Abstract
Strategies to promote bone repair have included exposure of cells to growth factor (GF) preparations from blood that generally include proteins as part of a complex mixture. This study aimed to evaluate the effects of such a mixture on different parameters of the development of the osteogenic phenotype in vitro. Osteoblastic cells were obtained by enzymatic digestion of human alveolar bone and cultured under standard osteogenic conditions until subconfluence. They were subcultured on Thermanox coverslips up to 14 days. Treated cultures were exposed during the first 7 days to osteogenic medium supplemented with a GFs + proteins mixture containing the major components found in platelet extracts [platelet-derived growth factor-BB, transforming growth factor (TGF)-β1, TGF-β2, albumin, fibronectin, and thrombospondin] and to osteogenic medium alone thereafter. Control cultures were exposed only to the osteogenic medium. Treated cultures exhibited a significantly higher number of adherent cells from day 4 onward and of cycling cells at days 1 and 4, weak alkaline phosphatase (ALP) labeling, and significantly decreased levels of ALP activity and mRNA expression. At day 14, no Alizarin red–stained nodular areas were detected in cultures treated with GFs + proteins. Results were confirmed in the rat calvaria–derived osteogenic cell culture model. The addition of bone morphogenetic protein 7 or growth and differentiation factor 5 to treated cultures upregulated Runx2 and ALP mRNA expression, but surprisingly, ALP activity was not restored. These results showed that a mixture of GFs + proteins affects the development of the osteogenic phenotype both in human and rat cultures, leading to an increase in the number of cells, but expressed a less differentiated state.
T
Because of the known effects of individual GFs on cell activities of different cell types, such proteins/peptides have been used in a series of strategies to promote tissue healing, usually in conjunction with delivery systems to control the speed, time, and quantity of release (reviewed in Lieberman et al. 2002; Luginbuehl et al. 2004; Rose et al. 2004; Anitua et al. 2007a; Lee and Shin 2007). In bone tissue, therapies with GFs have been applied to promote, and in some cases to induce, desirable amounts of new bone formation in bone defects, in sites of fracture healing, and adjacent to metal implant devices (Bessho et al. 1999; Ramoshebi et al. 2002; Schliephake 2002; Kloen et al. 2003). Considering that in the body, cells are exposed to diverse GFs simultaneously, ideally any therapy should involve a mixture of GFs with similarities to those implicated in bone tissue healing (Cheifetz et al. 1996; Roethy et al. 2001; Raiche and Puleo 2004; Arosarena and Puleo 2007). Indeed, a well-known therapy with GFs for bone defects is the application of autologous platelet-rich plasma (PRP) (Marx et al. 1998; Dugrillon et al. 2002; reviewed in Anitua et al. 2007b), a concentrate of platelets in plasma. These platelets liberate their content whose major components are platelet-derived growth factor (PDGF) and transforming growth factor β (TGF-β) (Gruber et al. 2003; Lucarelli et al. 2003; Graziani et al. 2006; van den Dolder et al. 2006). Although beneficial clinical results have been reported, some studies have shown that the use of PRP does not promote new bone formation (Ferreira et al. 2005; Hokugo et al. 2005; Gerard et al. 2006; Graziani et al., 2006; Sarkar et al. 2006; Hokugo et al. 2007; Mooren et al. 2007; Ranly et al. 2007; Roussy et al. 2007). These divergent outcomes have been attributed to intra- and interspecies variations in the relative proportions of PRP components (Lacoste et al. 2003; van den Dolder et al. 2006).
The objective of our study was to determine the effects of a PRP-like mixture of GFs and proteins (hereafter referred to as GFs + proteins) on human alveolar bone osteogenic cell cultures. To avoid the inherent variations in PRP preparations and results, we opted to experiment with a well-defined mixture formulated to contain the major components found in PRP extracts. Applications of these standardized mixtures show that they have inhibitory effects on in vitro osteogenic events, ultimately resulting in a complete lack of bone-like nodule formation.
Materials and Methods
Culture of Osteogenic Cells Derived From Human Alveolar Bone
Human alveolar bone fragments were obtained from adult healthy donors, using the research protocols approved by the Committee of Ethics in Research of the School of Dentistry of Ribeirão Preto of the University of São Paulo. Osteogenic cells were isolated by enzymatic digestion of the explants using type II collagenase (Gibco–Life Technologies; Grand Island, NY) as previously described (Mailhot and Borke 1998; Beloti et al. 2006). They were cultured in osteogenic medium, which was composed of Gibco α-MEM (Invitrogen; Carlsbad, CA), supplemented with 10% FBS (Gibco), 50 μg/ml gentamicin (Gibco), 0.3 μg/ml Fungizone (Gibco), 10-7 M dexamethasone (Sigma; St. Louis, MO), 5 μg/ml ascorbic acid (Gibco), and 7 mM β-glycerophosphate (Sigma). Subconfluent cells in primary cultures were harvested after treatment with 1 mM EDTA (Gibco) and 0.25% trypsin (Gibco) and subcultured on Thermanox coverslips (Nunc; Rochester, NY) in 24-well polystyrene plates (Falcon; Franklin Lakes, NJ) at a density of 2 × 104 cells/well. Cells were grown at 37C in a humidified atmosphere of 5% CO2 and 95% air, and the medium was changed every 3 or 4 days.
Exposure of Osteogenic Cultures to a Mixture of GFs and Proteins
The mixture of GFs and proteins tested was diluted in osteogenic medium and contained 27 ng/ml recombinant human PDGF-BB, 22 ng/ml recombinant human TGF-β1, 15 ng/ml recombinant human TGF-β2, 3.7 μg/ml serum-derived human albumin, 2 μg/ml plasma-derived human fibronectin, and 0.5 μg/ml platelet-derived thrombospondin, all purchased from Sigma. The GFs and proteins of the mixture were selected from major components found in porcine (Venne et al. 1999) and human (Lacoste et al. 2003; Martineau et al. 2004) platelet preparations. Treated cultures were exposed during the first 7 days to the GFs + proteins mixture, which was added at time 0 and at day 3 and to osteogenic medium alone thereafter. Control cultures were only exposed to the osteogenic medium. To evaluate the influence of known osteogenic factors on cells exposed to GFs + proteins, 100 ng/ml recombinant human bone morphogenetic protein 7 (BMP-7; Sigma) or 20 ng/ml recombinant human growth and differentiation factor 5 [GDF-5 or cartilage-derived morphogenetic protein-1 (CDMP-1); Pepro Tech, Rocky Hill, NJ) as added to GFs + proteins–treated cultures at time 0 and day 3 or at days 7 and 10. Cultures exposed to BMP-7 and GDF-5 were evaluated at day 14 in terms of runt-related transcription factor 2 (Runx2) and alkaline phosphatase (ALP) mRNA expression and ALP activity.
Fluorescence Labeling
At 1, 4, 7, and 14 days, cells were fixed for 10 min at room temperature with 4% paraformaldehyde in 0.1 M phosphate buffer (PB), pH 7.2. After washing in PB, they were processed for indirect immunofluorescence labeling (de Oliveira et al. 2007) for detection of ALP (Eghbali-Fatourechi et al. 2005) and the nuclear antigen Ki-67 (Robertson et al. 2005). In addition, cell morphology was evaluated by direct fluorescence with fluorophore-conjugated probes. Briefly, cells were permeabilized with 0.5% Triton X-100 in PB for 10 min, followed by blocking with 5% skimmed milk in PB for 30 min. Primary antibodies to human bone ALP (monoclonal B4-78, 1:100; Developmental Studies Hybridoma Bank, Iowa City, IA) and human Ki-67 (polyclonal, 1:70; Diagnostic Biosystems, Pleasanton, CA) were used, followed by a mixture of Alexa Fluor 594 (red fluorescence)–conjugated goat anti-mouse or goat anti-rabbit secondary antibody (1:200; Molecular Probes, Eugene, OR) and Alexa Fluor 488 (green fluorescence)–conjugated phalloidin (1:200; Molecular Probes), which labels the actin cytoskeleton. Replacement of the primary antibodies with PB was used as control. All antibody incubations were performed in a humidified environment for 60 min at room temperature. Between each incubation step, the samples were washed three times (5 min each) in PB. Before mounting for microscope observation, cell nuclei were stained with 300 nM 4′, 6-diamidino-2-phenylindole, dihydrochloride (DAPI; Molecular Probes) for 5 min and samples were briefly washed with dH2O. Thermanox coverslips were placed face up on glass slides and mounted with a Fisherbrand 12-mm-round glass coverslip (Fisher Scientific; Suwanee, GA) using an antifade mounting medium (Vectashield; Vector Laboratories, Burlingame, CA). The samples were examined under epifluorescence using a Leica DMLB light microscope, with N Plan (×10/0.25, ×20/0.40) and HCX PL Fluotar (×40/0.75) objectives, outfitted with a Leica DC 300F digital camera (Leica; Bensheim, Germany). The acquired digital images were processed with Adobe Photoshop software (version 7.0; Adobe Systems, San Jose, CA).
Growth Curve and Cell Viability
Cells grown for periods of 1, 4, 7, and 10 days were enzymatically detached from Thermanox coverslips using 1 mM EDTA, 1.3 mg/ml collagenase, and 0.25% trypsin solution (Gibco; Invitrogen). Total number of cells/well and percentage of viable and non-viable cells were determined after Trypan blue (Sigma) staining using a hemacytometer.
Proportion of Cycling Cells
The proportion of cycling cells at days 1 and 4 was determined by means of double Ki-67/DAPI labeling. The total number of nuclei (DAPI stained) and of cycling cells (Ki-67 positive; Scholzen and Gerdes 2000) on control and treated cultures was calculated by light microscope counting under epifluorescence at ×40 objective. A minimum of 200 total cells were counted on three Thermanox coverslips at each time point.
Total Protein Content
Total protein content was calculated at days 4, 7, 10, and 14, according to the method described by Lowry et al. (1951). The wells were filled with 2 ml of deionized water. After five cycles of thermal-shock (alternating temperature between 15 min at 37C and 20 min at −20C), 1 ml of the sample from each well was mixed with 1 ml of Lowry solution (Sigma) and left for 20 min at room temperature. Subsequently, 0.5 ml of phenol reagent of Folin and Ciocalteau (Sigma) was added. This was left for 30 min at room temperature, after which absorbance was measured using a spectrophotometer (CE3021; Cecil, Cambridge, UK) at 680 nm. Total protein content was calculated from a standard curve using BSA (Sigma), giving a range of 25–400 μg protein/ml, and data were expressed as micrograms of protein per milliliter.
ALP Activity
For cultures at days 4, 7, 10, and 14, aliquots of the same wells used for calculating total protein content were assayed to measure ALP activity. p-Nitrophenyl-phosphatase (PNPPase) activity was assayed discontinuously at 37C in a spectrophotometer (Spectronic Genesys 20; Thermo Electron, Madison, WI) by following the liberation of p-nitrophenolate ion (∊ = 1 M, pH 13 = 17,600 M-1 cm-1) at 410 nm. Standard conditions were 50 mM AMPOL buffer, pH 10.0, containing 2 mM MgCl2 and 10 mM PNPP in a final volume of 1.0 ml as previously described (Simão et al. 2007). The reaction was stopped by adding 1 ml of 1M NaOH. All determinations were carried out in duplicate, and the initial velocities were constant for at least 90 min provided that <5% of substrate was hydrolyzed. Controls without added enzyme were included in each experiment to allow the determination of non-enzymatic hydrolysis of substrate. Data were expressed as nmoles p-nitrophenyl per microgram protein per minute. Cultures were also stained with Fast red for in situ histochemical detection of ALP activity (Majors et al. 1997) at days 10 and 14.
RNA Extraction and Quantitative Real-time RT-PCR
The primer sequences, the predicted amplicon sizes, and the annealing and melting temperatures, which were designed using the PrimerExpress software (Applied Biosystems; Foster City, CA), are depicted in Table 1.
Total RNA from cultures at day 14 was extracted using the Promega RNA extraction kit (Promega; Madison, WI), according to the manufacturer's instructions. The concentration of RNA was determined by optical density at a wavelength of 260 nm, using the GeneQuant (Amersham Biosciences; Piscataway, NJ). cDNA was synthesized using 2 μg of RNA through a reverse transcription reaction (M-MLV reverse transcriptase; Promega). Real-time PCR quantitative mRNA analyses were performed in an ABI Prism 7000 Sequence Detection System using the SybrGreen system (Applied Biosystems). SybrGreen PCR Master-Mix (Applied Biosystems), specific primers, and 2.5 ng of cDNA were used in each reaction. The standard PCR conditions were 95C (10 min) and 40 cycles of 94C (1 min), 56C (1 min), and 72C (2 min), followed by the standard denaturation curve. For mRNA analyses, the relative level of gene expression was calculated in reference to β-actin expression and the control group using the cycle threshold (Ct) method (Livak and Schmittgen 2001).
Primers sequences and reaction properties
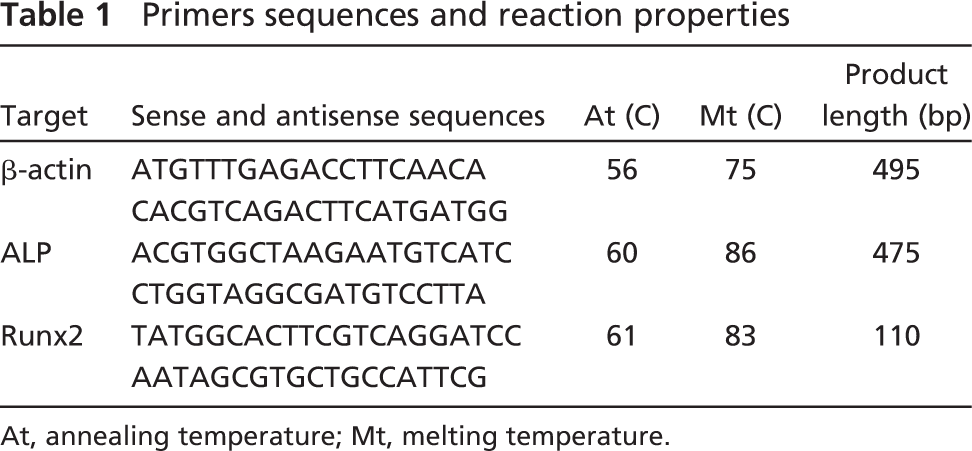
At, annealing temperature; Mt, melting temperature.
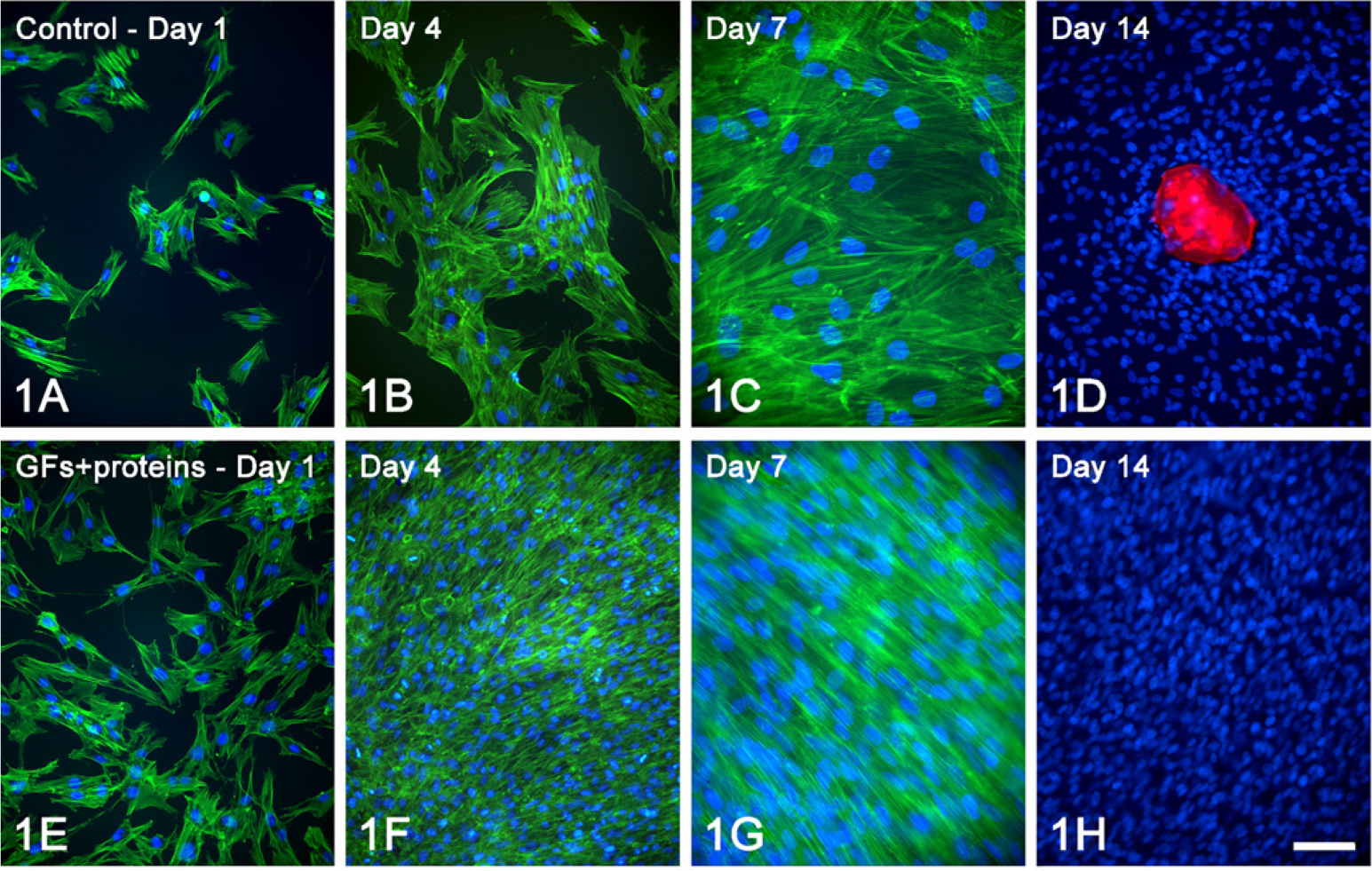
Epifluorescence to detect actin cytoskeleton (green fluorescence) and cell nuclei (blue fluorescence) of control (
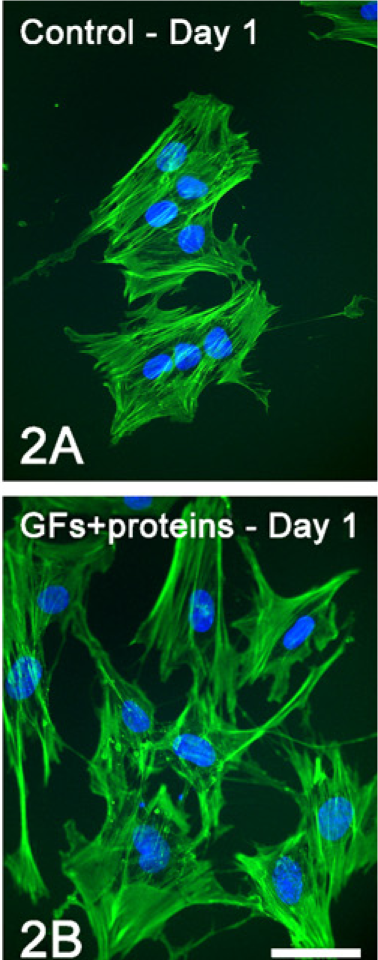
Epifluorescence to detect actin cytoskeleton (green fluorescence) and cell nuclei (blue fluorescence) at day 1 of control (
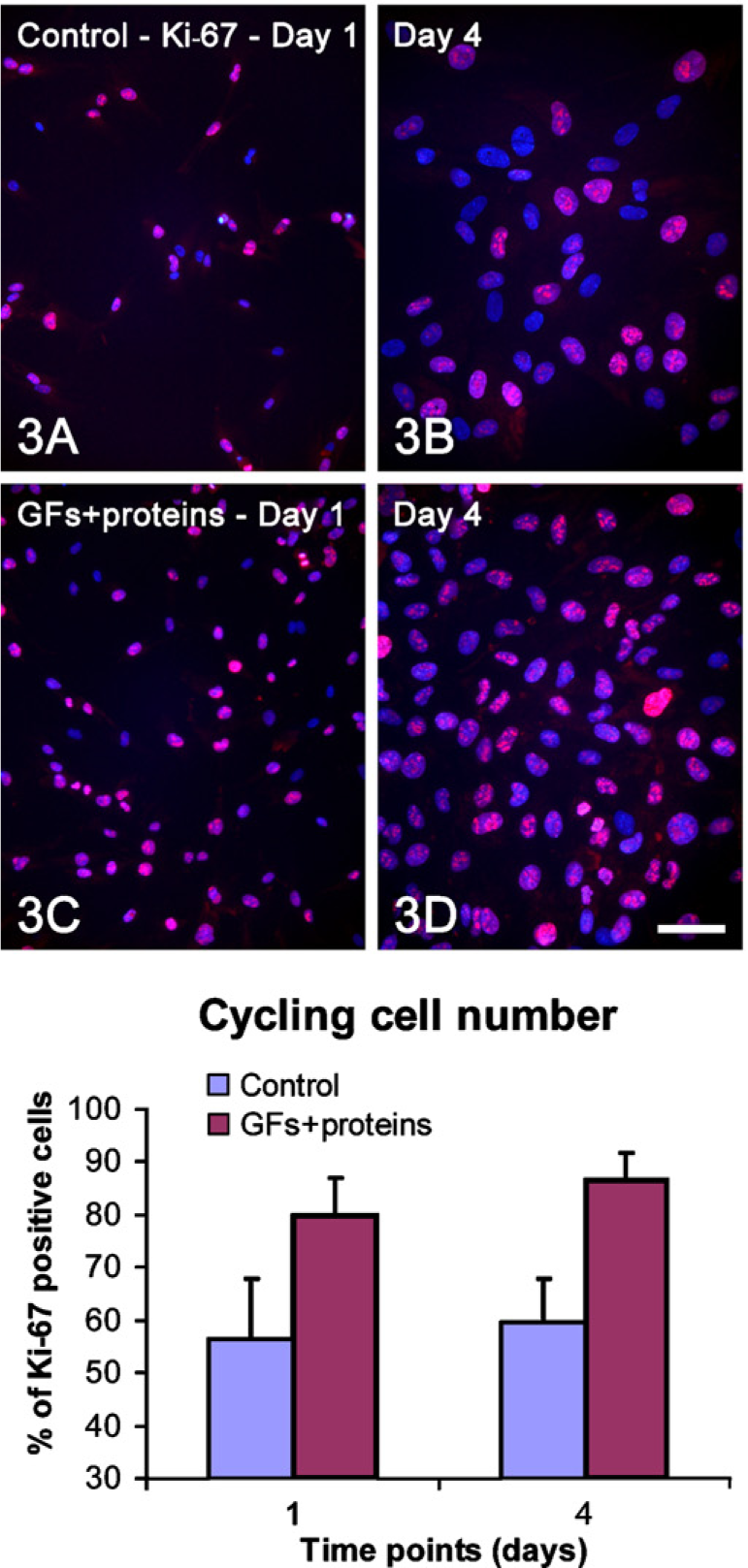
Double labeling Ki-67/4′, 6-diamidino-2-phenylindole, dihydrochloride (DAPI) (red and blue fluorescence, respectively) to determine the proportions of cycling cells at days 1 (
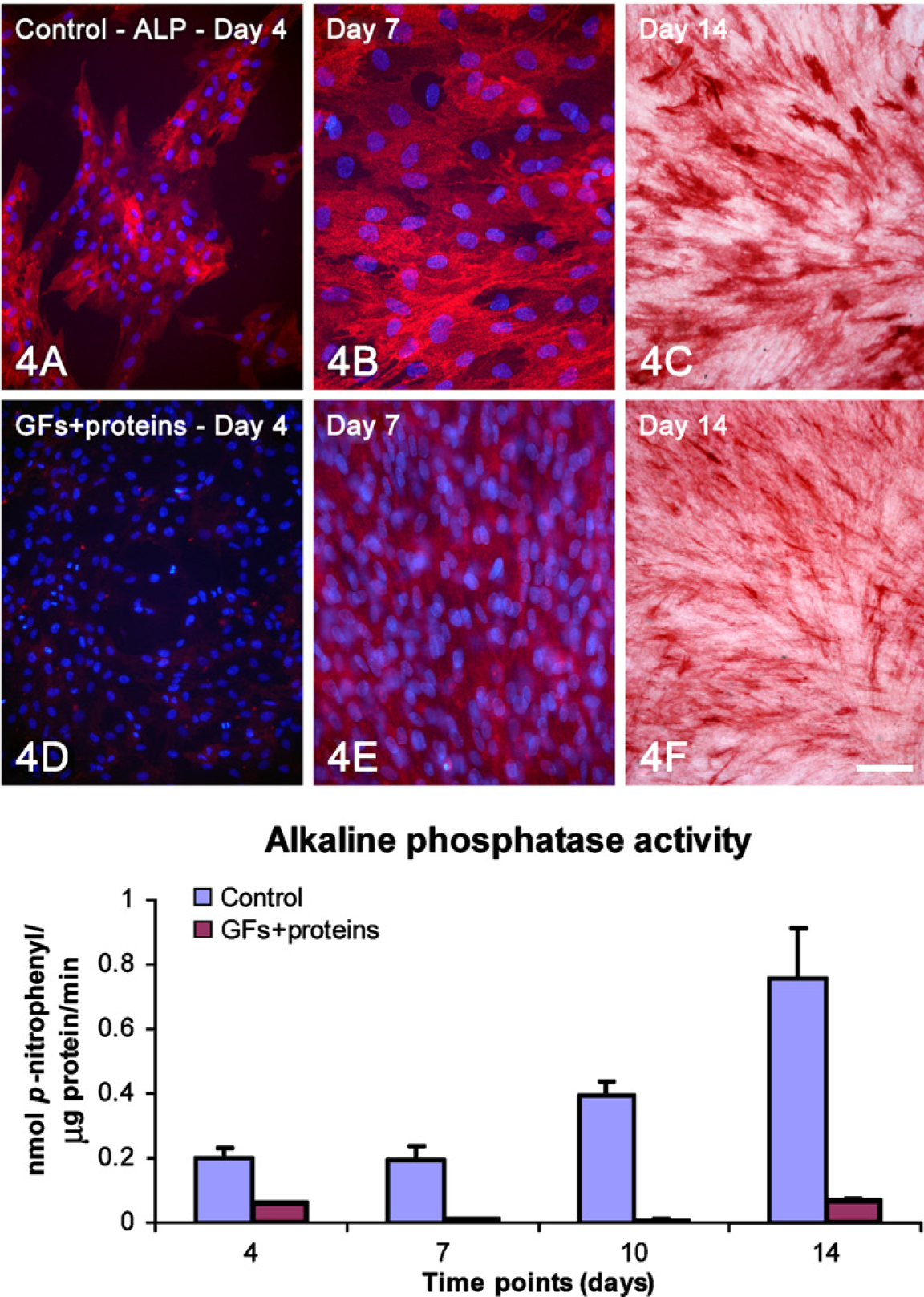
Double labeling alkaline phosphatase (ALP; red fluorescence)/DAPI (
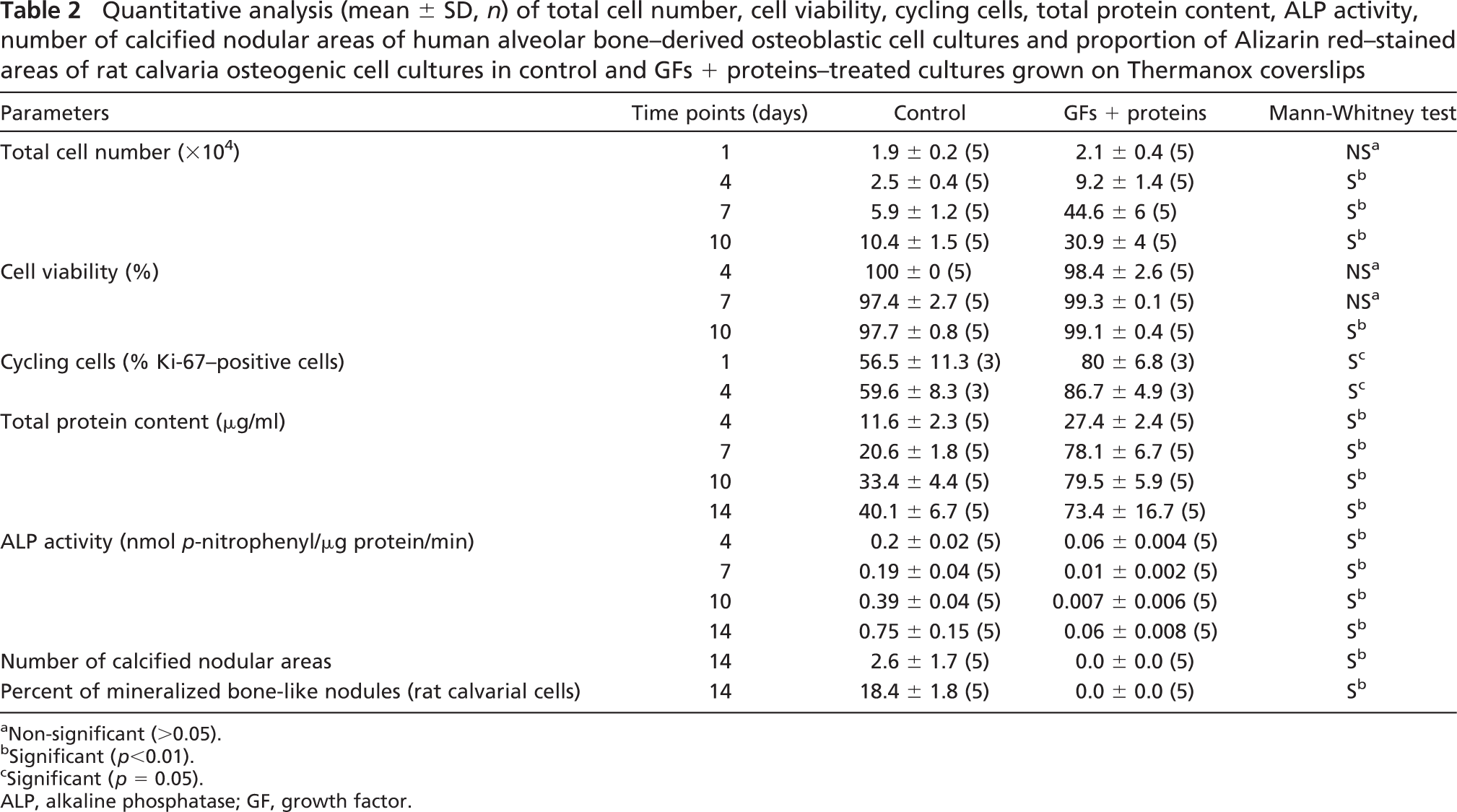
Quantitative analysis (mean ± SD, n) of total cell number, cell viability, cycling cells, total protein content, ALP activity, number of calcified nodular areas of human alveolar bone–derived osteoblastic cell cultures and proportion of Alizarin red–stained areas of rat calvaria osteogenic cell cultures in control and GFs + proteins–treated cultures grown on Thermanox coverslips
a Non-significant (>0.05).
b Significant (p<0.01).
c Significant (p = 0.05).
ALP, alkaline phosphatase; GF, growth factor.
Mineralized Bone-like Nodule Formation
At day 14, cultures were washed in Hanks' balanced salt solution (Sigma) and fixed in 70% ethanol for 60 min at 4C. The samples were washed in PBS and dH2O and stained with 2% Alizarin red (Sigma), pH 4.2, for 15 min at room temperature, followed by permeabilization with 0.5% Triton X-100 and DAPI staining for epifluorescence microscopic detection. To validate the results obtained with human alveolar osteogenic cells, the effect of the GFs + proteins mixture was also tested on the rat calvaria osteogenic cell culture system (Nanci et al. 1996; de Oliveira et al. 2007). Cells were grown under the same experimental conditions, and mineralized bone-like nodule formation was evaluated at day 14. Alizarin red–stained calvarial cultures were photographed with a high-resolution digital camera (Canon EOS Digital Rebel Camera, 6.3 Megapixel CMOS sensor, with a Canon EF 100 mm f/2.8 macro lens; Canon, Lake Success, NY), and the percentage of the substrate area occupied by Alizarin red–stained nodules was determined using Image Tool software (University of Texas Health Science Center; San Antonio, TX).
Statistical Analysis
Data are presented as mean ± SD. The χ2 test was used to determine normality of data sets. Comparisons were carried out using the non-parametrical Mann-Whitney test for two independent samples or Kruskal-Wallis test for multiple comparisons, followed by the Fischer test based on rank (level of significance: 5%), where appropriate. The results described below are representative of a minimum of two sets of human and rat osteogenic cell cultures. The real-time PCR analysis was carried out in triplicate.
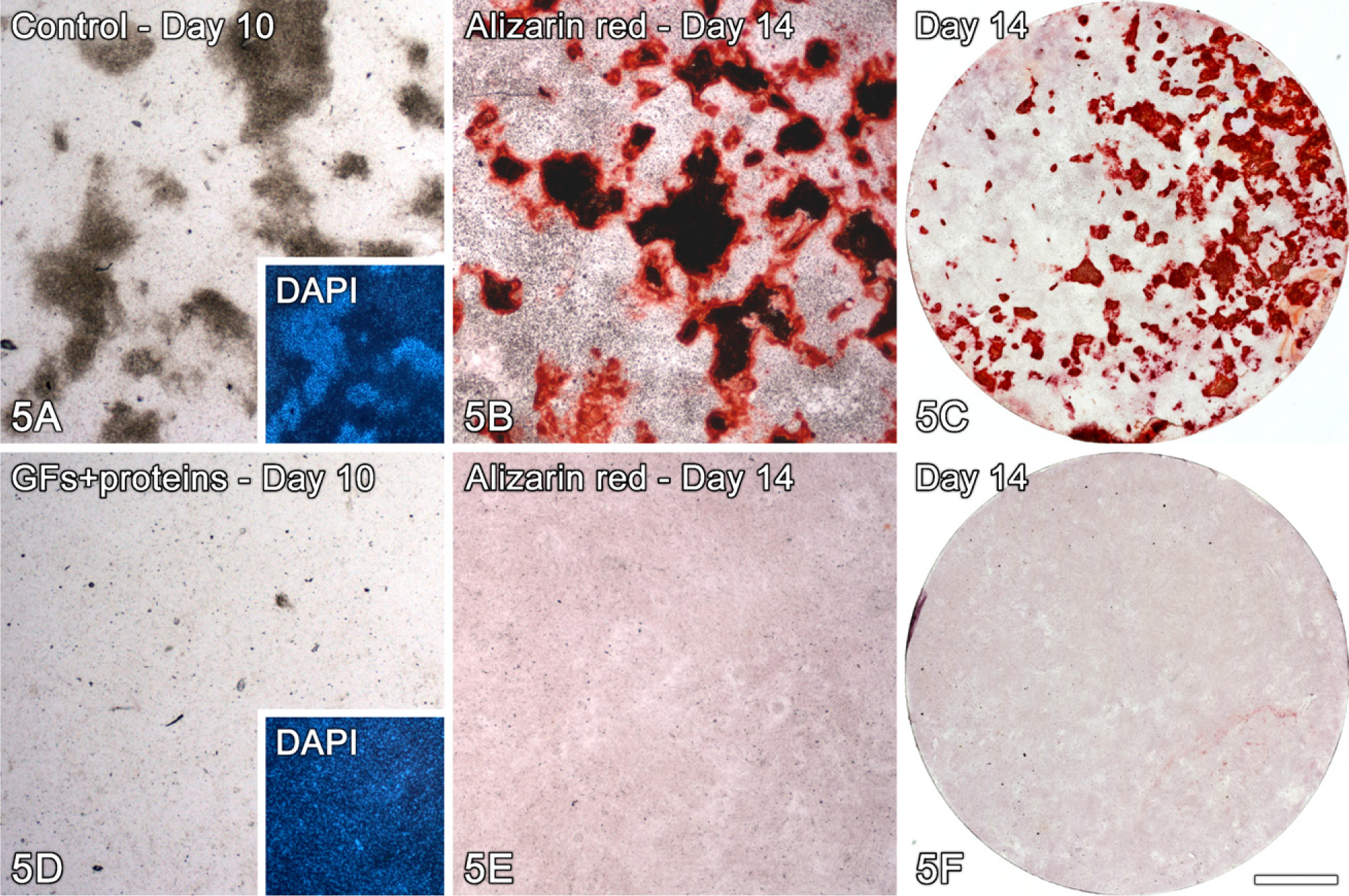
Bone-like nodule formation in control (
Results
Epifluorescence showed marked differences in cell morphology and tissue architecture during the progression of the cultures (Figure 1). At day 1, although both control and treated cells were polygonal in shape, cytoplasmic extensions were observed in a higher proportion of treated cells (Figures 1A, 1E, and 2). At day 4, whereas treated cultures were confluent with areas of cell multilayering and abundant mitotic figures, control ones formed discrete cell aggregates (Figures 1B and 1F). At day 7, control cells formed a confluent layer and were oriented randomly, whereas in the GFs + proteins–treated group, cells generally oriented in the same direction (Figures 1C and 1G). Noticeably, no areas of calcified matrix were detected in treated cultures at day 14, whereas control cultures exhibited occasional Alizarin red–stained areas (Figures 1D and 1H).
Growth analyses indicated that there were significantly more cells in treated cultures from day 4 onward (Table 2). Significantly higher values in cell viability were detected for treated cultures only at day 10 (Table 2). The proportion of cycling cells was significantly higher for treated cultures both at days 1 and 4 (Figure 3; Table 2) compared with the control. Indeed, an increase of at least 40% in the population of Ki-67–positive cells was detected during the first 7 days of treatment with GFs + proteins.
Total protein content was significantly higher in treated cultures at all time points (Table 2) and the amount of ALP, as judged by activity measurements, Fast red staining and immunolabeling of the protein was reduced (Figure 4; Table 2).
Both control human and rat cell cultures exhibited an osteogenic phenotype as determined by qualitative and quantitative analysis of mineralized nodules (Figures 1D and 5A–5C, respectively; Table 2). Conversely, treated cultures showed no bone-like nodule formation at days 10 (Figure 5D) and 14 (Figures 1H, 5E, and 5F; Table 2).
Expression of Runx2 mRNA was not affected by the exposure of cultures to the mixture of GFs + proteins (Figure 6A; Table 3). However, exposure of treated cultures to BMP-7 or GDF-5 generally resulted in a significant increase in Runx2 mRNA above levels found in untreated and GFs + proteins cultures (Figure 6A; Table 3). Strikingly, treatment with GFs + proteins resulted in the downregulation of ALP mRNA expression (Figure 6B; Table 3). Such inhibitory effect was reverted by the exposure of treated cultures to BMP-7 (0–7 days), GDF-5 (0–7 days), or GDF-5 (7–14 days) (Figure 6B; Table 3). However, ALP activity remained at significantly reduced levels for all groups compared with the control [control, 0.6 ± 0.14; GFs + proteins, 0.03 ± 0.03; BMP-7 (0–7 days), 0.009 ± 0.01; GDF-5 (0–7 days), 0.01 ± 0.006; BMP-7 (7–14 days), 0.05 ± 0.05; GDF-5 (7–14 days), 0.02 ± 0.01; Table 3).
Discussion
The results of this study showed that a mixture of GFs + proteins containing the major components found in platelet extracts promotes a significant increase in cycling cells and cell number but dramatically reduces the osteogenic potential of both human alveolar bone and rat calvaria–derived cell cultures. There was a reduction in ALP activity and mRNA expression and inhibition of bone-like nodule formation. In addition, we showed that, whereas the inhibitory effects on ALP mRNA expression can be reverted by exposure of cells to BMP-7 or GDF-5, ALP activity is not restored.
During the processes of bone formation and repair, a complex sequence of signaling events takes place, which includes interaction of a series of GFs and other matrix proteins with receptors in various target cells at different stages of differentiation. The ensuing cell signaling events will promote the development of the osteoblastic phenotype, which ultimately results in bone tissue formation and remodeling. Because bone repair is a multifactorial process, molecular therapies for the local delivery of mixtures of GFs have been devised, such as found in PRP extracts (Marx et al. 1998; Dugrillon et al. 2002; reviewed in Anitua et al. 2007b). These, however, have been reported to show significant intra- and interspecies variations in relative proportions of components, to which differences in the quality and quantity of new tissue formation and in clinical outcomes have been attributed (van den Dolder et al. 2006). To avoid this variability, we prepared from recombinant human GFs and proteins a well-defined mixture that contained the major components of PRP. Addition of our standardized mixture of GFs + proteins allowed obtaining consistent results throughout the various sets of experiments. Interestingly, despite interspecies variations, inhibition of bone-like nodule formation occurred in both human alveolar bone and rat calvaria osteogenic cell cultures. Although FBS in the culture medium contains GFs and proteins, the results obtained clearly correlate with the addition of our PRP-like mixture.
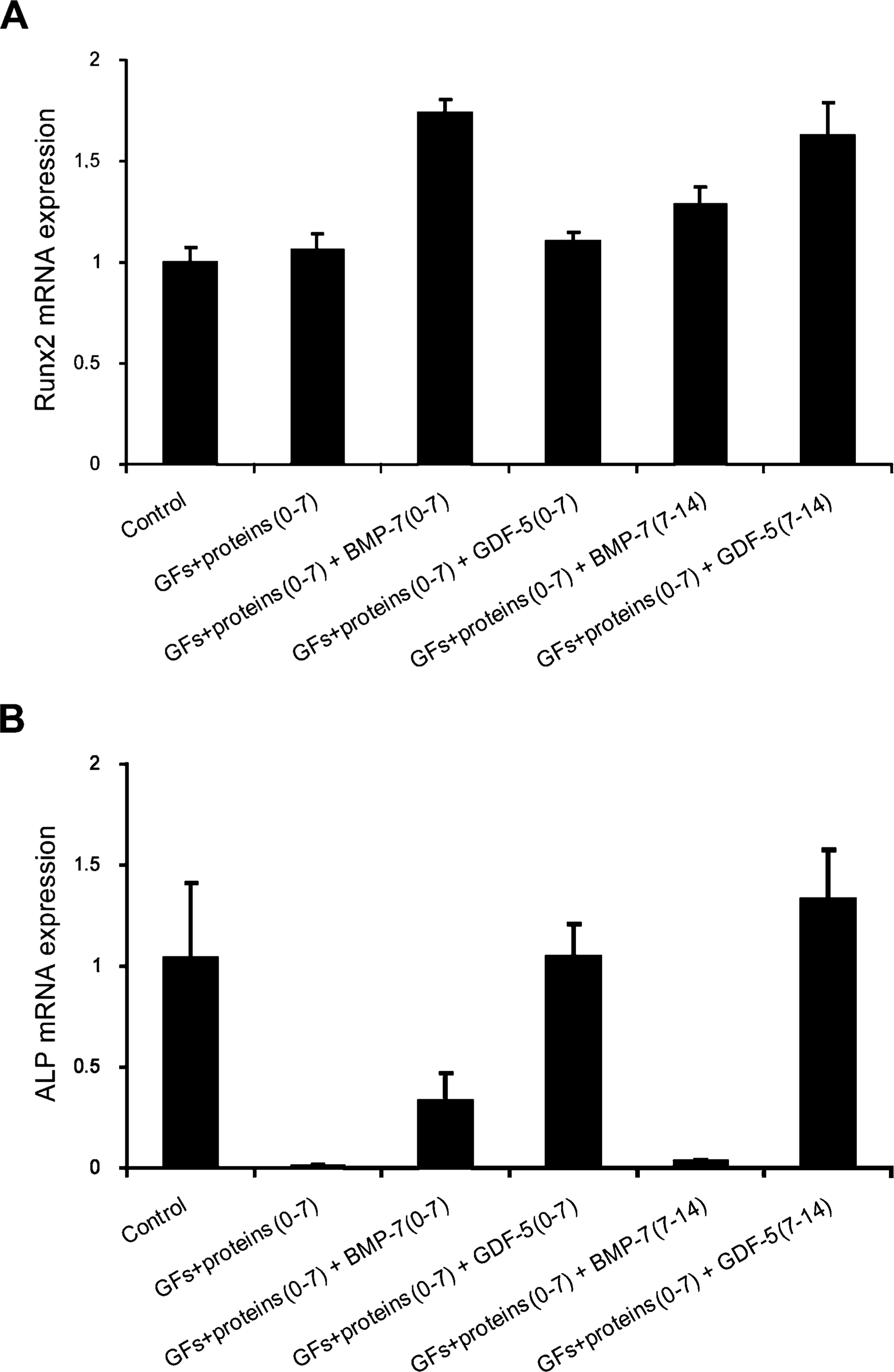
Effects of the exposure of GFs + proteins–treated cultures to bone morphogenetic protein (BMP)-7 or growth and differentiation factor (GDF)-5 (0–7 or 7–14 days) on Runx2 (
Comparative analysis (Fischer posttest) of the effect of BMP-7 or GDF-5 on the expression of Runx2 and ALP mRNA (Kruskal-Wallis test, p<0.01) and ALP activity (Kruskal-Wallis test, p<0.05) in GFs + proteins–treated human alveolar bone–derived osteoblastic cell cultures at day 14 on Thermanox coverslips
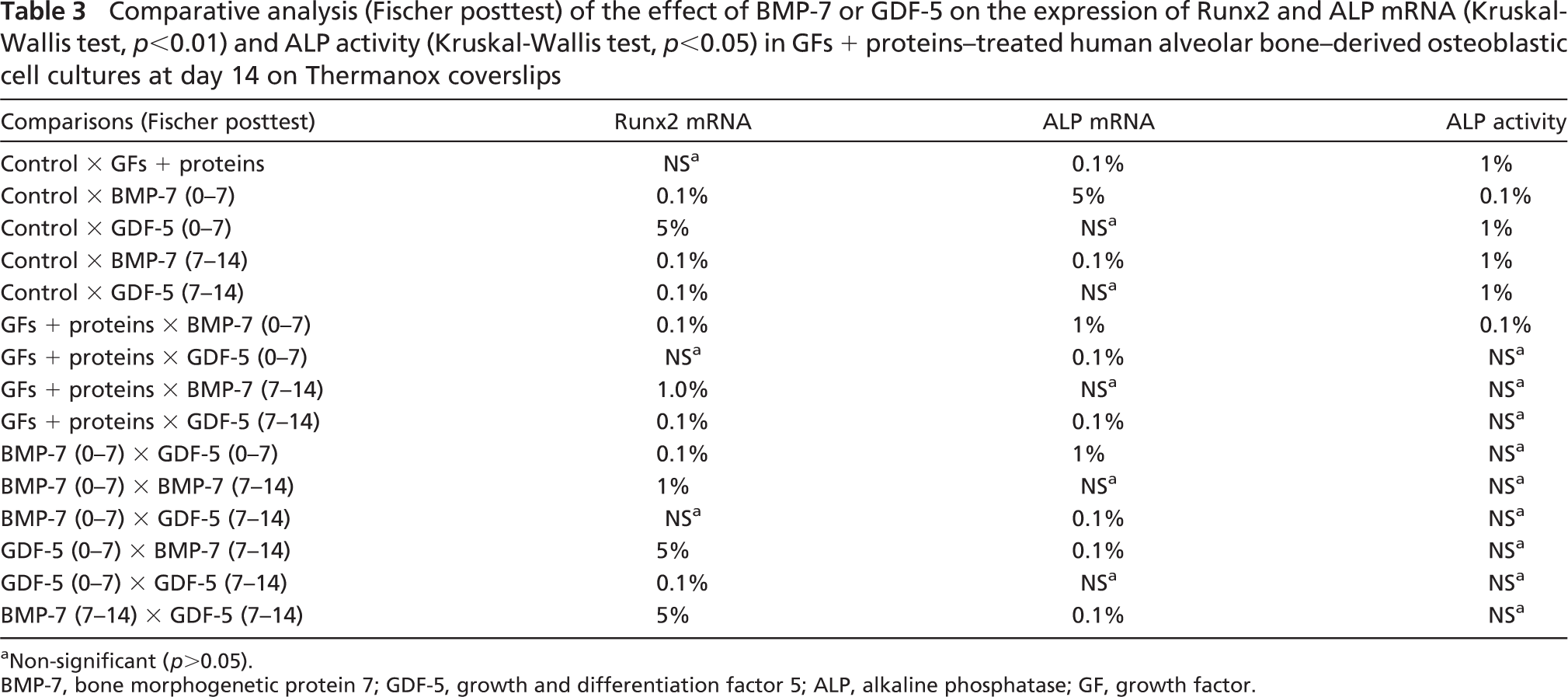
a Non-significant (p>0.05).
BMP-7, bone morphogenetic protein 7; GDF-5, growth and differentiation factor 5; ALP, alkaline phosphatase; GF, growth factor.
Inhibition of ALP activity and mRNA expression by GFs + proteins and no changes in Runx2 mRNA levels compared with control indicate that the expanding cell population was made up of cells with a less differentiated osteoblastic phenotype and thereby decreased capacity to lay down an osteogenic matrix and/or nucleate biological apatite. The lack of calvarial cell multilayering typically observed during development of bone-like nodules lends additional support to the interpretation that the treated osteogenic cells did not achieve a fully differentiated state. It has been proposed that the role of tissue nonspecific ALP (TNAP) in the bone matrix is to generate the Pi needed for hydroxyapatite crystallization (Majeska and Wuthier 1975; McComb et al. 1979; Fallon et al. 1980; reviewed in Bonucci 2007) and that such enzyme is crucial for the initiation, but not for the progression/maintenance, of the matrix mineralization process (Bellows et al. 1991). TNAP may also hydrolyze the mineralization inhibitor PPi to facilitate mineral precipitation and growth (Moss et al. 1967; McComb et al. 1979; Rezende et al. 1998; Millán 2006; Addison et al. 2007; Bonucci 2007). In addition, it has been shown that a specific level of TNAP activity has to be reached for the initiation of apatite nucleation and that a moderate reduction in TNAP expression levels and enzyme activity could be sufficient to impair the mineralization process (Wennberg et al. 2000). Therefore, in this study, the absence of bone-like nodule formation in GFs + proteins–treated cultures could be caused not only by an effect on the progression of osteoblastic differentiation (cells not reaching a fully differentiated state) but also by the reduced ALP levels they exhibited. Such an inhibitory effect has also been described after long-term exposure of calvarial cells to platelet extracts (Soffer et al. 2004).
In an attempt to rescue the osteogenic phenotype, treated cultures were exposed to BMP-7 or GDF-5, two osteogenic GFs known to promote development of the osteoblastic phenotype (King et al. 1996; Wozney 2002; Kuniyasu et al. 2003). Both BMP-7 and GDF-5 upregulated Runx2 mRNA expression and reverted downregulation of ALP mRNA expression. Surprisingly, despite upregulation of mRNA, ALP activity was not restored. These results indicate that monitoring of mRNA expression levels alone should be interpreted with caution and support the concept that the relation between transcription and translation is complex (Mehra et al. 2003). Indeed, the lack of correlation between mRNA and protein expression has been reported elsewhere (Lee et al. 2003).
In conclusion, this study showed that exposure of osteogenic cells to a well-defined mixture of GFs + proteins, such as found in platelet preparations, causes downregulation of the osteogenic phenotype. The data are consistent with previous reports showing that PRP preparations do not promote bone formation under certain conditions. Intriguingly, the inhibitory effect of GFs + proteins was expressed even in presence of BMP-7 and GDF-5. However, ALP mRNA levels were restored in cultures first treated with GFs + proteins followed by GDF-5. Our results support the concept that the timing and duration of multiple component molecular therapies play an important role in their outcome and indicate the need for more comprehensive studies using combinations of GFs and proteins aiming to enhance bone formation particularly under unfavorable/adverse conditions.
Footnotes
Acknowledgements
This study was supported by the State of São Paulo Research Foundation (FAPESP; Brazil) and by the National Council of Scientific and Technological Development (CNPq; Brazil). A.N. wishes to acknowledge support from the Canadian Institutes of Health Research and the Natural Sciences and Engineering Research Council of Canada. M.A.O. is the recipient of a Masters scholarship from FAPESP. W.M.A.M. and K.E.V.S. were recipients of internship scholarships from FAPESP and CNPq, respectively. G.E.C. is grateful for a postdoctoral fellowship from FAPESP. The mouse monoclonal anti-human bone ALP antibody (B4-78), developed by Jerry A. Katzmann, was obtained from the Developmental Studies Hybridoma Bank developed under the auspices of the NICHD and maintained by the Department of Biological Sciences of the University of Iowa (Iowa City, IA).
We thank Roger Rodrigo Fernandes and Júnia Ramos for invaluable technical assistance.
