Abstract
Islet transplantation has become an established method for the treatment of insulin-deficient diabetes such as type 1 and type 3C (pancreatogenic). An effective transplantation necessitates a thorough understanding of the islet architecture and related functions to improve engraftment outcomes. However, in chronic pancreatitis (CP), the structural and related functional information is inadequate. Hence, the present study is aimed to understand the cytoarchitecture of endocrine cells and their functional implications in CP with and without diabetes. Herein, a set of human pancreatic tissue specimens (normal, n=5 and CP, n=20) was collected and processed for islet isolation. Furthermore, immunohistochemistry was used to assess the vascular densities, cell mass, organization, and cell–cell interactions. The glucose-stimulated insulin release results revealed that in chronic pancreatitis without diabetes mellitus altered (CPNDA), at basal glucose concentration the insulin secretion was increased by 24.2%, whereas at high glucose concentration the insulin levels were reduced by 77.4%. The impaired insulin secretion may be caused by alterations in the cellular architecture of islets during CP progression, particularly in chronic pancreatitis with diabetes mellitus and CPNDA conditions. Based on the results, a deeper comprehension of islet architecture would be needed to enhance successful transplantation in CP patients:
Introduction
In the last few decades, a substantial number of individuals with type 1 and type 3C diabetes have received allogenic and autologous islet transplantations to cure diabetes. Islet transplantation plays a crucial role in regulating hormonal functions within the body and has the potential to effectively reverse diabetic conditions. After transplantation, several physiological parameters such as insufficient islet cell mass, toxicity of immunosuppressive drugs, disease recurrence, allograft rejection, quality of islets, and engraftment failure collectively reduce the survival rate of islets and hinder their metabolic functions.1,2 Even though the physiology of rodent and human islets has been extensively studied in the context of type 1 diabetes, research on type 3C diabetes remains relatively limited. The findings from rodent islets have been extrapolated to human islets, which however may not be applicable in entirety. 3 Hence, it is pertinent to examine the structural and functional aspects of human islets and their suitability for auto-transplantation in chronic pancreatitis (CP).
Islet of Langerhans act as a micro-organ that constitutes 1% or 2% of total cell mass scattered throughout the pancreas. It contains five different endocrine cell types such as alpha (α-glucagon), beta (β-insulin), delta (δ-somatostatin), pancreatic polypeptides, and epsilon cells (ghrelin).4,5 Each cell has their own typical secretory functions with paracrine and neuronal interactions.6,7 Most of the studies on rodents provide the comprehensive description of islet cell composition and structure. 8 Recent advancements in pancreatic research now recognize that the internal arrangement of human islets differs from that of rodent models.9,10 These variations may appear in vascular, core, as well as mantle arrangements of the endocrine cells.11,12 Structural arrangement of core–mantle is known to affect blood flow as well as sequential distribution of blood glucose to various cell types. 6 In addition, these arrangements are suggested to be essential in maintaining a stable cellular environment and transmitting blood-borne signals to the cells. 13 The islet non-β-cell populations first receive blood-borne signals that influence insulin secretion from the β-cells or vice versa. 6 When the islet cell mass fails to adapt to these changes, aberrations in glycemic control ensue. In CP, diabetes typically develops later and the condition14,15 can be triggered by variable degrees of inflammation, autoimmune destruction, exocrine atrophy, and fibrosis followed by endocrine insufficiency leading to diabetes. 16
In general, type 1 and type 2 diabetes are associated with insulin deficiency resulting from autoimmunity and insulin resistance, respectively, whereas type 3c diabetes exhibits a combination of both insulin deficiency and hepatic insulin resistance. 17 The most common causes of type 3c diabetes are CP, pancreatic cancer, cystic fibrosis, and other conditions that can cause inflammatory damage to the pancreas. Insulin therapy is the mainstay of treatment and islet transplantation is one of the alternative treatment strategies for both the types of diabetes. Unlike type 1 diabetes, type 3c diabetes typically develops later in life and may not have an obvious or sudden onset symptoms. In both type 1 and type 3c diabetes, the morphology of islets is altered. Hence, understanding the islet’s morphological changes in type 3c condition is crucial to comprehending the distinct etiologies and pathologies, which in turn may aid in developing diverse treatment strategies.
Patients with CP are prone to opt for interventional surgery like pancreatectomy for a variety of reasons, including persistent pain. With an increased prevalence of diabetes in CP patients, islet cell transplantation is one of the popular alternative therapies recommended by several clinicians. 18 During an islet auto-transplantation, a patient’s pancreas is excised, islets are separated, and then transplanted back to maintain blood glucose homeostasis. Thus, continuous efforts are being made to improve islet isolation processes and prevent islet loss, especially early during the post-transplant period. However, majority of transplantations have failed and the precise reasons of graft failure remain largely unknown. Considering this, the present work was envisaged to understand the cellular organization of the islet in relation to its architectural arrangements and functional aspects and their role in graft failure.
Materials and Methods
Pancreas Procurement
The CP patients (n=20) included in this study were diagnosed based on clinical, radiological, and endoscopic investigations and final confirmation was done by histopathological examination. Diabetes was defined as fasting glucose levels ≥120 mg/dl and/or glycosylated hemoglobin (HbA1c) levels ≥6.5% as per the American Diabetes Association criteria, and CP with diabetes mellitus (CPDM) and CP without diabetes mellitus (CPND) were confirmed based on these cut-off values. The unaltered and altered islets (CPNDU and CPNDA) were categorized based on their exhibited morphological changes and functions. Normal pancreas (n=5) was procured from heart-beating brain-dead donors (controls) and screened for fasting glucose levels <100 mg/dl, HbA1c levels <6.0%, and absence of any clinical history or pathological evidence of diabetes or CP. Specimens from all the subjects (control, CPND, and CPDM) were collected from the same area of the tail region (10–20 g) of pancreata upon distal pancreatectomy and were used for islet isolation and characterization. Patients below 18 years of age and those with acute exacerbation of CP, pancreatic cancer, and inability to give informed consent were excluded.
The patients who met the diagnostic criteria for type 3c diabetes, which includes individuals with exocrine pancreatic disease, where their diabetes is likely to be a consequence of their exocrine pancreatic disease, were included in this study. Selection was based on pathological pancreatic imaging, such as computed tomography scans and endoscopic ultrasonography, and excluded individuals with type 1 diabetes. All samples used in this study were obtained with the consent of the patients’ relatives and the permission of the Jeevan Daan committee (Cadaver Organ Transplantation program initiated by the Government of Telangana) in Hyderabad, India (Table 1).
Clinical Characteristics of Controls and Patients With CP.
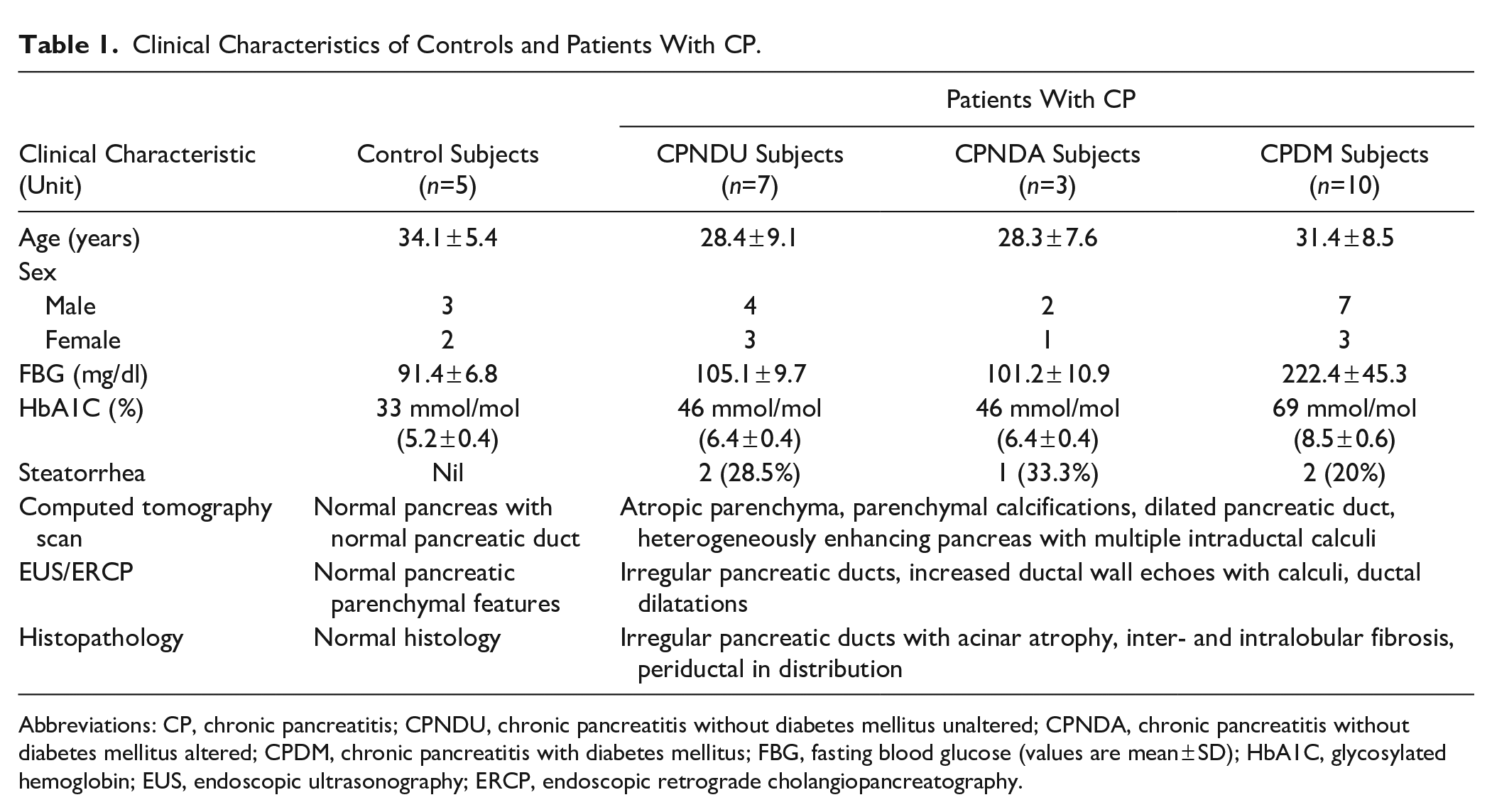
Abbreviations: CP, chronic pancreatitis; CPNDU, chronic pancreatitis without diabetes mellitus unaltered; CPNDA, chronic pancreatitis without diabetes mellitus altered; CPDM, chronic pancreatitis with diabetes mellitus; FBG, fasting blood glucose (values are mean ± SD); HbA1C, glycosylated hemoglobin; EUS, endoscopic ultrasonography; ERCP, endoscopic retrograde cholangiopancreatography.
Isolation of Islets
Islets were isolated from controls and CP patients as per our earlier protocol followed by Ricordi method with minor modifications.19 –21 Briefly, the resected pancreatic tissues were collected in University of Wisconsin solution and each tissue freed of surrounding muscle and adipose tissue. The pancreatic tissues were enzymatically digested with 1 mg/ml digestion media (collagenase V with Hank’s balanced salt solution).16,22 The process of digestion time depends on the nature of tissue fibrosis (5–10 or 10–15 min) and the process of digestion halts by using digestion stop media (cold RPMI 1640). After gradient centrifugation, the final islet pellet was cultured in complete media [CMRL with 10% fetal bovine serum (FBS) containing 5.6 mmol/l glucose with recommended antibiotics] for 48 hr at 37C in 5% CO2-humidified chamber.22 –24 The cultured islets were further processed for morphological and functional analysis. 25
Immunohistochemistry
Paraffin-embedded tissue blocks were sectioned at 5-µm interval. For each pancreas sample, a total of 60 consecutive serial sections were obtained. From each group (control, CPND, and CPDM), 150 islets were selected and examined. Hematoxylin and eosin (H&E) and Masson’s trichrome (MT) were used for the staining of tissue sections. Immunostaining of entire pancreatic islet was employed by our earlier protocol. 26 Briefly, the tissue sections were blocked with blocking buffer [4% FBS in 1× phosphate-buffered saline (PBS)] and incubated overnight at 4C with primary antibodies of anti-human insulin, glucagon, and CD34 [guinea pig anti-insulin (1:300; Sigma); mouse anti-glucagon (1:300); and rabbit anti-CD34 (1:300; Sigma, MO, USA)]. After incubation, the slides were rinsed with PBS and incubated with appropriate secondary antibodies [goat anti-guinea pig Alexa 488 for insulin and goat anti-mouse Alexa 546 for glucagon (Invitrogen, Shanghai; 1:300)] for 1 hr at 37C. For CD34, the conjugated secondary antibody (goat anti-rabbit) along with peroxidase was used (Sigma). All the slides were rinsed with PBS. After mounting, the images were captured using a CARV II bio imager (BD Biosciences) and analyzed by using IPLab software v4.0 (BD Biosciences, Rockville, MD).
Functional Assessment of Isolated Islets
Cytoarchitecture of islet and its related functions was assessed by using static method [glucose-stimulated insulin release (GSIR)].27,28 The selected islets (n=50) in each individual sample group were washed with Roswell Park Memorial Institute (RPMI) media, followed by Krebs–Ringer bicarbonate HEPES (NaCl, 120 mM; KCl, 5 mM; CaCl2, 2.5 mM; MgCl2, 1.1 mM; NaHCO3, 25 mM; and HEPES, 10 mM) containing 0.2% bovine serum albumin. The secreted insulin from isolated islets in the media in response to both basal (5.5 mmol/l) and high (16.5 mmol/l) glucose concentrations was measured by enzyme-linked immunosorbent assay method (Mercodia, Uppsala). The final results were compared with islet architecture associated with disease conditions.
Morphological Analysis
Microscopic sections of entire three-dimensional (3D) pancreatic islets and dissociated islet subpopulations (α-, β-cells) were captured using a CARV II semi-confocal bio-imager equipped with green, red, and blue fluorescence filters. The captured images were subsequently analyzed using a ProgResC5 cool camera and recorded. For image analysis, IPLab software (BD Biosciences) was utilized to evaluate the frequency of α- and β-cell distribution as well as vessel size. The distribution of α- and β-cell areas was identified by labeling with insulin (green fluorescence) and glucagon (red fluorescence) and quantified in three distinct subregions (mantle, core, and vessels) of each islet in all groups. The mantle area was precisely defined as a region of 20 µm in depth following the external perimeter of the islet. The core area was calculated by subtracting the mantle area from the total islet area. To delineate the vessel area, we relied on CD34 staining, a specific marker for endothelial cells, within the islet structure. The total islet area was measured using automated contouring of each islet.
Results were expressed as the b/(a + b) ratio, where “a” and “b” represented the α- and β-cell areas, respectively. Differences in means were calculated either by Student’s t-test and one-way analysis of variance (ANOVA). When ANOVA was applied, Schiffe least significant difference post hoc analysis was used to identify significant differences. The rationale for defining CPNDU and CPNDA groups and characterizing islet regions stems from the need for a comprehensive understanding of pancreatic islets. This holds significant clinical relevance and provides insights into diabetes progression and guiding treatment strategies, especially in the context of islet transplantation. Differentiating between CPNDU, which closely resembles nondiabetic islets, and CPNDA, representing the structural features associated with CP and diabetes, serves to illuminate significant structural distinctions. CPNDA exhibits alterations in islet structure, disrupted cell ratios, vascular abnormalities, and disturbed cell associations, thus underscoring its role as an intermediary stage between nondiabetic and diabetic islets. These distinctions are essential for the selection of optimally functioning islets, paving the way for a successful transplantation.
Results
A total of 25 patients, including control subjects (n=5; three males, 60%) and patients diagnosed with CP (n=20; 13 males, 65%), formed the study groups. Based on their glycemic status, patients with CP were categorized into those with diabetes (n=10; seven males, 70%) and without diabetes (n=10; six males, 60%; Table 1). Altered and unaltered islets were categorized based on the comparative examination of cellular organization using imaging techniques (H&E and MT staining; Fig. 1A to D).
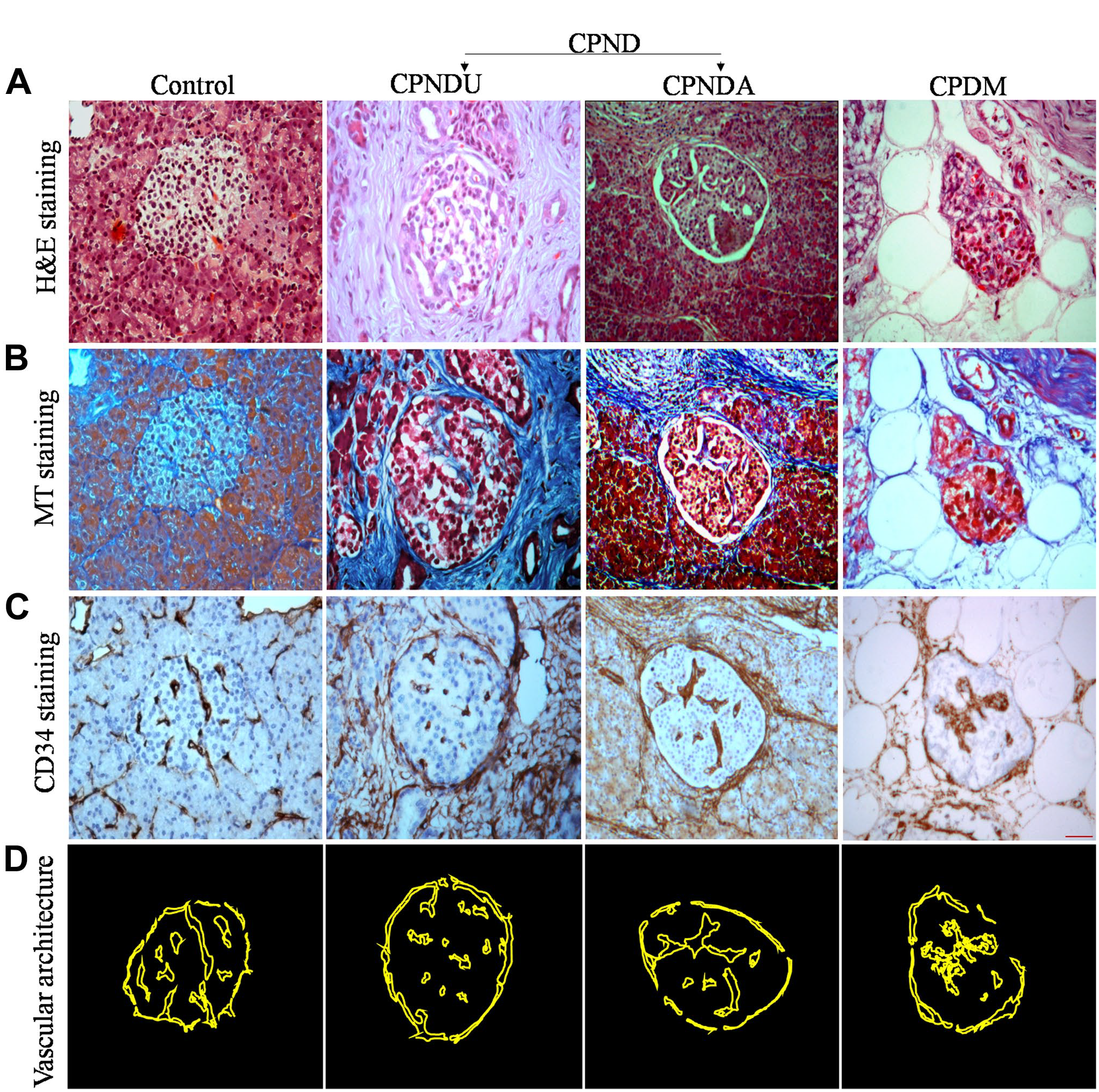
Histological examination of islet organization in control, chronic pancreatitis without diabetes mellitus (unaltered and altered), and chronic pancreatitis with diabetes mellitus cells during progression of disease in chronic pancreatitis. Hematoxylin and eosin-stained pancreatic tissue sections displayed exocrine loss by increasing trends of fibrosis along with adipose deposition (A). Masson’s trichrome-stained pancreatic tissue sections showed rising tendency of collagen fibers (B). Peri- and intra vascular endothelial cells in islets were highlighted with CD34 staining (C). Schematic showing islet vascular architecture in the examined disease samples along with control (D). Scale bar C = 20 µm. Abbreviations: CPNDU, chronic pancreatitis without diabetes mellitus unaltered; CPNDA, chronic pancreatitis without diabetes mellitus altered.
Staining of the pancreatic sections with dual stains for insulin (green for β-cells) and glucagon (red for α-cells; Fig. 2A to D) identified a segregated cell type scattering in different subregions such as whole, mantle, core, and vessels of the islets (Fig. 2E to H). The ratio of α-and β-cells, b/(a + b) ratio, was compared in all the mentioned disease categories in defined islet subregions. The α- and β-cell ratio remained similar in all the islet’s subregions in CPNDU group as compared with control, whereas in CPNDA condition, a significant reduction in the α- and β-cell ratio was observed. In CPND condition, the reduction rate of α- and β-cells in whole, mantle, core, and vessels parts of the islet was 19.1%, 27.5%, 3.2%, and 25.8%, respectively. The cell ratio in CPDM group was further diminished (except in mantle) in all the subregions of islet compared with control and CPNDA condition. In comparison to the control group, the reduction rate in CPDM condition was 26.9%, 21.8%, 7.6%, and 45.8% in the islet subregions of whole, mantle, core, and vessels, respectively (Fig. 2I). The results suggest that the α- (a) and β- (b) cell ratios gradually decreased over time in CPDM. The reduction of cells varied depending on the area of the islet; in all conditions, a greater reduction was seen in the islet’s vascular portion, whereas a lesser reduction was seen in the core region.
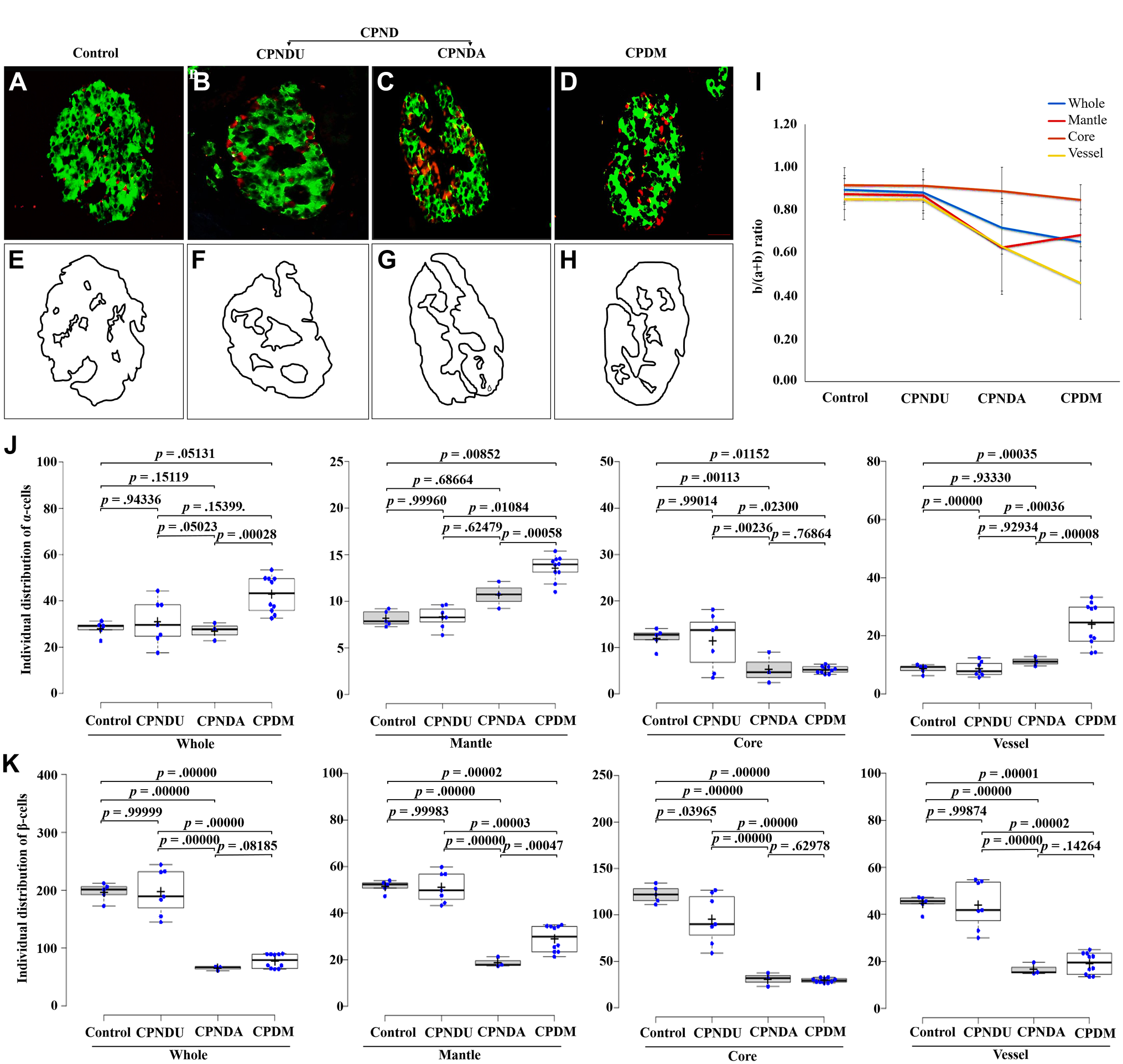
Distribution of α- and β-cells in different islet subregions among different test samples. Pancreatic islet labeled by immunofluorescence for insulin (green) and glucagon (red; A–D). Schematic lines (black lines) represent mantle, core, and vessel areas of immunofluorescent islet images of A to D (E–H). Ratio of α- and β-cells distribution in different islet subregions (whole, mantle, core, and vessel) among different test samples (I). Distribution of individual α-cells and β-cells in diverse islet subregions among different categories investigated (J and K). The results for distribution studies were expressed as b/(a + b) ratio, where a = α-cells and b = β-cells. The average was calculated for all the islets from a single subject. The results are expressed as mean ± SEM. A total of 450 islets were taken from 25 pancreata from all the groups; n=5 pancreata (control), n=7 pancreata from chronic pancreatitis without diabetes mellitus unaltered, n=3 pancreata from chronic pancreatitis without diabetes mellitus altered, and n=10 pancreata from chronic pancreatitis with diabetes mellitus. Bar graph of different groups showing statistical similarities and differences according to analysis of variance with post hoc test. Significant p values are depicted in the image. Scale bars A–D = 20 µm. Abbreviations: CPND, chronic pancreatitis without diabetes mellitus.
In addition, we looked at the apparent islet size-based distribution of α- and β-cells population in CPND and CPDM conditions. The segregation of both α- and β-cells was measured in three different subregions of each islet at various sizes (Fig. 2J and K). The 3D analysis of islet at various sizes showed the multiple vascular channels in it (Fig. 3A to C). Furthermore, the α- and β-cell ratio at the same diameter did not show any significant differences (p<0.05) among the control, CPNDA, CPNDU, and CPDM groups. However, islets with a diameter of 100–150 µm and above exhibited multiple ramified empty areas that were significantly increased in the CPDM group compared with the control group.
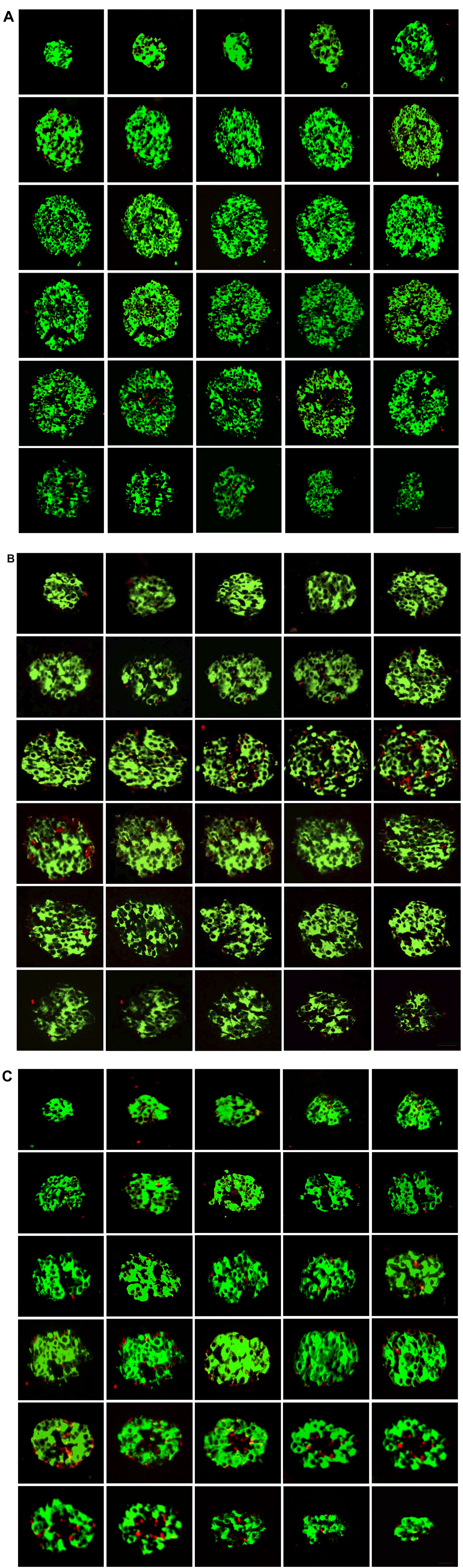
Histological three-dimensional (3D) representation of entire islet cell organization. Consecutive sections through an entire pancreatic islets labeled for α-cells (red) and β-cells (green) are organized into continuous 3D networks that span the entire islet of control (A), chronic pancreatitis without diabetes mellitus altered (B), and chronic pancreatitis with diabetes mellitus (C). The islet profile shows a deceptive similar core–mantle structure to that of 20 μm in diameter. These series of optical sections through an entire islet reveal continuous ramified structures with vascular channels. The frequency of heterologous contacts between α- and β-cells was scored, and the results show a higher frequency in the progression of disease in chronic pancreatitis.
The α- and β-cell ratio gradually decreased in the CPDM and CPNDA as compared with control and CPNDU group. Almost 13.4% reduction was observed in CPNDA and 33.6% reduction was observed in CPDM at 100–150 µm size. The decreasing trend of α- and β-cell ratio was observed with increasing islet diameter (150–200, 200–250, and >250 µm) in all the conditions. The depletion of α- and β-cell ratio at islet size diameter 250–300 µm exhibited 46% and 53.2% in CPNDA and CPDM groups, respectively (Fig. 4A).
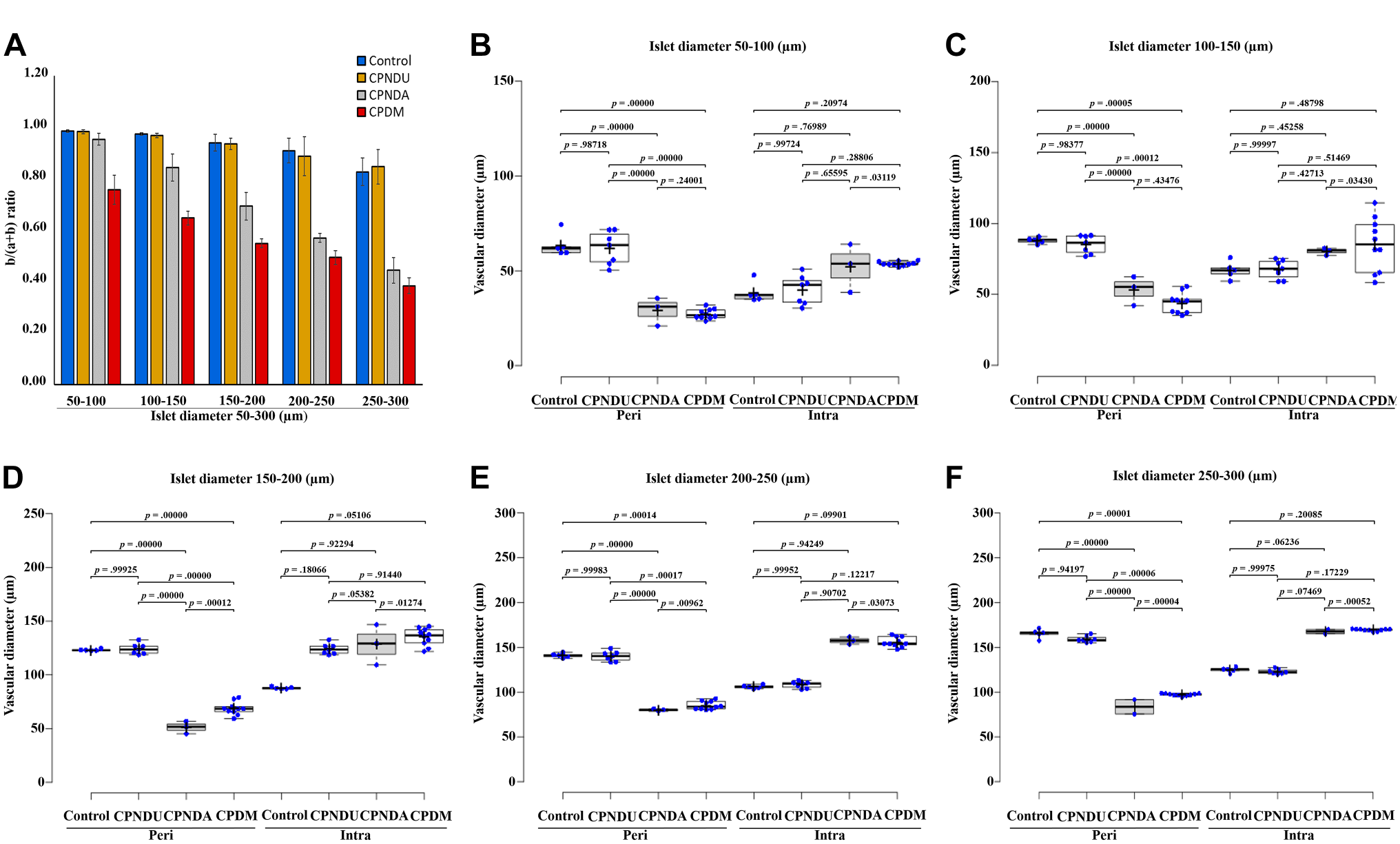
Islet apparent size-based regional distribution of α- and β-cells, along with vascular diameter in control, chronic pancreatitis without diabetes mellitus unaltered, chronic pancreatitis without diabetes mellitus altered, and chronic pancreatitis with diabetes mellitus cells. After immunofluorescence, β-cell (green) and α-cell (red) areas in islets were labeled and b/(a + b) ratio was calculated, where a and b represent glucagon- and insulin-labeled areas, respectively (A). Alteration of peri- and intra vascular diameter at various regions of islet size (B–F). Distribution studies were carried at different islet size diameters ranging from 50–100, 100–150, 150–200, 200–250, and 250–300 µm (B–F). Each experiment was performed in triplicates and the results are represented as mean ± SD. One-way analysis of variance was used for the analysis of results. The post hoc analysis revealed that the results of statistically significant p values are depicted in the image.
Assessment of vascular channels (endothelial cells) revealed that there was no altered vascularity in islet size diameter with ˂50 µm. However, as the islet size diameter increases, there is an increasing trend of intravascularity observed, especially in the CPDM and CPNDA groups, whereas the perivascularity was diminished in CPDM and CPNDA groups as compared with control and CPNDU groups. The islet’s intravascularity was increased 39% in CPDM and 35% in CPNDA at 50–100 µm diameter.
Furthermore, the width of intravascular channels (α-cells lineage on ramified empty areas) increased by 26–54% in the CPDM group and 19–48% in the CPNDA group in almost all islet sizes (100–150, 150–200, 200–250, and 250–300 µm). Conversely, the perivascular size decreased in disease conditions as the islet size increased. A decline of 39–57% in CPDM and 39–58% in CPNDA was observed across increasing islet diameters. The vascularity of islet endocrine cells was dramatically altered in disease conditions, with intravascularity increasing and perivascularity declining (Fig. 4B to F).
Immunofluorescence staining was carried to gain a thorough understanding of islet cell organization (homologous and heterologous interactions) and their alignment along with vascular lineages (Fig. 5A to H). The results of cell organization revealed that, there are more heterologous contacts between α- and β-cells around vessels than homologous contacts. When we examined the association of α- and β-cells in islets, we found that many α-cells were wrapped by β-cells in the CPNDA and CPDM. Among the examined, the heterologous contacts such as β-cells wrapping α-cells showed a reduction of 65.9% in CPNDA condition and 18.4% reduction in CPDM, whereas α-cell wrapping β-cell population was gradually amplified in CPDM and CPNDA. The frequency of α-cell wrapping β-cell was increased to 13 folds in CPDM condition and 8 folds in the CPNDA group as compared with control (Fig. 5M).
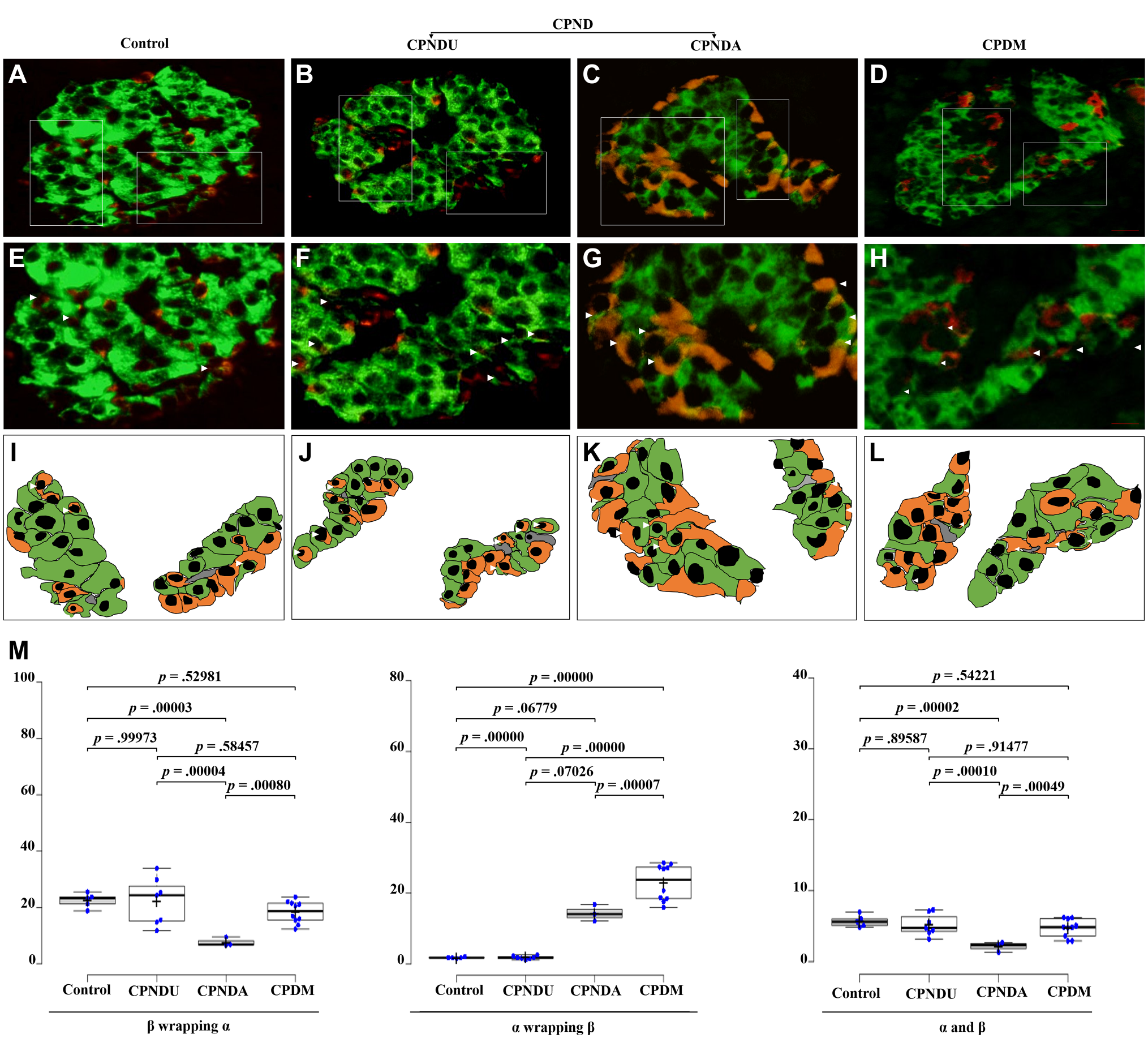
Wrapping association between α- and β-cells in pancreatic islets of control, chronic pancreatitis without diabetes mellitus unaltered (CPNDU), chronic pancreatitis without diabetes mellitus altered (CPNDA), and chronic pancreatitis with diabetes mellitus (CPDM). Islets immunolabeled for glucagon (red, α-cells) and insulin (green, β-cells) were captured at low magnification (A–D). Boxed areas are shown at higher magnification (E–H), depicting the α- and β-cells lining the mantle and vascular channels (I–L). Arrowheads indicate the wrapping associations between the α- and β-cells. The frequency of heterologous contacts between α- and β-cells was scored based on their association (M), which includes β wrapping α, α wrapping β, and neutral apposition between α- and β-cells (α–β). All experiments were performed in triplicate, and the relative frequencies are represented as mean ± SEM. The wrapping association between the cells of β wrapping α, α wrapping β, and α-, β-cells showed high significance (p<0.00035, p<0.00001, and p<0.000042, respectively; control vs CPNDU vs CPNDA vs CPDM). 1 graphs show statistical differences according to analysis of variance post hoc test. Scale bars A–D = 20 μm; scale bars E–H = 40 μm.
Functional assessment of islet architecture in terms of insulin secretion was investigated using GSIR assay at two different time intervals such as 0 and 12 h. Both low (basal: 5.5 mmol/l) and high glucose (stimulant: 16.5 mmol/l) concentrations were used for insulin secretion. The results revealed that cytoarchitecture of the islet in CPNDA conditions has functional implications and suggested increasing trends of insulin secretion in response to basal glucose concentration. As compared with control, the insulin secretion was raised nearly 24.2% in CPNDA condition at basal glucose concentration (Fig. 6A), whereas in CPNDU and CPDM conditions, the insulin secretion was not changed significantly. At high glucose (stimulant) concentrations, the insulin secretion was significantly decreased in CPNDA and CPDM conditions. In comparison to the control, insulin secretion was reduced by 77.4% and 78% in the CPNDA and CPDM conditions, respectively (Fig. 6B), whereas in CPNDU group, 26.8% reduction of insulin secretion was noticed (Fig. 6).
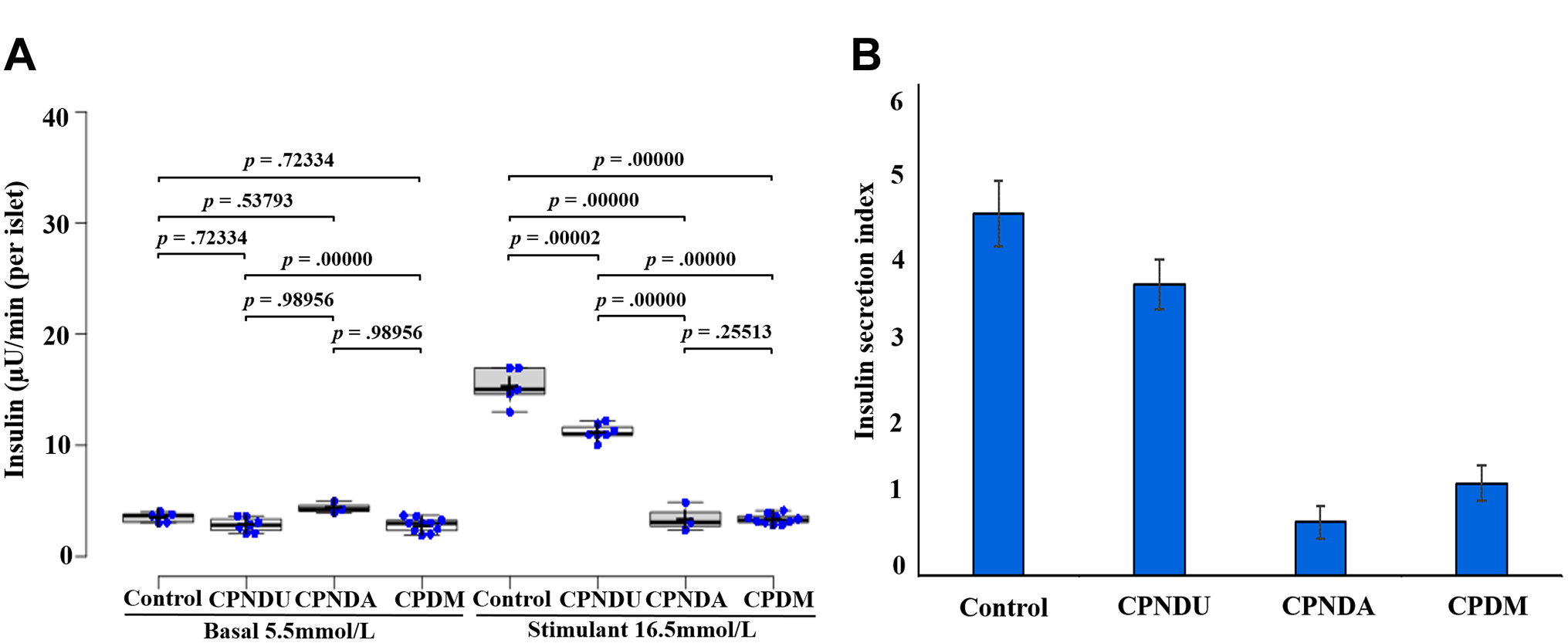
Islet functional observations in control and chronic pancreatitis groups. Islets were isolated from control, chronic pancreatitis without diabetes mellitus unaltered (CPNDU), chronic pancreatitis without diabetes mellitus altered (CPNDA), and chronic pancreatitis with diabetes mellitus (CPDM) groups. Islets from the three groups were stimulated with glucose [5.5 mmol/l (low glucose/basal) and 16.5 mmol/l (high glucose/stimulant)] for 1 hr (A). Insulin secretion was determined by enzyme-linked immunosorbent assay. Index of insulin is depicted in (B). The results are expressed as mean ± SEM (n=50 islet equivalency (IEQ) islets from each group). All experiments were performed in triplicate. The functional observations of basal and stimulant conditions were analyzed. The statistical significance (basal: p<0.630706; stimulant: p<0.00001) of the results was confirmed using analysis of variance post hoc analysis.
Discussion
The quality of life for a CPND patient can be significantly improved by an effective islet transplantation.29 –31 Islet cells, after transplantation, resume their function of releasing insulin and maintaining blood sugar levels. Previous studies have shown the beneficial effects of auto-transplantation of islets in CP patients. 32 Nevertheless, early islet function failure33,34 and insulin dependence were reported in a few patients, and the underlying causes are not clear. 35 Furthermore, the cellular and subcellular arrangement and composition of islets in CP patients have not been thoroughly investigated. Therefore, this study was conducted to gain a better understanding of the structural changes in islets and their impact on functional properties in patients with CP. We conducted a comparative investigation of islet cellular structure, composition, and related functional effects in CPNDA and CPDM patients to achieve effective islet cell transplantation.
Species-specific differences in islet composition and architecture exist, 36 with the human islet structure being different from animal models. 36 These differences can also exist within the same species under normal and diseased conditions. 9 CP is a persistent inflammatory condition that can cause islet cell dysfunction and subsequently lead to the development of diabetes. 15 Therefore, we examined the islet structural alterations and composition in CP with and without diabetes. The study provides new insights into the role of islet structure in the development of diabetes in CP patients. Furthermore, we have adopted various methods to analyze the islet structure and function, which increases the validity of the results. The histological representation of islet organization shows increasing trends of fibrosis with adipose deposition in CP. The islet is a highly vascularized micro-organ due to its endocrine role. 37 Hence, we performed CD34 staining, and the results revealed irregular morphological abnormalities in peri- and intra vascular endothelial cells in CP compared with normal cells. Our findings demonstrate that the islet vascular endothelial cells were segregated, fragmented, and condensed in CPNDA and CPDM patients (Fig. 1C). The increased intravascular density in CP may be associated with dysglycemia and lead to β-cell loss. In general, the loss of β-cell function could impair vascular endothelial growth factor signaling that leads to the development of diabetes. 38 Our findings support previous research that has emphasized endothelial dysfunction and alterations in diabetic conditions.39,40
It is known that the structural arrangement of the core–mantle influences blood flow and the sequential delivery of blood glucose to islet cells. 6 Furthermore, these configurations are shown to be critical in maintaining a stable cellular environment and insulin secretion. In this study, we compared the islet architecture and composition in defined islet subregions (whole, mantle, core, and vessels) in CPND and CPDM patients (Fig. 2A to K). The results showed a reduction in the ratio of α- and β-cells in both CPNDA and CPDM conditions, but the reduction was higher in CPDM than in CPNDA (except in the mantle) in all the islet subregions. Our results suggested that the α- and β-cell ratio gradually decreased. The vascular component of the islet showed the greatest decrease, whereas the core area, the least decrease in CPNDA and CPDM conditions. The overall α-cell population was greatly enhanced, and the β-cell population was diminished. Earlier studies suggested that diabetes-related changes in α-cell architecture could cause abnormal glucagon secretion and contribute to the pathophysiology of diabetes.6,41 The elevated α-cell population is well recognized in type 1 diabetes. 42 An increased α-cell population and a decreased β-cell population in the vascular region were observed in CPNDA and CPDM conditions, which could contribute to the development of diabetes. The major concern of the study was to focus on CPNDA, an intermediate condition of diabetes progression in CP that has received relatively less attention in the literature. Hence, we have carried a comparative analysis between CPNDA and CPDM conditions, which allows for a better understanding of the differences in islet structure and function.
Furthermore, our findings indicate that the distribution of α- and β-cells based on the apparent islet size is altered in CP patients. As the islet diameter increases, the ramified empty areas also increase. The α- and β-cell ratio gradually decreases in both CPNDA and CPDM groups (Fig. 4A). Assessment of vascular channels showed that islets with a size diameter of <50 µm did not show altered vascularity in CP conditions. However, in islets with a size diameter >50 µm, aberrant α- and β-cell ratios and increased vascular endothelial density were observed in CPNDA, CPNDU, and CPDM patients. Detailed observations of the vascularity of islet endocrine cells showed an increased intravascularity and a decreased perivascularity in CPDM and CPNDA groups (Fig. 4B). These findings are consistent with earlier reports indicating that vascular endothelial dysfunction is an early indication of diabetes. 43 Human islets have a unique architecture that allows all endocrine cells to be located close to blood vessels, promoting interactions between α- and β-cells. Previous studies have shown that glucagon has a positive effect on insulin release through heterologous contacts between α- and β-cells, which are prevalent in human islets.44 –46 Furthermore, the physical proximity between different α- and β-cells is physiologically important as these cells are likely to communicate through paracrine signaling to maintain glucose homeostasis. 47 It is well established that the potential for physiological heterologous contact between α- and β-cells is essential for proper paracrine interactions and glucose homeostasis. 48 In this study, we investigated the heterologous associations in CP progression and found that they are significantly altered. Our results indicated that the density of β-cells wrapping around α-cells decreased, and the density of α-cells wrapping around β-cells increased in CPNDA and CPDM groups, respectively. The increased density of α-cells wrapping around β-cells in CPNDA may result in islet functional abnormalities. Our findings suggest that these modifications could impact insulin secretion.
Overall, the aforementioned findings about islet function reveal that the distinct cellular architecture of CP islets has measurable functional effects. Our study showed that the altered cytoarchitecture of the islet in CPNDA had functional consequences, which were supported by rising trends in insulin production in response to baseline glucose concentration. The functional assessment through GSIR interval measurements demonstrated that islets under CP progression modify both insulin sensitivity and secretion. In addition, in our earlier study, we reported that a low number of β-cell density may lead to inadequate insulin production. In the current investigation, an increased α-cell wrapping β-cell density in the CPNDA condition was noted. Collectively, both the increased α-cell wrapping β-cell density and decreased β-cell population may contribute to the progression of diabetes in CP patients. The internal islet composition and structure may have a role in the pathogenesis of diabetes. Therefore, focusing on islet structural alterations and associated functional values might be a better alternative for achieving successful transplantation results. Pancreatic islet transplantation is a promising therapy that is rapidly evolving for individuals with CP. Currently, efforts are being made to improve the quality of transplantation by focusing on quality and long-term functionality of the islet. Certainly, islet isolation, architectural assessment, functional analysis, and selection of suitable islets are the critical parameters that played a significant role in islet transplantation. Our findings suggest that the islet subregions such as vessels, core, and mantle may play a major role in the functioning of islet. Furthermore, the unique arrangement between α- and β-cells is vital for the function of transplanted islets. The examination of islet cytoarchitecture in CP is critical and may aid in the selection of functional cells for efficient transplantation.
Footnotes
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
PPK designed the study, executed the experiments, carried out the data analysis, and drafted the manuscript; GVR revised the manuscript and provided important intellectual insights; MS provided clinical assistance for samples and interpretation of data; RP revised the manuscript; CPV performed immunohistochemistry; MS revised the manuscript; and DN provided intellectual insights in the design of work.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors would like acknowledge to the DST-SERB for the financial assistance in the form of research grant (CRG/2021/005760).
