Abstract
This study investigated the spatiotemporal dynamics of cardiac CD34⁺ stromal cells (SCs) during the reparative/proliferative phase of post-myocardial infarction (MI) healing. A transmural, non-reperfused MI was induced in middle-aged male Sprague-Dawley rats via left anterior coronary artery ligation, and proliferating cells were labeled with 5-bromo-2′-deoxyuridine. Hearts were collected at days 3, 7, and 14 after MI and analyzed using histology and immunohistochemistry. We found that the myocardial interstitium and coronary vessel adventitia harbored a population of cardiac CD34⁺ SCs. Following MI, activated CD34⁺ SCs expanded from the peri-infarct region across the healing wound through proliferation and migration, often alongside activated fibroblasts/myofibroblasts. While α-SMA⁺ myofibroblasts accumulated at pro-fibrotic granulation tissue sites, CD34⁺ SCs preferentially repopulated residual endomysial scaffolds spared by phagocytic macrophages. Over time, expanding fibrotic tissue progressively overtook these regions, leading to disappearance of CD34⁺ SCs. Importantly, clusters of CD34⁺ SCs accumulated at the scar border around the stumps of surviving cardiac myocytes, seemingly facilitating integration of endomysial connective tissue from non-infarcted myocardium into the developing fibrotic scar matrix. Collectively, these findings suggest that, unlike α-SMA⁺ myofibroblasts, cardiac CD34⁺ SCs seemed to support regenerative rather than fibrotic repair during post-MI wound healing by contributing to the preservation of myocardial stromal architecture.
Keywords
Introduction
Left ventricular (LV) scarring and adverse remodeling following a large myocardial infarction (MI) often lead to progressive heart failure (HF). 1 Despite ongoing advances in pharmacological strategies aimed at preserving LV chamber geometry and systolic function, 2 MI-induced HF remains a leading cause of morbidity and mortality among patients with coronary artery disease in the United States. 3 Consequently, recent years have seen numerous preclinical and clinical efforts to develop novel therapeutic approaches targeting key elements of repair and remodeling within the ischemia-damaged heart, with the goal of further reducing the rate of mortality associated with post-MI HF.4–6
Central to these efforts is the recognition that the size, structural composition, and mechanical properties of the post-MI scar are fundamental determinants of maladaptive LV chamber remodeling. 7 Accordingly, the three overlapping phases of myocardial wound healing—i.e., inflammatory, reparative/proliferative, and maturation/fibrotic 8 —remain the primary grounds for modern-day explorations, including studies of stem cell–based regenerative therapies5,9–11 and scar-modifying interventions6,7,12 aimed at restoring the ventricular wall to optimal structural and functional integrity.
Although some novel cardiac repair strategies have shown promises in improving myocardial wall healing and recovering LV function,9,13 the field of cardiac regenerative therapy, at large, has yet to overcome a number of serious challenges to continue its advances.4,5,11 One major obstacle is the incomplete understanding of the roles of diverse stromal cell (SC) populations involved in post-MI wound repair.14–16
In this regard, it is important to emphasize that the current literature demonstrates an apparent discrepancy pertaining specifically to the nature and role of cardiac resident CD34-positive (+) interstitial/SCs, often referred to as cardiac telocytes. 17 Surprisingly, even after two decades since the initial recognition of these cells within the stroma of mammalian heart, first, as the interstitial Cajal-like cells,18,19 and later, as cardiac telocytes or CD34+ telocytes/SCs,20,21 their existence as a distinct population of resident cells is frequently omitted from a number of recent studies categorizing the cellular composition of the myocardial stroma.15,16,22–25 Moreover, despite several published reports documenting the involvement of cardiac CD34+ SCs, presumably cardiac telocytes, in heart repair,26–28 they are persistently absent from all latest reviews describing the phases of post-MI wound healing.8,15,29
Certainly, the above-mentioned discrepancies in recognition of cardiac telocytes as a unique interstitial/stromal population in the mammalian hearts may largely stem from the lack of telocyte-specific markers30,31 on the one hand and the presence of a noticeable heterogeneity in their immunophenotypes on the other. 31 Therefore, to alleviate such predicament in our current study, we decided to solely utilize the term “cardiac CD34+ SCs” instead of “cardiac telocytes” or “cardiac CD34+ telocytes/SCs,” considering that CD34+ expression remains the most widely accepted marker for identifying these cells within the cardiac stroma.32,33
Most importantly, although nearly all prior studies have acknowledged the essential role of cardiac CD34+ SCs in the extent of ischemia-induced scarring and LV fibrosis,26–28,34 detailed information regarding their spatiotemporal dynamics, particularly during proliferation/reparative phase of post-MI wound healing, remains limited. Furthermore, even though it has been well-established that a large, transmural, nonreperfused MI is the optimal experimental model to examine the temporal sequence of the cellular events at the site of post-ischemic myocardial wound healing, 35 the absence of standardized approaches in earlier studies of cardiac CD34⁺ SC dynamics lead to noticeable inconsistencies with regard to interpretation of obtained results.36,37 Moreover, the exclusive use of young or young-adult animals in these studies,26–28 whose local and systemic regenerative potential remained yet unaffected by biological senescence,38,39 further limits the applicability of the reported findings to human, where the incidents of MI are more prevalent in middle-aged and older individuals. 1
Accordingly, the present study investigates the spatiotemporal dynamics of cardiac CD34+ SCs during post-ischemic wound healing following a large, transmural, nonreperfused MI in middle-aged Sprague-Dawley rats. Specific emphasis is placed on a thorough delineation of cellular events occurring during a reparative/proliferative phase of LV wall healing, focusing on the relationship between cardiac CD34+ SCs and the major reparative events taking place at this phase, i.e., macrophage-mediated wound clearance of necrotic tissue, SC proliferation and migration, granulation tissue formation, and integration of the developing scar tissue with the stroma of surviving myocardium.
Materials and Methods
All animal procedures were approved by the Institutional Animal Care and Use Committee and conducted in accordance with the Guide for the Care and Use of Laboratory Animals, 8th edition (NRC 2011, National Academic Press).
Animals and Experimental Protocol
A large transmural, nonreperfused MI was induced in middle-aged (12-month-old) male Sprague-Dawley rats (n=18; Charles River Laboratories, Inc., Wilmington, MA) under ketamine (100 mg/kg intraperitoneal [i.p.]) and xylazine (10 mg/kg i.p.) anesthesia by permanent ligation of the left anterior descending (LAD) coronary artery, as previously detailed. 40 Age-matched sham-operated rats (n=5) served as controls. Following surgery, rats were housed under climate-controlled conditions at a 12-hour light/dark cycle and provided with standard rat chow and water ad libitum. Post-MI mortality was ~26%, with all death occurring within first 48 hours after surgery.
On day 0, 4, and 11 after LAD artery ligation, three separate groups of rats (n=6 per group) received 5-bromo-2’-deoxyuridine (BrdU; Sigma, St. Louise, MO) in phosphate-buffered saline (PBS) via i.p. ALZET osmotic pumps (Durect, Cupertino, CA) at a dose of 12.5 mg/kg/day for 72 hours, as previously detailed. 41 Rats in these groups were euthanized on days 3, 7, and 14 after MI (n=5–6 per group), respectively, and their hearts were collected for further evaluations. Briefly, rats were anesthetized with 4% isoflurane in pure oxygen, and hearts were arrested in diastole via intracardiac injection of 100 mmol/L potassium chloride. Hearts were then excised, perfusion-fixed on a Langendorff apparatus with 4% paraformaldehyde (PFA) in PBS for 20 minutes, and subsequently stored in a fresh portion of 4% PFA in PBS for 48 hours at +4C, as previously detailed. 42 Each heart was transversely cut into 2-mm-thick parallel slices using a blade guillotine, and the two midventricular slices were processed into paraffin for histology, immunostaining, and quantitative morphometry.
Histology, Immunostaining, and Histochemistry
Transverse serial sections (8.0-µm-thick) were cut from each paraffin-embedded slice and mounted onto microscope slides. From each heart, multiple serial sections were routinely stained with hematoxylin and eosin (H&E), Masson’s trichrome, and picrosirius red stains. Additional sets of serial sections were subjected to immunolabeling using various combinations of primary (Table 1) and secondary antibodies (Table 2) following either protease- or heat-induced epitope (antigen) retrieval protocols.
Primary Antibodies Used for Immunostaining.
Abbreviations: DSHB, Developmental Studies Hybridoma Bank; HIER, heat-induced epitope retrieval using citrate buffer (pH6.0); PIER, protease-induced epitope retrieval using proteinase K (20µg/ml) or trypsin (0.025%).
Fluorophore- and Enzyme-Conjugated Secondary Antibodies Used for Immunostaining.
Abbreviations: IgG (H+L), gamma immunoglobulin heavy and light chains; HRP, horseradish peroxidase; RTU, ready to use.
Briefly, deparaffinized and rehydrated sections were treated with a proteolytic enzyme, either proteinase K (20 µg/ml; cat. no. P4850; Sigma, St. Louis, MO) or trypsin (0.025%; cat. no. SM-2001-C; MilliporeSigma, Burlington, MA) diluted in PBS containing calcium and magnesium ions, for 15 minutes at +37C. Alternatively, sections were subjected to heat-induced epitope retrieval by boiling in 0.01M sodium citrate buffer (pH 6.0) for 30 minutes, followed by cooling in the same buffer for 20 minutes at room temperature (RT). For immunofluorescence staining, sections were incubated with primary antibodies for 1.5 hours at +37C in a humidified chamber, followed by incubation with fluorophore-conjugated secondary antibodies for 45 minutes at +37C. Sections were then coverslipped using a ProLong Gold antifade mounting medium (cat. no. P36931; Molecular Probes, Eugene, OR) containing DAPI (4,’ 6-Diamidino-2-Phenylindole) to counterstain the nuclei, as previously detailed. 43 For immunohistochemistry, endogenous peroxidase activity was quenched by incubating deparaffinized and rehydrated sections in 0.3% hydrogen peroxide in distilled water for 15 minutes at RT. Sections were incubated with primary antibodies for 1.5 hours at +37C in a humidified chamber, followed by incubation with horseradish peroxidase (HRP)-conjugated secondary antibodies (Table 2) for 30 minutes at +37C. Immunoreactivity was visualized using 3,3'-diaminobenzidine (DAB) substrate (cat. no. SK-4103; ImmPACT DAB EqV Peroxidase Substrate kit; Vector Laboratories, Newark, CA) at RT. Nuclei were counterstained with Vector hematoxylin (cat. no. H-3401; Vector Laboratories, Newark, CA). Negative controls included omission of primary antibodies or substitution of primary antibodies with species-specific non-immune serum.
Lectin histochemistry was performed by incubating fluorophore-conjugated lectins concurrently with fluorophore-conjugated secondary antibodies. All washing steps were conducted using PBS containing calcium and magnesium ions. Biotin conjugated lectin was visualized using the VECTASTAIN ABC-HRP kit (cat. no. PK-4000; Vector Laboratories, Newark, CA) in combination with DAB substrate (cat. no. SK-4103; ImmPACT DAB EqV Peroxidase Substrate kit; Vector Laboratories, Newark, CA).
Importantly, selected immunostaining protocols employing a rabbit monoclonal anti-CD34 antibody (Table 1) were intentionally combined with lectin histochemistry to enable precise colocalized visualization of cardiac CD34+ SCs with other cell types, such as phagocytic macrophages and capillary endothelial cells, or with extracellular matrix (ECM) structures, such as endomysial connective tissue scaffolds. This approach was applied in situations where dual immunofluorescence labeling was not feasible due to incompatibility of certain cell/tissue-specific primary antibodies with the heat-induced epitope retrieval protocol required for CD34 antigen unmasking. Accordingly, to address this limitation, Griffonia simplicifolia lectin-IB4 was used to label both phagocytic macrophages 44 and capillary endothelial cells, 45 Lens culinaris agglutinin was employed to identify endomysial connective tissue structures, 46 and Sambucus nigra lectin was used to selectively label vascular endothelial cells 47 (Table 3). In selected cases, lectin cell/tissue specificity was further validated by dual staining with specific primary antibodies that could not be directly combined with anti-CD34 immunolabeling because they exclusively require protease-induced epitope retrieval. According to this validation, all CD68+ phagocytic macrophages within areas of necrotic tissue resorption were labeled by Griffonia simplicifolia lectin-IB4, while laminin+ endomysial scaffolds consistently colocalized with Lens culinaris agglutinin staining (data not shown).
Fluorophore- and Enzyme-Conjugated Lectins Used for Histochemical Staining.

Abbreviations: α, alpha; Gal/GalNAc, galactose/N-acetyl-galactosamine; GSL I, Griffonia Simplicifolia Lectin I.
Light and Fluorescence Microscopy
Stained sections were examined using a Leica DM4000 B microscope (Leica Microsystems, Deerfield, IL) equipped with 20x, 40x, and 63x objectives. Images from H&E, Masson’s trichrome, and picrosirius red-stained sections were acquired with a Leica K3C color digital camera (Leica Microsystems, Deerfield, IL), whereas double- and triple-fluorescence-labeled images were captured using a Leica K3M monochrome digital camera with Leica Application Suite X software. To ensure comparability between experimental groups, all images were acquired using identical microscopy settings, including exposure times, gain, and light intensity. An image analysis was performed using the same standardized procedures for all conditions to guarantee unbiased quantification. Final figures were digitally assembled from the captured images using Adobe Photoshop (version 25.4.0; Adobe Systems, San Jose, CA).
Quantitative Morphometry
From each heart, transverse sections encompassing the entire midventricular profile stained with Masson’s trichrome, picrosirius red or H&E stain were digitized using a PreciPoint M8 digital slide scanner (PreciPoint, Freising, Germany) equipped with 20x and 40x objectives. The resulting high-resolution, zoomable images from serial sections were used to precisely match corresponding LV regions stained with different histological, immunohistological, and histochemical techniques.
To determine infarct size, scanned images of Masson’s trichrome-stained midventricular sections were analyzed using Image-Pro 10 software (Media Cybernetics, L.P., Silver Spring, MD). Briefly, in each ventricular cross-section, the total LV circumference and the length of the infarcted segment were measured at the mid-wall level. Infarct size was expressed as the percentage of the infarcted region length relative to the total LV circumference.
The same digitized images were further evaluated to quantify additional LV structural parameters, including LV cross-sectional area (CSA) and mean diameter, LV cavity CSA and mean cavity diameter, average thickness of the non-infarcted free wall and interventricular septum, the average thickness and CSA of the infarcted region, as previously detailed. 48 All quantifications were performed in a blinded manner, with investigators unaware of the experimental groups to ensure an unbiased assessment.
Statistical Analysis
Data are presented as mean ± standard error of the mean (SEM). Statistical analyses were performed using Prism 6 software (IBM Corp., Armonk, NY). Comparisons among multiple groups were conducted using one-way analysis of variance (ANOVA) followed by Tukey’s post-hoc test. Unpaired one-tailed Student’s t-tests were used for comparisons between two groups. Statistical significance was defined as p≤0.05, with threshold of p≤0.01 and p≤0.001 indicating higher levels of significance.
Results
Experimental Rationale and Temporal Framework
Because a large, transmural, non-reperfused MI may significantly limit rapid infiltration of the infarcted LV region by circulating inflammatory and other nonresident cells, thereby enhancing the opportunity to observe the intrinsic behavior of resident cardiac SCs throughout the healing response, only hearts with an MI size ≥40% of the LV circumference were included in the final analysis (Table 4). Furthermore, because this MI model is characterized by a prolonged period of necrotic tissue clearance and wound healing, three sequential post-MI time points—days 3, 7, and 14—were deliberately selected to capture the dynamics of spatiotemporal changes within the injured myocardium as the reparative process progressed from the late inflammatory to early maturation phases.
Structural Parameters of the Left Ventricle in Sham-Operated and Post-MI Rats.
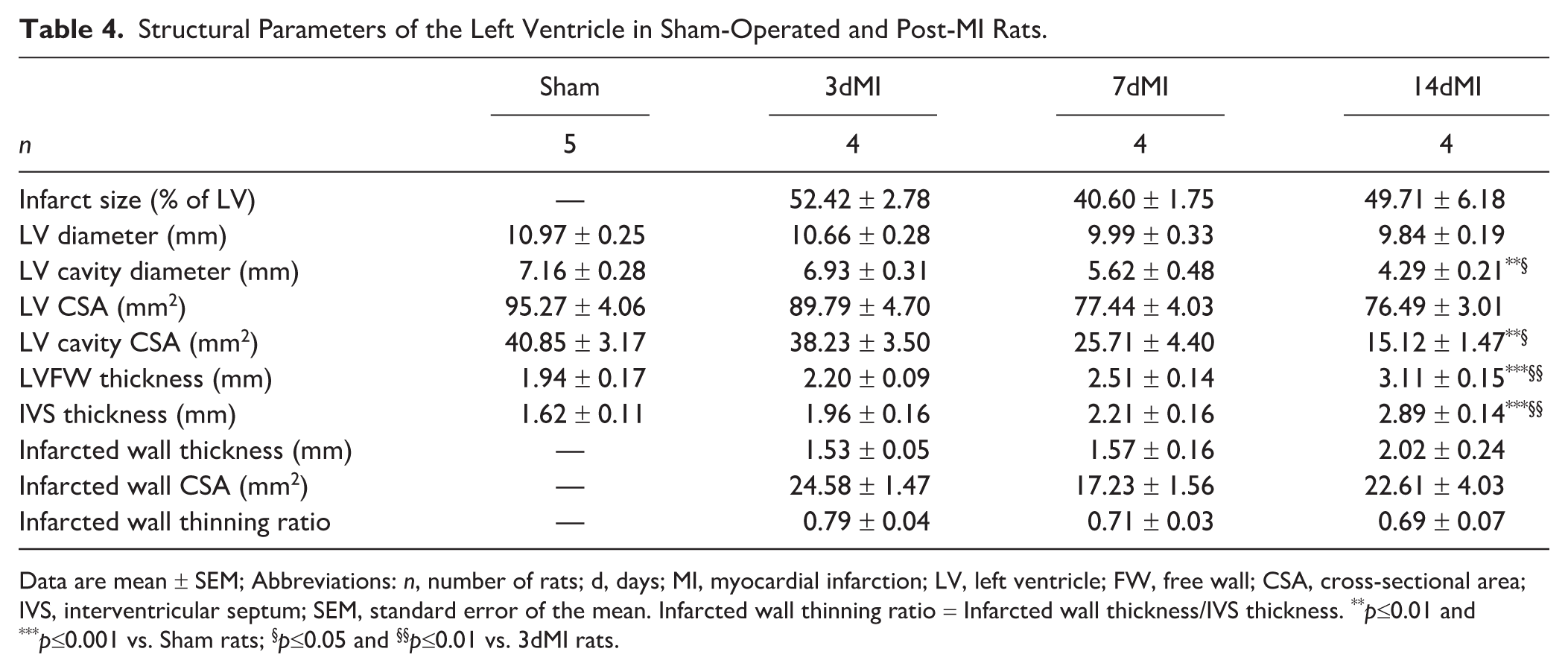
Data are mean ± SEM; Abbreviations: n, number of rats; d, days; MI, myocardial infarction; LV, left ventricle; FW, free wall; CSA, cross-sectional area; IVS, interventricular septum; SEM, standard error of the mean. Infarcted wall thinning ratio = Infarcted wall thickness/IVS thickness. **p≤0.01 and ***p≤0.001 vs. Sham rats; §p≤0.05 and §§p≤0.01 vs. 3dMI rats.
Intact Myocardium Contains a Distinct Population of Cardiac CD34+ SCs Residing in the Adventitial and Interstitial Connective Tissue
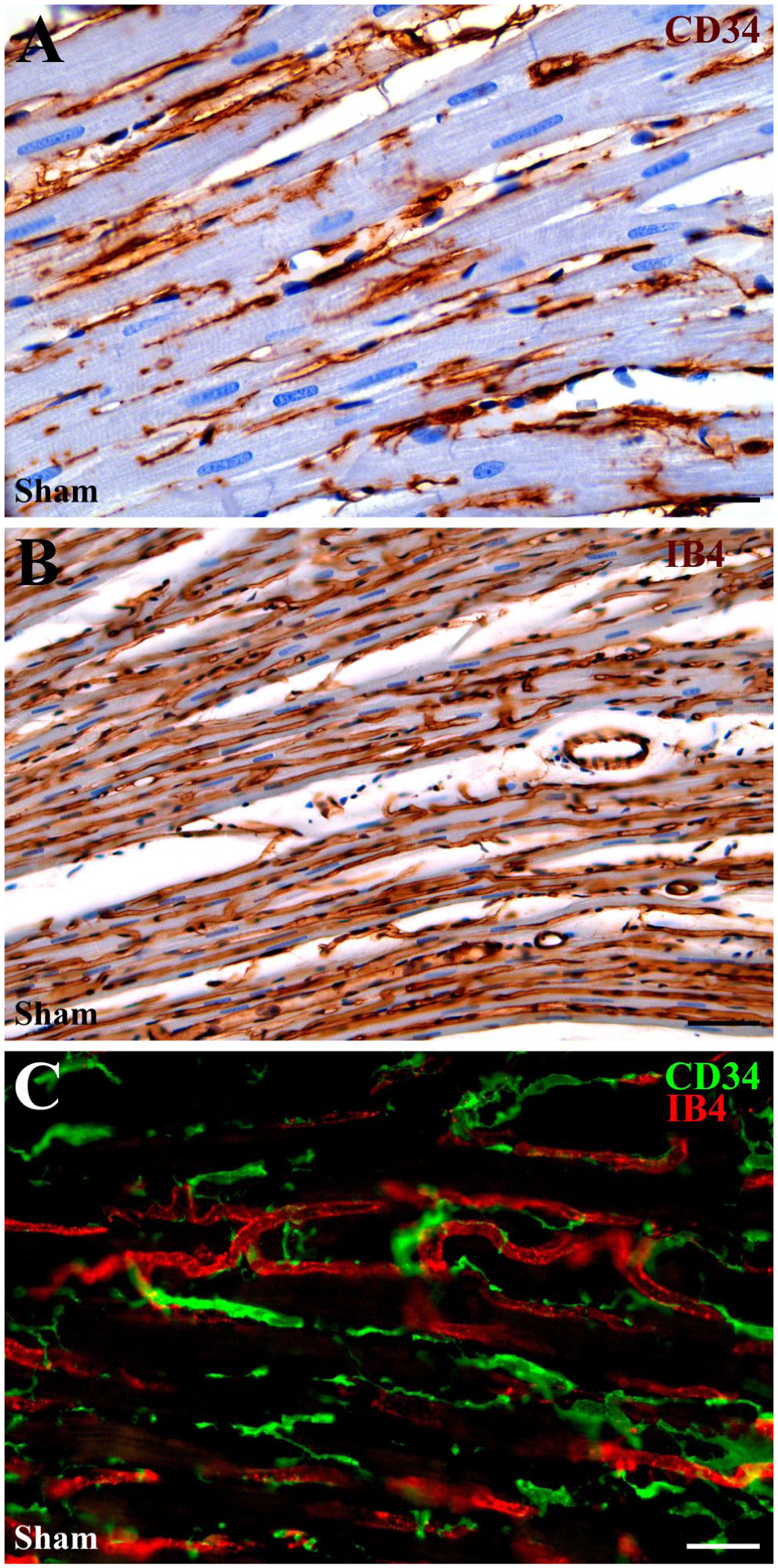
Examination of intact LV myocardium from sham-operated rats revealed that the resident cardiac CD34+ SCs were present in the adventitia of coronary arteries (Fig. 1A–C) and veins (Fig. 1C, D and G), as well as within the myocardial interstitium near capillaries (Fig. 1C, D) and in close association with cardiac myocytes (Fig. 1E–H). In the adventitia of large coronary arteries, cardiac CD34+ SCs formed a mesh-like network encircling the vessel circumference (Fig. 1A, B), whereas cells located within the myocardial interstitium and around small veins were distributed more sparsely and randomly, without an obvious organizational pattern (Fig. 1E–H). Furthermore, a detailed morphological assessment proved phenotypic heterogeneity within the cardiac CD34+ SC population, with cells exhibiting features reminiscent of both cardiac telocytes and activated fibroblasts (Fig. 1E–H).
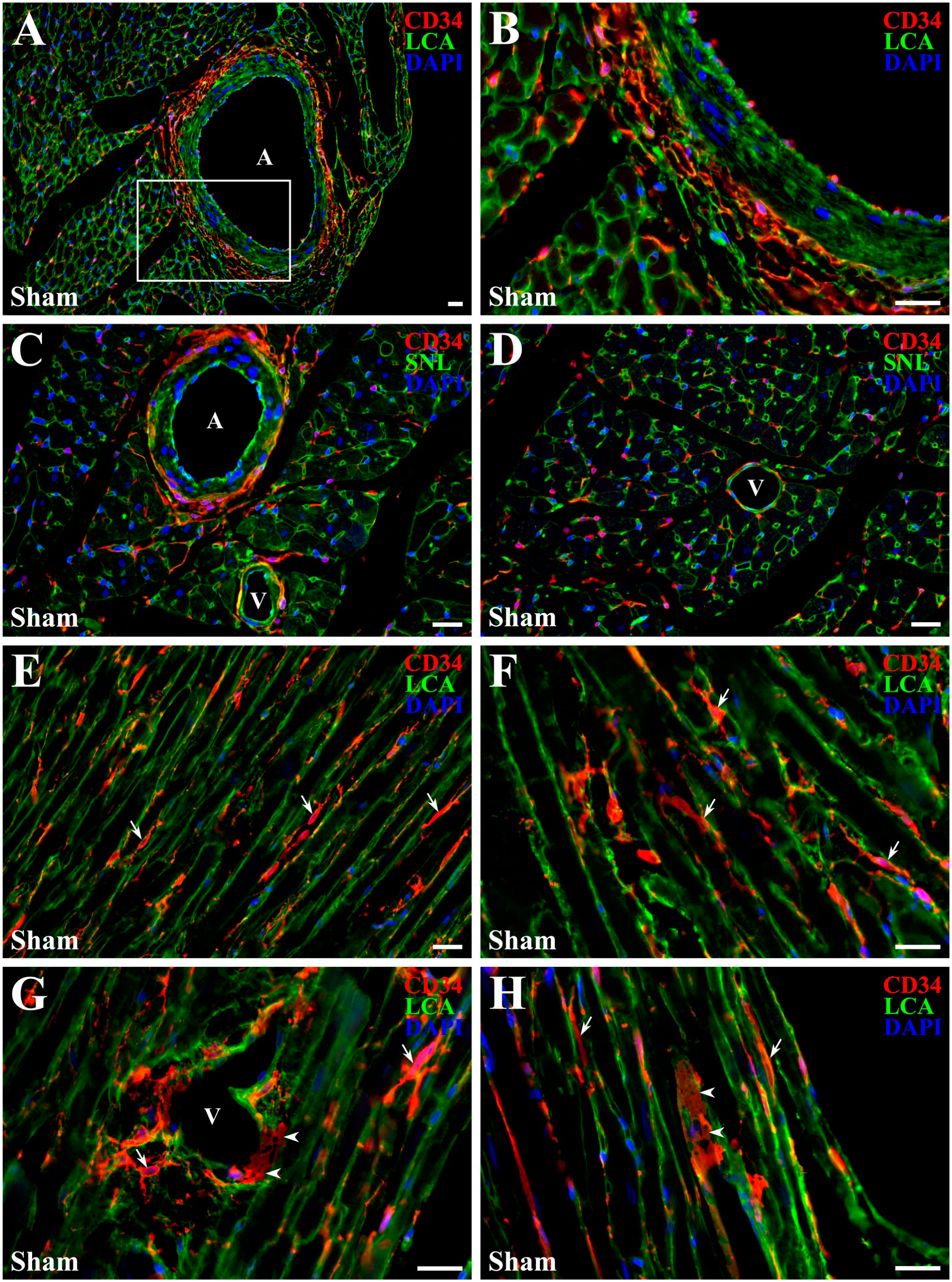
Spatial distribution of cardiac CD34+ SCs (red) in LV myocardium of sham-operated rats. In A, B, and E-H, endomysial ECM is visualized with a fluorescein-conjugated Lens Culinaris agglutinin (LCA, green); in C and D, endothelial cells are visualized with fluorescein-conjugated Sambucus Nigra lectin (SNL, green). Nuclei are counterstained with DAPI (blue). (A) Low-power view of a large coronary artery [A] showing aggregation of cardiac CD34+ SCs in the tunica adventitia. (B) High-power view of the rectangle area outlined in A, showing in a dense, web-like network of cardiac CD34+ SCs; some endothelial cells lining the vessel lumen also express CD34, while the tunica media, composed of vascular smooth muscle cells, remains CD34-negative. (C, D) High-power views of transversely sectioned LV myocardium showing cardiac CD34+ SCs in the adventitia of a medium-sized coronary artery (A) and small veins (V), as well as in interstitial spaces near capillaries and cardiac myocytes. Note that capillary endothelial cells are CD34-negative. (E–H) High-power views of longitudinally sectioned cardiac myocytes showing phenotypic heterogeneity among cardiac CD34+ SCs distributed in the myocardial interstitium and adventitia of a small vein (V). Some cells have small bodies with slim, elongated processes (arrows), typical of cardiac telocytes, while others appear enlarged and flattened along the cardiac myocyte or vessel wall surfaces (arrowheads), resembling activated fibroblasts. Scale bars: 20 µm.
Because vascular endothelial cells can express CD34 protein and thus potentially confound identification of cardiac CD34+ SCs, numerous vascular profiles were systemically examined. According to this analysis, CD34+ immunoreactivity was predominantly confined to endothelial cells lining the endocardium and the lumens of large coronary arteries (Fig. 1B), whereas endothelial cells of medium- and small-sized coronary arteries, arterioles, veins, venules, and myocardial capillaries were largely CD34 immunonegative (Fig. 1C, D, and G).
Given that cardiac CD34+ SCs were frequently observed near capillary profiles in transverse myocardial sections (Fig. 1C, D), raising the possibility of a pericytic identity, their spatial relationship to capillaries was further assessed in longitudinal sections using co-labeling with IB4 lectin (Fig. 2A, B). Although some cardiac CD34+ SCs were occasionally positioned adjacent to IB4+ capillary wall, more commonly their cell bodies and/or processes were seen at a distance from the capillary boundaries (Fig. 2C), arguing against the pericytic nature of these cells.
Arrangement of cardiac CD34+ SCs and capillaries in LV myocardium of sham-operated rats. (A) Distribution of cardiac CD34+ SCs visualized with an HRP-conjugated secondary antibody and DAB substrate (brown). (B) Distribution of the capillary network visualized with biotin-conjugated Griffonia Simplicifolia I isolectin B4 (IB4+), followed by labeling with HRP-conjugated avidin-biotin complex and DAB substrate (brown). Nuclei are counterstained with Gill hematoxylin. Note that cardiac CD34+ SCs and IB4+ capillaries exhibit distinct spatial patterns, with minimal alignment. (C) High-power fluorescence micrograph showing juxtaposition of CD34+ SCs (green) and IB4+ capillaries (red). Cardiac CD34+ SCs are rarely aligned with capillary walls, suggesting that they are unlikely to be pericytes. Scale bars: 20 µm (A, C) and 50 µm (B).
Clearance of Necrotic Tissue and Formation of Granulation Tissue Are Delayed Following Large, Transmural, Nonreperfused MI in Middle-Aged Rats
Quantitative morphometric analysis revealed that, while the size and thickness of the infarcted region remained relatively constant across the three post-MI time points, non-infarcted portions of the LV free wall and intraventricular septum exhibited progressive thickening consistent with compensatory hypertrophy (Table 4 and Fig. 3). Importantly, during the progressive inward resorption of necrotic myocardium, much of the endomysial connective tissue scaffolding remained preserved. These structures were readily identifiable as hollow, honeycomb-like ECM frameworks occupying the space between centrally positioned necrotic tissue (Fig. 3, asterisks and dotted outlines on high-power micrographs) and the infarct borders, which were delineated laterally by viable LV myocardium and subendocardially by a thin rim of surviving cardiac myocytes (Fig. 3). Notably, substantial accumulation of granulation tissue was not evident until approximately 14 days after MI, indicating delayed wound clearance and repair. Moreover, developing granulation tissue preferentially emerged adjacent to residual necrotic debris and around surviving coronary arteries and subsequently expanded outward into regions previously occupied by hollow endomysial scaffolds (Fig. 3). Together these distinctive spatiotemporal features provided a favorable framework for a detailed analysis of cardiac CD34+ SCs behavior during myocardial healing.
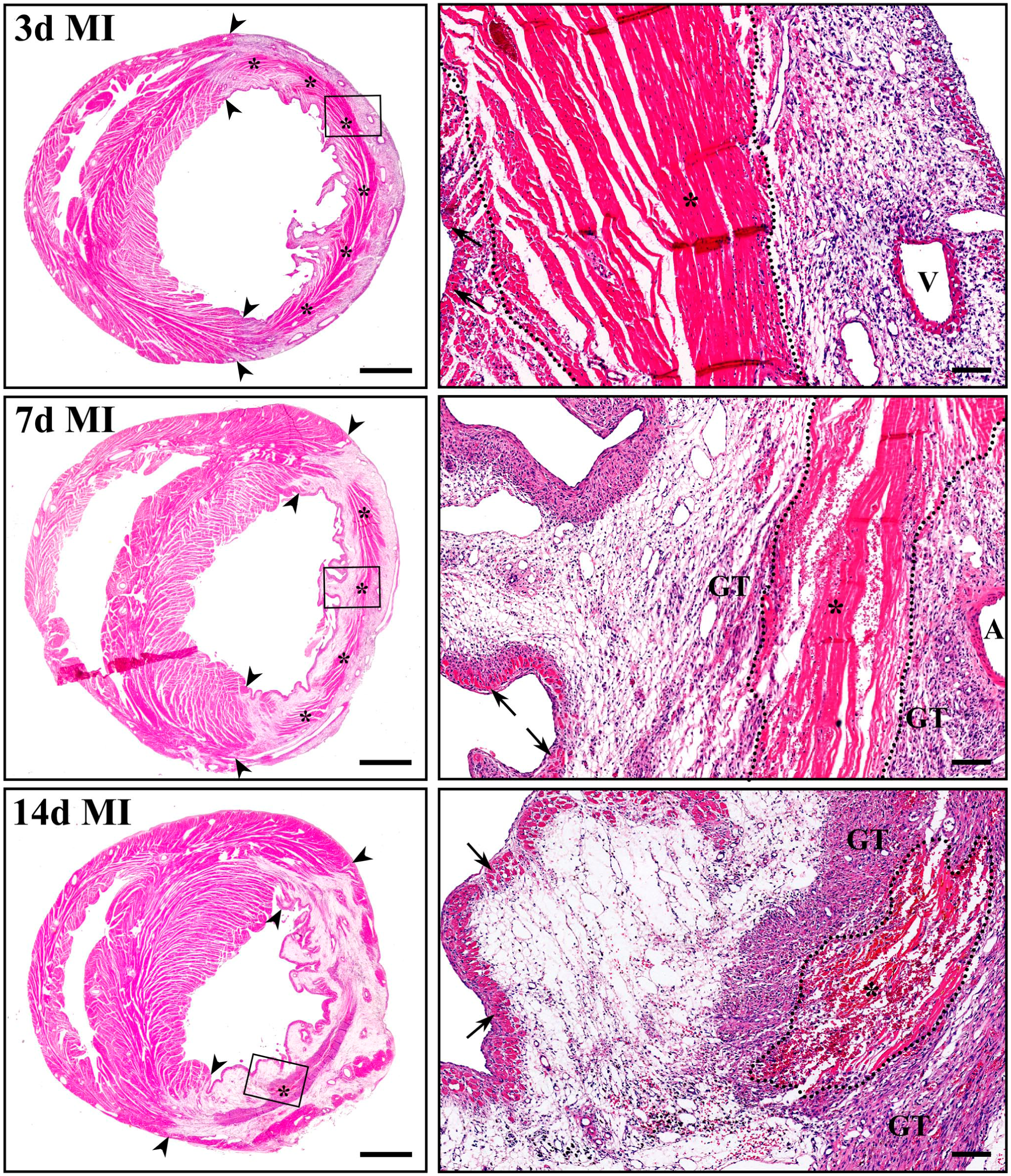
Hematoxylin and eosin–stained LV cross-sections showing the extent of transmural post-ischemic injury and the progression of necrotic tissue resorption at 3, 7, and 14 days (d) after MI. In the transmural region, surviving cardiac myocytes persist along the subendocardial layer (arrows) and near residual coronary veins (V). Arrowheads denote lateral MI borders, whereas asterisks and dotted outlines identify necrotic remnants. Note that necrotic debris removal is prolonged and does not result in thinning of the LV wall. Importantly, debris clearance preserves much of the original endomysial scaffolds, visible as hollow, honeycomb-like structures surrounding areas occupied by necrotic remnants or developing granulation tissue (GT). Granulation tissue preferentially evolves around surviving coronary arteries (A) and necrotic remnants (high-power micrographs of post-MI regions outlined by black boxes in the corresponding low-power views) and expands outward into regions filled with endomysial connective tissue scaffolds. Scale bars: 1.5 mm (low-power micrographs) and 100 µm (high-power micrographs).
Activated Cardiac CD34⁺ SCs Migrate Across the Myocardial Wound Along Endomysial Scaffolds Preserved During Macrophage-Mediated Necrotic Debris Clearance
Analysis of damaged myocardium at post-MI days 3, 7, and 14 demonstrated that activated cardiac CD34+ SCs originating from peri-infarcted regions progressively migrated inward into areas of necrotic tissue resorption containing preserved endomysial scaffolds (Fig. 4). Importantly, comparison of the spatiotemporal distribution of cardiac CD34+ SCs and phagocytic macrophages revealed a coordinated pattern: early in the reparative phase, both cell populations were densely intermingled near the infarct border (Fig. 5A, B), whereas at later stages, cardiac CD34+ SCs consistently trailed behind phagocytic macrophages (Fig. 5C), which advanced further into mid-wall regions still filled with necrotic remnants (Fig. 5D). Lectin labeling confirmed that regions cleared of cellular debris retained ECM components of the original endomysial frameworks (Fig. 6A, B). Moreover, the alignment of activated cardiac CD34+ SCs along these preserved scaffolds (Fig. 6C) suggests that maintenance of the endomysial ECM facilitates their migration across the healing region, potentially by providing structural support and/or directional cues (Fig. 6D).
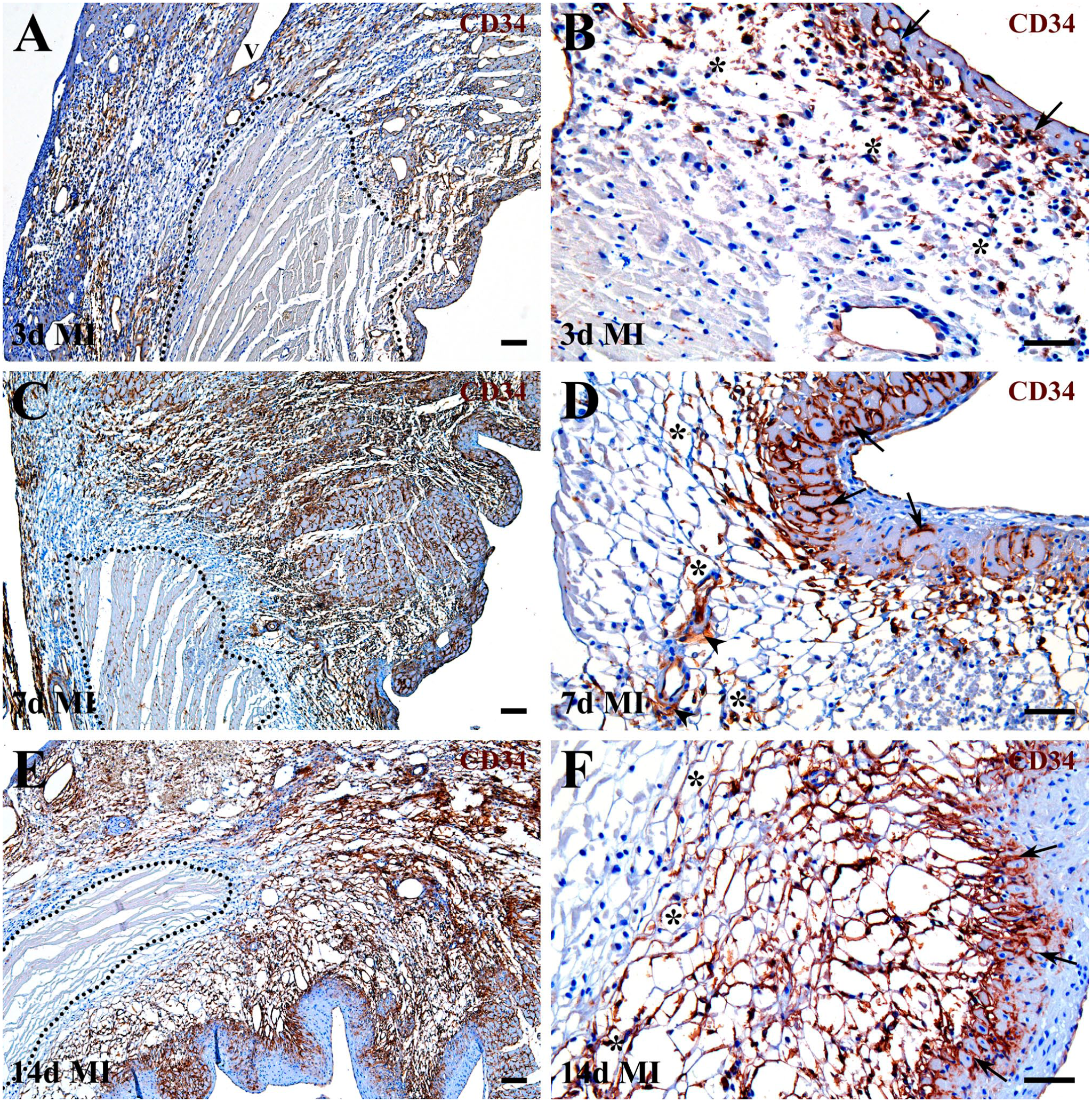
Spatiotemporal dynamics of activated cardiac CD34+ SCs in the healing region at 3 (A, B), 7 (C, D), and 14 (E, F) days after MI. Cardiac CD34+ SCs are visualized with an HRP-conjugated secondary antibody and DAB substrate (brown), whereas nuclei are counterstained with Gill hematoxylin (dark blue). Hematoxylin also provides a light-blue background for cardiac myocytes, non-myocyte cells, necrotic debris, and ECM scaffolds. Note that cardiac CD34+ SCs are absent from necrotic tissue (dotted outlines in A, C, and E) but expand into debris-cleared areas (asterisks in B, D, and F) from the lateral infarct borders, the interstitium surrounding cardiac myocytes surviving near subepicardial veins (V in the micrograph A) and beneath the endocardium (arrows in B, D, F), as well as from the adventitia of residual coronary vessels (arrowheads in D). Scale bars: 100 µm (A, C, and E) and 50 µm (B, D, and F).
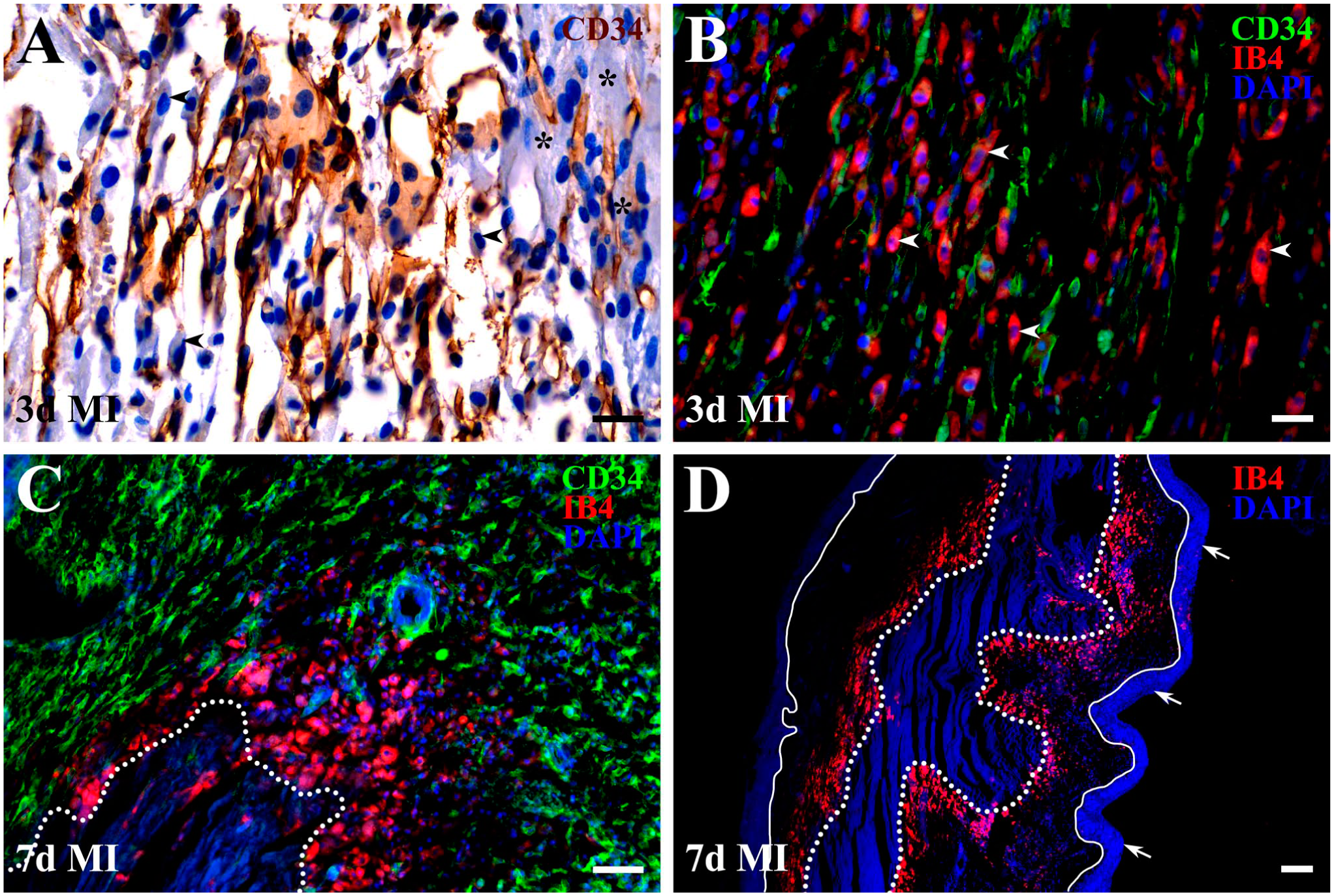
Juxtaposition of activated cardiac CD34+ SCs and phagocytic macrophages at 3 (A, B) and 7 (C, D) days after MI. (A) Activated cardiac CD34+ SCs, visualized with an HRP-conjugated secondary antibody and DAB substrate (brown), intermingle with large, oval-shaped macrophage-like cells (arrowheads) in regions of necrotic debris clearance near surviving myocardium (asterisks). Gill hematoxylin counterstains nuclei and provides a light-blue tone to the cytoplasm of cardiac myocytes and other cells. (B) Fluorescence micrograph shows cardiac CD34+ SCs (green) intermingled with IB4+ phagocytic macrophages (red) at the lateral MI border. Nuclei are counterstained with DAPI (blue). The parallel alignment of these two cell populations suggests coordinated migration across the wounded region. Importantly, the morphology of IB4+ phagocytic macrophages in B (arrowheads) closely resembles that of macrophage-like cells marked by arrowheads in A. (C) High-power fluorescence micrograph showing the spatial distribution of cardiac CD34+ SCs (green) and IB4+ phagocytic macrophages (red) in an area adjacent to necrotic tissue remnants. Nuclei are counterstained with DAPI, and the doted outline demarcates the area occupied by necrotic debris. Note that activated cardiac CD34+ SCs remain behind phagocytic macrophages actively involved in debris removal. (D) Low-power fluorescence micrograph depicting the IB4+ phagocytic macrophages (red) engulfing necrotic tissue in the transmural region. DAPI, in addition to serving as a nuclear counterstain, provides nonspecific background labeling of necrotic tissue in the center of the infarcted region (dotted outline), as well as viable tissue in the subendocardial (arrows) and subepicardial regions. Solid lines delineate surviving tissue from area of cellular debris resorption. The poorly stained space between the solid and dotted lines represents regions cleared of necrotic tissue and occupied by preserved endomysial scaffolds. Scale bars: 20 µm (A, B), 100 µm (C), and 50 µm (D).
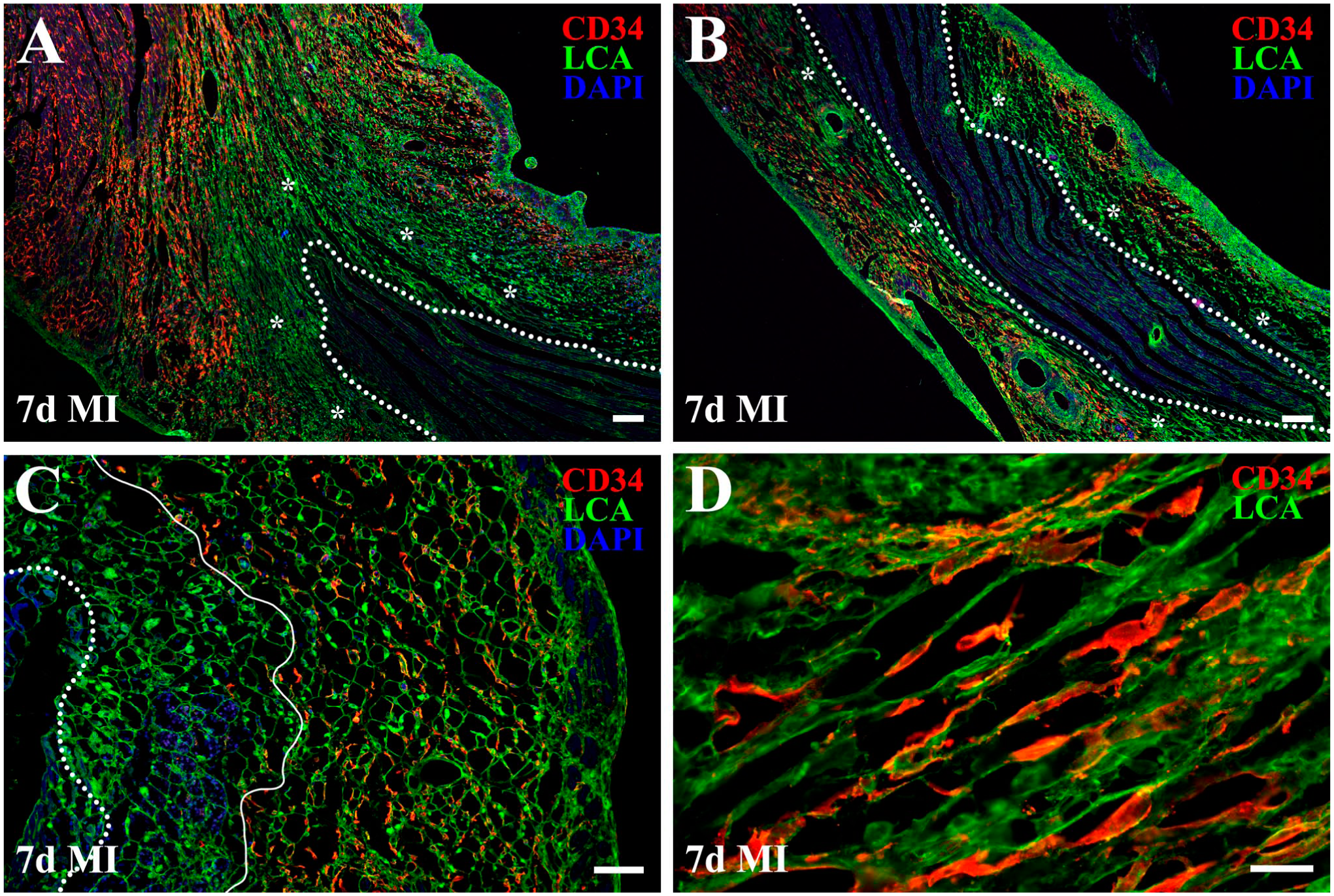
Spatial relationship between migrating cardiac CD34+ SCs (red) and endomysial ECM scaffolds, visualized with fluorescein-conjugated Lens Culinaris agglutinin (LCA, green), in post-MI region cleared of necrotic debris. In A-C, nuclei are counterstained with DAPI (blue), and doted outlines demarcate areas occupied by necrotic tissue remnants. Note that the two low-power fluorescence images, one from the lateral infarct border (A) and the other from the transmural portion of the infarct (B), demonstrate that migrating cardiac CD34+ SCs repopulate preserved endomysial scaffolds (asterisks). The spatial distribution of these cells across the healing region indicates that they advance centrally following the macrophage progression. (C) High-power fluorescence micrograph illustrating inward repopulation of hollow, honeycomb-like endomysial scaffolds by activated cardiac CD34+ SCs. Note that the narrow space between the solid and dotted outlines adjacent to necrotic tissue remains devoid of migrating cardiac CD34+ SCs. Importantly, the close alignment between activated cardiac CD34+ SCs and the LCA+ honeycomb-like endomysial scaffold supports the concept that preserved original ECM plays a pivotal role in cardiac CD34+ SC migration. (D) High-power fluorescence micrograph showing the distribution of cardiac CD34+ SCs along preserved endomysial scaffolds. The parallel orientation of migrating cells suggests coordinated movement in a common direction, while preserved ECM scaffolds appear to play a key role in guiding this process. Scale bars: 100 µm (A, B), 50 µm (C), and 20 µm (D).
Activated Cardiac CD34+ SCs Undergo Proliferation During Myocardial Wound Healing
To assess whether expansion of activated cardiac CD34+ SC within the post-MI region involved cell proliferation, BrdU incorporation was examined. Subsequently, BrdU-positive (+) cardiac CD34+ SCs were detected within regions cleared of necrotic debris (Fig. 7A–F, arrowheads), indicating mitotic activity. In several instances, BrdU+ cardiac CD34+ SCs were aligned in parallel rows, suggesting that proliferation occurred concomitantly with cell migration across the healing wound (Fig. 7E–F). Importantly, activated cardiac CD34+ SCs were frequently juxtaposed with numerous BrdU+ cells lacking CD34 immunoreactivity (Fig. 7A–D), consistent with the presence of proliferating fibroblasts and/or myofibroblasts—the principal cellular components of developing granulation tissue. Supporting this interpretation, subendocardial and subepicardial fibrotic thickenings in the post-MI region consistently contained only CD34− cells, some of which were BrdU+ (Fig. 7G–H, arrows), indicative of their fibroblastic or myofibroblastic identity.
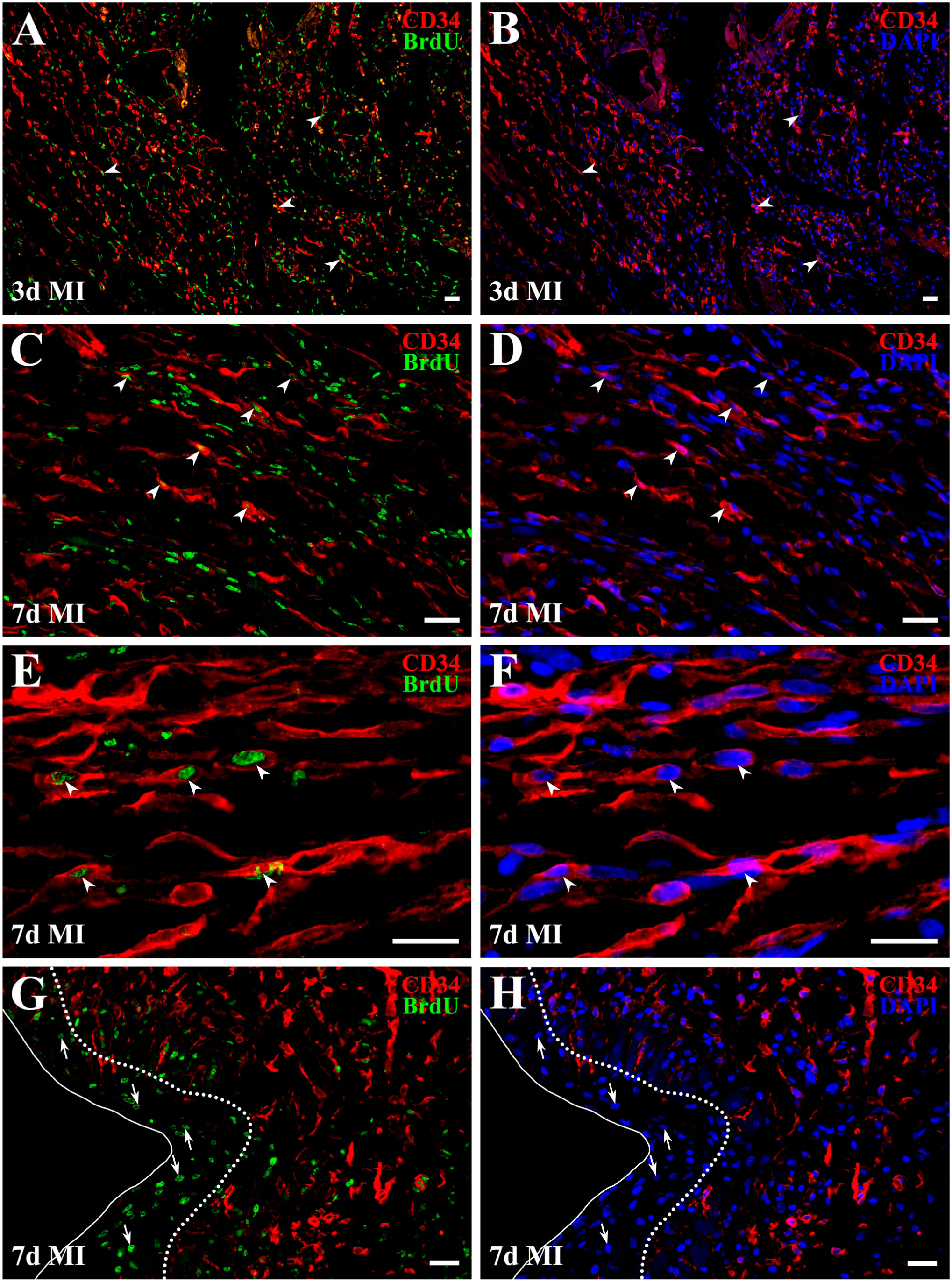
Proliferating cardiac CD34+ SCs in the healing myocardium following MI. In micrographs B, D, F, and H, nuclei are counterstain with DAPI (blue). Arrowheads and arrows in corresponding image pairs (A–B, C–D, E–F, and G–H) identify the same nuclei showing positive immunoreactivity for BrdU (green), indicating cell proliferative. On day 3 (A) and 7 (C, E, and G) after MI, regions cleared of necrotic debris contain numerous BrdU+ cells. A subset of these cells is CD34 immunopositive and therefore represent activated cardiac CD34+ SCs (arrowheads). BrdU+ cells lacking CD34+ immunoreactivity likely correspond to activated fibroblasts/myofibroblasts associated with granulation tissue development. (E–F) High-power micrographs demonstrate BrdU labeling in activated cardiac CD34+ SCs (arrowheads), which exhibit an elongated morphology and parallel alignment, consistent with proliferation during migration through the healing wound. (G–H) Representative post-MI region showing abundant BrdU+/CD34− cells (arrows) within the subendocardial space between the endothelial lining (solid line) and the former myocardial boundary (dotted line), consistent with the development of fibrotic subendocardial thickening. Scale bars: 20 µm.
Activated Cardiac CD34⁺ SCs Avoid Myofibroblast-Rich Areas During Myocardial Wound Healing
To further define the relationship between cardiac CD34+ SCs and myofibroblasts, alpha (α)-smooth muscle actin (α-SMA) immunostaining was employed. Early in the reparative phase, α-SMA+ myofibroblasts were largely confined to developing subepicardial and subendocardial fibrotic thickenings (Fig. 8A, D), whereas they were notably absent from mid-wall regions adjacent to the necrotic core (Fig. 8A) and from lateral infarct borders (Fig. 8B)—areas in the post-MI region typically associated with granulation tissue formation. Importantly, growing myofibroblast-rich thickenings lacked activated cardiac CD34+ SCs, whereas these cells remained abundant in the interstitium surrounding viable cardiac myocytes beneath the fibrotic layers and along preserved endomysial scaffolds in debris-cleared regions (Fig. 8C, D). However, at later stages, myofibroblasts-rich granulation tissue that became prominent near the necrotic core (Fig. 8E, F) similarly lacked activated cardiac CD34+ SCs (Fig. 8F). In contrast, cardiac CD34+ SCs persisted primarily within narrow zone containing residual endomysial scaffolds situated between expanding myofibroblast-rich structures (Fig. 8E, F). Furthermore, when residual coronary artery became enveloped by granulation tissue, the adventitial networks of cardiac CD34+ SCs gradually diminished (Fig. 8G). A comparable disappearance cardiac CD34+ SCs was also observed at the interface between outwardly expanding granulation tissue and adjacent regions still occupied by hollow endomysial frameworks (Fig. 8H). Consequently, as the reparative phase advanced, the spatial domain occupied by activated cardiac CD34+ SCs progressively decreased.
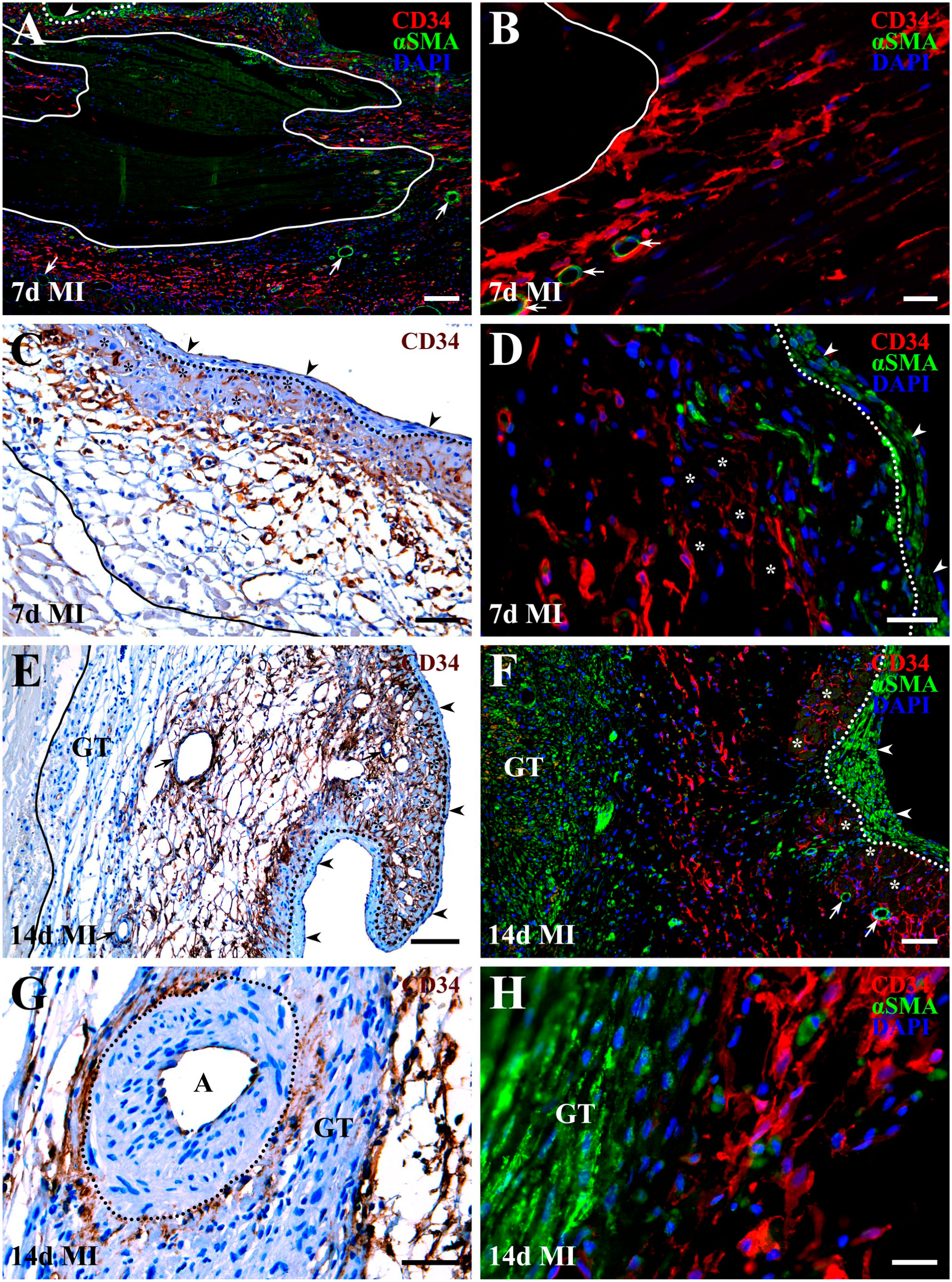
Spatial distribution of activated cardiac CD34+ SCs and developing myofibroblast-rich structures within the healing myocardial wound at 7 (A–D) and 14 (E–H) days following MI. (A, B, D, F, and H) Fluorescence micrographs showing the juxtaposed distribution of activated cardiac CD34+ SCs (red) and α-SMA+ myofibroblasts (green). Nuclei are counterstained with DAPI (blue). Note that α-SMA+ immunoreactivity is present in myofibroblasts accumulating within the subendocardial fibrotic thickening (area between dotted lines and arrowheads in A, D, and F) and within the granulation tissue (GT in F and H), as well as in the walls of residual coronary arteries/arterioles (arrows in A, B, and F), corresponding to vascular smooth muscle cells (vSMCs). (C, E, and G) Distribution of activated cardiac CD34+ SCs visualized using an HRP-conjugated secondary antibody and DAB substrate (brown). Nuclei are counterstained with Gill hematoxylin (dark blue). Gill hematoxylin also produces nonspecific, bluish staining of other tissue components, including necrotic cellular debris (degenerating structures to the left of the solid line in C and E), cytoplasm of surviving cardiac myocytes (asterisks in C and E), myofibroblasts within granulation tissue (GT in E and G) and subendocardial thickenings (area between the dotted lines and arrowheads in C and E), and vSMCs in the media of residual arteries (arrows in E and the region outlined by a dotted line around the arterial [A] lumen in G). Notably, at 7 days after MI, α-SMA+ myofibroblasts are not detected in regions typically associated with granulation tissue formation, including the transmural (A) and lateral border (B) regions adjacent to the necrotic core (areas outlined by solid lines in A and B). By 14 days after MI, α-SMA⁺ myofibroblasts become detectable only within the transmural portion of the infarct. Activated cardiac CD34+ SCs are consistently observed in areas of necrotic tissue resorption, particularly along preserved endomysial scaffolds (C and E) and within the interstitium surrounding surviving cardiac myocytes (asterisks in C–F) but remain absent from developing granulation tissue and from subepicardial and subendocardial fibrotic thickenings throughout the reparative phase. Importantly, CD34+ immunoreactivity appears reduced in cardiac CD34+ SCs located at the interface with developing α-SMA⁺ myofibroblast-rich structures (micrographs D, G, and H). Scale bars: 100 µm (A, E), 50 µm (C, F, and G) and 20 µm (B, D, and H).
Dedifferentiated Cardiac CD34+ SCs Seem to Contribute to the Growth of Myofibroblasts-Rich Structures During the Healing of Post-MI Wound
To assess the fate of cardiac CD34+ SCs in regions of myofibroblast-rich structure expansion, areas adjacent to granulation tissue (Fig. 9A) and subendocardial/subepicardial fibrotic thickenings (Fig. 9D) were examined. In these transitional zones, some cardiac CD34+ SCs adjacent to α-SMA+ myofibroblasts exhibited reduced CD34 expression, consistent with dedifferentiation, and appeared incorporated into myofibroblast-rich tissue (Fig. 9B, E). Importantly, the absence of pyknotic nuclei in these regions suggested that this process was not associated with overt cell death. Furthermore, as most cells within myofibroblast-rich regions were immunopositive for α-SMA (Fig. 9B, E) or vimentin (Fig. 9C, F), their findings support the possibility that a subset of dedifferentiated cardiac CD34+ SCs may acquire fibroblastic and/or myofibroblastic phenotype and contribute to post-MI scar formation.
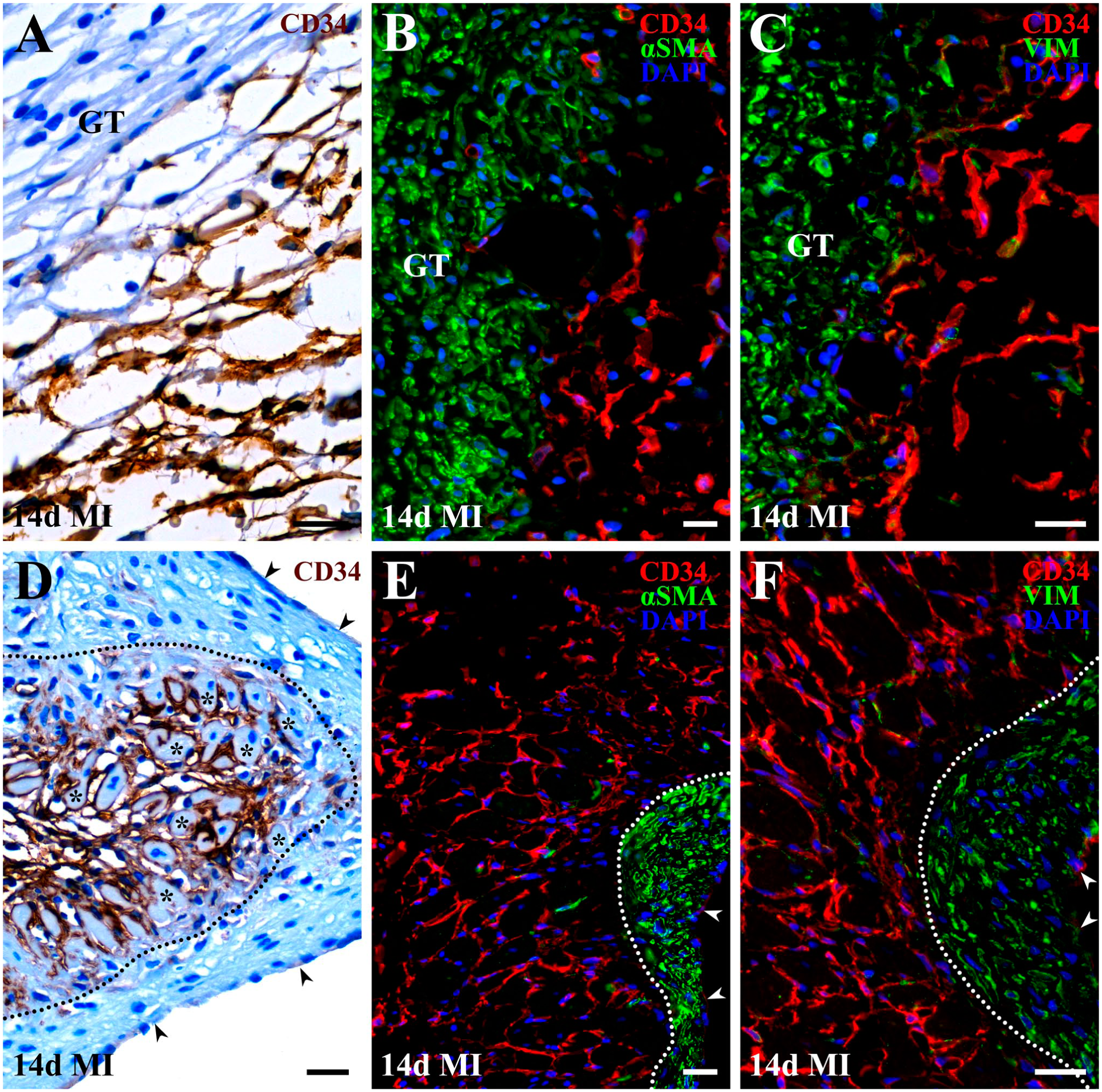
Spatial arrangement and structural features of activated cardiac CD34+ SCs in regions undergoing conversion into myofibroblasts-rich granulation tissue (A–C) and subendocardial fibrotic thickening (D–F) at 14 days after MI. (A and D) High-power micrographs showing activated cardiac CD34+ SCs visualized using an HRP-conjugated secondary antibody and DAB substrate (brown) along preserved endomysial scaffolds (A) and within the interstitium surrounding surviving cardiac myocytes (asterisks in D). Nuclei are counterstained with Gill hematoxylin. Gill hematoxylin also produces nonspecific, bluish staining of other tissue components, including myofibroblasts within granulation tissue (GT in A) and subendocardial fibrotic thickening (area between a dotted line and arrowheads in D), as well as the cytoplasm of surviving cardiac myocytes (asterisks in D). (B and E) High-power fluorescence micrographs showing the juxtaposed distribution of activated cardiac CD34+ SCs (red) and α-SMA+ myofibroblasts (green). (C and F) High-power fluorescence micrographs demonstrating the juxtaposed distribution of activated cardiac CD34+ SCs (red) and vimentin+ fibroblasts/myofibroblasts (green). In B, C, E, and F, nuclei are counterstained with DAPI (blue). Differences in α-SMA and vimentin immunostaining patterns within granulation tissue (GT) and subendocardial fibrotic thickenings (areas between dotted lines and arrowheads) are consistent with the presence of fibroblastic cells at different stages of differentiation. Importantly, activated cardiac CD34+ SCs located adjacent to α-SMA+ and/or vimentin+ fibroblasts/myofibroblasts-rich structures exhibit reduced CD34 immunoreactivity, indicative of phenotypic modulation. Moreover, the presence of cells lacking immunoreactivity for any markers used suggests a population of cells with an undetermined phenotype, potentially arising from cardiac CD34+ SCs which undergoing phenotypic changes consistent with dedifferentiation. Scale bars: 20 µm.
Activated Cardiac CD34⁺ SCs Accumulate at the Infarct Border and Mediate Stromal Integration Between the Developing Scar and Endomysial Tissue of the Non-Infarcted Myocardium
In contrast to their apparent disappearance within myofibroblast-rich regions, activated cardiac CD34+ SCs frequently accumulated at lateral infarct borders devoid of α-SMA+ myofibroblasts (Figs. 10A, B and 11A, B). Interestingly, expansion of the cardiac CD34+ SC population in these regions was closely associated with sites of newly deposited ECM enriched in fibrillar collagen (Figs. 10C–F and 11C–F). In many instances, these ECM deposits were located adjacent to truncated ends of surviving peri-infarct cardiac myocytes, suggesting that they may serve as anchoring interfaces between stromal components of the developing scar and the connective tissue framework of non-infarcted myocardium (Fig. 10G, H). Notably, these regions lacked a detectible population of α-SMA+ myofibroblasts (Figs. 11G, H and 12A) but contained numerous CD34−/α-SMA− cells (Figs. 10H and 11H), presumably activated fibroblasts, indicating that these cells are likely the principal contributors to ECM deposition at the infarct border. Concurrently, enlarged and interconnected cardiac CD34+ SCs formed loose cellular networks (Fig. 12B) that appeared interwoven with the newly deposited fibrillar matrix of the developing scar (Fig. 12C, D), consistent with their role as the spatial organizers of nascent ECM. Moreover, the extension of CD34+ SC cuffs from the truncated ends of surviving peri-infarcted cardiac myocytes toward the infarct border (Fig. 12B) further supports their involvement in structural integration of stromal elements between non-infarcted myocardium and the scar matrix. Finally, the presence of activated cardiac CD34+ SCs along microvessels extending from peri-infarcted myocardium into the developing scar (Fig. 12E, F) suggests a potential role for these cells in coordinating reparative angiogenesis and/or stromal-vascular remodeling during post-MI myocardial healing.
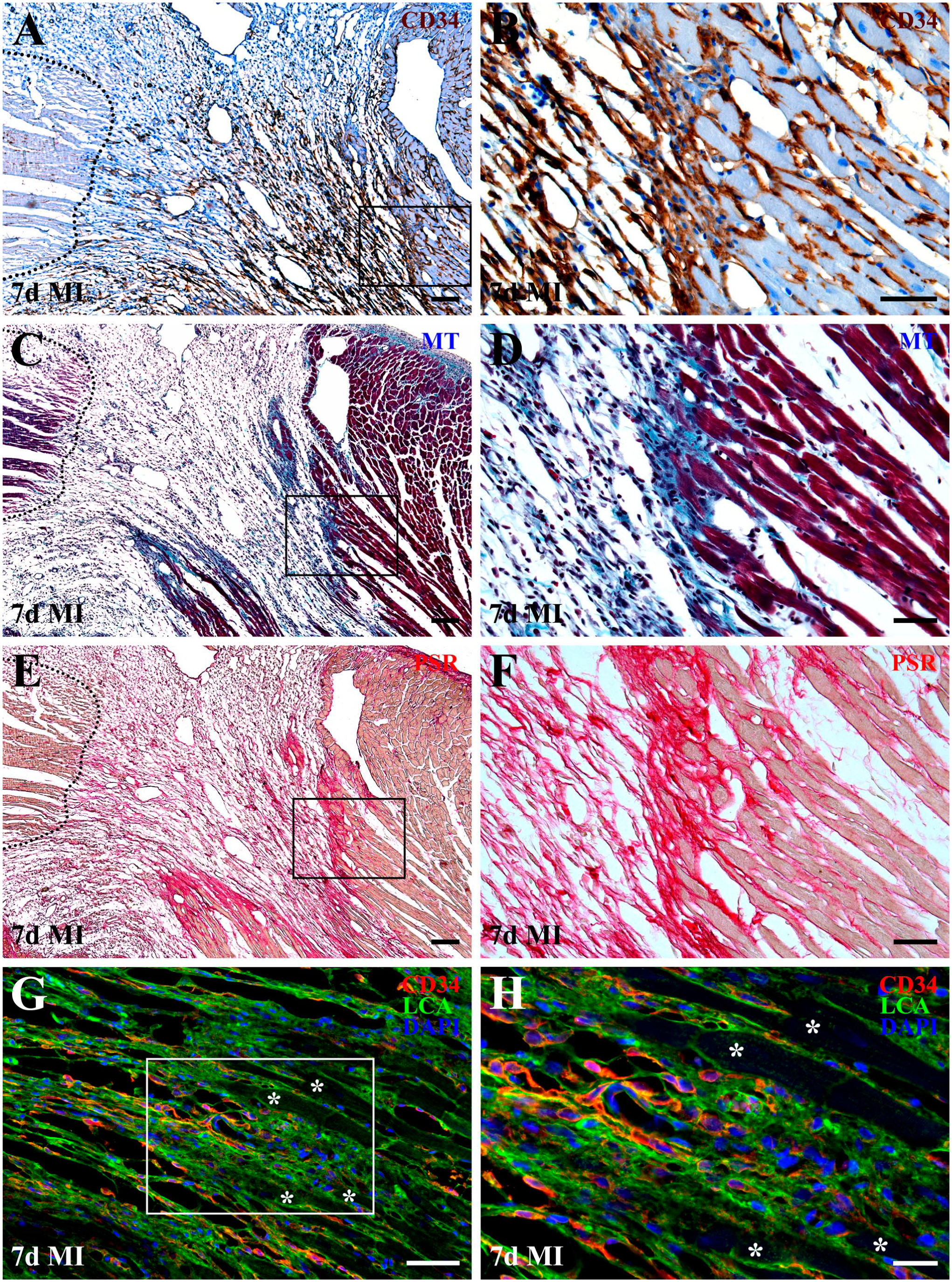
Spatial relationship between activated cardiac CD34+ SCs and newly deposited extracellular matrix (ECM) at the lateral infarct border 7 days after MI. Low-power micrographs (A, C, E, and G) show the same field of view within the healing post-MI region that is stained using different histological techniques on serial sections, whereas high-power micrographs (B, D, F, and H) depict selected regions from the corresponding low-power images (outlined by black boxes). In A, C, and E, dotted outlines delineate non-resorbed necrotic myocardium persisting within the healing area, while asterisks in G and H indicate profiles of corresponding cardiac myocytes. (A and B) Distribution of cardiac CD34+ SCs visualized using an HRP-conjugated secondary antibody and DAB substrate (brown). Importantly, activated cardiac CD34+ SCs are often observed around the stumps of surviving cardiac myocytes at the infarct border. (C and D) Spatial relationship of ECM components and peri-infarct cardiac myocytes visualized by Masson’s trichrome (MT) staining. (E and F) Distribution of fibrillar collagen near and around surviving cardiac myocytes revealed by picrosirius red (PSR) staining. Note that regions enriched in newly deposited collagen fibrils correspond spatially to areas containing accumulations of cardiac CD34+ SC, particularly near cardiac myocyte stump at the infarct border. (G and H) Spatial relationship between activated cardiac CD34+ SCs (red) and the connective tissue stroma of the developing scar and peri-infarct myocardium visualized using fluorescein-conjugated Lens Culinaris agglutinin (LCA, green). Nuclei are counterstained with DAPI (blue). Note that activated cardiac CD34+ SCs area distributed along connective tissue scaffolds bridging the endomysial framework of non-infarcted myocardium with stromal components of the developing scar. Moreover, at sites of newly deposited ECM adjacent to cardiac myocyte stumps (asterisks), activated cardiac CD34+ SCs are frequently juxtaposed with clusters of CD34-immunonegative cells, presumably fibroblasts, suggesting these regions may serve as anchoring points for stromal components of the developing scar. Scale bars: 100 µm (A, B, C, and E), 50 µm (D, F, and G), and 20 µm (H).
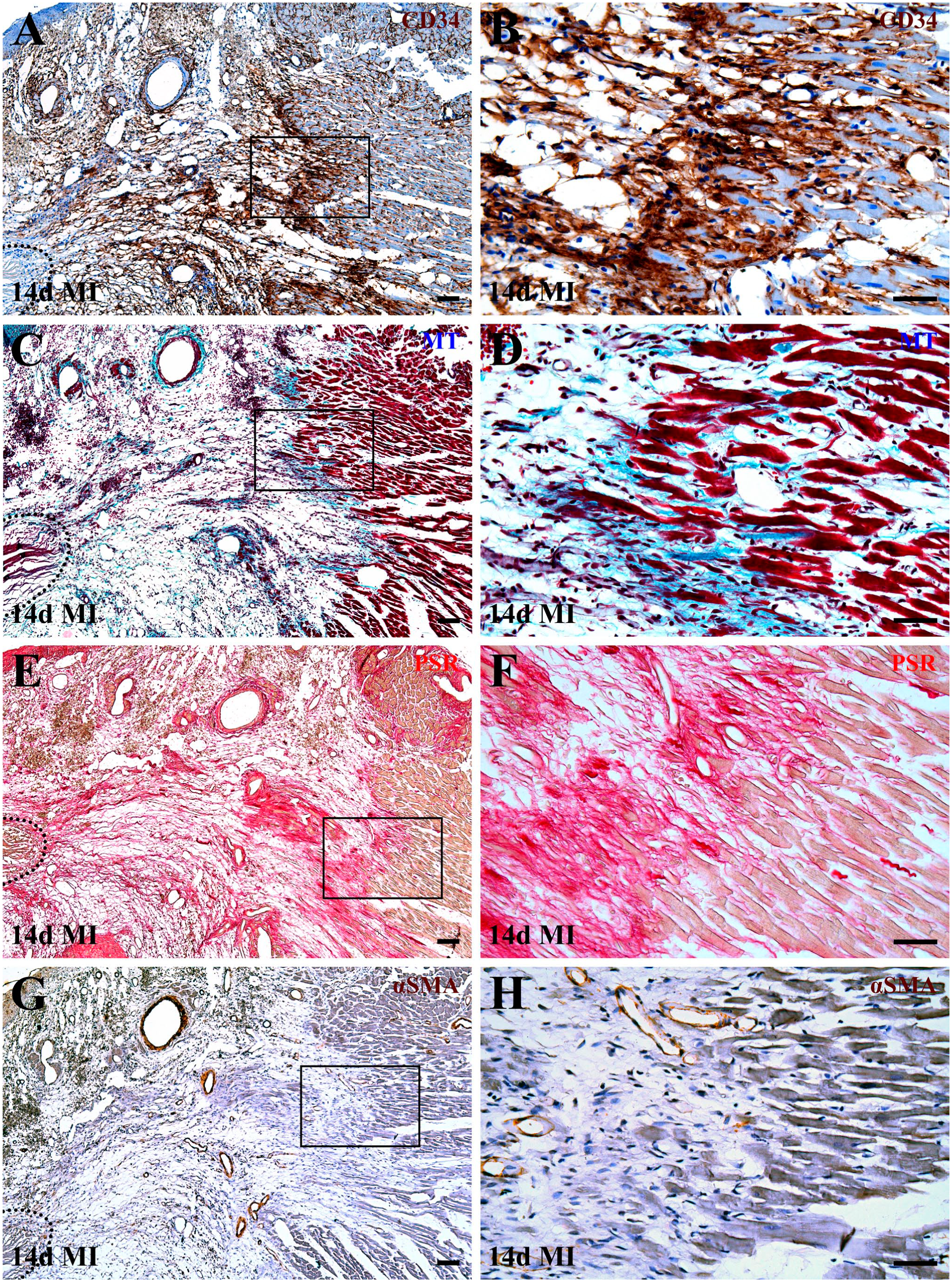
Spatial relationship between activated cardiac CD34+ SCs and the newly deposited ECM at the infarct border 14 days after MI. Low-power micrographs (A, C, E, and G) depict the same region stained using different histological techniques on serial sections, whereas high-power micrographs (B, D, F, and H) show selected areas from the corresponding low-power micrographs (outlined by black boxes). In A, C, E, and G, dotted outlines delineate remnants of necrotic myocardium. (A and B) Distribution of cardiac CD34+ SCs at the infarct border visualized with an HRP-conjugated secondary antibody and DAB substrate (brown). Note that the population of activated cardiac CD34+ SCs in this region appears markedly expanded compared to day 7 after MI (Fig. 10A, B). (C and D) Spatial arrangement of newly produced ECM and peri-infarct cardiac myocytes visualized by Masson’s trichrome (MT) staining. (E and F) Distribution of newly deposited collagen fibers near and around surviving cardiac myocytes revealed by picrosirius red (PSR) staining. Importantly, regions with expanded population of cardiac CD34+ SC at the infarct border coincide with the areas of progressive accumulation of newly deposited ECM rich in fibrillar collagen. (G and H) Spatial distribution of α-SMA+ cells within the infarct border zone. Notably, areas associated with newly deposited ECM near cardiac myocyte stumps lack α-SMA+ myofibroblasts, whereas α-SMA+ immunoreactivity is evident in subepicardial fibrotic thickening, containing myofibroblasts, and in arterial vessel walls, corresponding to vSMCs. Scale bars: 100 µm (A, C, E, and G) and 50 µm (B, D, F, and H).
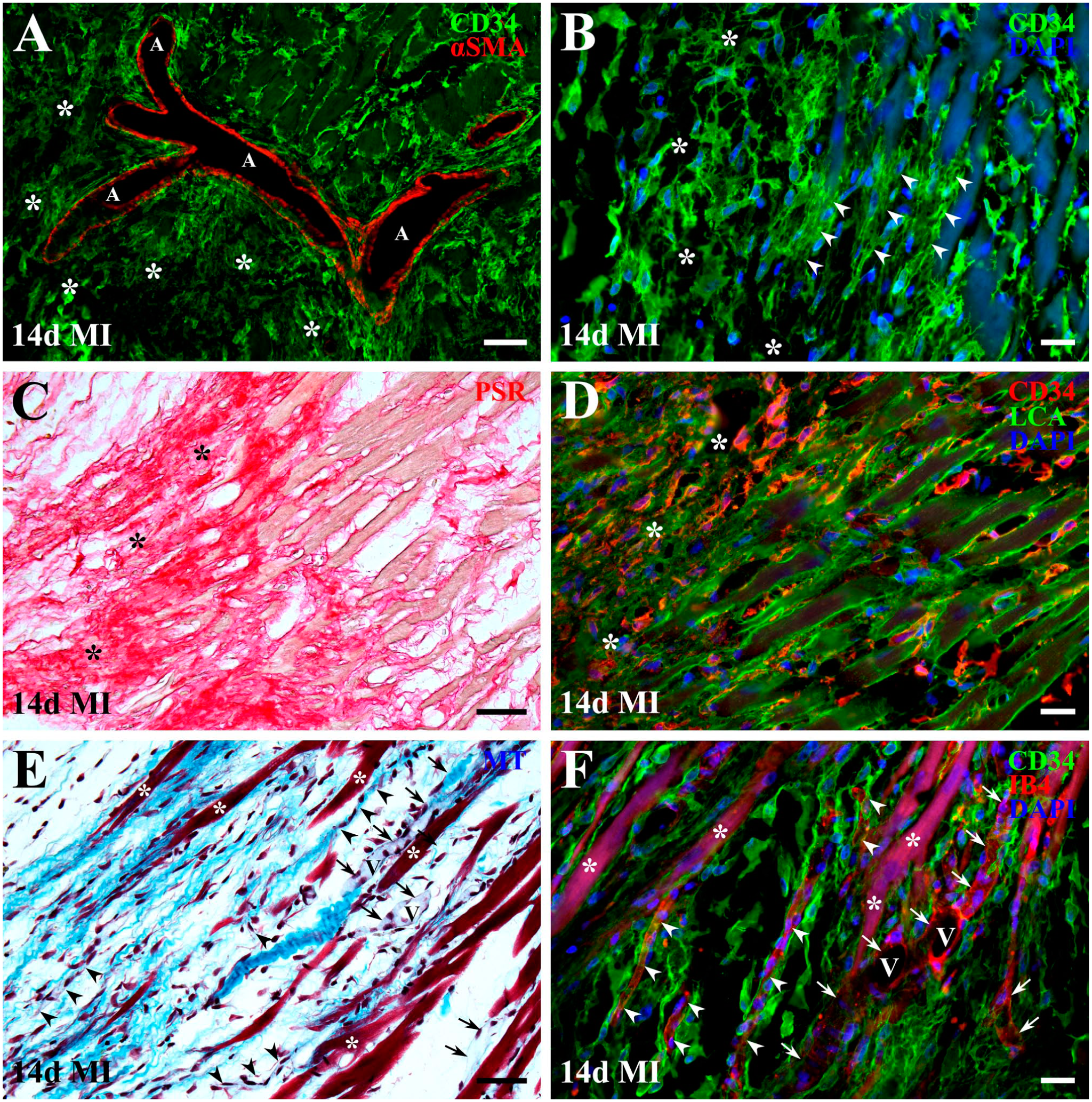
Structural appearance and spatial arrangement of activated cardiac CD34+ SCs at sites of stromal attachment between the developing scar and peri-infarcted myocardium 14 days after MI. In micrographs A–D, asterisks indicate the infarct border zone adjacent to non-infarcted myocardium, whereas in E and F, asterisks denote surviving cardiac myocytes; arrowheads mark residual capillaries, and arrows indicate venules (V). Nuclei are counterstained with DAPI (blue) in B, D, and F. (A) Juxtaposed arrangement of activated cardiac CD34+ SCs (green) and arterial (A) vessel walls containing α-SMA+ vSMCS (red) at the infarct border. Note that α-SMA+ myofibroblasts are absent in the region connecting the developing scar to non-infarcted myocardium. (B) Appearance and spatial arrangement of activated cardiac CD34+ SCs (green) near and around blunt-ended stumps of surviving cardiac myocytes. Importantly, at sites of newly deposited ECM (asterisks), cardiac CD34+ SCs display enlarged, flattened cell bodies with multiple processes forming an interconnect, loose cellular network. In contrast, cardiac CD34+ SCs surrounding cardiac myocyte stumps (bluish profiles) form continuous cuff-like ensheathments encircling the myocyte perimeter (arrowheads) that expand toward the infarct margin as multicellular cords. (C and D) Same area of the infarct border showing newly produced fibrillar collagen deposits stained with picrosirius red (PSR; C) and the spatial arrangement of activated cardiac CD34+ SCs (red) with LCA+ connective tissue stroma (green; D). Importantly, the interwoven arrangement of CD34⁺ SC bodies and processes with the components of newly deposited ECM suggest their involvement in organizing the connective tissue stroma at the infarct border. (E and F) Same field of view at the infarct border showing vascularized ECM deposits visualized by Masson’ trichrome (MT; E) and the spatial relationship of cardiac CD34+ SCs (green) with IB4+ endothelial cells (red) of residual or newly formed microvessels, including capillaries (arrowheads) and venules (V and arrows; F). Note that activated cardiac CD34+ SCs align along microvessels, suggesting a role in structural integration between developing scar stroma and ingrowing vessels. Note that in panel F, nonspecific fluorescence from DAPI and Alexa Fluor 594-conjugated IB4 lectins produces a magenta background in surviving cardiomyocytes. Scale bars: 50 µm (A, C, and E) and 20 µm (B, D, and F).
Discussion
The principal findings of this study are as follows: (1) the intact myocardium of middle-aged Sprague-Dawley rats contains a population of cardiac, non-endothelial CD34+ SCs residing within the interstitial connective tissue and the adventitia of coronary vessels; (2) following a large, transmural, nonreperfused MI, activated cardiac CD34+ SCs originating from the peri-infarct myocardium progressively expand throughout regions of necrotic tissue resorption during the early reparative phase via proliferation and migration; (3) to facilitate dissemination across the post-infarct region, activated cardiac CD34+ SCs utilize preserved endomysial connective tissue scaffolding left intact by phagocytic macrophages during wound clearance; (4) during later stages of repair process, intramural regions containing activated cardiac CD34+ SCs are progressively reduced by expansion of myofibroblast-rich structures, including developing granulation tissue and fibrotic thickenings of the subepicardial and subendocardial layers; (5) a subset of activated cardiac CD34+ SCs located at interfaces with myofibroblasts-rich structures appears to lose their CD34+ expression, consistent with cell dedifferentiation, and may contribute to fibrotic outgrowths; and (6) activated cardiac CD34+ SCs frequently accumulate at lateral infarct borders, surrounding the residual and newly formed vessels, and the stumps of truncated cardiac myocytes embedded in newly deposited ECM, suggesting a role in regulating structural coupling between the endomysial connective tissue of surviving myocardium and the stroma of the developing scar.
Although the presence of non-endothelial CD34+ SCs in the hearts of various mammalian species, including humans, has been well-documented over the past two decades,14,17,28 their recognition as a distinct population of resident cells within the myocardial interstitium has been repetitively overlooked in recent studies describing the cellular composition of the myocardial stroma.15,16,22–25 Moreover, despite multiple reports demonstrating the involvement of cardiac CD34+ SCs in myocardial repair,26–28 these cells are not explicitly addressed in latest reviews outlining the phases of post-MI wound healing.8,15,29 Surprisingly, even studies that proved a critical role for cardiac CD34+ SCs in ischemia-induced myocardial repair26–28,34 have provided limited insight into their dynamic behavior within the healing myocardial wound. Consequently, the present study is the first to offer a comprehensive characterization of the spatiotemporal dynamics of cardiac CD34+ SCs throughout the reparative/proliferative phase of post-ischemic wound healing following a large, transmural, nonreperfused MI in middle-aged rats.
Nature of Non-Endothelial CD34+ Cells in the Adult Myocardial Stroma
Our findings confirm the presence of substantial population of cardiac, non-endothelial CD34+ cells within the intact myocardial stroma of middle-aged Sprague-Dawley rats, occupying both interstitial and adventitial compartments. While this observation is consistent with prior reports across mammalian species, including humans,19,49–52 the precise identity of these cells remains the matter of ongoing debates.31,49 Whereas some studies have suggested that these cells could originate from a pool of circulating, bone-morrow-derived CD34+ fibrocytes, 50 others inclined to regard them as a subset of either cardiac pericytes 52 or cardiac telocytes.26,27,49,51,53
According to the present study (Figs. 1 and 2), the perivascular distribution of cardiac CD34+ SCs along capillary walls, as well as their spatial relationship to the endothelial cell basement membrane, differed from that typically observed for cardiac pericytes.54,55 Moreover, given that circulating CD34+ fibrocytes are typically recruited to peripheral tissues only during fibrosing disorders or aberrant wound repair, 56 it is unlikely that they account for the large population of resident cardiac CD34+ SC observed in the intact myocardium of middle-aged rats. Instead, the morphological features and tissue distribution of these cells closely resemble those previously described in the heart and other organs, initially as resident CD34+ stromal fibroblastic/fibrocytic cells 21 and later classified as CD34+ SCs/telocytes. 57 However, despite some similarity between cardiac CD34+ SCs observed here and features typical of cardiac telocytes,19,58 we deliberately avoided designating these cells as “telocytes” due to the absence of additional immunohistochemical markers and ultrastructural characteristics considered essential for definitive identification of this myocardial cell type. 17 Nonetheless, our findings provide sufficient evidence to challenge the pericytic and fibrocytic interpretations of cardiac CD34+ SCs while supporting their close association with the cardiac telocyte lineage.
Regenerative Versus Pro-Fibrotic Role of Cardiac CD34+ SCs During Post-MI Repair
Although numerous studies over the past several decades have methodically delineated the spatiotemporal sequence of post-MI healing events, the precise role of cardiac CD34⁺ SCs in this highly orchestrated process has remained incompletely understood.17,28 While some reports suggested that ischemia-induced death of these cells contributes to the replacement of highly structured endomysial stroma with disorganized fibrotic scar tissue,26,27 others provided evidence that CD34⁺ SCs may directly contribute to ischemia-associated myocardial fibrosis. 28 In contrast, we observed that following ischemia-induced cell loss in middle-aged Sprague-Dawley rats, a population of cardiac CD34⁺ SCs were recovered within the healing post-MI region via proliferation and migration. Notably, their distribution closely mirrored the spatial pattern of preserved endomysial scaffolds, including ECM components of cardiac myocyte basement membranes left intact by phagocytic macrophages after clearance of necrotic debris (Figs. 4 and 6). In this regard, it is important to highlight that although the persistence of stromal architecture following coagulative necrosis is a well-established fact in both cardiac43,59 and skeletal60,61 muscles, this study is the first to demonstrate that preserved endomysial scaffolds in the post-infarcted myocardium can be repopulated by cardiac CD34+ SCs derived from the border zone. Furthermore, because preservation of basement membranes and endomysial connective tissue is required for orderly regeneration of muscle fibers by myogenic precursors in skeletal muscle,60,61 it is reasonable to hypothesize that the mammalian heart might similarly regenerate functional myocardium in place of fibrotic scar if appropriate myocyte precursors were available. This concept is further supported by observations in species capable of scar-free cardiac regeneration, such as the African clawed frog (Xenopus tropicalis), in which cardiac telocytes—interstitial cells analogous to rat cardiac CD34+ SCs—repopulate preserved endomysium to reestablish the stromal network around newly regenerated cardiac myocytes. 62 In adult mammals, however, the absence of cardiac myocyte regeneration results in disruption of basement membranes and endomysial scaffolds, which subsequently become enveloped by developing fibrotic tissue. 59 Consistent with this, in middle-aged rats, we observed that replacement of the original cardiac stromal framework with granulation tissue led gradual disappearance of cardiac CD34+ SCs from these regions. Nonetheless, the preferential migration of cardiac CD34+ SCs into areas occupied by preserved endomysial scaffolds, in contrast to fibrotic regions, suggests that these cells are predisposed to support regenerative rather than fibrotic repair by maintaining stromal architecture characteristic of functional myocardium.
Contribution of Cardiac CD34+ SCs to Fibroblast and Myofibroblast Populations During Post-MI Repair
Activated fibroblasts and myofibroblasts are widely recognized as the principal cell types responsible for granulation tissue formation and expansion in the post-MI heart.8,15 Moreover, it has been demonstrated that the majority of fibroblasts/myofibroblasts contributing to the developing post-MI scar are derived from resident fibroblasts of the peri-infarct myocardium.63–66 In contrast, our observations indicate that regions cleared of necrotic debris by phagocytic macrophages are concurrently repopulated by two distinct cell populations: cardiac CD34⁺ SCs and CD34⁻ fibroblasts/myofibroblasts. Although initially colocalized in the infarct border zone, these populations subsequently segregate spatially, with cardiac CD34+ SCs occupying preserved endomysial scaffolds, whereas myofibroblasts accumulate at classical pro-fibrotic sites (Figs. 3, 8, and 9). This spatial distribution is consistent with previous reports demonstrating that myofibroblast-rich granulation tissue preferentially emerges at defined locations within the post-MI region, including areas surrounding the remnants of non-resorbed cardiac myocytes, the vicinity of large residual coronary arteries, and the ventricular wall compartments adjacent to the endocardial and epicardial linings.67–69 Importantly, our data further demonstrates that as α-SMA+ myofibroblast-rich granulation tissue expands from these pro-fibrotic foci, it progressively incorporates endomysial connective tissue regions initially populated by cardiac CD34+ SCs, suggesting that these cells may partially support fibrotic tissue growth within the post-MI myocardium. At these interfaces, a subset of cardiac CD34+ SCs exhibited loss of CD34 expression and became incorporated into the expanding fibrotic tissue (Figs. 8 and 9), predominantly as morphologically undifferentiated cell. This observation favors a process of cellular dedifferentiation rather than direct conversion into fibroblasts or myofibroblasts. Furthermore, in contrast to previous studies reporting that tissue-resident CD34+ SCs serve as a substantial source of myofibroblasts during fibrotic repair in other organs,70,71 direct differentiation of cardiac CD34+ SCs into α-SMA+ myofibroblasts was only rarely observed in the present study. Collectively, these findings indicate that cardiac CD34+ SCs are unlikely to represent a major source of ECM-producing myofibroblasts during post-MI wound repair, although they may indirectly contribute to granulation tissue formation and expansion.
Role of Cardiac CD34+ SCs in Structural Integration at the Infarct Border
Prominent accumulation of cardiac CD34+ SCs was observed at interfaces between viable myocardium and the developing scar, particularly around truncated cardiac myocyte stumps and residual or newly formed coronary vessels (Figs. 10–12). Because these sites are known to undergo extensive remodeling that leads to the formation of myotendinous-like junctions anchoring endomysial connective tissue, including basement membranes, of surviving cardiac myocyte to the collagenous stroma of the post-MI scar,72,73 the preferential accumulation of cardiac CD34+ SCs at these interfaces suggests a role in coordinating stromal integration between functional myocardium and scar tissue. In addition, enrichment of cardiac CD34+ SCs along microvessels extending into the infarcted region supports their potential involvement in integrating vascular and stromal compartments during post-MI healing. This interpretation is consistent with prior evidence indicating that structural incorporation of residual coronary vessels into the post-MI region is essential for effective granulation tissue formation and myocardial wound repair. 74
Study Limitations
This study has several limitations. The absence of genetic lineage tracing precluded definitive delineation of cardiac CD34+ SCs from other CD34-expressing cell lineages and prevented fate mapping of these cells following loss of CD34 expression and acquisition of alternative immunophenotypes. Technical constraints arising from differences in antigen-retrieval requirements among primary antibodies limited multiplex immunolabeling and necessitated partial reliance on less-specific lectin-based histochemistry. Functional manipulation of cardiac CD34+ SC migration or proliferation was not feasible in the present study, limiting the ability to directly assess their role in post-MI wound healing. Finally, because analyses were restricted to the reparative/proliferative phase, the study does not address the fate or function of cardiac CD34+ SCs during scar maturation and remodeling.
Conclusions
Collectively, our findings demonstrate that following a large, transmural, non-reperfused MI in middle-aged Sprague-Dawley rats, activated cardiac CD34+ SCs expand into necrotic regions through proliferation and migration, preferentially repopulating preserved endomysial scaffolds. Their spatial segregation from pro-fibrotic fibroblast and myofibroblast populations suggests a predisposition toward regenerative rather than fibrotic repair. Although transmural expansion of α-SMA+ myofibroblast-rich tissue ultimately incorporates some dedifferentiated CD34+ SCs into the scar, persistent CD34+ SCs at the lateral infarct borders appear to facilitate integration of myocardial stroma and coronary vasculature into the developing scar. These observations further support the recognition of cardiac CD34+ SCs—likely cardiac telocyte-related cells—as key contributors to post-MI repair26,75 and potential targets for regenerative therapeutic strategies.
Footnotes
Acknowledgements
During preparation of this manuscript, ChatGPT (OpenAI, GPT-5 mini) was used for language polishing and refinement. All AI-generated content was reviewed and edited by the authors, who take full responsibility for the accuracy and integrity of the final text.
Competing Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
EID conceptualized, designed, and conducted the experiments; collected, analyzed, and interpreted the data; designed and assembled the figures; wrote and revised the manuscript. DTS collected and analyzed data and contributed to manuscript revision.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the Camden Health Research Initiative (CHRI) award from Rowan University.
