Abstract
Processing of proglucagon into glucagon-like peptide-1 (GLP-1) and GLP-2 in intestinal L cells is mediated by the prohormone convertase 1/3 (PC1/3) while PC2 is responsible for the synthesis of glucagon in pancreatic alpha cells. While GLP-1 is also produced by alpha cells, the identity of the convertase involved in its synthesis is still unsettled. It also remains to be determined whether all alpha cells produce the incretin. The aims of this study were first, to elucidate the identity of the proconvertase responsible for GLP-1 production in human alpha cells, and second, to ascertain whether the number of glucagon cells expressing GLP-1 increase during diabetes. To answer these questions, sections of pancreas from donors’ non-diabetic controls, type 1 and type 2 diabetes were processed for double-labelled immunostaining of glucagon and GLP-1 and of each hormone and either PC1 or PC2. Stained sections were examined by confocal microscopy. It was found that all alpha cells of islets from those three groups expressed GLP-1 and PC2 but not PC1/3. This observation supports the view that PC2 is the convertase involved in GLP-1 synthesis in all human glucagon cells and suggests that the regulation of its activity may have important clinical application in diabetes.
Introduction
The preproglucagon gene generates the precursor protein preproglucagon, which undergoes posttranslational processing to form glucagon like-peptide–1 (GLP-1), GLP-2 and glucagon. 1 Although the identity of the enzymes involved in the conversion of proglucagon into the three hormones has been the source of intense scrutiny, there is now general agreement that processing of GLP-1 and GLP-2 in intestinal L cells is mediated by the prohormone convertase 1/3 (PC1/3) while PC2 is responsible for the synthesis of glucagon in pancreatic alpha cells.1,2 In addition, significant functional and immunohistochemical evidence indicates that human alpha cells also produce GLP-1. However, the identity of the convertase involved in GLP-1 synthesis in alpha cells is still unsettled. Initial observations indicating that islets of mice lacking PC2 lack the ability to process proglucagon 3 and that PC2 can convert proglucagon into glucagon and GLP-1 4 support the view that this convertase is responsible for the synthesis of both hormones. In contrast, other studies suggested that synthesis of the incretin was mediated by PC1/3.5–9 It also remains to be determined whether the ability to produce GLP-1 is a trait of a subset of alpha cells, as suggested by Campbell et al, 10 or, alternatively, it is shared by all alpha cells.
The goals of this study were, first, to elucidate the identity of the proconvertase responsible for GLP-1 production in human alpha cells, and second, to ascertain whether the number of glucagon cells expressing GLP-1 increase during diabetes. For this purpose, pancreatic islets of cadaveric non-diabetic controls, type 1 and type 2 diabetes were examined using immunohistochemistry for the coexpression of glucagon and GLP-1 and of each hormone and either PC1 or PC2.
Methods
Paraffin sections of human pancreas were provided by the network of Pancreatic Organ Donors (nPOD). Immunohistochemical staining of sections was carried out essentially as previously described. 11 Rabbit antibody to PC1/3, a generous gift from Iris Lindberg (U. of Maryland), was generated by D.F. Steiner (U. of Chicago). This antibody recognizes the active 87 and 66 kDa forms of the enzyme and has been characterized in human tissues. 12 An additional rabbit antibody to PC1/3 was purchased from Abcam, Waltham, MA (Ab3532). Antibodies to PC2 were from D.S. Steiner (U. of Chicago) and from Cell Signaling, Danvers, MA (rabbit Mab #14013). Guinea pig antibodies to insulin were purchased from Linco Res (END Millipore, Burlington, MA). Rabbit ab to glucagon was purchased from Peninsula (Glu1-29, T-4357 (BMA, Basel Switzerland)). Antibody to GLP-1 (7-36) amide was purchased from Abcam, Waltham, MA (Ab# 26278). This antibody was reported to detect the C-terminal amidated of the 1-36, 7-36, and 9-36 GLP-1 isoforms but not glucagon. 10 Functional analysis confirmed that the molecule recognized by this antibody is GLP-1.6,10,13–15
Secondary antibodies: Antirabbit Alexa Fluor 488 IgG (green) and goat-anti mouse Alexa Fluor 594 (red) were purchased from Molecular Probes (Eugene, Oregon). For immunostaining, 5 µm paraffin sections were heated in 10 mmol/L citrate buffer, pH 6, for 30 min in a water bath at 100C and incubated overnight at 4C with two primary antibodies, which were produced by different hosts. The following day, sections were incubated with the corresponding secondary antibodies for 2 hr at room temperature and cover slipped.
Fluorescence microscopy: Images were capture with a Leica SP5 confocal microscope and with an Axio Observer 7/LSM 800 Confocal Carl Zeiss microscope. At least, six islets from each stained section were captured. Confocal single plane images were acquired with an Argon (488 nm) and HeNe (543 nm) laser source using an oil immersion 40× lens (Leica) or a 20× lens (Zeiss). Images were scanned sequentially to prevent crosstalk between the fluorophores. The same confocal microscope setting for laser power, photomultiplier voltage gain, offset, and pinhole value were used to collect the signals. Characteristics of the donors examined are presented in Table 1.
Table Donors.
Results
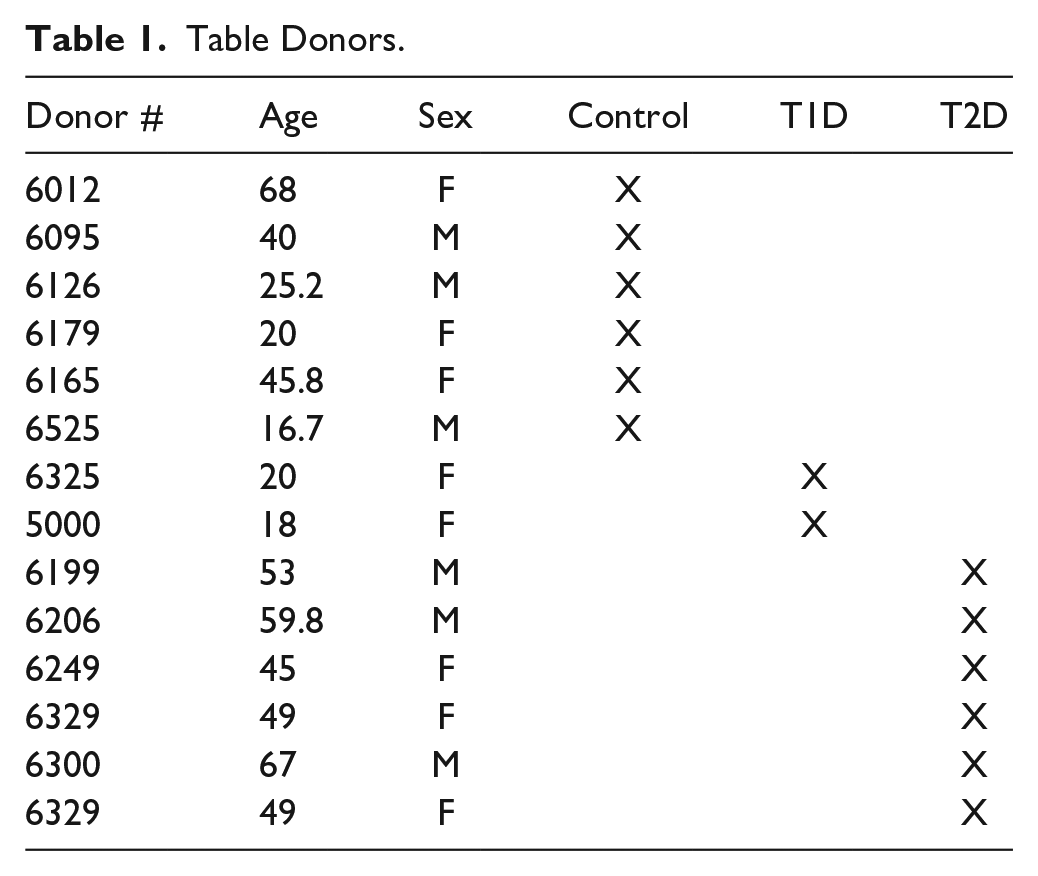
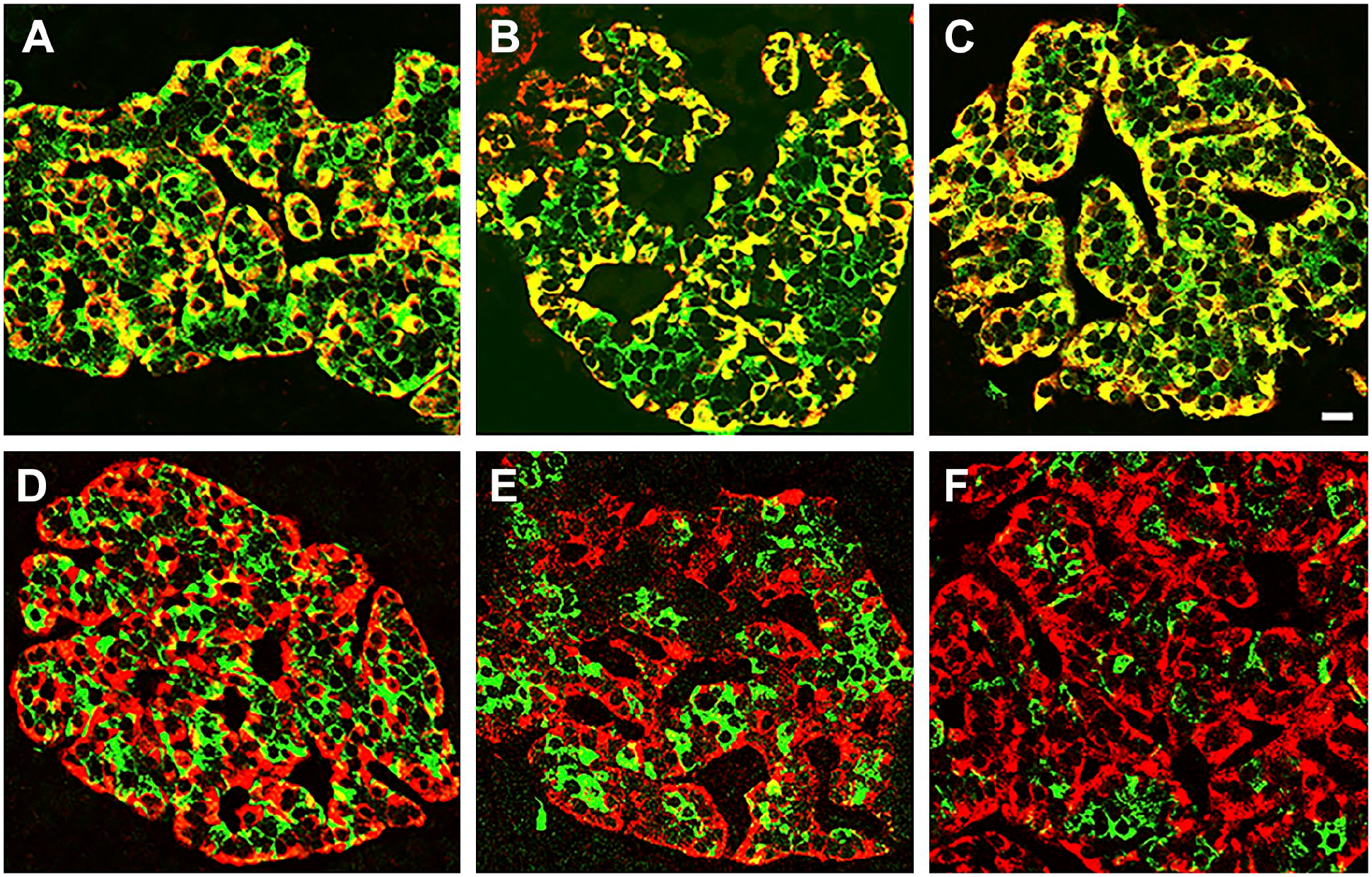
To ascertain the identity of the convertase/s involved in GLP-1 synthesis, pancreas slices from non-diabetic control, T1D and T2D cadaveric donors were immunostained for amidated GLP-1 and either PC1/3 or PC2. Results, illustrated in Fig. 1, revealed that PC2 is expressed by GLP-1+ cells of non-diabetic control (A), of Type 1 (B) and Type 2 diabetic donors (C). This result was obtained with both PC2 antibodies tested. In contrast, immunostaining for PC1/3 and GLP-1 revealed that alpha cells of islets of control (D), Type 1D diabetic (E), and most T2D diabetic (F) did not express this convertase. The exception was some islets of T2D donor #6249 which appeared to have cells coexpressing both markers. Because several studies reported the expression of PC1/3 in mouse and human alpha cells,6–10 a detailed analysis of these presumptive PC1/3+GLP-1+ cells were performed. As illustrated in Fig. 2A, some alpha cells of this islet appear to express both markers. To further ascertain whether both markers overlap in the cytoplasm of the same cell, the region containing these alpha cells was enlarged. This analysis, documented in Fig. 2A to C, indicate that the GLP-1 and PC1/3 antibodies labeled different cells that are within the same optical image, resulting in the overlapping fluorescence. Analysis of other presumptive GLP-1+ or GLU+ and PC1/3 gave similar results. It should also be noted that Steiner’s PC1/3 antibody was far more sensitive than the commercial one, providing highly precise cellular images at low concentration.
Human alpha cells express PC2 but not PC1/3− Pancreas sections from donors non-diabetic control (A and D), type 1 diabetic (B and E) and type 2 diabetic (C and F) were immunostained for GLP-1(red) and a convertase (green). Photomicrographs A, B, and C illustrate islets stained for PC2 and GLP-1. Photomicrographs D, E, and F show islets stained for PC1/3 and GLP-1. Bar: 15 µm.
Analysis of a fluorescent image produced by overlapping cells. Photomicrograph illustrates a pancreatic islet of a control donor (#6102) immunostained for glucagon (red) and PC1/3 (green). Yellow cell indicated with an arrow in (A) was enlarged and the green and red images shown in (B) and (C). Note that each label identifies a different cell, indicating that the fluorescent image in (A) is due to the overlap of two different cells. Bar in (A): 15 µm; bar in (B): 5 µm.
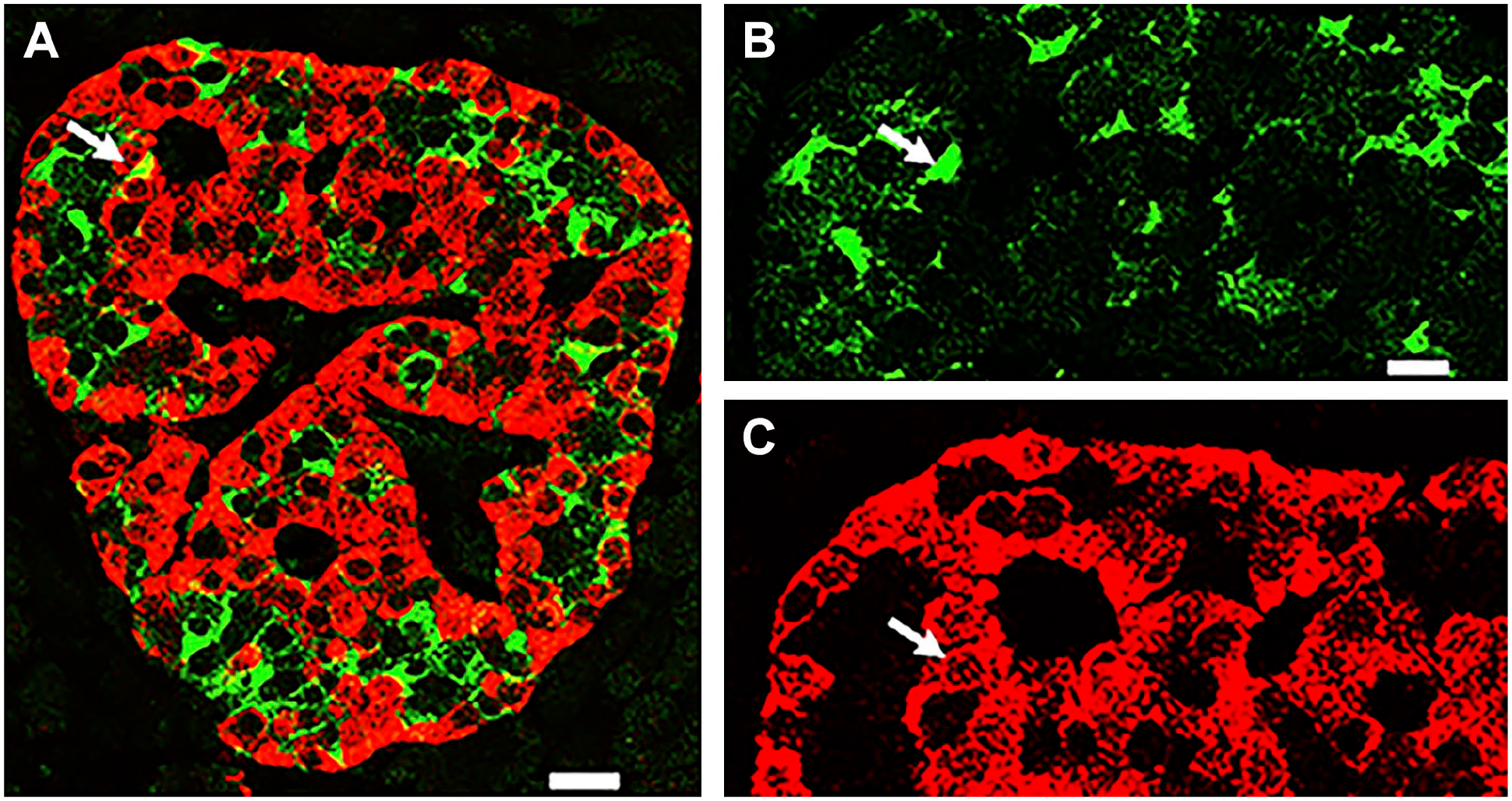
To determine whether GLP-1 is only expressed by a subset of alpha cells, slices of pancreas from donors’ adult control, Type 1 and Type 2 diabetes donors were immunostained for glucagon and amidated GLP-1. The results documented in Fig. 3 indicate that all islet glucagon cells of the three groups examined express the incretin.
All human alpha cells express GLP-1 and glucagon. Photomicrographs illustrate pancreas sections from donors’ control (A–C), Type 1 Diabetic (D–F) and Type 2 Diabetic (G–I). Sections were immunostained for glucagon (red) and GLP-1 (green). The overlap of the two labels is illustrated in Figures C, F, and I respectively. Bar: 15 um.
Discussion
The findings reported here indicate that the expression of the convertases is cell-type specific. Thus, while beta cells express both PC1/3 and PC2, alpha cells only express PC2, confirming previous observations by others. 16 These findings support the view that PC2 is responsible for the conversion of proglucagon into both GLP-1 and glucagon and confirms previous observation in mice that the lack of PC2 results in the absence of proglucagon processing. The present observations differ from previous reports that concluded that PC1/3 was expressed by alpha cells. Some the studies that used double label immunohistochemistry failed to provide evidence of overlap of both labels, which would indicate that individual alpha cells coexpress both PC1/3 and GLP-1.5,9 Other studies employed immunostaining of GLP-1 and PC1/3 in consecutive sections of mouse pancreas6,7 and then overlapped those two sections to identify coexpressing cells. However, this approach fails to distinguish between overlapping GLP-1 and PC1/3 cells, and individual cells that contained both antigens.
Our analysis, performed using double label immunohistochemistry for visualization of PC1/3 and a either glucagon or GLP-1, detected few areas with combined immunofluorescence. However, upon detailed examination of enlarged images of these areas, revealed that the mixed fluorescent signal was produced by overlying cells, each labeled with a different marker. While the present analysis indicates that alpha cells expressing PC1/3 may be extremely rare, the report of PCSK1 transcripts in isolated alpha cells 6 calls for further studies to confirm the identity of the convertase/s involved in GLP-1 synthesis.
While this study determined that all alpha cells express the incretin, two previous reports indicate that only a subset of alpha cells were Glu+Glp-1+.5,10 The fact that the present analysis and one such previous report 10 used the same antibody to GLP-1 suggest that the discrepancy between these results may be related to the sensitivity of the technical approach. Since all glucagon cells express GLP-1 and the fact that alpha cell mass appears unchanged in both Type 1 and Type 2 Diabetes,17–20 suggests that reported increase in GLP-1 secretion5,10 is likely due the surge in the levels of incretin produced per cell rather than to an increase in cell number.
The correct identification of the enzyme responsible for the synthesis of GLP-1 could lead to a better understanding of the mechanisms that regulate endogenous levels of the incretin. While GLP-1r agonists have emerged as targets for the treatment of diabetes and obesity, they also are correlated with adverse events, which appear to entail differences between GLP-1 and the incretin mimetics, in the identity of the signaling networks elicited. 21 Circumventing these problems with an islet source of GLP-1 would be of significant clinical relevance.
Footnotes
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
GT designed the study, performed the image analysis and wrote the manuscript.
Funding
The author disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The author(s) received intramural funding for the research, authorship, and/or publication of this article.
