Abstract
Connexin 43 (Cx43) is a potential target for accelerating wound healing. This study aimed at evaluating the therapeutic efficiency of topical application of Gap27, a Cx43 mimetic peptide, on corneal tissue reorganization during wound healing in streptozocin-induced Diabetes in albino rats and its effect on the infiltration of inflammatory cells. Fifty adult male albino Wistar rats were divided equally into two groups: non-diabetic and diabetic. Twenty rats from each group were subjected to corneal injury: 10 untreated and 10 treated with Gap27. The remaining five rats from each group served as negative controls (intact corneas). All rats were sacrificed 3 days after injury. Histological studies were performed to assess signs of cell degeneration, the infiltration of inflammatory cells. Histomorphometric studies were performed to quantify the expression of Cx43. Gap27 promoted corneal wound healing in non-diabetic and diabetic rats. It reduced mononuclear cell infiltration and improved corneal tissue remodeling. However, minor structural changes were still seen in diabetic corneas after treatment with Gap27. Blocking Cx43 was a valuable tool to restore corneal tissue structure, reduce the infiltration of inflammatory cells in non-diabetic and diabetic rats.
Introduction
Corneal transparency is crucial for optimal vision. The most common corneal injuries occur in the superficial corneal epithelium, which may also involve the Bowman’s membrane or deeper into the anterior part of the stroma. Corneal opacity accounts for approximately 4.2% of the causes of blindness annually.
1
Diabetic keratopathy is relatively frequent in persons with Diabetes mellitus. Delayed corneal epithelial wound healing is common and may compromise patient’s vision. This predisposes patients to sight-threatening complications, such as stromal opacification, corneal surface irregularities, and microbial keratitis.
2
Regardless of the etiology or the depth of the injury, the main therapeutic goal is to promote structural restoration during wound healing with minimal scarring.
3
The effect of Diabetes mellitus on the cornea has many aspects. Delayed corneal epithelial wound healing is a common complication which can involve an inflammatory invasion of antigen presenting cells.
4
Corneal epithelial cells form gap junction channels assembled by connexin proteins. Gap junctions are thought to influence tissue injury and repair mechanisms, with connexin 43 (Cx43) playing a major role in corneal wound healing.5–7 Cx43 blocking with Gap27 has been reported to accelerate wound healing in
Materials and methods
Experimental design
Fifty adult Wistar male albino rats, weighing about 200–220 g, were purchased and housed in the Medical Research Center, Faculty of Medicine, Ain Shams University, Cairo, Egypt. They were housed in wire mesh plastic cages under standard conditions of breading, with
The rats were randomly allocated into two equal groups; Group I (non-diabetic group): Rats of this group (n = 25) received intraperitoneal injection of a single dose of citrate buffer equivalent to Group II. Group II (diabetic group): Diabetes mellitus was induced in rats of this group (n = 25) using single intraperitoneal injection of freshly prepared solution of 40 mg/kg Streptozocin (STZ, Sigma-Aldrich, Italy). STZ was dissolved in ice-cold 0.5 mL of 0.1 M/L citrate buffer at a pH of 4.5. Fasting blood glucose level in rats of the diabetic group was monitored daily by the glucometer, using a drop of rat tail vein blood, after sterilization of the tail with Betadine solution followed by scraping of the tail hair. Three days post-injection, STZ-treated rats which developed glucose levels higher than 400 mg/dL were considered diabetic. Rats were maintained diabetic for 3 months before starting the experiments (Diagram 1).
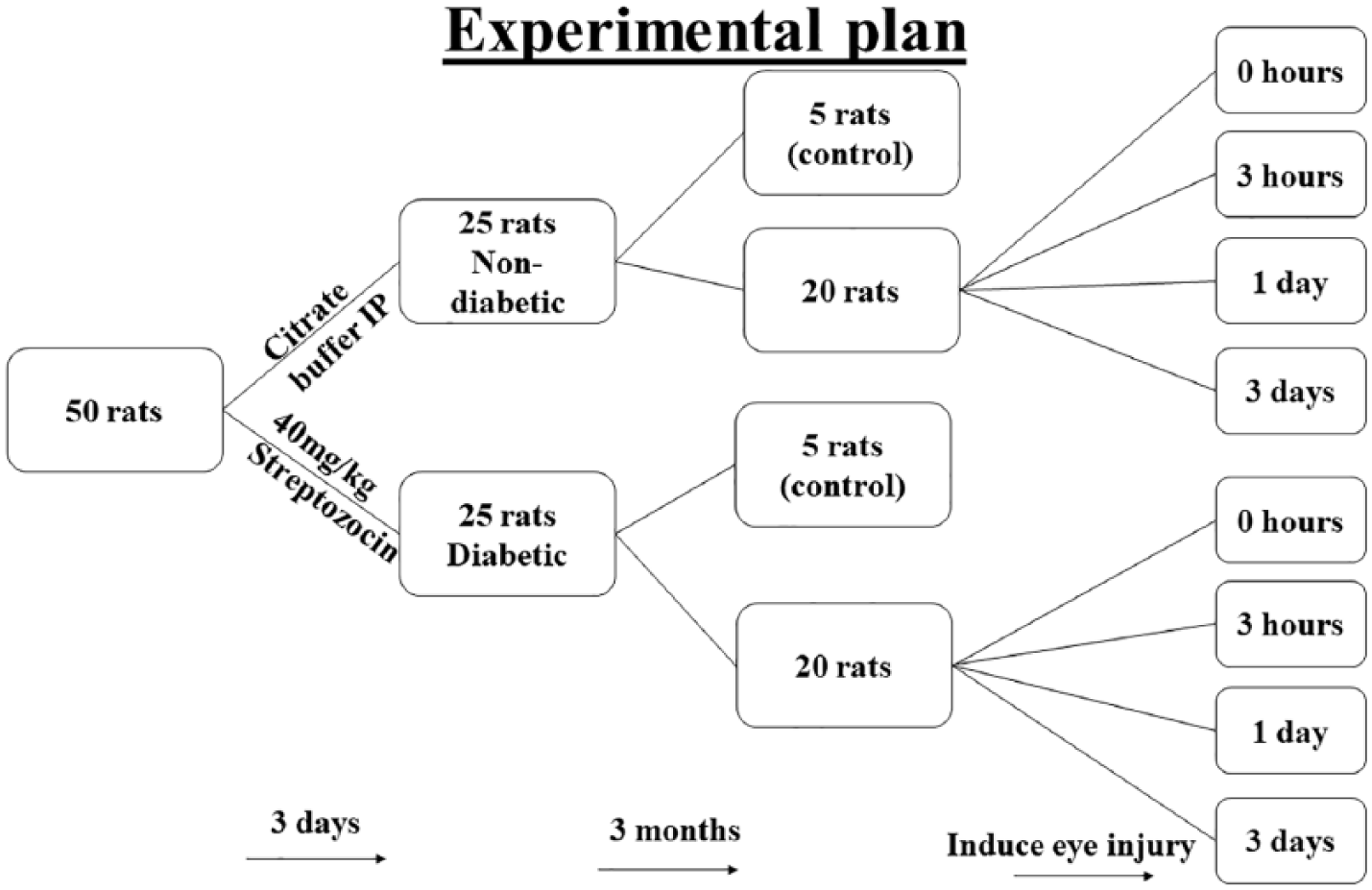
Flow chart for experimental design.
Five rats from each group were used as negative controls and received phosphate buffered saline (PBS) cold eye drops, 2 drops/cornea, at same time intervals as the test group. Corneal wound injury was induced in the right eye corneas of the remaining 20 rats of each group. In the non-diabetic group, all left eyes received PBS cold eye drops (2 drops/cornea), while the treatment group received Gap27 (Tocris, Bristol, UK) eye drops prepared in ice-cold PBS (1 mM) at the same time intervals (right eyes). All procedures were performed following the European Union (EU) directive of 2010/63/EU for animal experiments. Ethical approval was obtained before the commencement of the work.
Fluorescein penetration test to study wound closure rates
Wound healing assays were performed using fluorescein dye (Sigma-Aldrich, Italy). Twelve adult Wistar male albino rats were used and injuries were induced in the right eye of each animal. Corneal debridement was performed using Alger brush II to gently remove the corneal epithelium from the whole corneal surface. Neutral fluorescein dye was applied to the cornea of each rat for 1 min before washing out with PBS drops three times. Corneas were then photographed with a digital camera, PowerShot G7 X Mark II (Canon, USA) under ultraviolet (UV) light. All 12 animals were sacrificed after the experiment. The fluorescent area was calculated as percentage from initial injury using ImageJ software (USA).
Corneal wound healing model
Twenty experimental animals from each group (non-diabetic and diabetic) were anesthetized by ether inhalation. One drop of Akten® topical anesthetic (Lidocaine hydrochloride ophthalmic gel 3.5%, Akorn Inc., Lake Forest, IL 60045, USA) was applied to each eye before wound induction. A full-length steady linear corneal epithelial debridement was created across the cornea of both eyes in the nasal-temporal direction, with a corneal rust ring remover equipped with a rotating 0.5 mm burr (Alger brush II; Alger Equipment Co., Inc., Lago Vista, TX, USA). This instrument is designed to remove approximately 500 μm of corneal epithelium without penetrating the Bowman’s layer. After application of either PBS or Gap27 eye drops, trypan blue test was performed immediately after injury. Fucithalmic® eye antibiotic (Fusidic acid 1% viscous eye drops, Leo laboratories limited, 285 Cashel Road, Dublin, Ireland) was applied to both eyes to prevent infection.
Cx43 blocking by Gap27 peptide
Gap27 peptide (SRPTEKTIFII) was purchased from Tocris Bioscience (Bristol, UK). Gap27 peptide (1 mM) was made fresh for each treatment and maintained in 1 mL syringes on ice until used (Pollok et al, 2011; Elbadawy et al., 2016). Designated corneas of the experimental rats were treated with Gap27 eye drops, 2 drops/cornea, at selected time intervals; one drop just prior to corneal injury induction, one drop immediately after injury, two drops 2 h after injury, and then two drops every 12 h on Day 2.
Scarification of rats and processing of corneas
Five animals from each group were sacrificed at each time point after epithelial debridement and treatment, at 0 h (immediately after injury), 3 h, Day 1 and Day 3 after injury (Diagram 1). Negative control rats were sacrificed on Day 3. On the specified time of scarification, rats were anesthetized by intraperitoneal administration of sodium thiopental (45 mg/kg). 9 Corneas were carefully dissected out, washed in PBS, and fixed in 10% neutral buffered formalin. Corneas were then included in paraffin blocks, orienting the axis of the wound in perpendicular position to the bottom of the mold. Serial sections of 5 μm thickness were prepared and subjected to Hematoxylin and Eosin stain (H&E stain). Identities of samples were masked after cutting and before the analysis of images. Three main parameters were investigated: corneal inflammation as indicated by the infiltration of mononuclear inflammatory cells. Karyolitic nuclei and vacuolated cytoplasm as two indicators of cell death. In addition, general histopathological findings were studied to evaluate the overall health of the corneal layers.
Immunohistochemical staining was done for Cx43 (Rabbit Polyclonal Anti Cx43 antibody Keratin 12 (K12) (
Morphometric and statistical studies were performed on area percentage of Cx43 positive cells. Measurements were performed using Leica QWin software program (Leica Microsystems, Heerbrugg, Switzerland). These parameters were measured from the center of the corneas where the wound was induced. Measurements were taken from five fields from each of five non-overlapping sections from all animals of each group (40×). The mean values were estimated and the standard deviation (SD) was calculated using SPSS statistical software version 21, IBM Corporation, NY, USA. Data were evaluated using the one-way analysis of variance (ANOVA) test with post hoc test. The least significant level of probability used was at
Results
Gap27 accelerates wound closure rates
Connexin blocking by Gap27 managed to accelerate corneal wound injury closure rate in both diabetic and non-diabetic rats. The healing process was noted 3-h post injury and progressed so that the gap was completely closed in 1 day in all groups except diabetic untreated corneas (Figure 1(a)). Diabetic rats showed slower rate of wound closure (Figure 1(b)). Gap27 treatment accelerated the healing rate in both non-diabetic and diabetic rats, however, the increase was to a lesser extent in diabetic rats.

(a) Cornea stained with fluorescein dye just after epithelial removal, after 0 h, 3 h and 1 day. Decreased wound gap (green) is evident in non-diabetic and diabetic corneas after 3 h and 1 day from injury. Each image is a representative for three images from three animals per group. (b) Wound closure rates calculated as percentage wound closure from initial wound. Error bars represents standard deviation from three measurements (n = 3).
Histological findings of intact corneas of negative control rats
Corneas of normal healthy rats showed intact regular five layers of the cornea arranged in their normal histological pattern, and a full thickness avascular corneal stroma with spindle shaped keratocytes in-between fibers. The endothelium appeared as a continuous regular single layer of flat endothelial cells with flat nuclei, resting on the non-cellular Descemet’s membrane, which separated them from the stroma (Figure 2(a)).
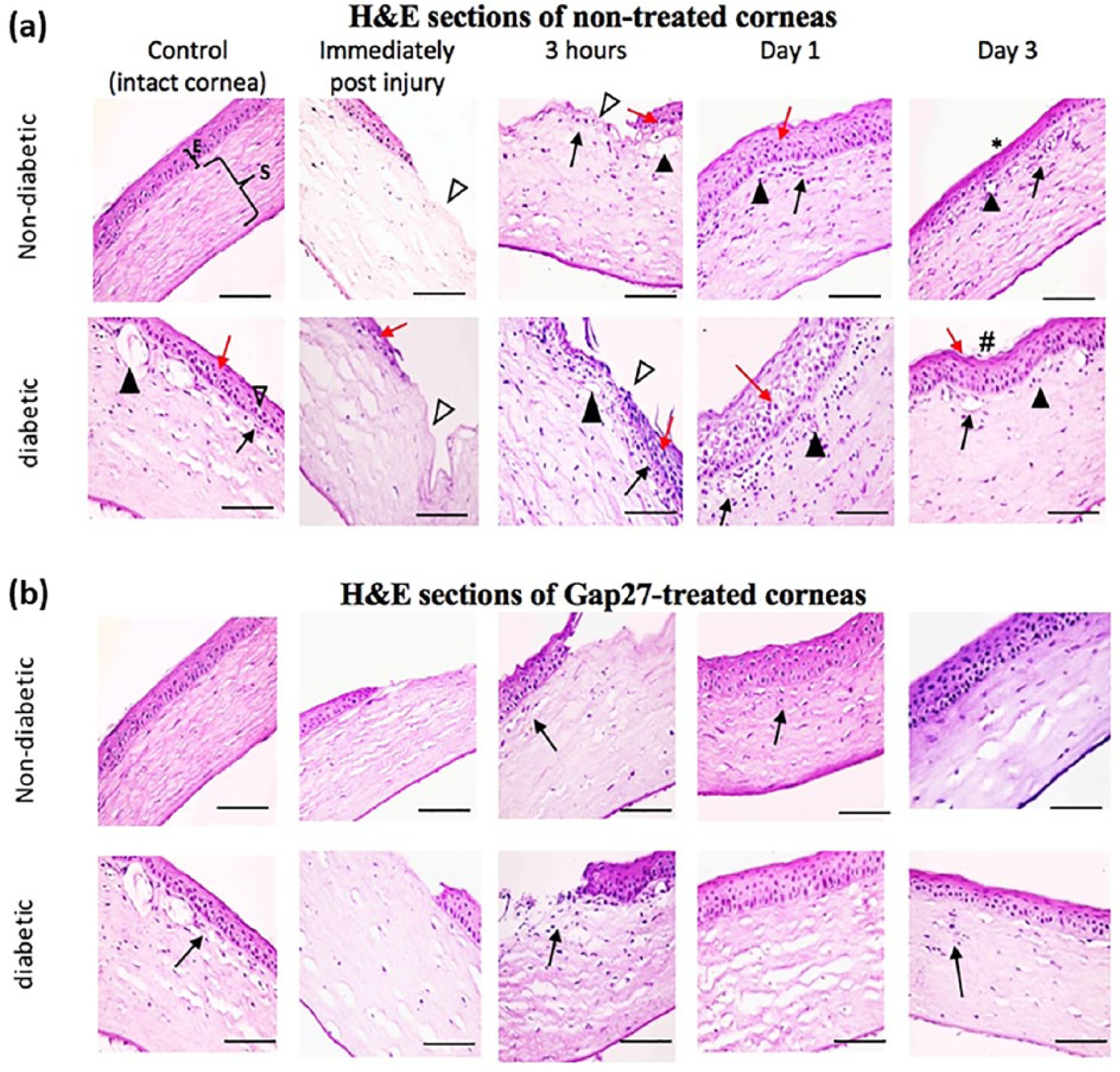
Sections of non-treated and Gap27-treated corneas. (a) Showing control non-diabetic cornea having regular epithelium (E) and regular stroma (S). Corneal wound injury is seen immediately after and 3-h post injury as denuded epithelial area (Δ) in non-diabetic and diabetic corneas. Corneal epithelial degenerative changes can be seen in non-diabetic corneas at 3 h and 1 day after injury. However, they can be clearly noticed in all time points in diabetic cornea shown by red arrows (↑). Neovascularization of corneal stroma (▲), and mononuclear inflammatory cells shown by black arrows (↑) can be seen in non-diabetic corneas from 3 h after injury onwards. They can be more evidently seen in control diabetic corneas and in all time points after injury. Some minor surface irregularities can be seen in non-diabetic corneas 3 days post injury (*). Notice the depressed corneal surface denoting highly irregular wound healing site in diabetic corneas (#). H&E x 400, scale bar = 50 µm. (b) GAP27-treated corneas showing the non-diabetic corneas comparable to the control with regular stratified epithelium after 3 days of treatment. However, diabetic corneas appear with slight surface irregularities and few mononuclear inflammatory cells (↑). H&E x 400, scale bar = 50 µm.
In diabetic rats, the corneas exhibited focal areas of minute surface irregularities. Many cells in basal and intermediate layers had vacuolated cytoplasm and deeply stained irregular nuclei. In addition, many cells of the surface epithelium showed karyolitic nuclei. Intercellular spaces were observed, especially between wing cells and the overlying squamous cells. The anterior stroma showed wider areas separating their collagen fibers, with the presence of mononuclear inflammatory cells’ infiltration. Descemet’s endothelium was desquamated in focal areas (Figure 2(a)).
Histological changes in injured rat corneas
Immediately after injury, corneal sections of the non-diabetic and diabetic groups showed completely denuded corneal epithelium not extending to Bowman’s layer (Figure 2(a)). The remaining layers were comparable to intact non-injured controls. After 3-h time point post injury in non-diabetic rats, many of the corneal epithelial cells showed vacuolated cytoplasm, others showed irregular or karyolitic nuclei. Few mononuclear inflammatory cells were seen extending beneath the wound area and wound margin in the anterior stroma. In diabetic rats, these findings were more apparent. Surface cells were seen migrating to the denuded area. The stroma showed mononuclear cellular infiltration, especially under the wound area, and beneath the wound edge. At Day 1 post injury, complete, yet irregular, surface re-epithelization was observed in both groups. Apparently, thick epithelial covering was seen lying over irregular basement membrane (Figure 2(a)). Degeneration of many wing cells in the diabetic group was evident.
Mononuclear inflammatory cells were still present in anterior stroma in non-diabetic rats and were numerously seen in most of the stroma in the diabetic rats. In the diabetic corneas, wavy basement membrane was seen underneath the epithelium (Figure 2(a)).
Treatment with Gap27 reduced inflammatory cell infiltration and cell degeneration
Gap27 treatment of injured corneas of both non-diabetic and diabetic groups showed better corneal parameters at all time points as compared with non-treated corneas. Gap27 treatment showed that very few mononuclear inflammatory cells were encountered in the anterior stroma of the diabetic group (Figure 2(b)).
Cx43 expression increased in diabetic rat corneas
Cx43 immunoreactivity was detected in the cytoplasm and few nuclei of corneal epithelium in the basal layer and in keratocytes of the anterior stroma in both non-diabetic and diabetic control rats just after injury and up to 3 h (Figure 3(a) and (b)). One day after injury, Cx43 immunoreactivity became distributed in all layers of the newly stratified epithelium, nevertheless, its area percentage was significantly less than the noted levels at 3-h post injury in both groups (Figure 3(c)). By Day 3, significant decrease in Cx43 immunoreactivity was detected as compared with the 1-day diabetic corneas. However, this reaction was still significantly increased when compared with the intact control diabetic corneas, and to the matching time point of the non-diabetic corneas (Figure 3(a) and (c)).
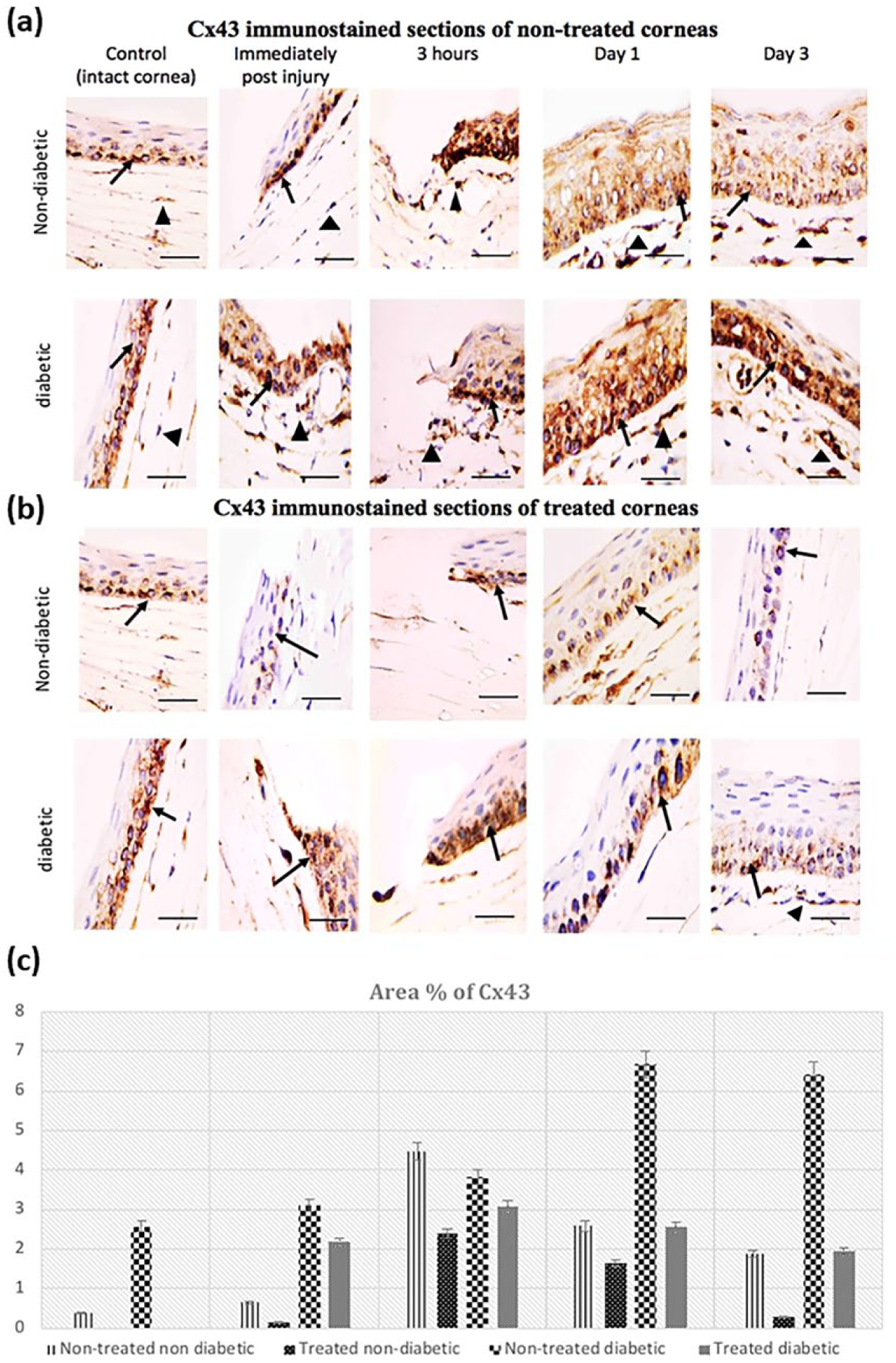
Cx43 immunoreactivity: (a) the reaction in basal layer of corneal epithelium (↑) and in some keratocytes of anterior stroma (▲) of non-treated non-diabetic and diabetic rats. The increased immunoreactivity appears more evidently in the diabetic corneas in all time points. (b) Decreased Cx43 immunoreactivity in epithelium of non-diabetic and diabetic corneas after treatment by Cx43 blocking using GAP27 eye drops. Avidin-Biotin peroxidase technique 1000×, scale bar = 25 µm. (c) Mean area % of Cx43 in corneal epithelium of non-diabetic and diabetic corneas in different time points. Significance was considered at
Of note, Cx43 area percentage was significantly higher in the diabetic corneas as compared with the non-diabetic ones (Figure 3(c)).
Treatment with Gap27 decreased cytoplasmic Cx43
Cx43 blocking by Gap27 showed mainly cytoplasmic Cx43 immunoreactivity, with few immunopositive nuclei (Figure 3(b)). Gap27 treated corneas exhibited a reduction in the overall expression of Cx43 after 1 and 3 days post injury. However, Cx43 expression was significantly higher in the treated diabetic group when compared with their matching non-diabetic corneas (Figure 3(c))
Immune reaction of keratin 12 in normal and diabetic rat corneas
The immunoreactivity of the epithelial marker Keratin 12 (K12) was used as an indicator of corneal epithelial differentiation. Cytoplasmic K12 immunoreactivity was seen in all layers of the non-diabetic control intact corneas indicating good cellular quality (Supplemental figure 1). Although K12 positive cells were detected in all layers of the diabetic intact corneas, it was significantly less (
Discussion
Diabetes contributes to numerous defects in tissue regeneration and remodeling during wound healing. Gap27 was shown to regulate wound closure rate, cell motility and accelerate the formation of fully differentiated epithelium in a previous work done by Pollok, Pfeiffer et al. (2011). Regarding the expression of Cx43 during corneal wound healing, decreased Cx43 expression is important for initiation of epithelial migration by uncoupling adjacent connections between cells. In our hands, this was confirmed by the increased expression of Cx43 in diabetic corneas as compared with non-diabetic group. Cx43 downregulation was previously shown
Supplemental Material
Figure_4 – Supplemental material for Blocking connexin 43 accelerates corneal healing and improves tissue remodeling during the healing of diabetic rat corneas: A histological and immunohistochemical study
Supplemental material, Figure_4 for Blocking connexin 43 accelerates corneal healing and improves tissue remodeling during the healing of diabetic rat corneas: A histological and immunohistochemical study by Hossein M Elbadawy, Amany Elshawarby, Mona H Raafat, Nevine Bahaa, Mohi IM Abdul, Naif Aljuhani, Saleh Bahashwan, Heba M Eltahir, Maan Albarry, Mohit Parekh and Stefano Ferrari in European Journal of Inflammation
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
According the EU directive of 2010/63/EU for animal experiments.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
