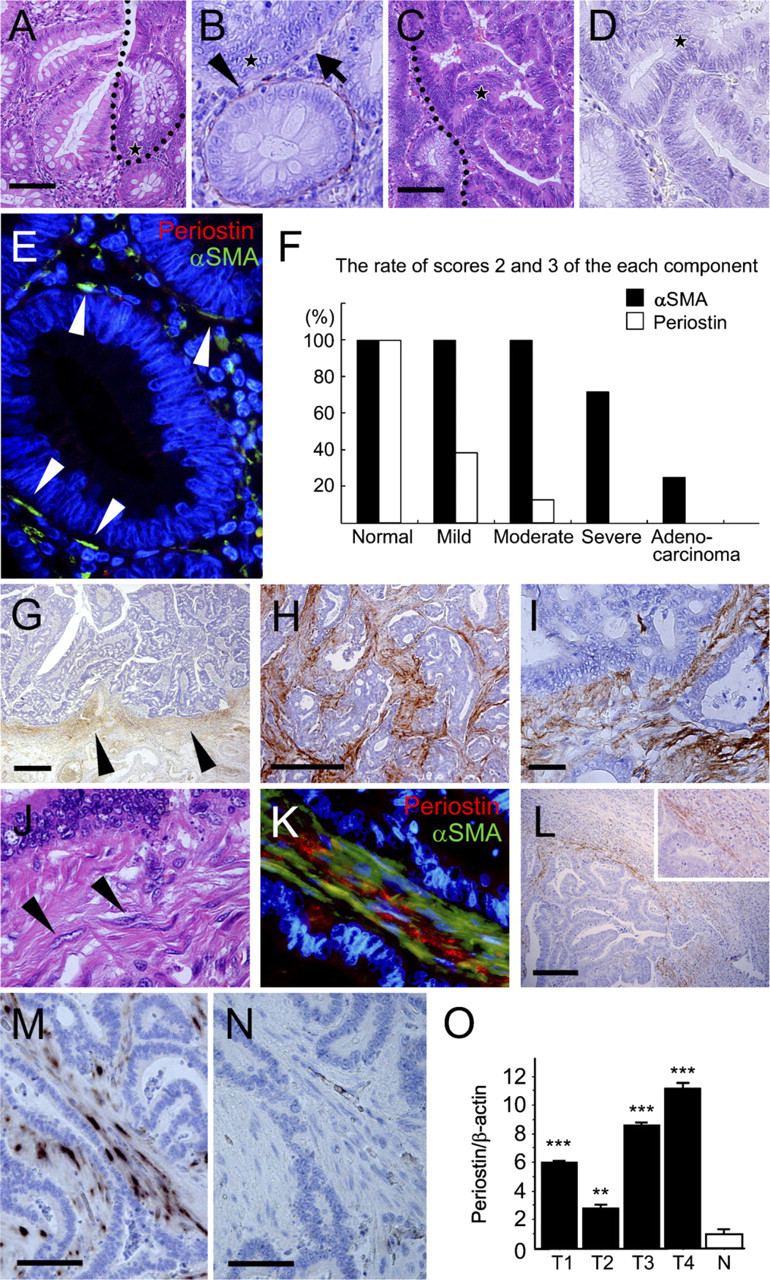
Periostin in adenoma and carcinoma of the colon. (
Periostin deposition and αSMA-expressing cells in normal and neoplastic colon
αSMA, α smooth muscle actin.
Fibroblasts in cancer stroma, showing characteristics of myofibroblasts, are frequently called CAFs, and one of the well-used immunohistochemical markers for CAF is αSMA, similar to PCFs. The immunofluorescent double staining for αSMA and periostin in advanced invasive cancer (Figure 3K) showed that fibroblast immunoreactive for αSMA appeared in the cancer stroma, and immunoreactive periostin was observed around these cells.
In the metastatic carcinoma of the liver, periostin immunoreactivity was observed in the capsular stroma (Figure 3L). There was little periostin immunoreactivity in normal liver tissue, including the portal areas.
Evaluation of Periostin mRNA Expression by ISH and RT-PCR
To evaluate periostin expression at the mRNA level, ISH was applied to the representative sections of normal and neoplastic colon with antisense (Figure 3M) and sense probes (Figure 3N). There were positive signals of periostin mRNA in the cytoplasm of fibroblastic cells in the stroma of the invasive carcinomas (Figure 3M). No signal was observed in the carcinoma cells. Thus, the finding confirmed that periostin was expressed in CAFs but not in neoplastic epithelial cells.
Quantitative evaluation of mRNA of periostin was performed by real-time RT-PCR in normal colonic mucosa and carcinoma tissues. The amount of periostin mRNA in normal tissue was apparently lower than that in colorectal carcinomas (Figure 3O).
Epithelial Proliferation and Periostin in Normal and Neoplastic Colon
To obtain an insight on the function of periostin produced by PCFs, we performed histological analysis of colon crypts in periostin−/- and periostin+/+ mice. There was no significant difference in the number of epithelial cells or PCFs per crypt and the distribution pattern of PCFs between periostin−/- mice and periostin+/+ mice (data not shown). PCFs in periostin−/- mice also showed immunoreactivity for αSMA. There was also no difference in the thickness of lamina propria between them. However, the number of Ki-67–positive epithelial cells per crypt was significantly decreased in periostin−/- mice (25.32 ± 8.29/crypt) compared with that in periostin+/+ mice (43.04 ± 12.38/crypt) (Figure 4A). Immunohistochemical study for single-strand DNA was also performed to examine the levels of apoptosis, and there was no significant difference in the number of apoptotic cells per crypt between periostin−/- mice (0.13 ± 0.08/crypt) and periostin+/+ mice (0.07 ± 0.02/crypt).
To further extend this observation to the neoplastic colon, we evaluated the proliferative effect of periostin on human cancer cells, HCT116, using a proliferation assay with human recombinant periostin. The proliferation of HCT116 cells showed a significant increase in periostin-supplemented conditions at 100 ng/ml after 6 hr of incubation (Figure 4B). The proliferation promoting effect was observed at 10 ng/ml and showed significant difference at 50-1000 ng/ml when evaluated at 24 hr of incubation (Figure 4C).
Cancer–Stromal Interaction Through Periostin in Type I Collagen Gel Three-dimensional Co-culture
For the assessment of cancer–stromal interaction through periostin produced by fibroblasts, we performed a colony formation assay under the three-dimensional co-culture of HCT116 cells and fibroblasts in type I collagen gel. In HCT116 cells co-cultured with periostin+/+ fibroblasts, both colony number and size of HCT116 cells were significantly larger (1.30- and 1.30-fold, respectively) than in the co-culture with periostin−/- fibroblasts. We also tested this experiment using transfectants of the fibroblast cell line, periostin-transfected NIH3T3-secreting periostin and EGFP-transfected NIH3T3 cells. Culture supernatant of the latter NIH3T3 did not show any amount of immunoreactive periostin. As a result, the difference was much more striking (1.48- and 1.54-fold, respectively) when compared between the co-cultures with periostin- and EGFP-transfected NIH3T3 cells (Figure 5; Table 2).
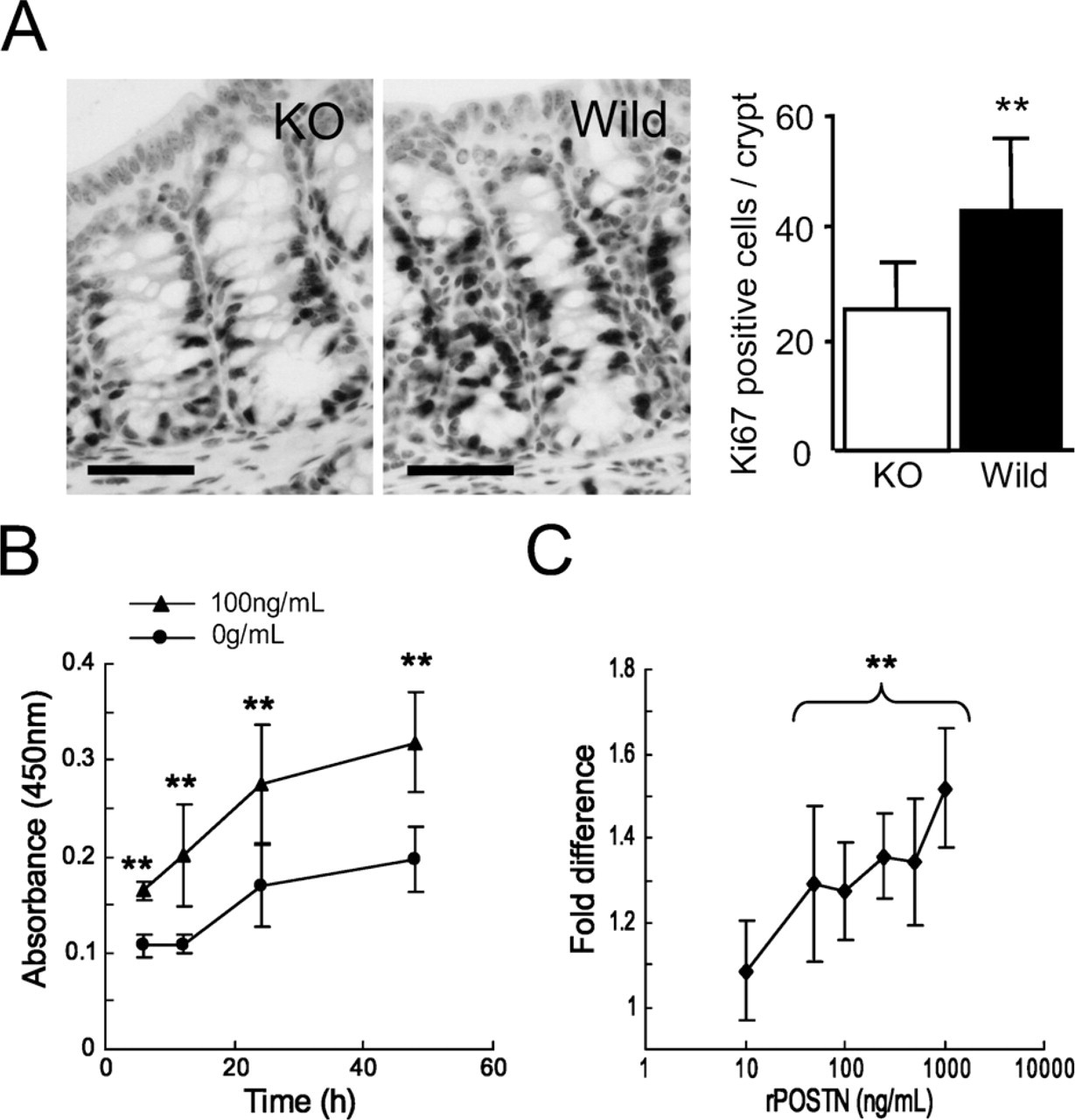
Evaluation of effect of periostin on epithelial proliferation of normal and neoplastic colon. (
Discussion
Periostin is primarily an extracellular matrix protein, and its function has been intensively studied in non-neoplastic and neoplastic tissues. In this study, we showed that there were several patterns of periostin deposition, such as pericryptal pattern, stromal mesh pattern, and infiltrating pattern, which are shown in Table 3.
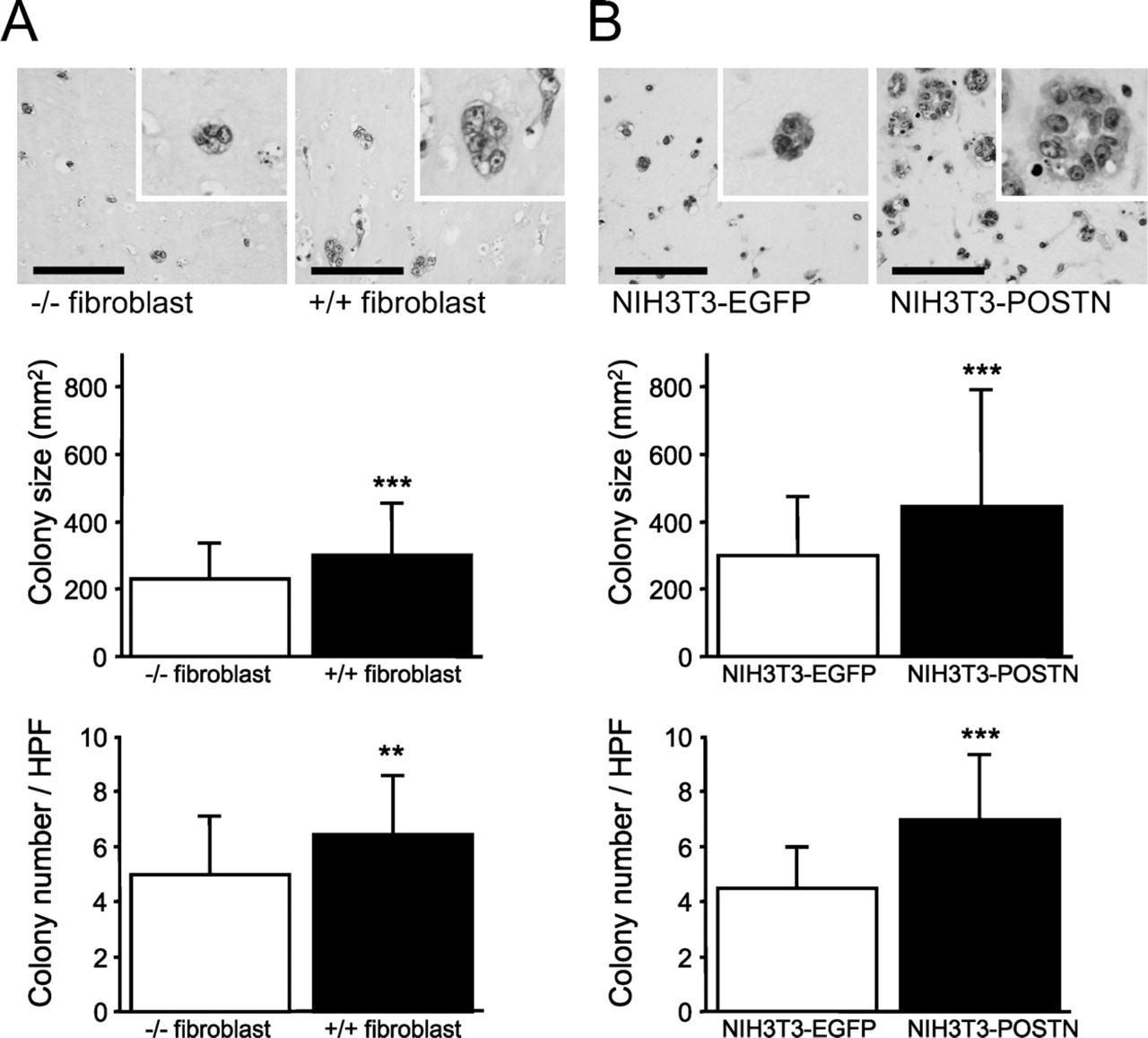
Colony formation assay of HCT116 co-embedded with fibroblasts in type I collagen gel. (
Periostin deposition showed a pericryptal pattern in normal colonic mucosa. We showed by immuno-electron microscopy that a specific type of myofibroblasts, PCFs, produces and secretes periostin within the myofibroblastic sheath of the cryptic glands. PCFs in the colon play a fundamental role in the differentiation and in the absorptive function of the crypts (Birchmeier and Birchmeier 1993; Kedinger et al. 1999; Naftalin and Pedley 1999; Powell et al. 1999), interacting with epithelial cells through a paracrine mechanism by transforming growth factor β (TGFβ), interleukin 6 (IL-6), and leukemia inhibitory factor (Powell et al. 1999; Rockman et al. 2001). We showed the difference of Ki67-labeling indexes in the colonic crypts of periostin−/- and periostin+/+ mice, suggesting that periostin also plays a role in regulating epithelial proliferation in normal colon. Furthermore, periostin in the lamina propria in the inflammatory process showing a mesh pattern may play a role in maintaining the integrity of the colonic mucosa. We have previously shown that periostin is fundamental for the tissue repair process of the myocardial infarction (Shimazaki et al. 2008).
Colony size and number of HCT116 cells in three-dimensional co-cultures with fibroblasts
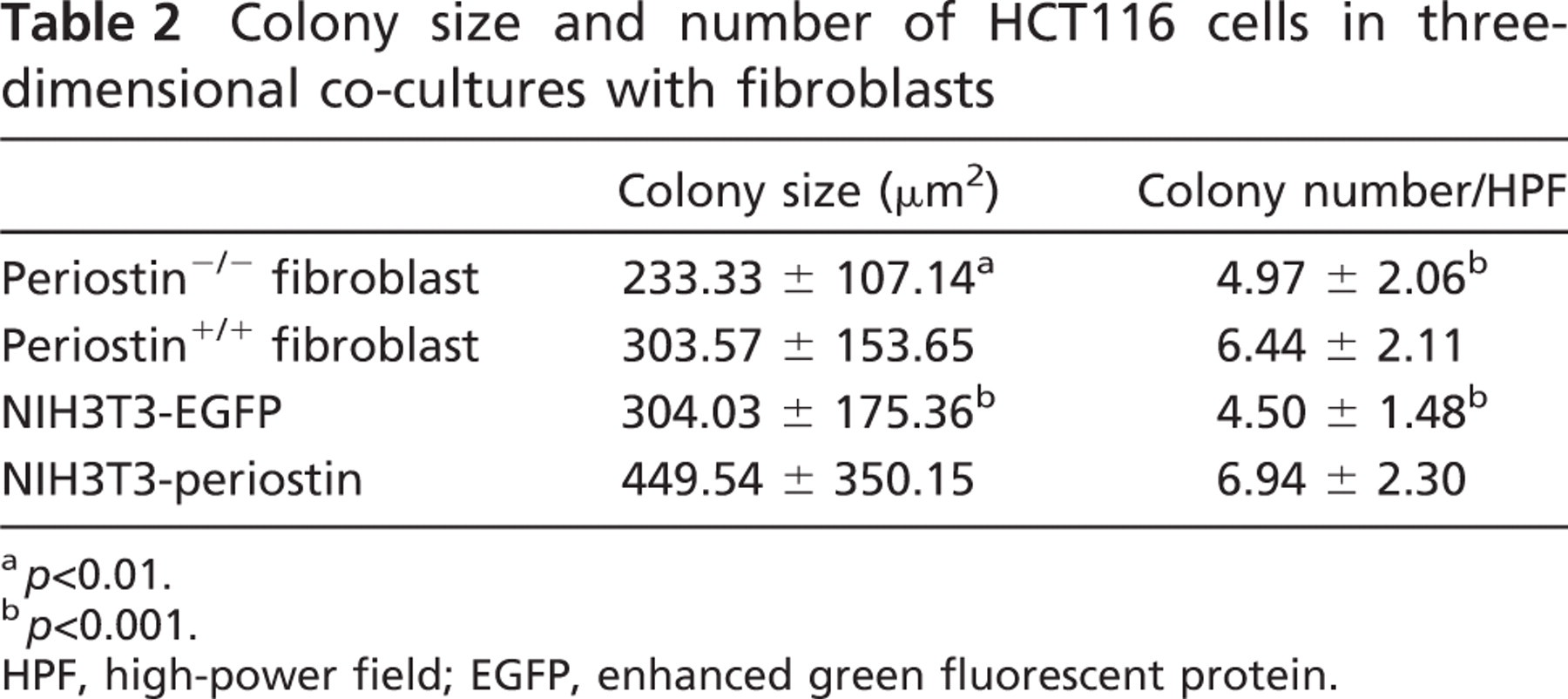
a p<0.01.
b p<0.001.
HPF, high-power field; EGFP, enhanced green fluorescent protein.
Periostin deposition decreased before the number of PCFs became lower in the adenomas and completely disappeared at the stage of intramucosal carcinoma. Periostin is induced by TGFβ, bone morphologic protein-2 (BMP-2), IL-4, or IL-13 in mesenchymal cells (Horiuchi et al. 1999; Ji et al. 2000; Takayama et al. 2006), and it was previously reported that the level of TGFβ and BMP-2 was lower or lost in adenoma-bearing mucosa (Kushiyama et al. 2000), in microadenomas of a mouse model of familial adenomatous polyposis (FAP) (Oshima et al. 1997), and in adenomas of patients with FAP (Hardwick et al. 2004). Thus, it is possible that periostin secretion decreases from PCFs, before PCFs decrease in the adenoma. The finding of periostin decrease heralds the neoplastic growth of colonic epithelial cells and may be useful for pathological diagnosis of the neoplastic change of the epithelial cells of the colon.
There has been controversy regarding the localization of periostin in neoplastic tissues. In this study, the primary cells to produce and secrete periostin are the fibroblastic cells in the interstitium of the carcinoma, i.e., CAFs, which have characteristics of myofibroblasts (Orimo et al. 2005). As for the results of ISH, Baril et al. (2007) showed mRNA of periostin in cancer cells in pancreatic cancer. In contrast, Sasaki et al. (2001, 2003) observed weak signals in the carcinoma cells of lung cancers (Sasaki et al. 2001) and breast cancers (Sasaki et al. 2003), and we recently observed that the signals were non-existent in pancreatic cancer (Fukushima et al. in press). According to Erkan et al. (2007), it is the stellate cells that produced periostin in pancreatic cancer. We have no definite explanation for these differences at present, but we frequently observed that endogenous alkaline phosphatase activity in the pancreatic acinar cells disturbs the tissue localization when the alkaline phosphatase method is used for visualization of the signals in the pancreas. Nevertheless, because periostin-transfected carcinoma cells facilitates its motility and metastasis, the induction of periostin in the CAFs was suggested to serve in the growth and development of the carcinomas in a paracrine loop. In this study, recombinant periostin accelerated the growth of HCT116. Both colony size and number of HCT116 cells were significantly larger when cultured with periostin-producing fibroblasts than with non-producing ones in a three-dimensional co-culture system. The fibroblasts used in the study were not derived from the colon, and therefore, the experimental condition was not a true recapitulation of the colon carcinoma. However, these facts strongly suggest that the growth of cancer cells is promoted by periostin through the paracrine route from the coexisting fibroblasts.
Pattern of periostin distribution in the normal and neoplastic colon

++, Strong expression; +, expression; +/−, weak expression; −, no expression.
In conclusion, periostin is a matrix protein secreted by both PCFs and CAFs. PCF–periostin and CAF–periostin may have a similar function, although their regulation is different; the former is regulated by the integrity of the concrete structure of pericryptal sheath, and the latter is unregulated at the invasive site of the stroma of the carcinoma.
Footnotes
Acknowledgements
This work was supported by Grants-in-Aid from Ministry of Education, Culture, Sports, Science and Technology or Japan Society for the Promotion of Science and in part by grants from The Yasuda Medical Foundation to Y.K. No conflicts of interest were declared.
We thank Dr. H. Niwa (RIKEN Center for Developmental Biology, Kobe, Japan) for kind provision of pCAGIPuro vector and M. Fujiwara, K. Sakuma, H. Yamamura, and A. Ogiwara (Department of Pathology and Diagnostic Pathology, Graduate School of Medicine, The University of Tokyo) for technical support.
