Abstract
Summary
Tissue preparation for paraffin embedding is a crucial step in histological processes. Standardized methods are required to ensure the accuracy of research and clinical diagnostic results. However, standardization is particularly challenging for long luminal tissues. Conventional methods such as single/serial sections and the Swiss roll, often have drawbacks including the risk of missing or misaligned sections, excess consumption of materials, and high workload. They also require significant expertise and are difficult to standardize. To address these issues, we developed the Histo-LOOP embedding tool—a novel tool designed to standardize, simplify, and improve histological processing. Histo-LOOP is suitable for various tissue types including long tubular tissue, allowing for a complete overview in cross-sectional and longitudinal views. It is also suitable for punch biopsies or small sections, and enables the assessment of multiple punch biopsies or sections within one paraffin block, and in multiple cutting planes, for example, for liver and prostate biopsies. Histo-LOOP does not interfere with the sectioning and staining process and does not cause artifacts.
Here, we introduce the novel tool Histo-LOOP and describe preparation techniques for tubular tissue and small tissue samples using this tool, along with examples of their histological evaluation.
Keywords
Histopathological evaluation of tissue specimens is essential for both research and diagnostic purposes. Tissue processing involves multiple steps including sample preparation, paraffin embedding, sectioning with a microtome, staining, mounting, and microscopic analysis.1,2 Paraffin embedding is a standard technique for a complete enclosure of tissue specimens in a firm, supporting medium to facilitate cutting thin sections as a prerequisite for staining, mounting, and microscopic analysis. The preparation of tissue for paraffin embedding is a critical step, as incorrect tissue embedding could jeopardize research data and the overall outcome. Histopathological processing of tubular structures, such as vascular or intestinal tissue can be particularly challenging. Firstly, the wall of tubular constructs usually consists of several tissue layers. Therefore, specific tissue orientation is necessary to visualize the desired layers, and incorrect orientation of the tissue may result in diagnostically important elements being missed or damaged during sectioning. Secondly, some tubular structures such as vessels or intestine are too long to be embedded in one single paraffin block, and standardized sample processing can be challenging.
Various techniques have been used to prepare small luminal tissues and organs for paraffin embedding. While short tubular structures can be embedded as singular specimens, longer or tortuous tissue samples have to be cut in serial sections. Subsequent sections of one tubular tissue structure can either be embedded in multiple blocks, which, however, requires lots of consumables and materials. Alternatively, multiple tissue sections can be embedded in one single paraffin block. This method, however, can be challenging because single tubular segments might easily be misaligned in paraffin. In addition, there is a risk of swapping the pieces or tipping over of pieces before the paraffin hardens. Furthermore, it can be challenging to keep all pieces in the right order. These issues also arise in histological processing of punch biopsies.
The Swiss rolling technique was first introduced by Magnus in 1937 3 and further refined by Moolenbeck and Ruitenberg 4 and Park et al. 5 as a processing method for the rodent intestine. Allowing for a complete longitudinal visualization of the intestine, all portions of the intestine can be captured within one slide. However, this technique requires expertise to avoid tissue overlap or non-aligned coils, resulting in missing tissue segments. Some protocols, however, require longitudinal opening of the intestine before rolling, resulting in potential tissue loss and artifacts due to the manipulation. Moreover, the conventional Swiss Roll technique is not suitable for completely fixed tissue due to its stiffness, making it difficult to form a Swiss Roll. 6 Previous studies have reported tissue tears after processing tissue fixed in formalin or with Bouin’s solution. 7
Thus, conventional techniques for the preparation of luminal tissue for paraffin embedding have several drawbacks. To overcome these limitations, we developed the Histo-LOOP, a novel tool to standardize, simplify and improve the processing and histological evaluation of tubular and longitudinal tissues. In addition, Histo-LOOP can be used for punch biopsies or small specimens with reliable and reproducible production of a multi-block for evaluation of multiple punch biopsies or in several cutting planes in one paraffin block. This can be particularly useful, for example, for liver and prostate biopsies. Misalignment and mix-ups can be avoided, and multiple biopsies can be analyzed simultaneously.
Here, we describe preparation techniques for histological evaluation of tubular tissue structures using the novel Histo-LOOP in three different manners. We show how to create cross-sectional and longitudinal sections of tubular structures using (1) spiral and (2) serpentine tissue wrapping and give an overview of the preparation of small tissue samples and punch biopsies using Histo-LOOP.
In many cases tissues are not processed immediately after dissection but are preserved in fixative solutions. These solutions prevent the decomposition or autolysis of the tissue but may alter tissue properties by causing tissue hardening or brittleness. We therefore assess the performance of Histo-LOOP in the preparation of non-fixed tissue and tissue fixed in formalin, Bouin’s, and Carnoy’s solutions. Moreover, we evaluate whether two different materials from which the Histo-Loop is made, i.e., sponge and elastomer, may affect the fixation process and thus the histological staining quality.
Materials and Methods
Tissue Specimens
Exemplary for tubular tissues, rat and mouse intestine and human mammary artery specimens were used for tissue preparation with Histo-LOOP. Human mammary arteries were obtained from the official body donation program of the Institute of Molecular and Cellular Anatomy—Prosecture, University Hospital RWTH Aachen. An ethic votum from the ethic committee was not required, as each person in this program has provided written informed consent for their body to be used for medical education and research by signing a personal will. Residual tissue of rat and mouse intestines were obtained from approved animal experiments conducted for scientific purposes in accordance with § 4 para. 3 of the Animal Protection Act of 18 May 2006 in the currently valid version.
Tissue Fixation
Human mammary arteries (n=2) were obtained as fresh specimens. One was processed as non-fixed tissue and one was fixed in formalin for 24 hours before being prepared with Histo-LOOP. A rat intestine (n=1) was fixed in formalin prior to tissue preparation. Mouse intestines (n=3) were received as fresh specimens; one remained unfixed, while the other two were fixed in Bouin’s and Carnoy’s solution, respectively, for 24 hours before tissue preparation.
Histo-LOOP Embedding Tool
Histo-LOOP was designed using Autodesk Inventor software (3D-CAD-Software Inventor) (Fig. 1A) and manufactured at the Scientific Workshop of the University Hospital RWTH Aachen. To enable proper fixation and positioning of tissue before paraffin embedding with defined and standardized distances between tissue cutting planes, Histo-LOOP was designed with equispaced recesses and notches at the top and bottom edges (Fig. 1B).

Histo-LOOP (A) 3D Computer-Aided Design of a small size Histo-LOOP (left). (B) photomicrograph of a small size Histo-LOOP (right), scale bar indicates 1 cm.
Longer luminal tissue can be placed within the recesses and secured by closed notches in two different ways: The tissue can be wrapped (1) spirally or (2) serpentine around the device (Fig. 2A and B). Spiral wrapping allows for cross-sectional views on the tissue, whereas serpentine wrapping results in longitudinal views. Punch biopsies or small sections can be fixed within the Histo-LOOP recesses (Fig. 2C), enabling tissue visualization in a cross-sectional view.

Schematic drawing of tissue preparation using Histo-LOOP in different manners. (A) spiral winding of luminal tissue within Histo-LOOP recesses for a cross-sectional view. (B) serpentine winding of luminal tissue within Histo-LOOP recesses for a longitudinal view. (C) small sections/punch biopsies fixed within Histo-LOOP recesses for a cross-sectional view.
Histo-LOOP is available in various sizes and materials, depending on sample size and requirements. Examples include a small Histo-LOOP with nine recesses (3 cm long, 1.7 cm wide) and a larger Histo-LOOP with five recesses (4 cm long, 2.3 cm wide). Histo-LOOPs can be injection-molded from elastomer. Allowing greater flexibility and easier sectioning, Histo-LOOPs can also be manufactured from sponge using a punch-out technique, which is particularly advantageous for spiral tissue wrapping and punch biopsies.
Histo-LOOP is registered at the German Patent and Trademark Office (DPMA) under the following number: DE 10 2020 119 764.
Tissue Preparation With Histo-LOOP Embedding Tool
Spiral Wrapping of Tubular Tissue for a Cross-Sectional View
To obtain a cross-sectional view, a specimen of human mammary artery was wound and pulled in a spiral through the Histo-LOOP recesses (i.e., diagonally from a recess on the upper left to a nearby recess on the lower right) and secured with closed notches (Fig. 3A). Histo-LOOP-secured tissue was then transferred to a standard embedding cassette and infiltrated with liquid paraffin (Fig. 3B). Following paraffin infiltration, a cut was made in the longitudinal midline of the Histo-LOOP (Fig. 3C). The cut side of one half was placed facing down into the mold filled with liquid paraffin (Fig. 3D). Paraffin embedding and histological staining were performed following a standard protocol 2 (Fig. 3E).

Preparation of human mammary arterty using Histo-LOOP for cross-sectional view. (A) Human mammary artery wrapped around the elastomer Histo-LOOP in a spiral manner (small size; 9 recesses, 3 cm length, 1.7 cm width). (B) Histo-LOOP after paraffin infiltration. (C) Paraffin-infiltrated Histo-LOOP after longitudinal midline cut. (D) the cut edges placed facing down into the histology mold filled with liquid paraffin. (E) Histo-LOOP-secured tissue embedded in a paraffin block. (F) Paraffin section mounted on glass slide: cross-sectional view of human mammary artery stained with H&E. All scale bars indicate 1 cm.
Serpentine Wrapping of Tubular Tissue for a Longitudinal View
For a longitudinal view, human mammary artery was wound and pulled through the Histo-LOOP recesses in a serpentine manner and secured by closed notches (Fig. 4A). The Histo-LOOP was then cut in halves longitudinally along the midline. One half of Histo-LOOP without tissue was removed. The other half of Histo-LOOP, which held the tissue was transferred to an embedding cassette and further processed according to a standard paraffin embedding protocol. 2 For longitudinal tissue sections, Histo-LOOP was placed in an upright position in the histology mold for production of the paraffin block (Fig. 4B).

Preparation of formalin-fixed human mammary arterty using Histo-LOOP for longitudinal view. (A) Human mammary artery, wound through the elastomer Histo-LOOP recesses in a serpentine manner (large size; 5 recesses, 4 cm length, 2.3 cm width). (B) Histo-LOOP-secured tissue positioned in an upright position in the paraffin embedding mold. (C) Longitudinal view of human mammary artery (H&E-stained section, mounted on a glass slide). All scale bars indicate 1 cm.
Processing of Punch Biopsies and Smaller Specimens
For processing of punch biopsies and smaller specimens, a Histo-LOOP made of sponge material was used. Small pieces of formalin-fixed rat intestine were positioned vertically into the Histo-LOOP recesses (Fig. 5A). Before transferring the sample to a standard cassette for paraffin embedding, the correct alignment of all tissue pieces was ensured. For cross-sections, Histo-LOOP was aligned horizontally in the histology mold for the production of the paraffin block (Fig. 5B).

Preparation of small sections of formalin-fixed rat intestine using Histo-LOOP for cross-sectional view. (A) Sections of rat intestine, positioned in sponge Histo-LOOP (large size; 5 recesses, 4 cm length, 2.3 cm width). (B) Paraffin-infiltrated Histo-LOOP placed into the histology mold filled with liquid paraffin. (C) Paraffin section of rat intestine mounted on glass slide, cross-sectional view of rat intestine stained with H&E. All scale bars indicate 1 cm.
Tissue Preparation Using Conventional Techniques—The Swiss Roll
The Swiss Roll technique is primarily used for the preparation of fresh intestinal tissue or for immediate tissue fixation by perfusion but not for tissue that has already been completely fixed beforehand6,7; therefore, a non-fixed mouse intestine was used as an example. An independent experienced experimenter performed the technique by dissecting the intestine and preparing it for paraffin embedding. Only the part from the duodenum to the ileum was used. Without cutting open the tissue, it was rolled around a stick from the duodenum to the ileum, creating the “Swiss Roll” shape. The tissue was then removed from the stick and placed in the embedding cassette before being transferred for paraffin embedding (Fig. 6).
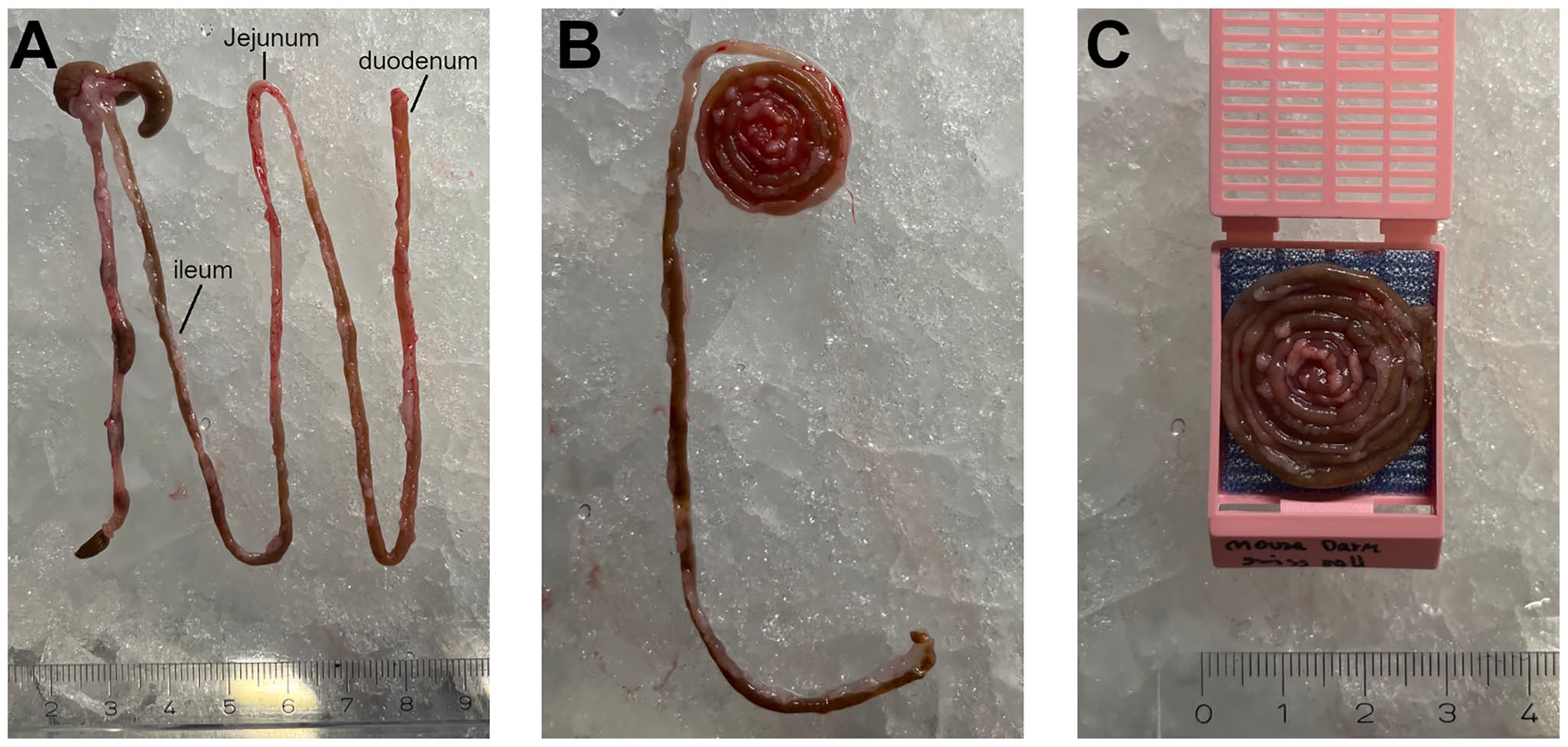
Preparation of tissue using the Swiss Roll technique. (A) Non-fixed mouse intestine oriented from the ileum (left) to the duodenum (right). (B) Mouse intestine rolled from the duodenum (inside) to the ileum (outside). (C) The Swiss Roll inside the embedding cassette.
Paraffin Embedding
After the tissue was positioned in the Histo-LOOP or in the Swiss Roll, all samples were subjected to a standardized processing protocol, which included dehydration using alcohol with increasing gradation, clarification with xylene, and paraffin-embedding. 2 Subsequently, the specimens were sectioned to 3–5 µm using a Leica RM 2155 microtome (Leica, Wetzlar, Germany). All specimens were stained with Hematoxylin & Eosin (H&E) (Dako REA, Santa Clara, CA), except for one sample of a human mammary artery, which was stained with Elastica van Gieson staining (EVG). Stained sections were examined with a light microscope (Leica DMI-3000, Wetzlar, Germany).
Results
Tissue Alignment Using Histo-LOOP
Tissue samples processed with Histo-LOOP were aligned exactly as planned. Spiral wrapping of the human mammary artery around Histo-LOOP resulted in cross-sectional views (Fig. 7A), while serpentine wrapping produced longitudinal views (Fig. 7B), as desired. Punch biopsies of rat intestine appeared as cross-sections (Fig. 7C).
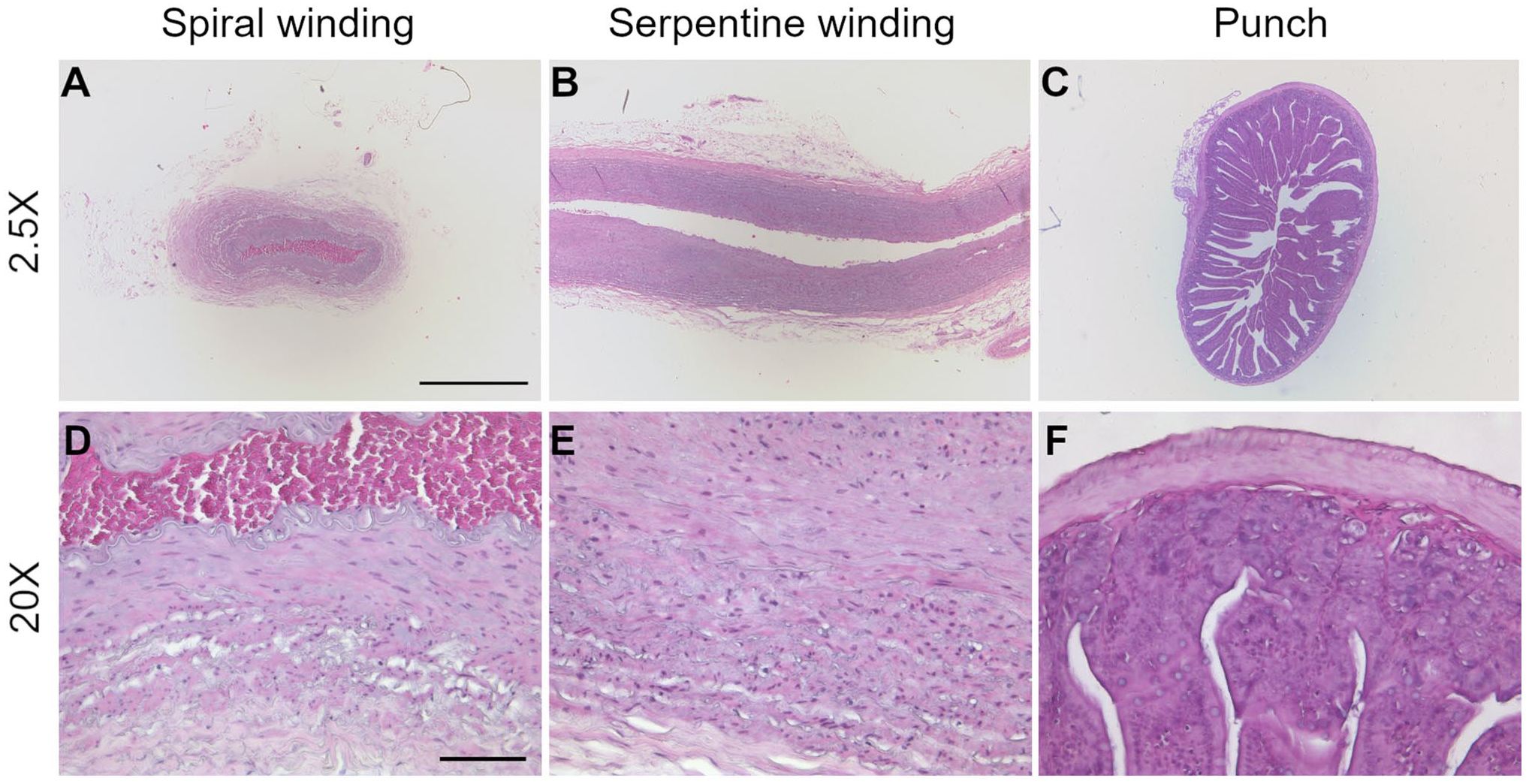
Histological images of tissue prepared with Histo-LOOP, H&E stained. (A and D) microscopical cross-sectional view of human mammary artery. (B and E) microscopical longitudinal view of human mammary artery. (C and F) microscopical cross-sectional view of rat intestine. (A, B and C): 25× magnification, scale bar indicates 1000 µm. (D, E, and F): 200× magnification, scale bar indicate 100 µm.
Figure 8 presents histological images of mouse intestine prepared using the Swiss Roll technique. While different segments of the intestine can be visualized longitudinally within a single section (Fig. 8A), the inner part of the “Swiss Roll” reveals a missing segment of the duodenum (Fig. 8B).
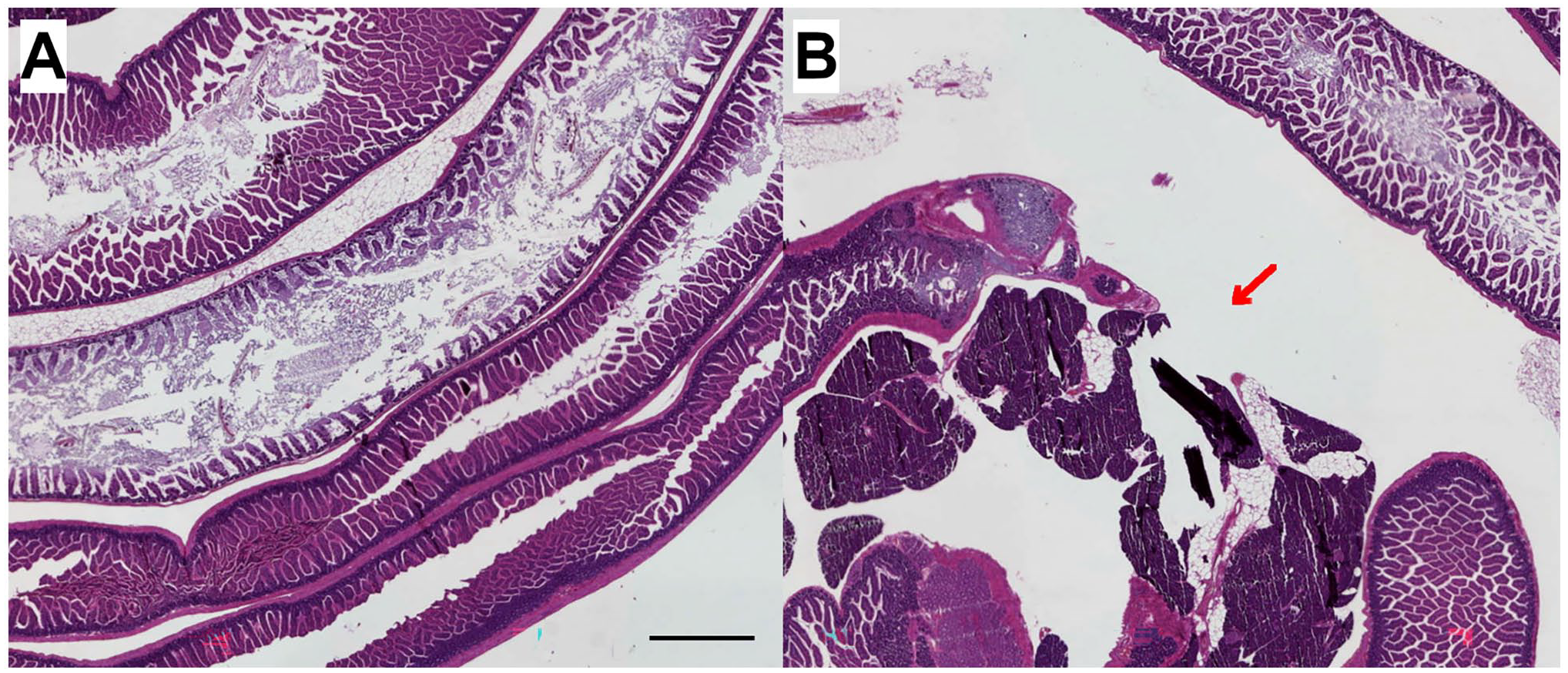
Histological images of mouse intestine prepared using the Swiss Roll technique; 20× magnification, scale bars indicate 1000 µm. (A) Segments of the mouse intestine in longitudinal view. (B) Missing segment of mouse intestine (red arrow). Image contrast and brightness were enhanced and applied to the entire image without altering the interpretation of the original image.
Effect of Fixatives on Tissue Preparation With Histo-LOOP
Fixation with formalin, Carnoy’s, or Bouin’s solution led to stiffening of the tissue. Still, both the elastomer and the sponge Histo-LOOPs enabled formalin-fixed and Carnoy-fixed tissue preparation in a similar way to non-fixed tissue, without fractures occurring (Figs. 4, 5, and 9). However, tissue fixed using Bouin’s fixative became more rigid and brittle, leading to tissue breakage when wrapped around Histo-LOOP recesses (Fig. 10B and D). This occurred both with the elastomer and with the sponge Histo-LOOP, despite the sponge material being softer and more flexible.
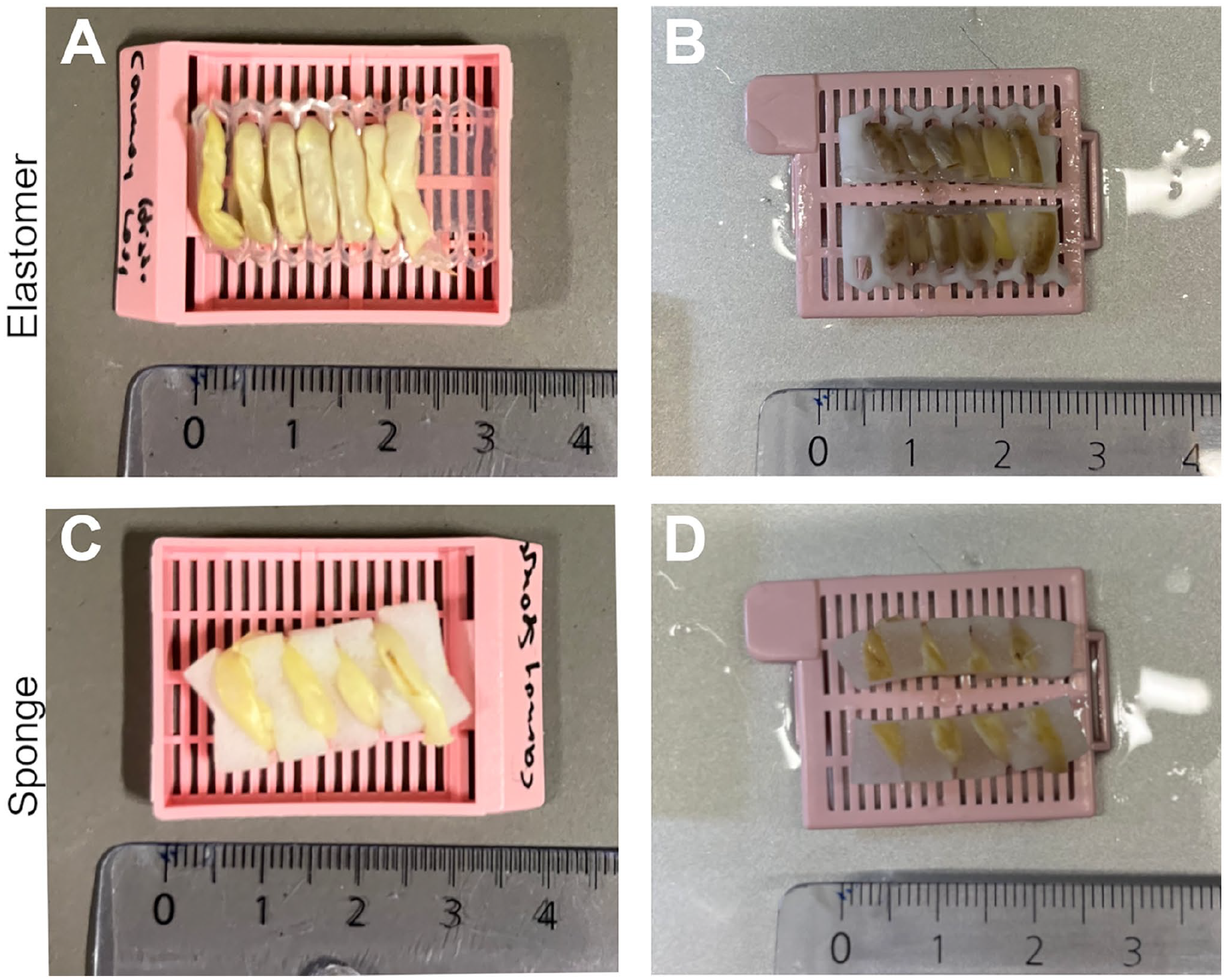
Carnoy-fixed mouse intestine prepared with elastomer and sponge Histo-LOOPs. (A) Mouse intestine wrapped around the elastomer Histo-LOOP. (B) Midline cut of Histo-LOOP before paraffin embedding. (C) Mouse intestine wrapped around the sponge Histo-LOOP. (D) Midline cut of Histo-LOOP before paraffin embedding.

Preparation of mouse intestine fixed with Bouin’s solution using elastomer and sponge Histo-LOOPs. (A) Mouse intestine wrapped around the elastomer Histo-LOOP. (B) Broken pieces of tissue (red arrow) before paraffin infiltration. (C) Mouse intestine wrapped around the sponge Histo-LOOP. (D) Tissue breakage after midline cut of Histo-LOOP.
Histological Evaluation
Standard H&E staining of tubular tissue prepared with Histo-LOOP performed as desired, in both non-fixed specimens and in specimens fixed in formalin, Bouin’s or in Carnoy’s solution. Histology images of non-fixed and formalin-fixed H&E-stained human mammary arteries are shown in Fig. 7A and D and Fig. 7B and E, respectively; H&E-stained specimens of formalin-fixed rat intestine punch biopsies are shown in Fig.7C and F. Histology images of Bounin-fixed and Carnoy-fixed tissue prepared with Histo-LOOP from elastomer or sponge and stained with H&E are shown in Figs. 11 and 12, respectively. A special staining of the fresh human mammary artery with EVG also turned out well (Fig. 13).
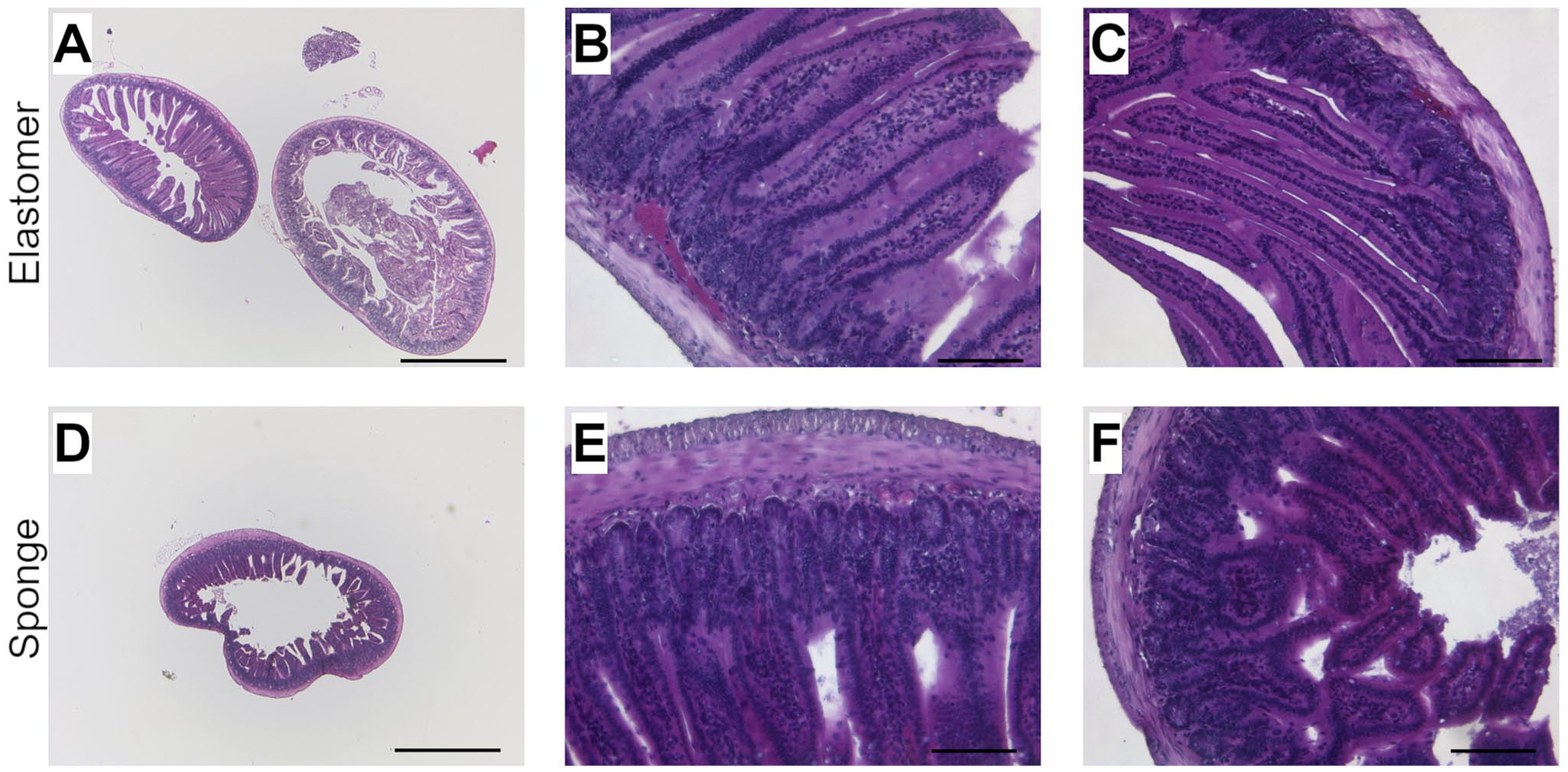
Histological images of Bouin-fixed mouse intestine prepared with elastomer Histo-LOOP (A) 25× magnification, scale bar = 1000 µm. (B and C) 200× magnification, scale bar = 100 µm; with sponge Histo-LOOP. (D) 25× magnification, scale bar = 1000 µm. (E and F) 200× magnification, scale bar = 100 µm, H&E stained.
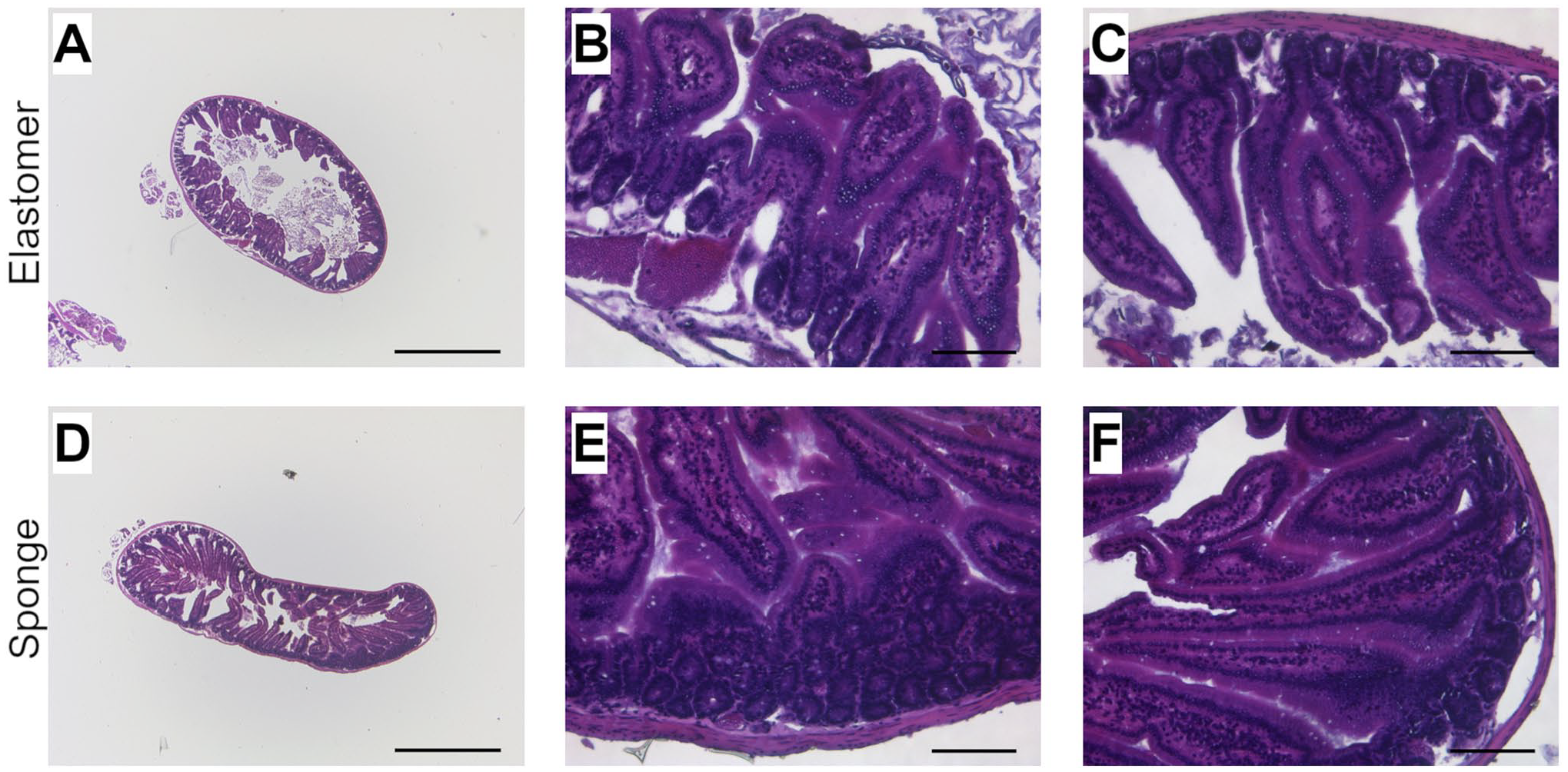
Histological images of Carnoy’s-fixed mouse intestine prepared with elastomer Histo-LOOP (A) 25× magnification, scale bar = 1000 µm. (B and C) 200× magnification, scale bar = 100 µm; with sponge Histo-LOOP. (D) 25× magnification, scale bar = 1000 µm. (E and F) 200× magnification, scale bar = 100 µm, H&E stained.
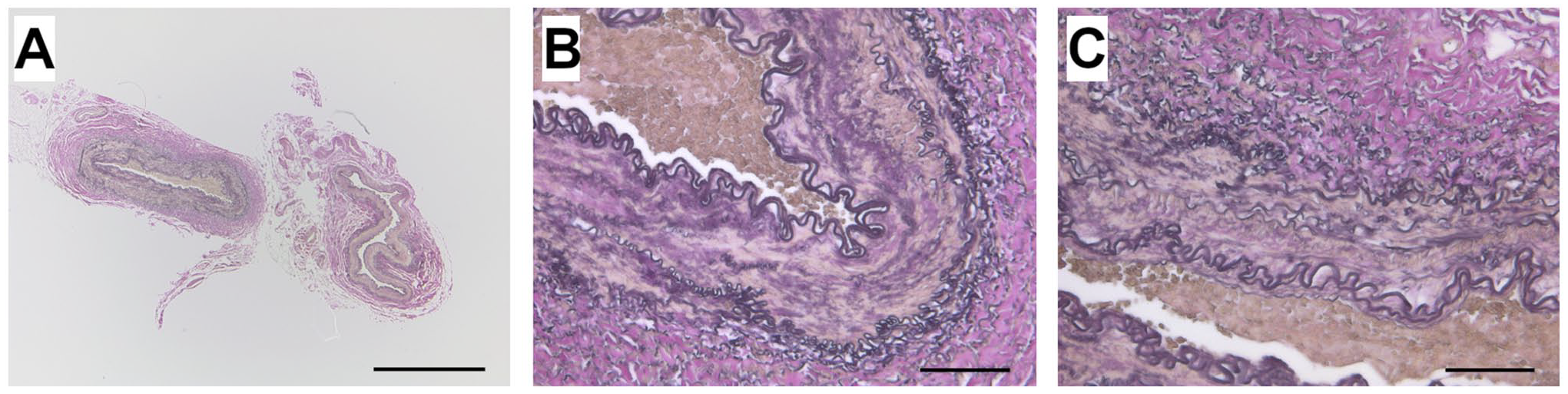
Histological images of human mammary artery prepared with Histo-LOOP, EVG stained. (A) 25× magnification, scale bar = 1000 µm. (B and C) 200× magnification, scale bar = 100 µm.
The staining quality was not impaired with either the Histo-LOOP made of elastomer or the Histo-LOOP sponge. Both materials are invisible on histology images and neither interfere with the tissue nor with the histologic staining. No artifacts were observed on the background or tissue samples.
Discussion
Histo-LOOP was designed to overcome limitations of conventional preparation techniques for paraffin embedding. It enables the production of both longitudinal and cross-sections, and thus provides a complete and reproducible overview of long tubular tissue. In addition, missing tissue segments due to misalignment during the rolling process as observed with the conventional Swiss Roll technique, are avoided. Histo-LOOP is also suitable for generating cross-sections of punch biopsies or small tissue samples. In contrast to conventional techniques, where one paraffin block is required for each punch, Histo-LOOP enables the processing of multiple punch biopsies or small tissue samples at once. In addition, smaller tissue samples can be fixed in the desired orientation, allowing for histopathologic assessment in multiple focal levels unlike conventional techniques, where assessment is often limited to a single level.
In comparison to the Swiss Roll technique, which is only suitable for the preparation of fresh tissue or for immediate fixation by perfusion,6,7 we demonstrate the feasibility of elastomer and sponge Histo-LOOP in preparation of both fresh and fixed tissue, including tissue fixed with formalin and Carnoy’s solutions. However, Histo-LOOP may not be suitable for tissue that has been fixed in Bouin’s solution, as it may become rigid and brittle, causing the tissue to break when wound through the Histo-LOOP recesses. Bouin-fixed tissue or other highly rigid and brittle tissue may only be suitable for punch preparation with the Histo-LOOP, where small pieces of tissue are fixed at each recess, avoiding tissue breakage from the wrapping process. When using Histo-LOOP with fixed tissue, we recommend testing the effect of fixatives beforehand, as the outcome may vary depending on the fixative and individual protocol.
Tissue structures that have been placed in Histo-LOOP can be embedded using standard paraffin embedding methods without interfering with sectioning or histological staining procedures, including both standard staining (H&E) and special staining (e.g., EVG). Both elastomer and sponge Histo-LOOP do not leave any artifacts on the tissue or in the background after staining.
Furthermore, Histo-LOOP can be manufactured in multiple sizes or materials depending on the requirements. Especially sponge material may be used when more flexibility is required or when preparing tissue in a spiral and punch fashion, as it easier to section. Thus, the tool is suitable for processing tubular tissue structures of various diameters.
Histo-LOOP helps standardize, simplify, and improve the processing and histological evaluation of tubular structures. Thanks to a very simple handling, manipulation-induced artifacts and tissue loss can be minimized. With its cost-efficient production and availability in multiple sizes, it is a promising tool for the investigation of tubular, longitudinal tissues and structures as well as solid tissues in diagnostics and research.
Footnotes
Acknowledgements
We would like to thank Christoph Otten for producing the computer-aided design file as well as the first prototypes and the Scientific Workshop at of the University Hospital RWTH Aachen for manufacturing the Histo-LOOP.
Competing Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors declare the following competing of interests: F.J.V., M.D., M.A., D.V.M., and N.S. hold a patent for the Histo-LOOP embedding tool. The remaining authors have no conflicts of interest to declare.
Author Contributions
P.N.: Writing—original draft and preparation, review & editing, M.A.: Writing—review & editing, conceptualization, N.S.: Methodology, Conceptualization, review & editing, C.N.: Writing—review & editing, R.S.: Writing—review & editing, A.P.: Writing—review & editing, F.J.V.: Writing—review & editing, Validation, A.T.G.: Writing—review & editing, Supervision.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the German Research Foundation (DFG) (grant numbers 465213526). The working group is supported by the North Rhine Westphalia Ministry of Culture and Science as part of the “NRW Return Program” (NRW-Rückkehrprogramm) (grant numbers 700706). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript. ![]() .
.
