Abstract
Hematoxylin and eosin (H&E)-stained slides inevitably deteriorate over time, frequently becoming unreadable. Reutilizing these slides can reduce the need for additional serial sections, particularly when the target region is no longer available in the tissue block. This study aims to develop efficient protocols for recycling faded H&E-stained slides, providing benefits for future research on stored samples. Seventy-one faded slides, representing a variety of tissue types and pathologies, were randomly divided into two groups. Slides were de-stained and re-stained using the conventional procedure and a modified Tris and HCl procedure. Three observers independently assessed all slides based on predefined parameters. The stability of the re-stained slides was re-assessed in 6 months. The modified Tris and HCl method yielded significantly higher scores compared to the conventional method for crispness of staining, nuclear staining, cytoplasmic staining, and vibrancy of staining (p < 0.05), as well as greater durability, as evidenced by minimal score reduction 6 months after staining. Thus, incorporating a Tris and HCl step into the process effectively enhances and restores faded H&E slides, offering a valuable technique for revitalizing histology slides for future research and educational purposes.
Introduction
A pathologist’s diagnostic toolkit relies on the histological examination of biopsy samples to identify various lesions. Histopathology slides are invaluable for medical diagnosis, research, and education.1,2 In histology, pathology, and laboratory medicine, medical students encounter microscopy to develop their skills and deepen their academic comprehension. However, it is both challenging and costly to maintain and preserve high-quality teaching histology materials. 3 To ensure their integrity and availability for future studies, these slides must be securely stored and preserved.
Despite the availability of advanced staining methods, hematoxylin and eosin (H&E) staining remains the most favored and widely used technique for diagnostic purposes. 4 H&E staining is employed in histopathology laboratories worldwide, enabling clear visualization of tissue architecture, structural details, and assessment of cellular and morphological structures as well as pathological alterations. 5 Hematein, the basic dye produced by the oxidation of hematoxylin, stains acidic (basophilic) tissue components, such as nuclei, rough endoplasmic reticulum, and ribosomes, a dark blue or purple. The acidic eosin dye stains protein structures such as muscle fibers, stroma, and cytoplasm in shades of pink.6,7 Accurate interpretation is facilitated by the application of H&E staining. Even with careful preservation, slides gradually fade and become unreadable. According to previous research, light exposure and the time elapsed since slide preparation are identified as the two primary factors contributing to the fading of H&E-stained sections.8,9
Reusing H&E-stained slides can alleviate the need for additional consecutively sections when the region of interest is no longer available in the tissue block. Removing the original stain from these slides and re-staining them may also help restore slide quality and reduce the need for re-biopsy. Current de-staining protocols are primarily designed for slides that are insufficiently or excessively stained with hematoxylin. These protocols often involve extensive steps that can extend the process from hours to days. 5 In histopathology, xylene and alcohol are frequently used in the dewaxing, clearing, and dehydration steps in the H&E staining process. 10 Acid reagents, such as acid or acid alcohol, are employed for washing out excess stain or removing nonspecific hematoxylin staining; however, caution is necessary, as excessively strong acids can damage tissue. 11 Some methods focus on removing attached primary antibodies, but the chemicals employed are not designed to eliminate H&E stains. 12 Reaction buffers, including Tris buffer at pH 7.6, have been reported to effectively remove hematoxylin stain, and if further stain removal is needed, the number of washing cycles can be increased. 5
In the conventional de-staining method, xylene is used to remove any remaining adhesive, while 95% alcohol is used to remove the eosin stain. 11 Tap water, which often contains elements that can de-stain hematoxylin, is used to gently remove excess eosin from the slide before reapplying the H&E stain. 11 However, re-staining using these methods leads to obscured images and color inconsistencies in some areas. The goal of these studies was to improve the efficiency of removing old stains and ensure that the new H&E staining adheres clearly and accurately to the tissue without color distortion. As a result, this study aims to develop practical protocols using Tris buffer and HCl for revitalizing old H&E-stained slides, offering significant benefits for future research on stored samples.
Materials and Methods
The study was conducted in the Department of Pathology. Seventy-one section slides, originally stained using the conventional Mayer’s hematoxylin and 1% eosin method from routine biopsy specimens archived with coverslips for 3 to 5 years, showing fading and scoring criteria of 2 to 3 (Table 2), were retrieved from the hospital. Tissue samples were from unidentified patients and provided without any patient identification data. This procedure details the steps for H&E de-staining methods and the reutilization of archived H&E-stained slides for re-staining using standard laboratory equipment and reagents.
This research is an experimental comparative study. A randomized selection of 71 faded sections was assessed under a microscope by a pathologist. The slides were then divided into two groups, each containing a variety of tissue types and pathologies, ensuring similar diversity between the groups. Thirty-four tissue sections were de-stained using the conventional procedure (Group A), and 37 tissue sections were de-stained using the modified Tris and HCl procedure (Group B) (Table 1). All slides, both before and 1 week after re-staining (Re-stained), were documented and microphotographed using a Pannoramic Desk II DW Scanner (3DHISTECH, Hungary), with images viewed using CaseViewer 2.4 software (3DHISTECH, Hungary). The slides were then scored and analyzed by two blinded pathologists and one blinded anatomist. The H&E re-stained sections were graded on a scale from 1 to 5 based on parameters such as crispness of staining, uniformity of staining, nuclear staining, cytoplasmic staining, and vibrancy of staining, as described in Table 2. Another evaluation was conducted at 6 months after re-staining (Re-stained 6 mo) and 1 year after re-staining (Re-stained 1 yr) to determine the durability of the re-stained color for both de-staining methods.
De-Staining and Re-Staining Procedure.

Slide Scoring Criteria.
The total scores of the slides from two de-staining methods were compared at four different time points (before staining, 1 week after re-staining, 6 months after re-staining, and 1 year after re-staining) using repeated measures analysis of variance (ANOVA) and independent samples t-test. Changes in scores for each slide criterion within the same method group were compared across the four time points using a multiple comparison test. A p value <0.05 was considered as significant. In addition, the reliability and consistency of the three evaluators were compared using the intraclass correlation coefficient (ICC). Statistical analyses were performed using STATA/BE 18.0 Software (StataCorp LLC, College Station, TX, USA). This study was approved by the Institutional Review Board and the Medical Ethics Committee, Royal Thai Army Medical Department, Thailand.
Results
The slide ratings from the three evaluators were highly reliable and consistent, as indicated by the ICC, which approached 1 for four criteria: crispness of staining (0.93), uniformity of staining (0.96), nuclear staining (0.93), and cytoplasmic staining (0.95). However, there was variability in one criterion: vibrancy of staining (0.58). There was no significant difference in the scores for a randomized set of faded slides before staining between the two experimental groups, except for the nuclear staining and vibrancy of staining criteria (p < 0.05). Scores were lower in Group B for these criteria (nuclear staining = 2.71, vibrancy of staining = 2.15; Fig. 1C and E). Despite this, all slides showed fading with partial staining of the nucleus and cytoplasm, which complicated diagnosis (Figs. 3 to 5A and E).
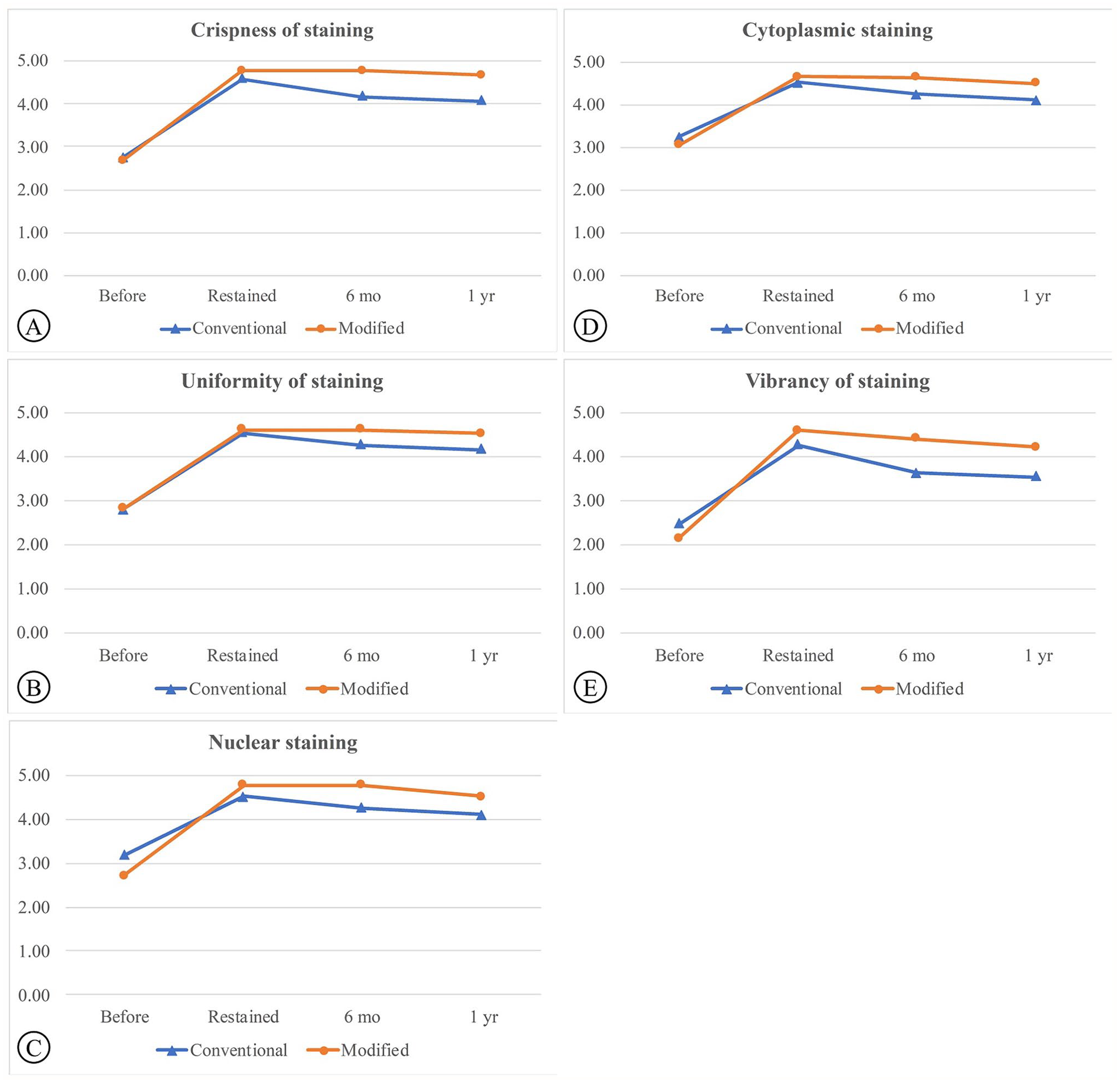
Comparative scores for the conventional and modified Tris and HCl procedures across defined parameters: (A) Crispness of staining, (B) Uniformity of staining, (C) Nuclear staining, (D) Cytoplasmic staining, and (E) Vibrancy of staining.
The effectiveness of two de-staining methods (conventional and modified Tris-HCl) on faded H&E-stained slides is shown in Fig. 2. The modified Tris-HCl method resulted in a more complete removal of faded H&E stains compared to the conventional method, as demonstrated in the after de-staining images (Fig. 2D). In both methods, the scores of the slides after re-staining at 6 months and at 1 year were significantly higher than the before re-staining scores (Table 3). After re-staining, scores were significantly higher in Group B for all criteria (p < 0.05), except for uniformity of staining (p > 0.05; Group A = 4.55, Group B = 4.62; Table 3; Fig. 1). The conventional group exhibited out-of-focus areas (Fig. 4B) and lower scores in crispness, uniformity, and vibrancy (Figs. 3 to 5B) compared to the modified group (Figs. 3 to 5F). The durability of the re-stained color was assessed 6 months (Figs. 3 to 5C and G) and 1-year after staining (Figs. 3 to 5D and H) for both de-staining techniques. Slides in Group B, subjected to the modified Tris and HCl procedure, showed minimal score reduction at 6 months, whereas slides in Group A experienced a more notable decrease. Specifically, Group A sections showed a decline in crispness of staining from a score of 4.59 to 4.17 and in vibrancy of staining from 4.27 to 3.64 (Table 3; Fig. 1A and E). However, there was a significant decrease in scores from the 6-months to the 1-year time point for all criteria (p < 0.05).
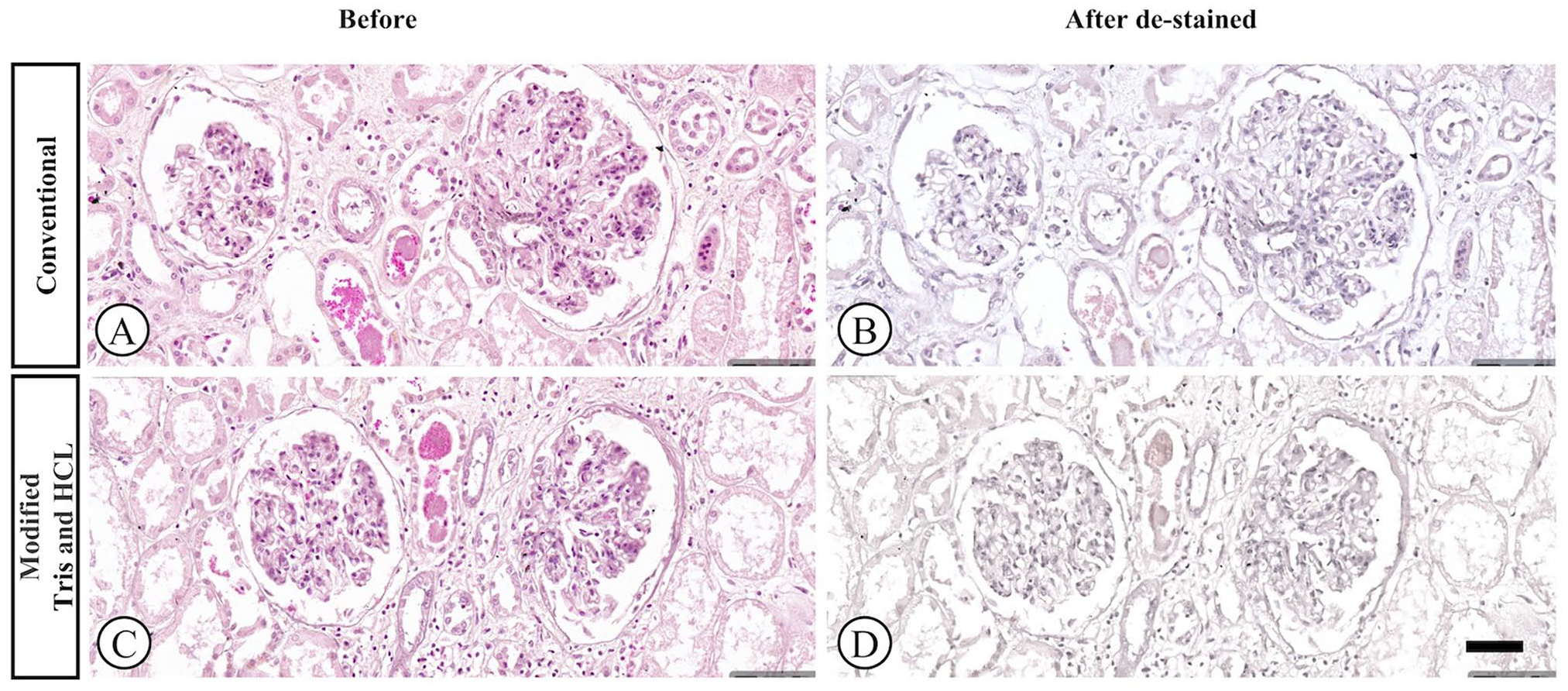
The impact of the de-staining process on faded H&E slides was compared between the conventional and modified Tris-HCl methods. The images illustrate the faded slide before (A and C) and after the de-staining and rehydration step (B and D), prior to re-staining with H&E. [H&E, 20× magnification].
Comparative Scores for the Conventional and Modified Tris and HCl Procedures.

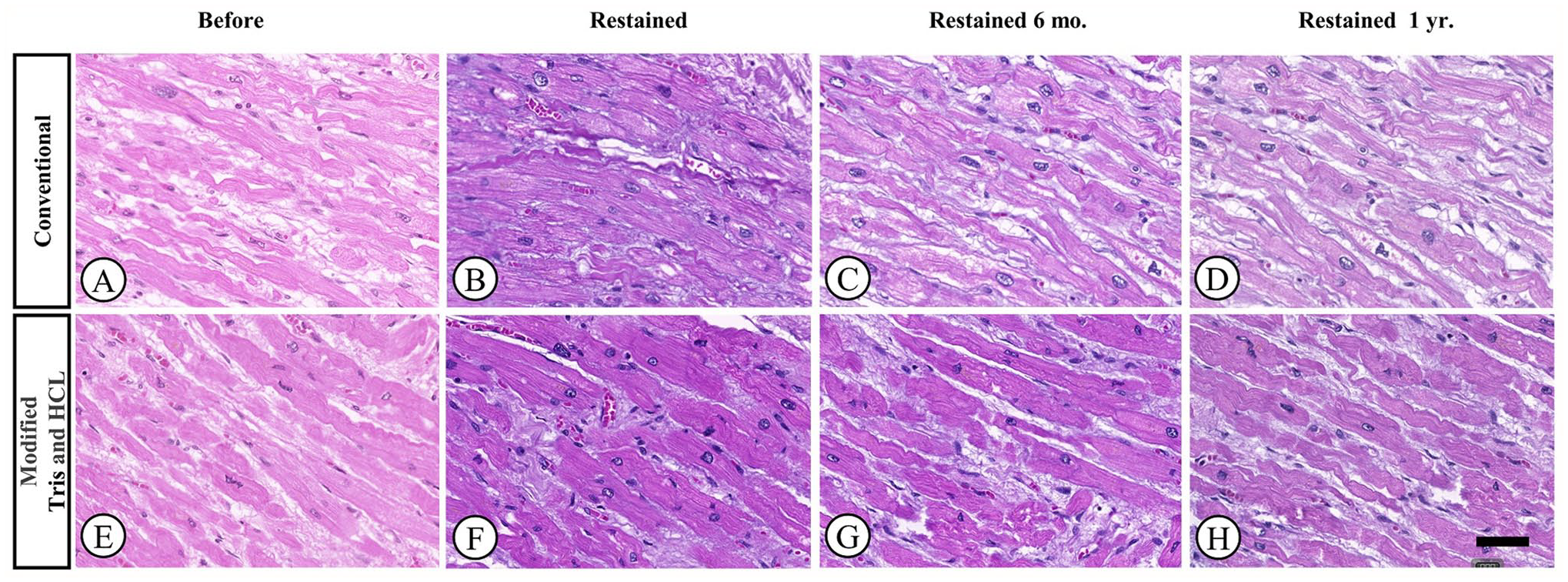
Photomicrographs of cardiac muscle samples stained with H&E. Samples were de-stained and re-stained using either conventional or modified Tris and HCl methods. Representative images were taken before re-staining (A and E), 1 week after re-staining (B and F), 6 months after re-staining (C and G), and 1 year after re-staining (D and H). [H&E, 20× magnification].
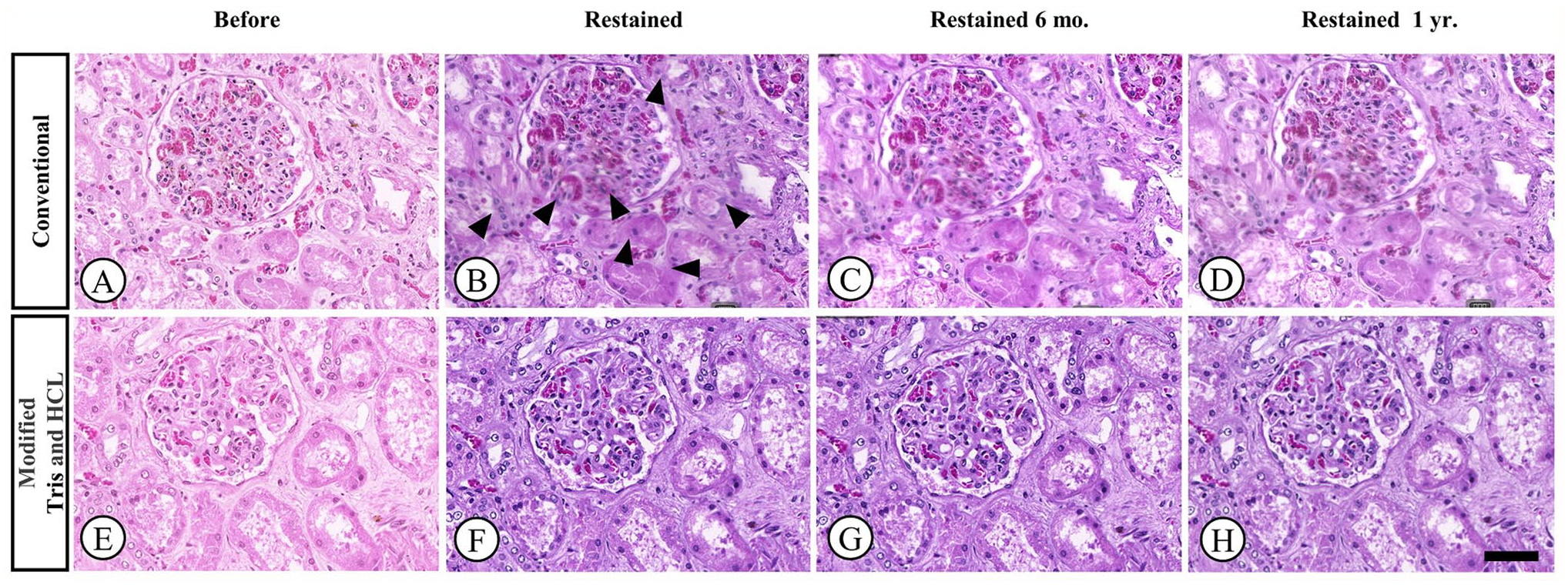
Photomicrographs of renal tissue samples stained with H&E. Samples were de-stained and re-stained using either conventional or modified Tris and HCl methods. Representative images were taken before re-staining (A and E), 1 week after re-staining (B and F), 6 months after re-staining (C and G), and one year after re-staining (D and H). Obscurity in some areas is noted in the image taken after re-staining using the conventional method (black arrowheads). [H&E, 20× magnification].
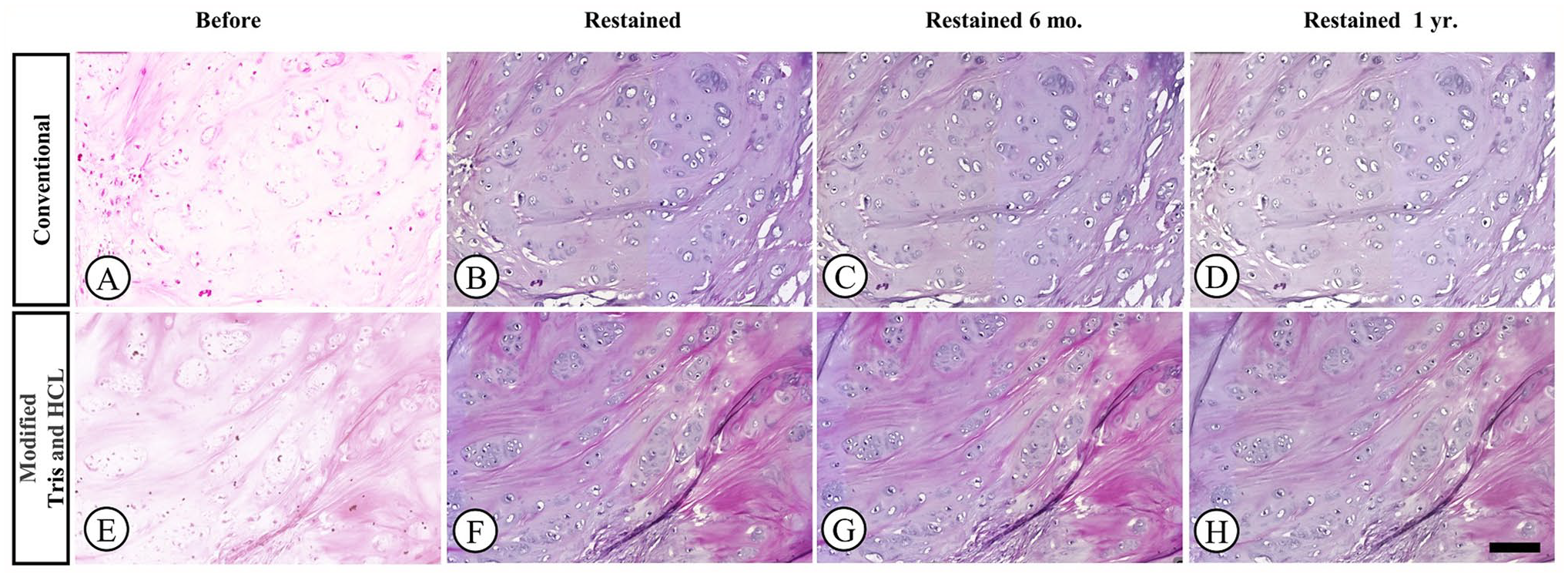
Photomicrographs of cartilage samples stained with H&E. Samples were de-stained and re-stained using either conventional or modified Tris and HCl methods. Representative images were taken before re-staining (A and E), 1 week after re-staining (B and F), 6 months after re-staining (C and G), and 1 year after re-staining (D and H). [H&E, 10× magnification].
Discussion
This study aims to develop efficient methods for recycling old H&E-stained slides by optimizing de-staining protocols, achieving optimal re-staining quality, and exploring their potential applications in research and education. A key finding of this study is that the incorporating Tris and HCl into the process significantly enhances the quality and revitalization of H&E slides. Compared to the conventional method, the modified technique demonstrated superior crispness, uniformity, nuclear staining, cytoplasmic staining, and vibrancy. Moreover, the re-stained color exhibited increased resistance to fading, rendering it a practical, efficient, and cost-effective protocol.
While digital scanning and archiving offer numerous benefits for remote collaboration, accessibility, and preservation, they may not be an affordable or practical option for all laboratories. 13 By providing a thorough re-staining procedure, this method can be especially helpful in educational and research settings, where hands-on experience with microscopes is essential for learning histological techniques, without the need for specialized digital equipment. Currently, protocols that provide instructions for efficiently reutilizing deteriorated H&E-stained slides have not been described. In fact, existing protocols primarily address de-staining procedures for slides with staining discrepancies, such as understaining or overstaining with hematoxylin. These protocols often involve lengthy steps that can extend the process from hours to days.10,11
The literature on H&E stain removal is limited, with only a few protocols available. Complete elimination of these dyes from tissue sections remains a significant technical challenge. 14 Among the explored methods, Tris buffer-based procedures have been used to optimize the decolorization process. However, their main purpose is to preserve target epitopes for subsequent immunohistochemistry, enabling slide reuse. 5 These Tris buffer-based procedures have been effective in removing most of the visible stain while preserving tissue structure and morphology, enabling subsequent immunohistochemical staining according to the desired protocol. In this study, slides de-stained using the modified Tris and HCl procedure demonstrated improved efficiency in clearing old stains compared to conventional methods. While tap water is used in conventional methods for diluting alcohol during de-staining and as a bluing reagent. 11 It is subject to daily and seasonal pH fluctuations and may not be sufficiently alkaline to act as an effective bluing agent. In addition, particulate matter in tap water can adhere to tissue sections. 11 Tris buffer, with its constant pH of 7.6, therefore offers a more reliable alternative.
Alternative approaches involve more corrosive reagents such as HCl solutions. 15 A previous study used 1% HCl in 70% ethanol as a differentiating agent to remove excess hematoxylin, which improved nuclear delineation and stain differentiation, resulting in clearer visualization of various cell structures. 16 Other methods employing guanidinium hydrochloride (GnHCl) (12), beta-mercaptoethanol/sodium dodecyl sulfate (2ME/SDS), or urea have been demonstrated to elute antibodies from immunostained tissue sections on glass slides or coverslips for sequential immunostaining. 17 However, these techniques primarily focus on target antibody removal rather than H&E removal.
This study found that incorporating Tris buffer and HCl into the de-staining and re-staining process can effectively revitalize H&E-stained slides. Our study utilized Mayer’s hematoxylin, a commonly used stain known for its reliable nuclear staining properties, controllable staining depth, and pale nuclear staining in a progressive stain. 10 Two other traditional formulas for nuclear staining, such as Gill’s and Harris’s, offer different properties.10,18,19 While we did not directly compare Gill’s or Harris’s hematoxylin in this study, we believe that our re-staining technique is likely applicable to a wide range of hematoxylin stains commonly used in histology laboratories.
Each slide was scored, with slides receiving a score of 2 or less considered inadequate for diagnosis, while those receiving a score between 3 and 5 considered adequate for interpretation and diagnosis. By successfully restoring faded slides from various tissue types, we aimed to demonstrate the broad applicability of our method and its potential to preserve valuable histological data across a wide range of specimens. This versatility is especially important for laboratories that handle diverse tissue collections. Discrepancies in vibrancy assessments among the three evaluators may be attributed to the subjective nature of freshness, influenced by individual preferences. Variations in human visual systems, which affect color perception tasks, stem from genetic differences in cone photopigments. 20
Before staining, all slides exhibited fading and were randomly assigned to each group. While the modified Tris and HCl procedure initially yielded lower scores for nuclear staining and vibrancy of staining compared to the conventional group, subsequent re-staining resulted in significantly higher scores for all evaluation criteria. This suggests that regardless of the initial poor quality of the slides, the revival process using Tris buffer and HCl can effectively restore them. Even after being stored under the same conditions as the conventional group and shielded from sunlight, the slides treated with this modified technique exhibited very good color durability, with minimal changes in staining scores.
H&E slide fading is influenced by various factors, including the type of hematoxylin, staining method, mounting media, and clearing agents such as xylene and toluene, as well as acids and alkalis. Discolored slides are particularly susceptible to further fading with prolonged exposure to air and sunlight.8,9,21 Employing reactive oxygen species scavengers-enriched mounting media, such as N-propyl gallate (NPG), 1,4-diazabicyclo[2.2.2]octane (DABCO), 4-pirydyl-1-oxide)-N-tert-butyl nitrone (4-POBN) and p-phenylanediamine (PPD), can effectively reduce bleaching and preserve histology slides even with continuous natural light exposure for a year. 22 Despite various mounting media options, none are completely ideal. 23
This study aimed to evaluate whether a simpler, less time-consuming, and cost-effective alternative method using Tris buffer and HCl could serve as an effective method for revitalizing faded H&E staining. The durability of the H&E stain after re-staining is particularly valuable for long-term research and preservation, especially when samples are scarce. Despite the relatively small sample size of approximately 35 per treatment group, our qualitative analysis, coupled with rigorous selection, randomization, a standardized scoring system, and observer consistency, provides a strong basis for our findings and makes a valuable contribution to the field. The high ICC of greater than 0.90 among our three observers further supports the reliability and consistency of our visual assessments. 24
The study’s limitations should be addressed. First, the evaluation of re-stained slides may be subjective, as it often relies on visual assessments by individual researchers. Artificial intelligence (AI)-based digital assessment, while promising, requires significant resources and expertise that were unavailable for this study. Implementing standardized scoring systems could help mitigate this subjectivity and provide more consistent evaluations. Second, the study might not have controlled for the specific causes of fading, such as exposure to light, temperature variations, or the quality of staining. Including a broader range of slide types and varying conditions of fading could improve the generalizability of the findings. Third, achieving complete removal of the original stain without damaging tissue morphology can be challenging and may vary depending on the slide’s condition, including tissue type, age, and storage conditions. Fourth, specific properties of different hematoxylin stains, such as their oxidation state or affinity for tissue components, could influence the effectiveness of our re-staining protocol. Future studies could explore the specific differences in re-staining outcomes when using various hematoxylin stains, such as Gill’s or Harris’s hematoxylin, to provide additional insights into the applicability of our method. Finally, the long-term effects of re-staining on slide durability and color fastness are not yet fully understood and warrant further investigation.
Footnotes
Acknowledgements
The authors would like to express their gratitude to Dr. Kittisak Wongchansom, Army Institute of Pathology, Phramongkutklao Medical Center, for providing histopathology slides, and to Ms. Natchanan Samatthakitkhajonh, the Pathology Staff at Phramongkutklao College of Medicine, for her technical assistance. They are also grateful to Ms. Supak Ukritchon from the Office of Research and Development, Phramongkutklao College of Medicine, for her support with the statistical analysis.
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
All authors have contributed to this article as follows: study design (NL, AK, MC), methodology and inspection (NL, CR, PA, TN). NL contributed as the first author by drafting the original manuscript, generating the tables and figures, performing data analysis and interpretation, and formatting the manuscript. TN participated in editing and grammar checking the manuscript. All the above authors have read and approved the final manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Phramongkutklao College of Medicine (ORD2565-PCM 3.5.13).
