Abstract
Photodynamic therapy (PDT) is an effective and cosmetically beneficial treatment of low-risk basal cell carcinomas (BCCs). To optimize PDT response, it is important to correctly select tumors. We sought to find markers that could identify such tumors beyond contributions from clinical and histological examination. Studies have shown that β-catenin, E-cadherin, and α-smooth muscle actin (SMA) expression can indicate BCC aggressiveness/BCC invasiveness. We wanted to use these markers in an explorative study to investigate whether they were differently expressed among non-recurring compared with recurring BCCs, to evaluate their ability of predicting PDT outcome. Fifty-two BCCs were stained with antibodies against β-catenin, E-cadherin, and α-SMA, and evaluated using immunoreactive score (IRS), subcellular localization, and stromal protein expression. Results showed that IRS of E-cadherin was significantly different among recurring compared with non-recurring BCCs and with area under a receiver operating characteristic curve of 0.71 (95% confidence interval: 0.56–0.86, p=0.025). Stromal β-catenin expression significantly increased among recurring BCCs. Some recurring BCCs had intense expression in the deep invading tumor edge. In conclusion, E-cadherin, and stromal and deep edge β-catenin expression were most prominent in BCCs that recurred post-PDT, suggesting they could potentially predict PDT outcome. Further studies are needed to investigate whether these results are of clinical value:
Introduction
Basal cell carcinoma (BCC) is the most common type of skin cancer in the White population worldwide and has a high and rising incidence. 1 Even though it grows slowly, BCCs can have local invasive properties that might cause significant tissue destruction and patient morbidity and place a large burden on health care systems.1,2 BCCs have different clinical and histological characteristics that help divide them into high- and low-risk types. Histologically, BCC is usually classified into superficial, nodular, and aggressive subtypes. 3 Low-risk BCCs are typically small-sized primary tumors located outside the mid-face-area and of histologically superficial or nodular subtypes. 4 Surgical excision is often regarded as the preferred treatment, but other, less invasive treatment options are available for low-risk BCCs.
Photodynamic therapy (PDT) is such a treatment and is effective and cosmetically advantageous with few side effects. 5 It is based on red light activation of a topical photosensitizer that in the presence of oxygen forms reactive oxygen species (ROS) that selectively by necrosis and apoptosis destroy tumor cells. 6 The treatment is limited by the depth of penetration of the photosensitizer and red light. 7 Although PDT has a high 3-month response rate of 91–92% for BCCs, it can be reduced to 63% by 5-year follow-up after treatment.6,8 To date, subtype and thickness are the most important histological factors for selection of BCCs for PDT. 6 Despite careful selection of tumors, treatment failure among low-risk BCCs has led to a search for other predictive markers on PDT outcome, including immunohistochemical (IHC) markers.9,10 IHC markers reflect protein expression which can potentially provide important information about how cancer cells will respond to treatment and thereby give additional information that can assist in the selection of BCCs for PDT. 11 Some studies have shown that changes in protein expression of β-catenin, E-cadherin, and α-smooth muscle actin (SMA) can be used as a sign of aggressiveness and local invasiveness in BCCs.12–16 We wanted to investigate this further by using these markers in an explorative study to see whether they are expressed differently in BCCs that were later followed up after PDT, to see whether they can serve as predictors of PDT outcome.
The primary objective was to investigate whether possible differences in protein expression of β-catenin, E-cadherin, and α-SMA in BCC tissue obtained before PDT and later found to be non-recurring and recurring could be used as predictors of PDT outcome. The secondary objective was to investigate whether there were differences in subcellular localization and stromal protein expression of these markers between non-recurring and recurring BCCs.
Materials and Methods
The biopsy material emanates from a previous clinical dimethyl sulfoxide (DMSO) supported 5-aminolevulinic acid (ALA) PDT study of 60 BCCs with 72 months follow-up in which the main outcome was the complete response rate of treated tumors. 17 A 2 to 3 mm punch biopsy was taken from each tumor before treatment. The results from this study provided information on which tumors recurred after PDT treatment.
In the present study, the original formalin-fixed paraffin blocks were retrieved from the Department of Pathology at St. Olavs Hospital and prepared at the Cellular and Molecular Imaging Core Facility, at the Norwegian University of Science and Technology. Histological examination was performed by one pathologist (PM), and BCCs were subclassified into superficial, nodular, and aggressive types. 3 Typically, superficial BCCs grow in small buds from the epidermis into the superficial dermis, while maintaining their attachment to the base of the epidermis. Nodular BCCs grow as rounded masses within the dermis, typically with peripheral palisading and a surrounding retraction artifact. Aggressive BCCs typically show groups of cells of various sizes with irregular outlines and pointy projections that infiltrate collagen bundles in the dermis. Peripheral palisading of tumor cells is often absent. This study was approved by the Regional Committees for Medical and Health Research Ethics Central in Norway, number 2011/1868, and patients provided written informed consent.
Immunohistological Protocol
To preserve the tissue well, paraffin sections of 4 µm were applied on Superfrost Plus slides (Thermo Fisher Scientific, Germany) and dried overnight at 37C, and then stored in a freezer (–20C) from 2018 until staining in November 2021. After being taken out of the freezer, three slides from each tumor were thawed for 1 hr at 60C in accordance with the established procedure of our laboratory. Sections were then deparaffinized with TissueClear for 15 min and then rehydrated in decreasing concentrations of ethanol (100%, 96%, and 80%) followed by rinsing in tap water for 5 min. Heat-induced epitope retrieval was performed in target retrieval solution pH 9 for 20 min at 97C and then cooled to 65C, conducted by PT Link (DAKO Denmark A/S; Glostrup, Denmark). After incubation of primary antibodies, sections were treated for 10 min in DAKO Wash Buffer S3006, 1:10 (DAKO Denmark A/S). Immunodetection was done using DAKO Autostainer Plus (DAKO Denmark A/S), incubated at room temperature. All steps of the immunostaining process were done in the autostainer.
To prevent endogenous peroxidase activity, each slide was exposed to Dako REAL Peroxidase Blocking Solution (S2023) and then washed in Dako Wash Buffer S3006 1:10. Slides were incubated with primary antibody at room temperature diluted with Dako REAL Antibody Diluent S2022. Info on each antibody is shown in Table 1. This was followed by secondary antibody incubation for 30 min using HRP Rabbit/Mouse EnVision—Polymer from Dako REAL EnVision Detection System K 5007. Slides were then developed using DAB+ Chromogen from Dako REAL EnVision Detection System and then washed two times in deionized water. The slides were then removed from the autostainer and contrast stained in an automated expression machine (Tissue-Tek Prisma) using Hematoxylin for 30 sec and then dehydrated in increasing concentrations of ethanol (80%, 96%, and 100%). Cover glass was then attached to slides using Tissue-Tek Prisma Glas g2 with TissueMount (Sakura Finetek, Netherlands) as a medium.
Details of the Primary Antibodies Used in This Study.

Abbreviation: Ig, immunoglobulin.
Control tissues were chosen based on recommendations from the antibody manufacturer and reviewing recommended tissues from Human Protein Atlas (https://www.proteinatlas.org/). Duodenum, liver, and tonsillar mucosa were positive control tissues for β-catenin, E-cadherin, and α-SMA, respectively. Cerebellum was negative control tissue for all three antibodies. To determine whether nonspecific binding occurred, a negative reagent control (NRC) with omitted primary antibody was used on an additional slide. The NRCs for α-SMA and β-catenin were Mouse IgG2a kappa isotype control, monoclonal (Antibodies -online Inc.), and for E-cadherin Mouse IgG1 kappa isotype control, monoclonal (Antibodes-online).
Scoring and Reporting
Slides were evaluated using a Nikon Eclipse 80i upright light microscope (Japan). The stained sections were examined by two investigators (PM and EM), and each tumor was given an immunoreactive score (IRS). IRS is a recognized scoring system for immunoreactivity 18 and is defined as the product of the percentage of positive tumor cells and the intensity of expression in tumor cells. The percentage of positive cells was scored as <1% (0), 1–10% (1), 10–50% (2), or >50% (3), while intensity was scored as no expression (0), weak (1), moderate (2), and strong (3). This gave an IRS scale from 0 to 9. The investigators also reported cellular location of expression (cell membrane, cytoplasm, and/or nucleus) and presence of stromal expression (yes/no). Stromal expression was defined by the investigators as positive staining in the connective tissue adjacent to tumor cells.
Two investigators (PM and EM) evaluated each slide separately and were blinded to treatment outcome. Consensus was reached in situations of disagreement.
Statistical Analyses
All analyses were carried out using SPSS Statistics (Version 28, IBM Corp., USA). Descriptive statistics were used to count the number of BCC subtypes, and non-recurring and recurring BCCs. Histograms of IRS for β-catenin, E-cadherin, and α-SMA were evaluated not to be normally distributed. The IRS results are presented in box plots.
The IRS scale of 0 to 9 was based on products from the percentage of cells and intensity; this was recoded to a scale of 0 to 7 to reflect the number of possible product outcomes. The mean differences in IRS between non-recurring and recurring BCCs were tested using the Mann–Whitney U test. Receiver operating characteristic (ROC) analyses were used to assess the ability of predicting PDT outcome to be non-recurring or recurring based on differences in IRS-protein expression. Areas under the curves (AUC) were calculated, presented with 95% confidence interval (lower–upper), and statistically tested to see if the areas were larger than 0.5.
Fisher’s exact test was used to test differences in cellular location of expression or presence of stromal expression. Observed and expected values in the 2 × 2 table were compared to evaluate the direction of significance. The statistical significance level was set as <0.05.
Results
Fifty-two BCCs were included, of which 39 were non-recurring and 13 were recurring tumors. Among recurring BCCs, there were histologically three superficial, four nodular, and six aggressive BCCs. Of non-recurring, there were histologically 9 superficial, 12 nodular, and 18 aggressive BCCs.
IRS and ROC Analyses for Non-recurring and Recurring BCCs
Figure 1 shows box plots of the results of IRS for β-catenin, E-cadherin, and α-SMA among non-recurring and recurring BCCs. The mean and mean difference of IRS and their respective p values are shown in Table 2. Figure 2 shows the results from the ROC curves. The AUCs were as follows: β-catenin had 0.59 (0.41–0.77, p=0.320), E-cadherin had 0.71 (0.56–0.86, p=0.025), and α-SMA had 0.61 (0.43–0.78, p=0.248). Figure 3 shows low and high IHC expression of the three markers in six BCCs.
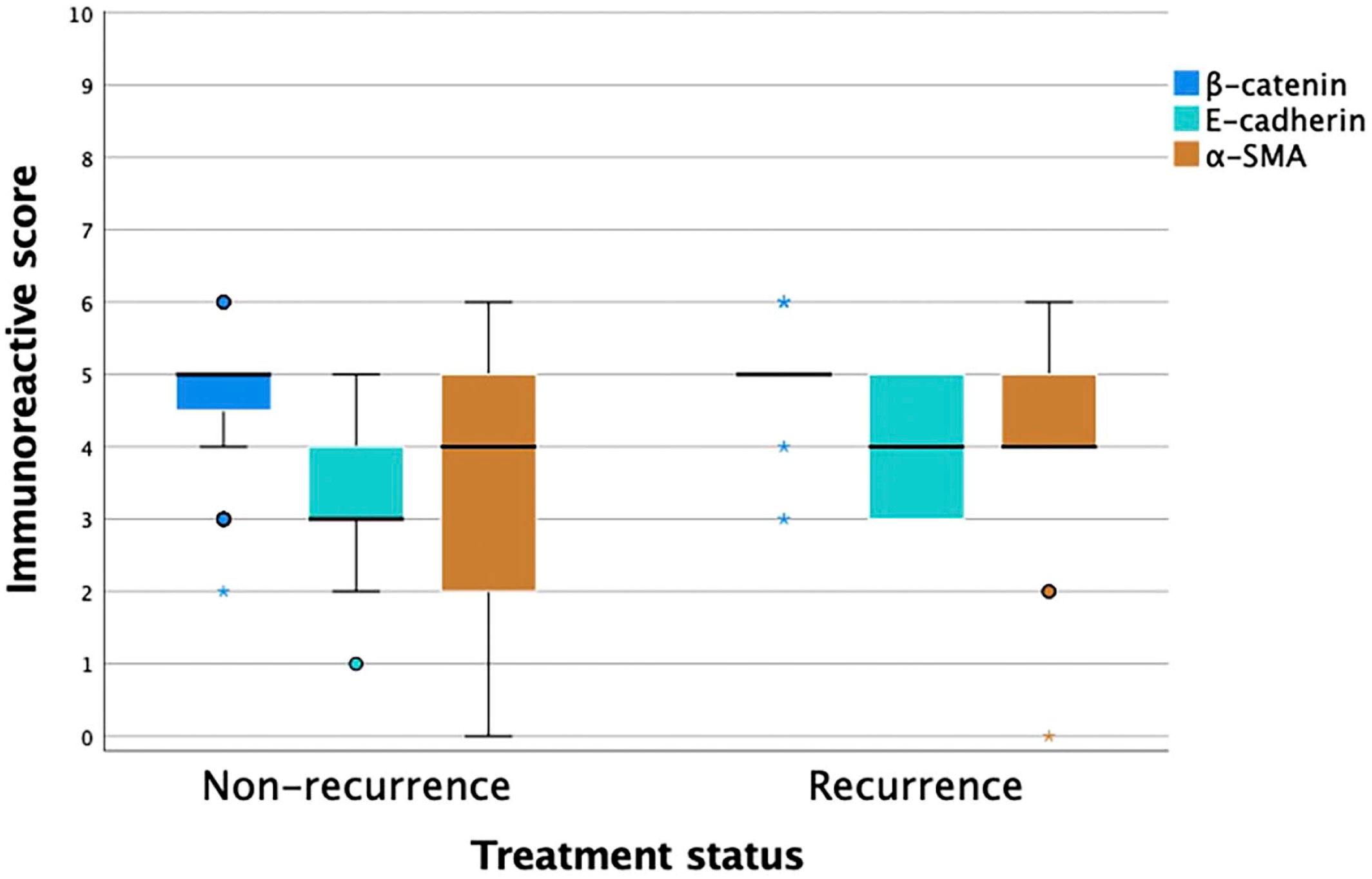
Box plot of immunoreactivity score for β-catenin, E-cadherin, and α-SMA among non-recurring and recurring BCCs. Circles represent “out” values and asterisks represent “far out” values. Abbreviations: BCC, basal cell carcinomas; SMA, smooth muscle actin.
Difference in IRS of Tumor Cells and Stromal Expression of Non-recurring and Recurring BCCs for β-Catenin, E-Cadherin, and α-SMA.
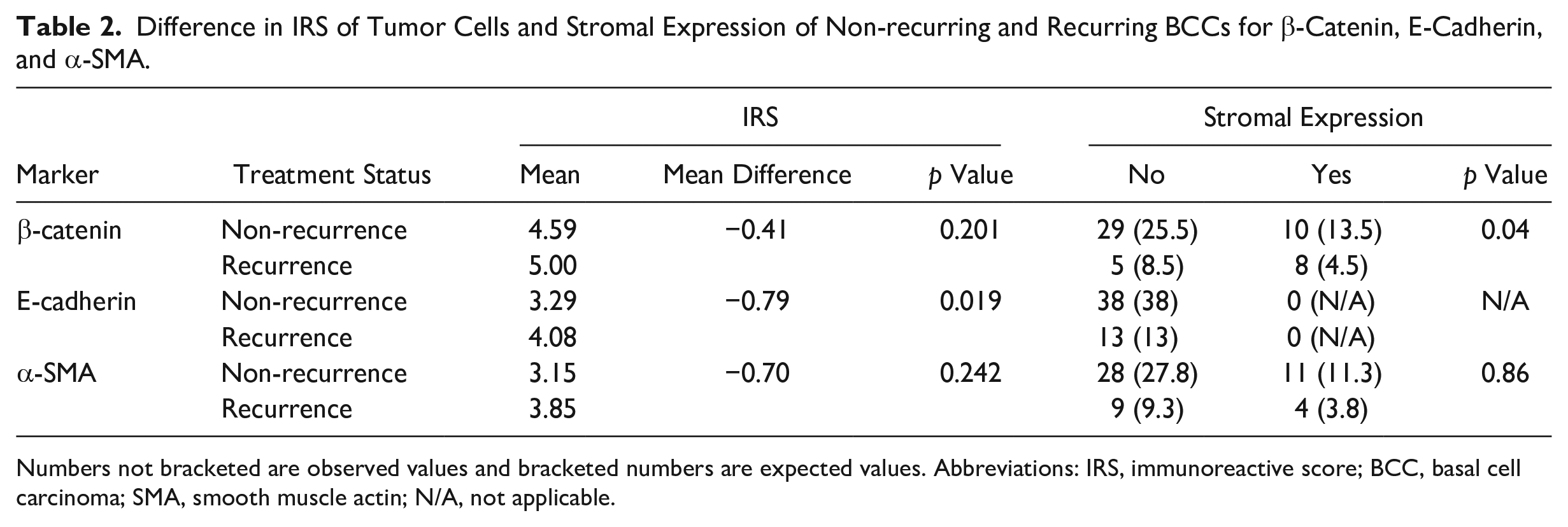
Numbers not bracketed are observed values and bracketed numbers are expected values. Abbreviations: IRS, immunoreactive score; BCC, basal cell carcinoma; SMA, smooth muscle actin; N/A, not applicable.
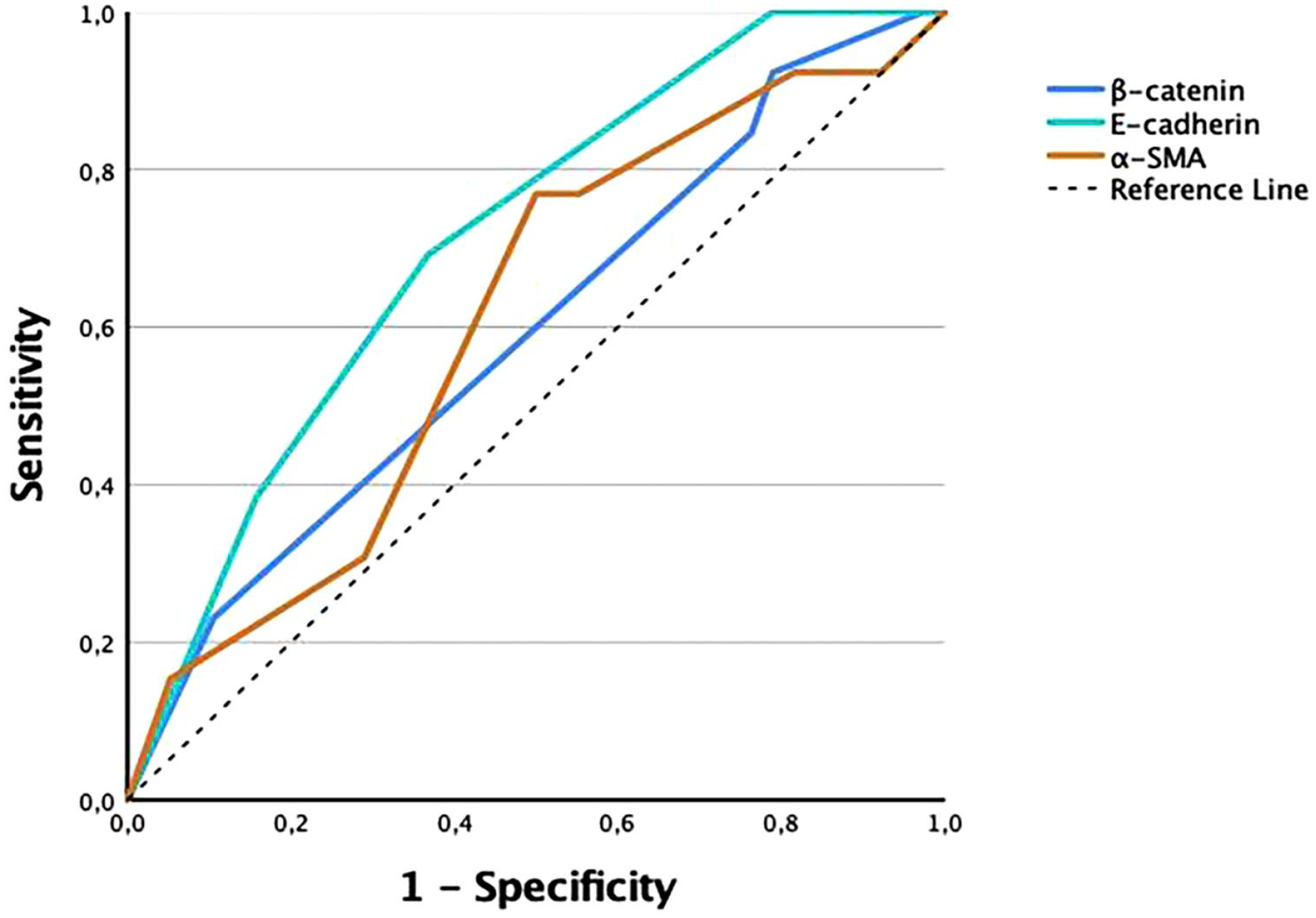
Receiver operating characteristic curves of immunoreactive scores of tumor cells for β-catenin, E-cadherin, and α-SMA. Abbreviation: SMA, smooth muscle actin.
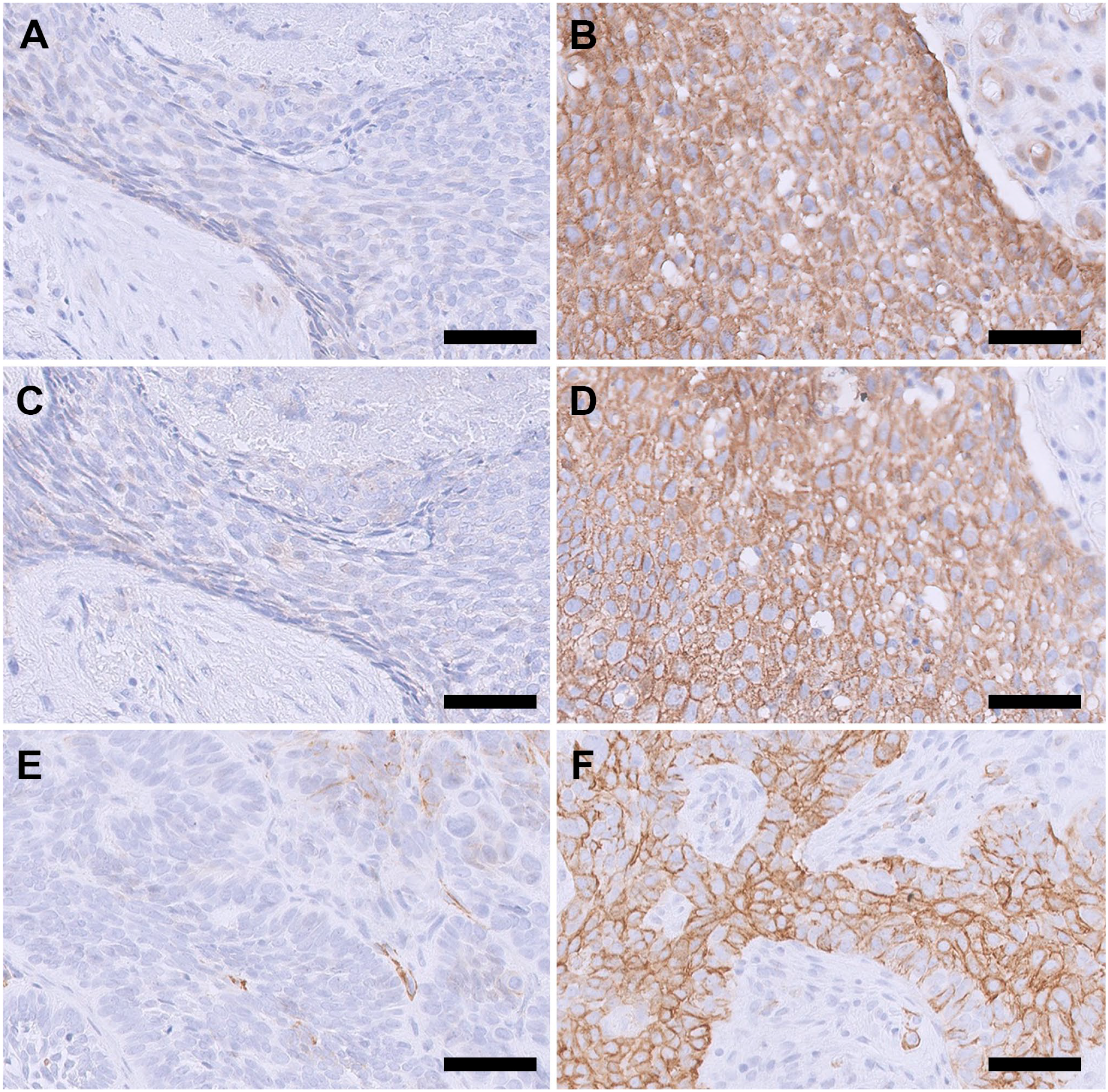
Tissue images of immunohistological expression of β-catenin, E-cadherin, and α-smooth muscle actin (SMA) in basal cell carcinomas (BCCs). (A) Negative β-catenin expression in an aggressive BCC. (B) High β-catenin expression in a superficial BCC. (C) Negative E-cadherin expression in an aggressive BCC. (D) High E-cadherin expression in a superficial BCC. (E) Negative α-SMA expression in a nodular BCC. The positive stain represents endothelial cells. (F) High α-SMA expression in an aggressive BCC. Magnification is 20× and scale bar is 50 µm for all images.
Cellular Location for Non-recurring versus Recurring BCCs
Table 3 shows the number of stains in the different cellular locations for β-catenin. E-cadherin was only observed in the cell membrane and α-SMA only expressed in cytoplasm except in one case with expression in both membrane and cytoplasm.
Number of Stains in Different Cellular Locations for β-Catenin.

For β-catenin, we observed specific expressions in recurring BCCs. In four recurring tumors with both membranous and cytoplasmatic expression, cytoplasmatic expression was more prevalent at the deep invading edge of the tumors with more intense expression in the same area for two of these tumors. In one recurring superficial and one recurring aggressive tumor, we saw expression of β-catenin in small groups of cells detached from the main tumor that was situated in the deep portion of the tissue. This is shown in Figure. 4B.
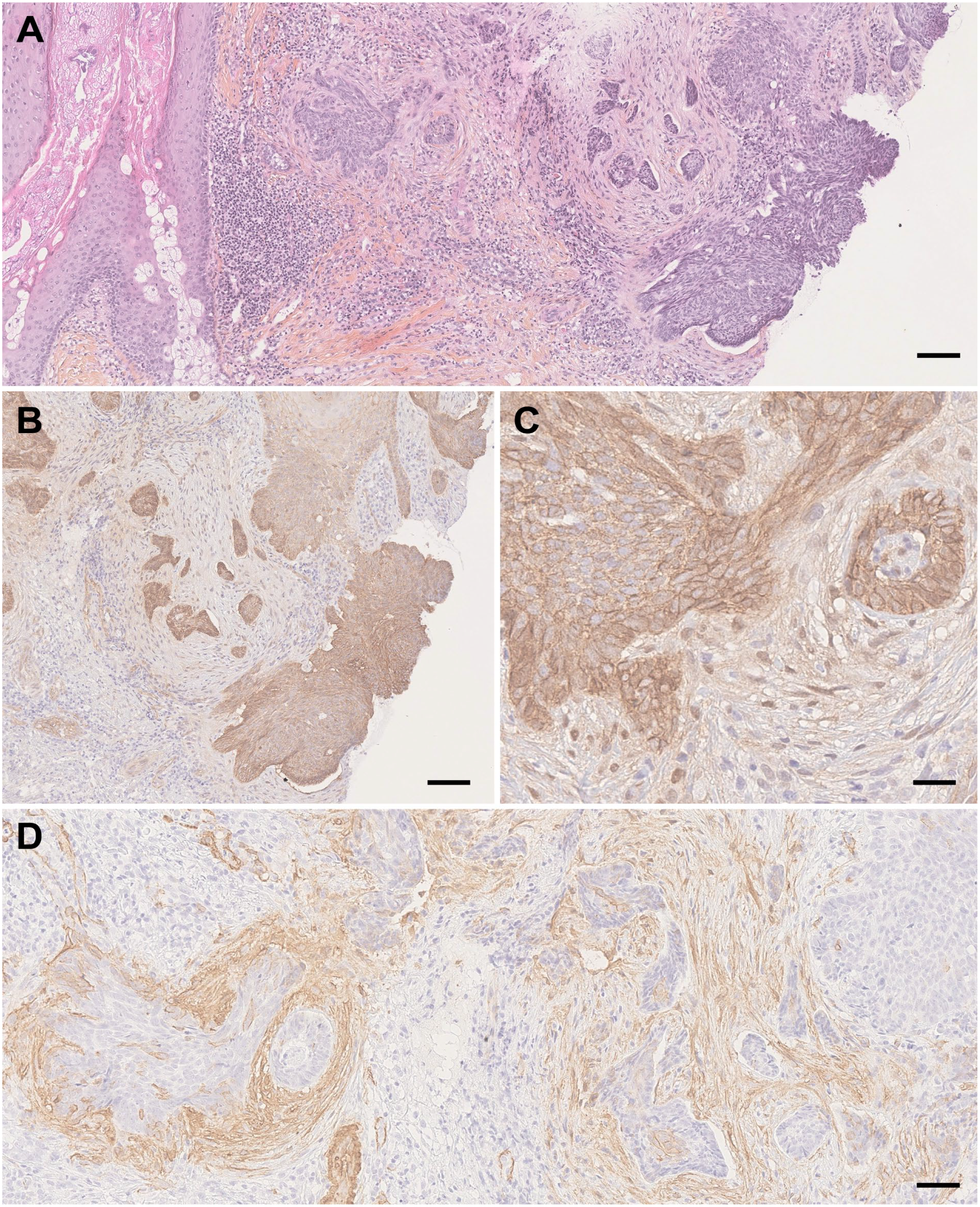
Tissue images of the same aggressive basal cell carcinoma (BCC) which later recurred after photodynamic therapy. (A) Hematoxylin-eosin-saffron (HES)-stain. Note the deep tumor islands at the right and center of the image. Magnification 5×, scale bar 100 µm. (B) β-catenin staining in tumor islands at the deep invading edge of the tumor. Magnification 20×, scale bare 50 µm. (C) Weak stromal expression of β-catenin adjacent to tumor island. Magnification 40×, scale bar 50 µm. (D) Moderate stromal expression of α-smooth muscle actin in stroma surrounding tumor islands. Magnification 20×, scale bar 50 µm.
Stromal Expression for Non-recurring versus Recurring BCCs
Table 2 shows the number of cases with stromal expression for β-catenin, E-cadherin, and α-SMA. It shows that only β-catenin had a significant difference in stromal expression between non-recurring and recurring BCCs. It also shows that recurring BCCs more often expressed stromal β-catenin compared with non-recurring BCCs and that absence of stromal staining was more often present among non-recurring compared with recurring BCCs. Stromal expressions of β-catenin and α-SMA are shown in Figure. 4C and D.
Discussion
In this study, we investigated the protein expression of β-catenin, E-cadherin, and α-SMA in BCC tissue obtained before PDT and their ability of predicting PDT outcome. Results showed that only the protein expression of E-cadherin was significantly different in tumor cells among recurring compared with non-recurring tumors. The AUC of E-cadherin was 0.71 which is significantly higher than 0.5 and thus can be considered an acceptable level of discrimination between the two treatment response groups. 19 The AUCs of β-catenin and α-SMA were small and not statistically higher than 0.5 meaning that the markers had no predictive ability. Even though stromal β-catenin expression was weak, it was significantly increased among recurring BCCs. Some of the recurring BCCs had cytoplasmatic and intense β-catenin expression in deep tumor edges, as well as isolated groups of tumor cells separated from the main tumor, not found in non-recurring BCCs.
E-cadherin is a transmembranous polypeptide and is important in cell junctions as a cell-adhesion molecule. 20 In BCCs, loss of membranous E-cadherin expression or presence of nuclear expression has been shown to be related to aggressive and invasive tumor behavior.13,16,21,22 In our material, we observed significantly increased E-cadherin expression in BCCs recurring after PDT. All tumors, except one with cytoplasmatic staining, demonstrated membranous staining indicating that subcellular localization of E-cadherin did not discern between non-recurring and recurring BCCs. None of the tumors had stromal E-cadherin expression. Increased E-cadherin expression among recurring BCCs could be an example of cell adhesion–mediated drug resistance, understood as tumor cells adhering to extra-cellular matrix ligands, protecting them from undergoing apoptosis. 23 Results from a previous study have shown that PDT-responding cells have a loss of E-cadherin expression and that PDT-resistant cells have recovered distribution and reactivity of E-cadherin. Blocking E-cadherin function by using a blocking antibody resulted in PDT-induced apoptosis. 24 Similarly, in another study, PDT-resistant clones of mammary adenocarcinoma have shown E-cadherin expression with formation of multiple interdigitations between cell membranes indicating a stronger link between tumor cells. 25
β-catenin and α-SMA showed no statistical difference in IRS between the two treatment response groups. β-catenin is a multifunctional protein involved in controlling several cell activities, such as cell-to-cell interaction 26 at a membrane level and cell proliferation and differentiation at a nuclear level. 27 It is therefore important to recognize subcellular localization of expression (membrane, cytoplasm, and/or nucleus). The release of β-catenin from membranous E-cadherin decreases cell-to-cell adhesion and increases cytoplasmatic and nuclear β-catenin, which both have been linked to increased invasiveness among BCCs.12 –14,16,28 We found no difference in IRS or subcellular localization of β-catenin between the two treatment response groups. Similarly, Gracia-Cazaña et al. 9 found no statistical difference in the expression intensity of β-catenin among non-responders and responders to PDT. However, they did not evaluate subcellular localization of expression in their study. To our knowledge, no previous studies have compared subcellular or stromal expression of β-catenin in BCCs before PDT. However, the observation of stromal β-catenin expression and cytoplasmatic and intense β-catenin expression at the deep invading edge of the tumor has been described earlier in aggressive BCCs.12,28,29 In the present study, statistically, stromal β-catenin expression was more present in recurring BCCs, and absence of stromal β-catenin expression was more present among non-recurring BCCs.
α-SMA is a protein produced by myofibroblasts. 30 Myofibroblasts are important in tissue remodeling and wound healing, but they also produce cytokines and growth factors which can stimulate tumor growth and angiogenesis, and prevent tumor apoptosis. 31 α-SMA is an accepted marker for myofibroblasts31,32 and has been shown to increase in aggressive and recurring BCCs.13,15,33,34 We found no statistical difference in IRS, subcellular localization, or stromal expression of α-SMA between the two treatment response groups.
In this study, we included a wide range of BCCs, including recurring tumors after previous treatment, tumors from the mid-face region, histologically aggressive tumors, and tumors with thickness up to 3.5 mm that are outside current recommendations for PDT.6,35 These inclusions were intentional to reduce limitations in the number of lesions we could add to this explorative study. Strengths of this study include the use of a recognized scoring system and two investigators for scoring. Limitations include small sample size, long storage time for tissue sections before staining which for some antibodies may have led to loss of antigen reactivity, 36 and the use of polymer-based secondary antibodies which can reduce the ability to detect intracellular antigens. 37
Further studies are necessary to investigate whether the present results are clinically significant. Such studies should also take specific note of where in the tumor tissue and cells staining is present, as well as stromal staining. Additionally, exploration of other potential markers for predicting PDT outcome would be interesting as it is possible that mechanisms outside tumor invasiveness or aggressiveness may be related to PDT response. 38
In conclusion, the results show that E-cadherin expression is increased in BCCs recurring after PDT, as well as increased expression of β-catenin in the stroma and lower tumor edge, suggesting they could be potential predictive markers for PDT outcome. Further studies are necessary to investigate whether these results are of clinical value.
Footnotes
Acknowledgements
The authors thank all the patients and dermatologists for their contribution to this study. They thank the Department of Pathology, St. Olavs Hospital, Trondheim University Hospital, for scanning slides to digital images. They thank Cellular and Molecular Imaging Core Facility (CMIC) for the immunostaining.
Author Contributions
EM administered the project, applied for funding, designed the study, evaluated the material, analyzed the data, composed the digital image, and wrote the original draft. PM designed the study and evaluated the material. OAF analyzed data, interpreted results, helped structuring the manuscript, and prepared the graphic figures. IMB designed the study. EC administered the project, provided the material, and designed the study. All authors reviewed and edited the manuscript and approved it before submission. EM is accountable for all aspects of the work and ensures that aspects related to the accuracy and integrity of the work are appropriately investigated and resolved.
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Fylkesmann H.B. Guldahl og hustru Lucy Guldahls legat til bekjempelse av kreft og andre alvorlige sykdommer, June 8, 2021 (30.500 NOK) and The Liaison Committee for Education, Research and Innovation in Central Norway (Samarbeidsorganet).
