Abstract
The morphology of the osteocyte changes during the cell's lifetime. Shortly after becoming buried in the matrix, an osteocyte is plump with a rich rough endoplasmic reticulum and a well-developed Golgi complex. This “immature” osteocyte reduces its number of organelles to become a “mature” osteocyte when it comes to reside deeper in the bone matrix. We hypothesized that mineralization of the surrounding matrix is the trigger for osteocyte maturation. To verify this, we prevented mineralization of newly formed matrix by administration of 1-hydroxyethylidene-1,1-bisphosphonate (HEBP) and then examined the morphological changes in the osteocytes in rats. In the HEBP group, matrix mineralization was disturbed, but matrix formation was not affected. The osteocytes found in the unmineralized matrix were immature. Mature osteocytes were seen in the corresponding mineralized matrix in the control group. The immature osteocytes in the unmineralized matrix failed to show immunoreactivity with anti-sclerostin antibody, whereas mature osteocytes in the mineralized matrix showed immunoreactivity in both control and HEBP groups. These findings suggest that mineralization of the matrix surrounding the osteocyte is the trigger for cytodifferentiation from a plump immature form to a mature osteocyte. The osteocyte appears to start secreting sclerostin only after it matures in the mineralized bone matrix.
O
Sclerostin is the product of the SOST gene and is reported to be a negative regulator of osteoblastic bone formation (Winkler et al. 2003; van Bezooijen et al. 2005, 2007). Loss of sclerostin in humans causes sclerosteosis and Van Buchem disease, both of which are characterized by a progressively sclerosing bone dysplasia with increased bone formation (Balemans et al. 2001; Brunkow et al. 2001). Recently, it was reported that the cell mainly responsible for secreting sclerostin in bone tissue is the osteocyte. Relationships between matrix mineralization and the expression of sclerostin in osteocytes have been suggested (van Bezooijen et al. 2004; Poole et al. 2005).
A newly buried osteocyte has morphological features similar to those of an osteoblast on the bone surface, such as a rich rough endoplasmic reticulum (rER) and a well-developed Golgi complex. These “young” osteocytes seen near the bone surface or in osteoid are also referred to as formative, osteoblastic, or osteoid osteocytes. After a certain period of time, the osteocyte comes to reside in the mineralized bone matrix at a distance from the bone surface. Such an osteocyte is characterized by fewer cytoplasmic organelles and a thin cell body, and is called a “mature” osteocyte (Jande and Belanger 1973; Irie et al. 2000). The majority of osteocytes seen in the bone matrix are of this type. In light of the recent evidence on sclerostin expression, this classic morphological evidence points to the cooperative occurrence of morphological and functional changes in osteocytes. However, the mechanism by which immature osteocytes mature is obscure. We hypothesized that mineralization of the surrounding matrix is the trigger for osteocyte maturation. To verify this, we used bisphosphonates to prevent bone matrix mineralization. Bisphosphonates are a group of synthetic compounds characterized by P-C-P bonds, and they have a structural similarity to pyrophosphate. They bind to the apatite crystals of hard tissues to inhibit the precipitation, growth, and dissolution of the mineral phase (Fleisch and Russell 1972; Fleisch 1981), and they also inhibit osteoclastic bone resorption. One of them, 1-hydroxyethylidene-1,1-bisphosphonate (HEBP), is well known for its inhibitory effect on mineralization (Schenk et al. 1973). Therefore, to investigate the effect of matrix mineralization on maturation of the osteocyte, we prevented mineralization of the bone matrix by administration of HEBP and examined the morphological and functional changes in osteocytes. The results suggest the importance of matrix mineralization surrounding the osteocytes as a trigger of their morphological and functional changes.
Materials and Methods
All protocols for the current animal experiments were approved by the Animal Ethics and Research Committee, and the study was conducted in accordance with the Guidelines for the Care and Use of Laboratory Animals of the Health Sciences University of Hokkaido.
Four-week-old male Wistar rats were used for the experiment. Rats in the experimental group were subjected for 5 days to daily dorsal subcutaneous injections of HEBP (Sumitomo Pharmaceutical Co.; Osaka, Japan) at 40 mg/kg body weight (HEBP group). Rats of the same age were subcutaneously injected with the same volume of normal saline solution and served as controls (control group). Six hr after the final injections of HEBP or normal saline solution, each animal was fixed by perfusion through the ascending aorta via the left ventricle with either 4% paraformaldehyde in 0.1 M sodium phosphate buffer (pH 7.4) or a mixture of 2% paraformaldehyde and 2% glutaraldehyde 0.05% calcium chloride in 0.08 M cacodylate buffer (pH 7.4). The mandibles were dissected and further fixed by immersion in the same fixative overnight at 4C.
Calcein Labeling
For chronological labeling, calcein (10 mg/kg; Wako, Osaka, Japan) was injected subcutaneously 6 hr before the first injection of HEBP or normal saline solution. Following the procedure described above, the mandibles were fixed in 70% ethanol for 7 days. A cross section ∼2 mm thick at the second molar was made with a diamond disk and stained by being placed in Villanueva bone stain solution (Maruto; Tokyo, Japan) for 10 days. The specimens were then dehydrated with ethanol and embedded in Technovit 7200 VLC (Heraeus Kulzer; Wehrheim, Germany). The block was ground at the second molar, then the calcein labeling was observed by mounting the ground surface of the block on the stage of a fluorescence digital microscope (VB-6000; Keyence, Tokyo, Japan).
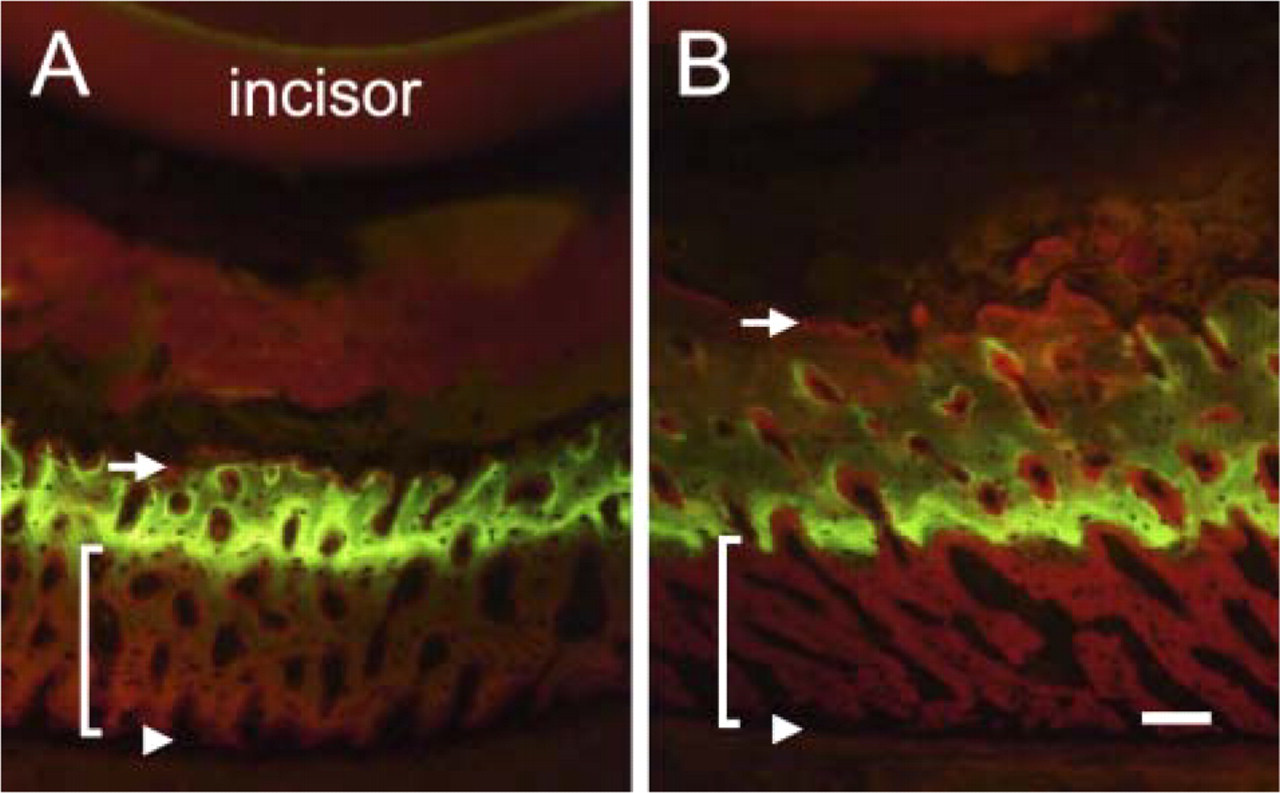
Calcein labeling in the enamel-organ–covering bone (ECB) in the control (
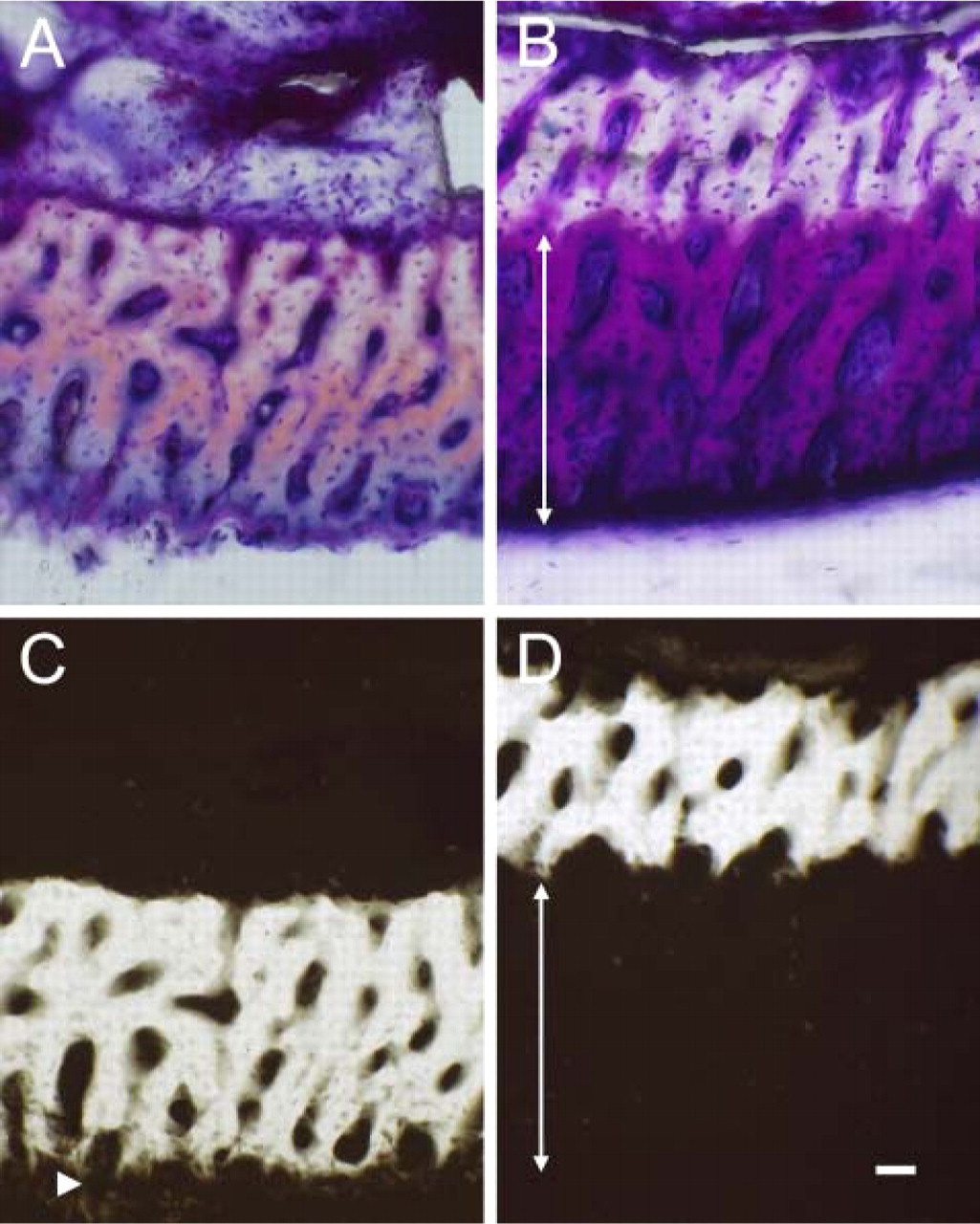
Villanueva bone-stain sections (
Contact Microradiogram
To examine the mineral density of the matrix in mandible, we prepared sections for contact microradiogram. The mandibles were fixed in 70% ethanol for 7 days. A cross section ∼2 mm thick at the second molar was made with a diamond disk and stained by being placed in Villanueva bone stain solution for 10 days. The specimens were then dehydrated with ethanol and embedded in Technovit 7200 VLC, and the blocks were ground to a thickness of 100 μm. Contact microradiographs were taken with a Sofron SRO-405C (Sofron; Tokyo, Japan), employing Kodak SO-181 high-resolution film (Eastman Kodak; Rochester, NY). Irradiation was done at 10 kV and 3.5 mA for 25 min.
Paraffin Immunohistochemistry
For immunohistochemical and histocytological observations, the fixed mandibles were decalcified with 10% EDTA for 10 days at 4C. Then the decalcified mandibles were cut into frontal sections ∼3 mm thick at the second molar. The specimens were dehydrated in ethanol and embedded in paraffin. Sections 6 μm thick were obtained and subjected to immunohistochemistry.
The sections were preincubated for 1 hr at room temperature in 0.01 M phosphate-buffered saline (PBS) containing 1% bovine serum albumin (BSA). Then the specimens were incubated for 3 hr at room temperature with goat anti-mouse SOST antibody (R and D Systems; Minneapolis, MN) diluted 1:100 in 0.01 M PBS; this was followed by incubation with the Simple Stain MAX-PO(G) (Histofine; Nichirei, Tokyo, Japan) for 30 min. Immunoreactivity was visualized with a DAB substrate kit (Histofine) and observed under a light microscope.
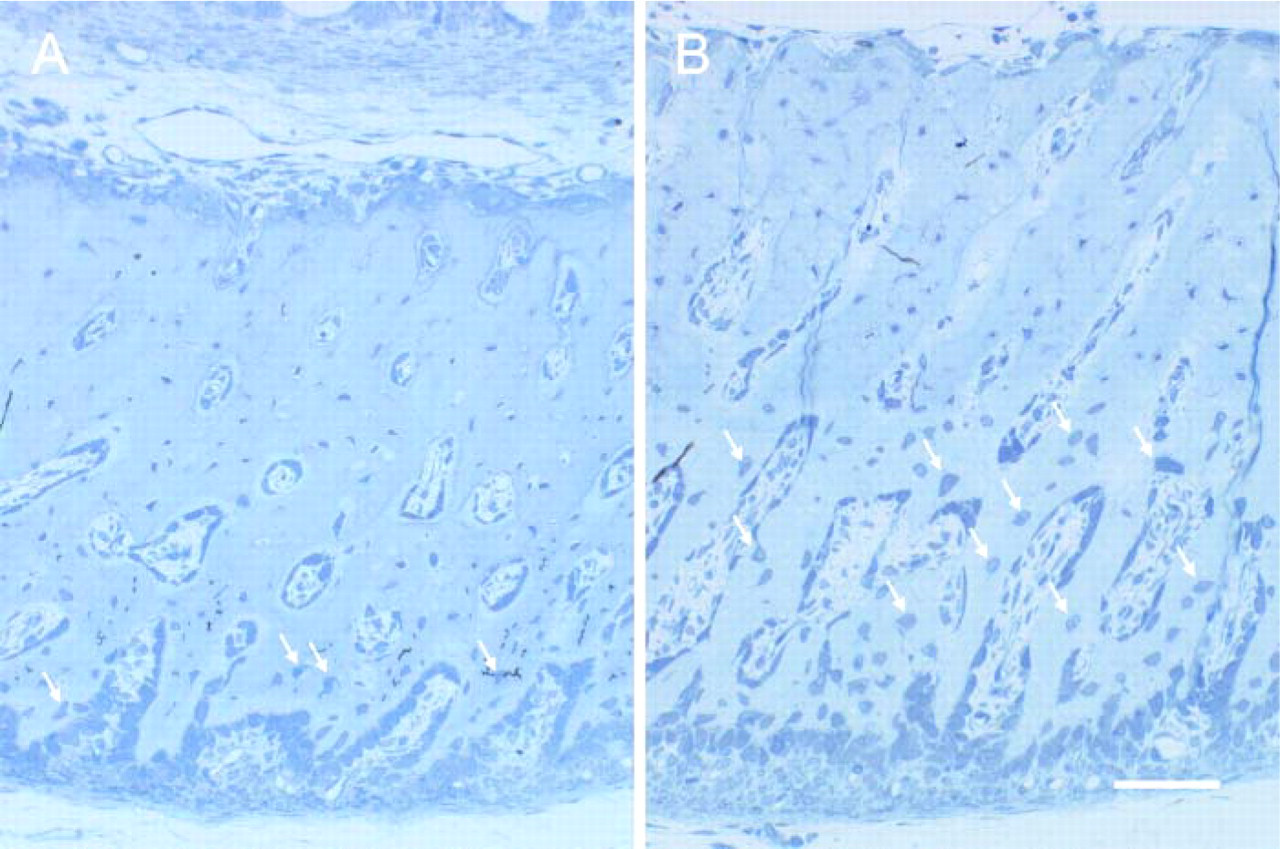
Histological sections of the ECB. In the control group, only the osteocytes (arrows) close to the periosteal surface are plump and immature (
Electron Microscopic Observation
Some mandibles fixed with glutaraldehyde were decalcified with 4.13% EDTA for 2 weeks at 4C. These specimens were then cut into frontal sections ∼1 mm thick at the second molar and postfixed with 1% osmium tetroxide, dehydrated in acetone, and embedded in Taab 812 epoxy resin (Okenshoji Co., Ltd; Tokyo, Japan).
Pre-embedding Immunocytochemistry
For immunohistochemical detection of SOST, 5-mm–thick blocks at the second molar were obtained from the decalcified mandible, and a frontal section ∼80 μm thick was obtained from each block with a Microslicer (D.S.K. DTK-1000; Kyoto, Japan). The sections were preincubated for 3 hr at 4C in 0.01 M PBS containing 1% BSA. The specimens were then incubated for 48 hr at 4C with goat anti-mouse SOST antibody diluted 1:100 in 0.01 M PBS. After incubation of the specimens with horseradish-peroxidase–conjugated rabbit anti-goat IgG F(ab′)2 diluted 1:100 in 0.01 M PBS, the site of antigen–antibody reaction was visualized with a DAB substrate kit. Sections were post-fixed with 1% osmium tetroxide containing 1.5% potassium ferrocyanide and then dehydrated in graded acetone and embedded in Taab 812 epoxy resin.
Semi-thin and ultrathin sections from the resin blocks were obtained and respectively examined under a light or transmission electron microscope.
Results
The area that we examined was the enamel-organ–covering bone (ECB) of the lower incisor. In the ECB of the control group, intense calcein labeling of the matrix was observed at a depth of two-thirds the distance from the periosteal surface (Figure 1A). The labeling became diffuse toward the surface facing the enamel organ (EO). In the HEBP group, intense labeling was observed in the bone matrix in the middle of the ECB (Figure 1B). The thickness of the bone matrix from the most intense part of the calcein label to the periosteal surface, indicating the amount of bone matrix formed during the experimental period, was about the same in the control and HEBP groups. The bone matrix from the most intense part of the calcein label to the EO-facing surface was thicker in the HEBP group than in the control as a result of inhibition of bone resorption on the EO-facing surface by the HEBP.
In the control ECB, only a thin layer of the bone matrix close to the periosteal surface showed radiolucency (Figures 2A and 2C), whereas in the HEBP group, the entire periosteal half of the ECB—all of the bone matrix formed during the experiment—was radiolucent (Figures 2B and 2D).
Histologically, in the control ECB, plump osteocytes were observed only in the bone matrix near the periosteal surface of the ECB (Figure 3A). These osteocytes had features similar to those of osteoblasts on the periosteal surface: they were rich in rER and had a well-developed Golgi complex in their plump cytoplasm (Figure 4A). In the ECB of controls, most of the osteocytes, other than those seen near the periosteal surface, were thin and possessed fewer organelles (Figure 4C). In contrast, in the HEBP group, large, plump osteocytes were observed in the periosteal half of the ECB (Figure 3B). These osteocytes were similar to those seen near the periosteal surface in the control ECB (Figure 4B). In the HEBP group, the osteocytes in the calcified half of the ECB, which faced the EO, were thin and had features typical of mature osteocytes, with fewer organelles (Figure 4D).
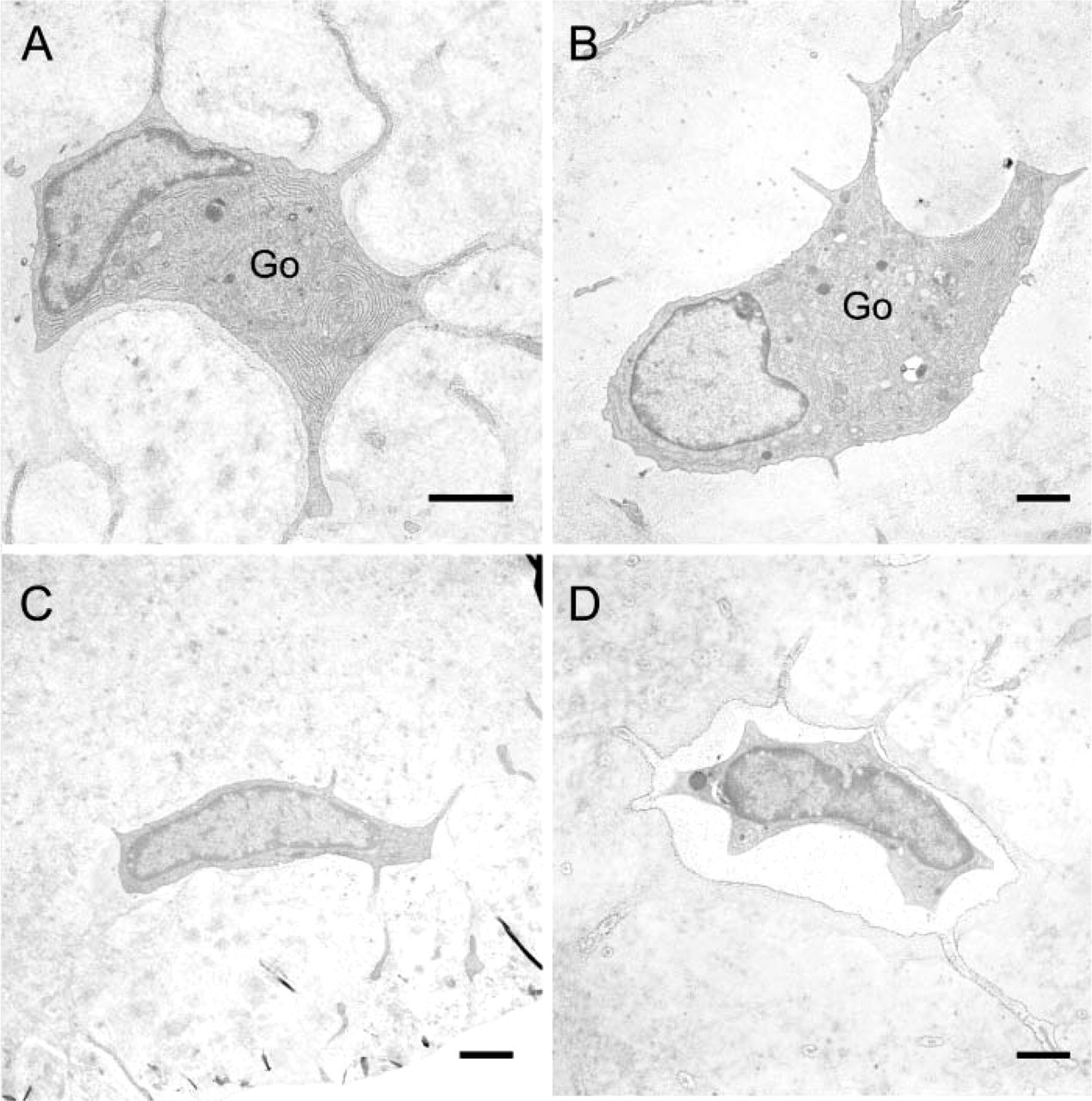
Electron micrographs of osteocytes seen in the ECB. (
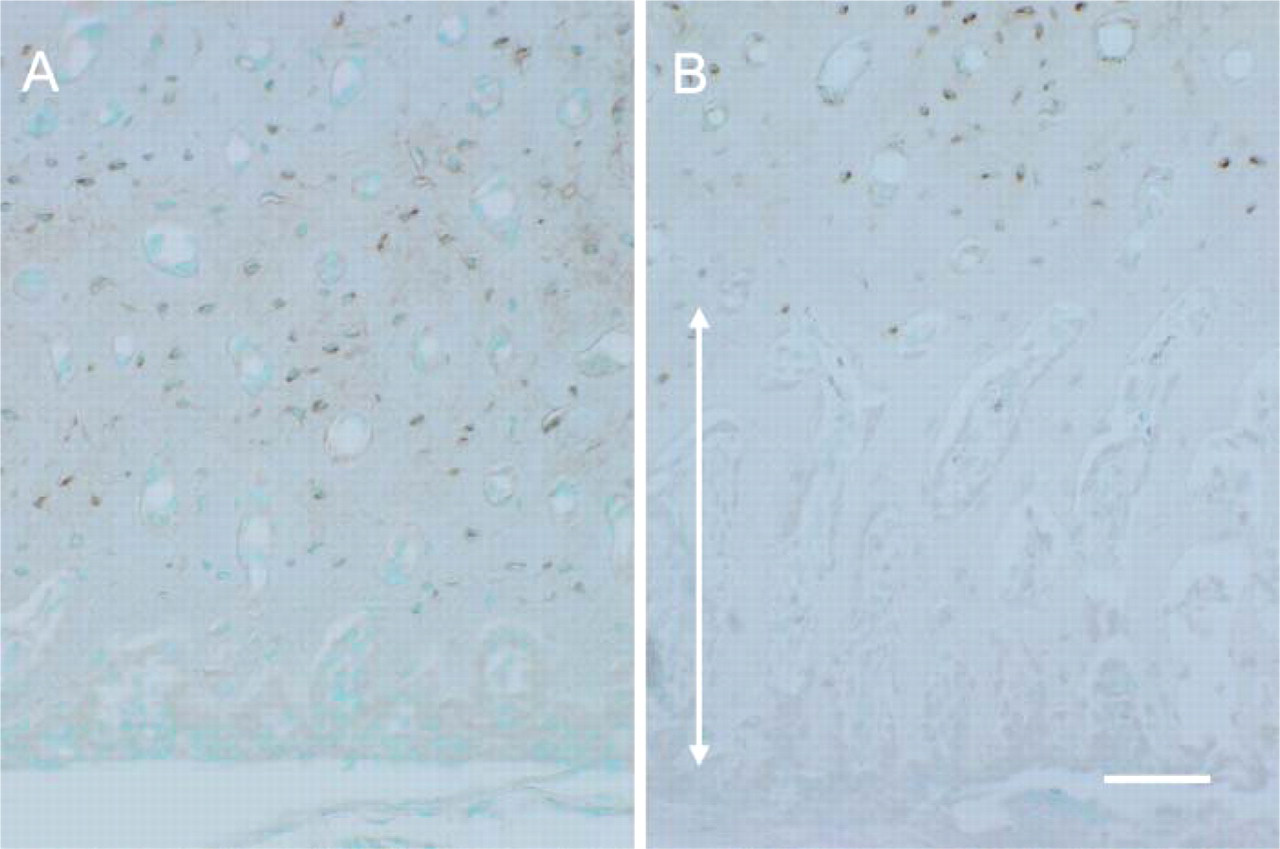
Anti-SOST immunohistochemistry. In the control group (
Anti-SOST immunolabeling was observed in the osteocytes and lacunocanalicular system. In the control group, immunopositive osteocytes were found throughout the ECB, except near the periosteal surface (Figure 5A). In the HEBP group, the osteocytes in the periosteal half of the ECB where the bone matrix was not calcified failed to show immunoreactivity (Figure 5B). Under the electron microscope, immunolabeling for anti-SOST was detected on the surface of the bone lacunae and canalicules and in the space between the lacunar surface and the plasma membrane of the thin, matured osteocytes in control animals (Figure 6).
Discussion
In the ECB of the rat, bone formation on the periosteal surface is coupled with bone resorption on the EO-facing surface, resulting in expansion of the bone toward the outside with controlled thickness (Irie and Ozawa 1990). As a consequence, in the ECB, the young osteocytes reside near the periosteal surface and the mature ones in the inner bone matrix (Irie et al. 2000). Thus the ECB is a suitable site for observing osteocyte maturation.
The thickness of the bone matrix between the periosteal surface and the calcein labeling was about the same in the control and HEBP groups, suggesting that the amount of matrix formed was not affected by the administration of HEBP. However, the corresponding area was highly radiolucent in the HEBP group, whereas the area in the control group was radiopaque, indicating that the matrix formed during the experimental period was unmineralized in the HEBP group. These findings are consistent with the demonstration by Nakahara et al. (1986) that HEBP inhibits mineralization of the bone matrix but does not prevent formation of the bone matrix during ectopic bone formation.
Histologically, the osteocytes seen in this unmineralized bone matrix were large and plump. Ultrastructural observation confirmed that they had rich rER and a well-developed Golgi complex, resembling osteoblasts on the periosteal bone surface. These features are characteristic of young osteocytes, and in the control group, they were seen only in the bone matrix near the periosteal surface. In the HEBP group, even at mid-depth in the ECB where the matrix was not mineralized, the majority of osteocytes seen were of the immature type, with rich rER and a well-developed Golgi apparatus. Morris et al. (1990) also observed osteoblast-like cells surrounded by osteoid matrix in an HEBP-treated, osteosarcoma-derived ectopic bone formation model. These findings suggest that the osteocytes in unmineralized bone matrix stay immature. The fact that mature-type osteocytes were found in mineralized bone matrix in both the control and the HEBP groups suggests the critical role of matrix mineralization in osteocyte maturation.

Electron micrograph showing immunolabeling for anti-SOST antibody in a control animal. Immunolabeling as seen on the bone lacunar surface and in the space between the lacunar surface and plasma membrane of a mature osteocyte. Bar = 1 μm.
It is noteworthy that the plump osteocytes in the unmineralized bone matrix in the HEBP group failed to show immunoreactivity to anti-SOST antibody. Van Bezooijen et al. (2005) reported that SOST mRNA expression was restricted to the mineralization phase of osteoblastic cultures. An immunohistochemical study by Poole et al. (2005) demonstrated that sclerostin-negative osteocytes were significantly closer to bone surfaces than were the more numerous sclerostin-positive osteocytes; they concluded that newly embedded osteocytes start secreting sclerostin after they mature in some way. These results, together with our findings that osteocytes in the mineralized bone matrix of both the control and the HEBP group showed immunoreactivity to anti-SOST antibody, suggest that sclerostin-secreting osteocytes are mature osteocytes. Moreover, we confirmed by electron microscopy that immunolabeling for sclerostin was detected only in mature osteocytes.
The mechanism by which an osteocyte senses the mineralization of the surrounding matrix is obscure. Recently, Hirao et al. (2007) reported that hypoxic conditions promoted the transformation from osteoblast to osteocyte. Thus, it is possible to suppose that a change in access to oxygen is a trigger for the cytodifferentiation of osteocytes. An osteocyte in the mineralized matrix is in a relatively hypoxic condition compared with an osteocyte in unmineralized matrix, even at the same distance from the bone surface. Shear stress produced by fluid flow through the lacuno-canalicular system in bone matrix has also been acknowledged to affect osteocyte function (Bonewald 2006a; Zhang et al. 2006). Assuming that fluid flow in mineralized matrix differs from that in unmineralized matrix, an altered set of genes might be activated in osteocytes. Moreover, the mechanics of the mineralized matrix itself must be significantly different from that of unmineralized matrix. Thus mechanosensing osteocytes (Bonewald 2006b) may sense the difference of the mechanics between mineralized and unmineralized matrix to start the transformation (Mikuni-Takagaki et al. 1996; Khatiwala et al. 2006; Peyton et al. 2007). Another appealing idea is the existence of an unknown mineralization-sensing mechanism in osteocytes, because osteocytes have long been acknowledged to play a role in calcium homeostasis. In this context, dentin matrix protein 1, which is reported to be involved in matrix mineralization and osteocyte differentiation (Feng et al. 2006), is an interesting candidate player in the mineralization-sensing mechanism of osteocytes. In any case, directly or indirectly, matrix mineralization induces osteocyte maturation.
In conclusion, we prevented the mineralization of newly formed bone matrix and examined the morphological and functional changes in the osteocytes. We found plump, immature osteocytes in the unmineralized bone matrix, even far from the bone surface. Only the mature-type osteocytes in the mineralized bone matrix showed immunoreactivity for the anti-SOST antibody. These findings strongly suggest that neither the time elapsed since burial in the bone matrix, nor the distance from the bone surface, but the mineralization of the matrix surrounding the osteocyte is the trigger of cytodifferentiation from young osteocyte to mature osteocyte. They also suggest that osteocytes start secreting sclerostin only after they mature in the mineralized bone matrix.
