Abstract
Neutral buffered formalin (NBF) is the most common fixative in clinical applications. However, NBF damages proteins and nucleic acids, limiting the quality of proteomic and nucleic acid-based assays. Prior studies have demonstrated that BE70, a fixative of buffered 70% ethanol, has many benefits over NBF but the degradation of proteins and nucleic acids in archival paraffin blocks remain a challenge. Thus, we evaluated the addition of guanidinium salts to BE70 with the hypothesis that this may protect RNA and protein. Guanidinium salt supplemented BE70 (BE70G)-fixed tissue is comparable with that of BE70 via histology and immunohistochemistry. Western blot analysis also revealed that HSP70, AKT, and glyceraldehyde 3-phosphate dehydrogenase (GAPDH) expression signals in BE70G-fixed tissue were higher than those in BE70-fixed tissue. The quality of nucleic acids extracted from BE70G-fixed, paraffin-embedded tissue was also superior, and BE70G provides improved protein and RNA quality at shorter fixation times than its predecessors. The degradation of proteins, AKT and GAPDH, in archival tissue blocks is also decreased with the addition of guanidinium salt to BE70. In conclusion, BE70G fixative improves the quality of molecular analysis with more rapid fixation of tissue and enhanced long-term storage of paraffin blocks at room temperature for evaluation of protein epitopes:
Introduction
Neutral buffered formalin (NBF, 3.7–4.0% phosphate-buffered formaldehyde) fixation and paraffin embedding is the gold standard technique for tissue preservation in surgical pathology enabling long-term storage of surgical specimens allowing high-quality histomorphological evaluation. However, formalin is a crosslinking fixative and impairs the extraction efficiency and quality of macromolecules (RNA, DNA, and protein).1–5 In the clinical setting, the stability of protein epitopes is generally sufficient for markers of cell lineage; however, prognostic and predictive markers, such as human epidermal growth factor receptor-2 (HER-2) and programmed death ligand 1 (PD-L1), were well documented to degrade in archival blocks over time.6–8 In addition, RNA is well documented to degrade in archival blocks, presenting an ever growing challenge for the application of RNA-based assays in the clinical setting. 3 The net result is that the formalin-fixed paraffin-embedded (FFPE) tissue specimens can be unreliable for the quantitative and qualitative measurement of molecular biomarkers. The molecular analysis of clinical specimens is based on fresh or snap-frozen tissues which are cost-intensive, limited in storage times and histomorphological features. 9 Furthermore, formaldehyde was classified as a human carcinogen by the International Agency for Research on Cancer (IARC) and also acknowledged for its carcinogenic role by other authorities, including the U.S. Environmental Protection Agency (EPA).10,11 The prior studies reported a positive association between formaldehyde exposure and lymphohematopoietic malignancies.12–14 Thus, there is a need for an “optimal fixative” that is inexpensive, allows preservation of histomorphological features similar to those seen in NBF-fixed tissues, and protects biomolecules in a manner comparable with fresh or snap-frozen specimens.
To date, two types (crosslinking and coagulant) of chemical fixation have been generally utilized in histological diagnosis and molecular analysis. These two classes of fixatives have certain advantages and disadvantages. The crosslinking fixatives, for example, formaldehyde and glutaraldehyde, induce crosslinks between proteins, individual protein moieties, and between nucleic acids and proteins. 15 These fixatives allow long-term stable tissue preservation and compatibility with downstream histological applications, but impair the extraction efficiency of biomolecules and produces fragmented and chemically modified nucleic acids.1,5,16 On the contrary, the coagulant fixatives are commonly used alcohols and dehydrate from the tissues which result in protein precipitation and coagulation. 17 These alcohol-based fixatives have advantages including rapid fixation times, 18 lack of carcinogenic chemicals, and better preservation of biomolecules; however, these fixatives also have disadvantages such as tissue shrinkage and hardening, lysis of erythrocytes, and issues concerning flammability.19,20 Previous studies have demonstrated that NBF, formulated with phosphate buffer, is an optimal formulation of formaldehyde fixatives. 5 Both classes of fixatives are associated with long-term degradation of biomolecules, believed to be related to incomplete dehydration.21,22 Alternative fixatives have failed to replace formaldehyde fixation in routine clinical use. Formaldehyde-base fixatives are associated with certain reproducible artifacts of formalin chemistry such as nuclear grooves in papillary carcinoma of the thyroid and the absence of eosinophil or basophilic granules in eosinophils and basophils.
We have previously described a phosphate-buffered ethanol-based fixative (called BE70) which showed general compatible histomorphology to the NBF fixative and improved biomolecule preservation. 18 Here, we pursued the improvement of BE70 fixative for enhanced biomolecular preservation as well as maintenance of comparable histomorphology with that of NBF-fixed tissue. Thus, we described a modular fixative in which guanidinium salt is supplemented to the buffered ethanol fixative (termed BE70G). Employing BE70G fixative, we evaluated histomorphology, immunohistochemistry, Western blotting, and nucleic acid quantity and quality, and compared the results with those of NBF and BE70. In addition, we also evaluated fixation times from 4 to 24 hr with respect to histochemical and molecular features. Furthermore, we examined the storage effect on precut tissue sections after storage times from 1 day to 3 months at room temperature with combined vac-seal and vac drierite (VD) or humidity chamber (HC) conditions.
Materials and Methods
Fixatives and Reagents
To examine histomorphological and molecular features of alternative molecular fixative, we used three different fixatives including 10% NBF, BE70 (buffered ethanol 70%; 70% ethanol with 1% glycerol + 0.5% glacial acetic acid + 0.5 × PBS, pH 6.1), and BE70G (70% ethanol with 0.5 × PBS + 1.69 M guanidine thiocyanate, pH 7.0). The final fixative formulation was adjusted by volume. Ethanol (200 proof/100%) and NBF were purchased from VWR (Radnor, PA). Glycerol and glacial acetic acid were purchased from Sigma-Aldrich (St. Louis, MO). Guanidine thiocyanate (Ambion; cat. no. AM9422) was obtained from ThermoFisher Scientific (Grand Island, NY).
Fixation and Tissue Specimen
C57BL/6 (wild-type) mice were purchased from Charles River Laboratories (Fredrick, MD) and used between 8- and 12-week-old mice of both sexes. Animals were housed and euthanized in accordance with National Institutes of Health (NIH) guidelines for the care and use of laboratory animals. The tissues were collected within 5 min of euthanasia, and were fixed in one of the three different fixatives (10% NBF, BE70, or BE70G) for various fixation times (4, 8, 12, 16, and 24 hr). Tissue was dissected to section of 2 to 3 mm thickness, and incubated tissues with fixative (exceeding a 1:30 tissue fixative volume). The tissues were rinsed in 70% alcohol, processed for 8 hr (30 min/stage) and embedded in paraffin as described previously.18,21
To compare the storage impact according to the fixatives, the tissue (5 µm thickness) slides were either vacuum-packed (VD) using a “Seal-a-meal” vacuum packaging unit (FoodSaver; Jarden Corporation, Providence, RI) with desiccant [Drierite, anhydrous calcium sulfate (CaSO4); W.A. Hammond Company, Xenia, Ohio] or placed in a HC and stored at room temperature for 3 months. The HC was a plastic container (11 × 8 × 6 cm3) containing 100 ml distilled water with the slides placed on a rack above the water and sealed in a plastic sack of the same material used for the vacuum-sealed specimens.
Histochemical and Immunohistochemical Analysis
Paraffin block of each entity was cut on a microtome (ASP300; Leica, Germany), mounted on superfrost plus slide (ThermoFisher Scientific; Grand Island, NY) and baked at 65C for 1 hr. The tissue sections were stained with hematoxylin and eosin (H&E) in the Leica Auto Stainer XL (Leica Biosystems Inc.) and evaluated by two experienced pathologists (K.K. and S.M.H.).
Immunohistochemistry was performed in automated immunostainer (Autostainer Link 48; Dako, Carpinteria, CA) according to manufacturer’s instructions. In brief, whole sections were deparaffinized and rehydrated through xylenes and decreasing alcohol gradient. Heat-induced epitope retrieval was conducted with antigen-retrieval buffer (pH 6.0; Dako) for 20 min by means of a pressure cooker. The slides were incubated for 10 min in 3% (V/V) H2O2 to block for endogenous peroxidase. The section was incubated with rabbit anti-keratin 14 (K14) polyclonal antibodies (cat. no. 905301; dilution 1:1000; BioLegend, Dedham, MA), rabbit anti-E-cadherin monoclonal antibodies (clone no. 24E10; dilution 1:500; Cell Signaling; Denver, CA), rabbit anti-CD3 monoclonal antibodies (clone no. SP7; dilution 1:100; Abcam), or rabbit monoclonal anti-Ki-67 antibodies (clone no. SP6; dilution 1:100; Abcam, Cambridge, MA) for 1 hr at room temperature. The reaction of antigen and antibody was visualized by the DAKO Envision+ peroxidase kit and visualized with 3,3-diaminobenzadine (Dako). Negative controls (IgG and omission of primary antibody) were concurrently performed. Thyroid tissue was used as a positive control.
Protein Extraction and Western Blotting
Protein was extracted from differently fixed and paraffin-embedded tissues as described previously.21,23,24 To examine the storage impact according to the fixatives, five replicates of differently fixed and paraffin-embedded tissue sections, 5-µm thick, were deparaffinized in xylene and rehydrated in a graded alcohol series. Liver tissue sections from VD and HC storage conditions were removed with a sterile scalpel and placed in microcentrifuge tubes. The samples were homogenized in 100 µl protein extraction solution (high pH AgR buffer, pH 9.9; Dako, Carpinteria, CA), 1% NaN3, 1% SDS, 10% glycerol, and protease inhibitor (1 tablet/25 ml; Roche, Indianapolis, IN), and high-temperature extraction was applied to all samples within a pressure cooker at 115C (Dako) for 15 min, and then centrifuged at 15,000 × g for 30 min at 4C. The supernatants were collected, and protein concentration was quantified using a BCA Protein Assay kit (Pierce Biotechnology; Rockford, IL).
For Western blotting, electrophoresed proteins (20 µg) were transferred onto a nitrocellulose membrane according to the manufacturer’s protocol (Invitrogen; Carlsbad, CA). To block unspecific reactions, the membranes were incubated in 5% non-fat dry milk in Tris-buffered saline with Tween (TBST; 50 mM Tris, pH 7.5, 150 mM NaCl, and 0.05% Tween-20) for 1 hr, and then probed overnight at 4C in TBST with 5% bovine serum albumin (BSA) containing rabbit anti-AKT polyclonal antibody (cat. no. 9272; dilution 1:1000; Cell signaling technology), anti-HSP70 polyclonal antibody (cat. no. 4872; dilution 1:1000; Cell signaling technology), or mouse anti-GAPDH (glyceraldehyde 3-phosphate dehydrogenase) monoclonal antibody (clone no. 6C5; dilution 1:2000; Calbiochem, Gibbstown, NJ). Membranes were washed and incubated for 1 hr at room temperature with horseradish peroxidase (HRP)–labeled anti-rabbit or anti-mouse secondary antibodies. Protein expression was visualized with a chemiluminescent reagent (SuperSignal Chemiluminescence Kit; Pierce Biotechnology), and the signals were detected using Kodak Biomax MR X-ray film (Kodak; Rochester, NY). Blot intensity was quantified using ImageQuant version 5.2 (Molecular Dynamics; Sunnyvale, CA).
RNA Quantity and Quality
Total RNA was isolated from two 1-mm tissue cores as previously reported.4,18 RNA quantity and quality were assessed by means of the NanoDrop ND-1000 UV spectrophotometer (NanoDrop Technologies; Wilmington, DE) and the Agilent 2100 bioanalyzer (Agilent Technologies; Palo Alto, CA) with the RNA 6000 LabChip kit (Agilent Technologies), respectively. We also measured RNA integrity with the Paraffin-Embedded RNA Metric (PERM) number as previously reported.24,25
Approximately, 4 μg DNase-treated RNA was transcribed into cDNA by reverse transcriptase using the QuantiTect Reverse Transcription (Qiagen; Valencia, CA) according to the manufacturer’s recommended protocol. All samples were reverse transcribed under the same conditions. The synthesized cDNA was stored at –20C and used in quantitative real-time PCR reactions as a template. We performed quantitative real-time PCR using the TaqMan Gene Expression reagent (Applied Biosystems; Foster City, CA) as previously reported.18,21 Briefly, quantitative real-time PCR was performed with 1 and 2 μg cDNA for housekeeping genes (beta-actin, ACTB) and mouse tumor necrosis factor (TNF), respectively. We examined the cycle threshold (Ct) values for ACTB and TNF genes to assess RNA integrity in triplicate.
DNA Quantity and Quality
DNA extraction from differently fixed, and paraffin-embedded tissues DNA was extracted from two 1-mm tissue cores using the QIAamp DNA FFPE Tissue kit (Qiagen) according to the manufacturer’s instruction. DNA extraction yield was assessed using the NanoDrop ND-1000 UV spectrophotometer (NanoDrop Technologies) and Qubit 4 Fluorometer (Invitrogen). DNA quality was evaluated by amplified DNA amounts using a BioScore Screening and Amplification kit (Enzo Life Sciences; Farmingdale, NY) as previously reported.21,24,26
Statistical Analysis
All data are representative of at least three separate experiments. Statistical differences were tested using ANOVA and the Student’s t-test. Statistical analysis was performed using the SPSS version 21.0 (SPSS Inc.; Chicago, IL). A value of p<0.05 was considered statistically significant.
Results
Protein Quantity and Quality in Different Fixatives
Mouse kidney and liver tissues were fixed with NBF, BE70, or BE70G for 24 hr at room temperature and subsequently paraffin-embedded following standardized protocols that have been used in prior studies of fixation.18,21 During preliminary experiments, we tested several different guanidine thiocyanate concentrations (0.51, 1.02, and 1.69 M). The protein immunoreactivity of BE70G was better than that of BE70, with the highest signal in the highest concentration of guanidine thiocyanate. In terms of RNA quality, the highest concentration of guanidine thiocyanate was the best. In addition, the highest concentration of guanidine thiocyanate showed the best histomorphological features among tested conditions, except for NBF (Supplementary Fig. S1). To assess the impact of fixative on protein quantity, we extracted protein from the three different fixatives-fixed and paraffin-embedded tissues. There was no significant difference in overall recovery, among tested fixatives in both kidney (NBF, BE70, and BE70G; mean: 6.22 ± 0.725 µg/mm3, 7.04 ± 0.323 µg/mm3, and 7.69 ± 0.783 µg/mm3, respectively) and liver (NBF, BE70, and BE70G; mean: 6.12 ± 0.748 µg/mm3, 6.84 ± 0.864 µg/mm3, and 7.52 ± 0.976 µg/mm3, respectively) tissues (Fig. 1A).
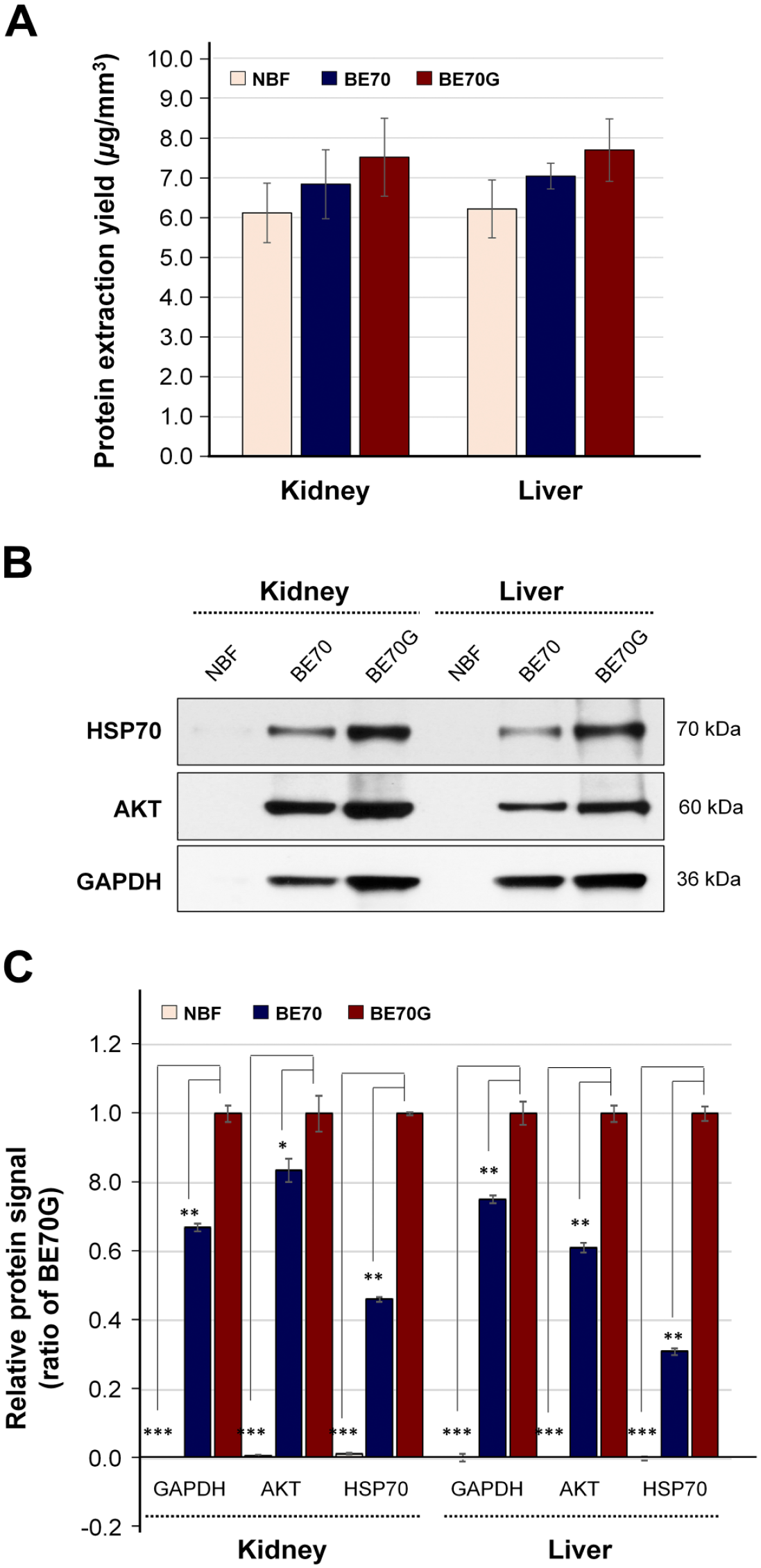
Quantity and quality of protein extracted from NBF-, BE70-, or BE70G-fixed tissues. (A) Protein extraction yield from each condition. The protein was extracted from mouse kidney and liver in NBF-, BE70-, or BE70G-fixed and paraffin-embedded tissue cores. The bar graph shows the relative means of protein extraction yields. (B) Protein integrities of different fixatives-fixed tissues were assessed by Western blotting. Protein extracted from each condition was probed with antiserine/threonine protein kinase (AKT) and GAPDH antibodies. (C) Relative expression signal of AKT and GAPDH expression signal under each condition is presented as a bar graph. The relative protein signal of each entity was normalized to 1-day BE70G-fixed tissue (1.00). Quantitative analyses were performed using ImageQuant version 5.2. Data are presented as the mean ± standard deviation (SD) from three independent experiments. Abbreviations: NBF, neutral buffered formalin; GAPDH, glyceraldehyde 3-phosphate dehydrogenase. *p<0.05, **p<0.01, ***p<0.001.
We next examined protein immunoreactivity of NBF-, BE70-, and BE70G-fixed kidney and liver tissues by Western blotting. The HSP70, AKT, and GAPDH expressional signals of BE70G-fixed tissues were significantly stronger in both kidney (2.2-, 1.2-, and 1.5-fold increase, respectively) and liver tissues (3.2-, 1.6-, and 1.3-fold increase, respectively) than in BE70-fixed tissues (Fig. 1B and C). However, these three molecules’ signals of NBF-fixed tissue were detected with only trace bands in both organ tissues. These results suggest that the BE70G fixative allowed better protein quality over BE70 and NBF, but did not impact to protein recovery yields from the fixed and paraffin-embedded tissues.
Immunoreactivity According to the Fixation Time in Different Fixatives
The fixation time of fixative highly impact on biomolecule quality, and the fixation period optimization of new fixative, is a fundamental factor for research and clinical usage. We examined the immunoreactivity of the protein recovered from the different fixatives-fixed mouse liver tissues at different fixation times (4, 8, 12, 16, and 24 hr) using Western blotting. The proteins extracted from BE70G- and BE70-fixed tissues were in similar amounts between 4 and 24 hr (BE70G vs BE70; mean: 7.98 ± 0.236 vs 7.68 ± 0.475 µg/mm3), while there was observed slightly decrease tendency in NBF-fixed tissues (mean: 6.28 ± 0.288 µg/mm3) (Fig. 2A). AKT and GAPDH expressional signals were detectable at all fixative times in BE70G and BE70, whereas both the signals were detected weak or traceable band in the NBF (Fig. 2B and C). As observed in a previous study, 21 the signal of AKT expression was the highest at 24-hr fixation period for all three fixatives. The AKT signal of the BE70G was significantly stronger than BE70, 2.39-fold increase at 4 hr (p<0.01), 2.26-fold increase at 8 hr (p<0.05), 2.22-fold increase at 12 hr (p<0.05), 2.46-fold increase at 16 hr (p<0.01), and 1.36-fold increase at 24 hr (p<0.05). Notably, the GAPDH immunoreactivity is relatively well preserved from 4- to 24-hr fixation with BE70G, while the BE70 gradually decreased according to the fixation periods (Fig. 2B).
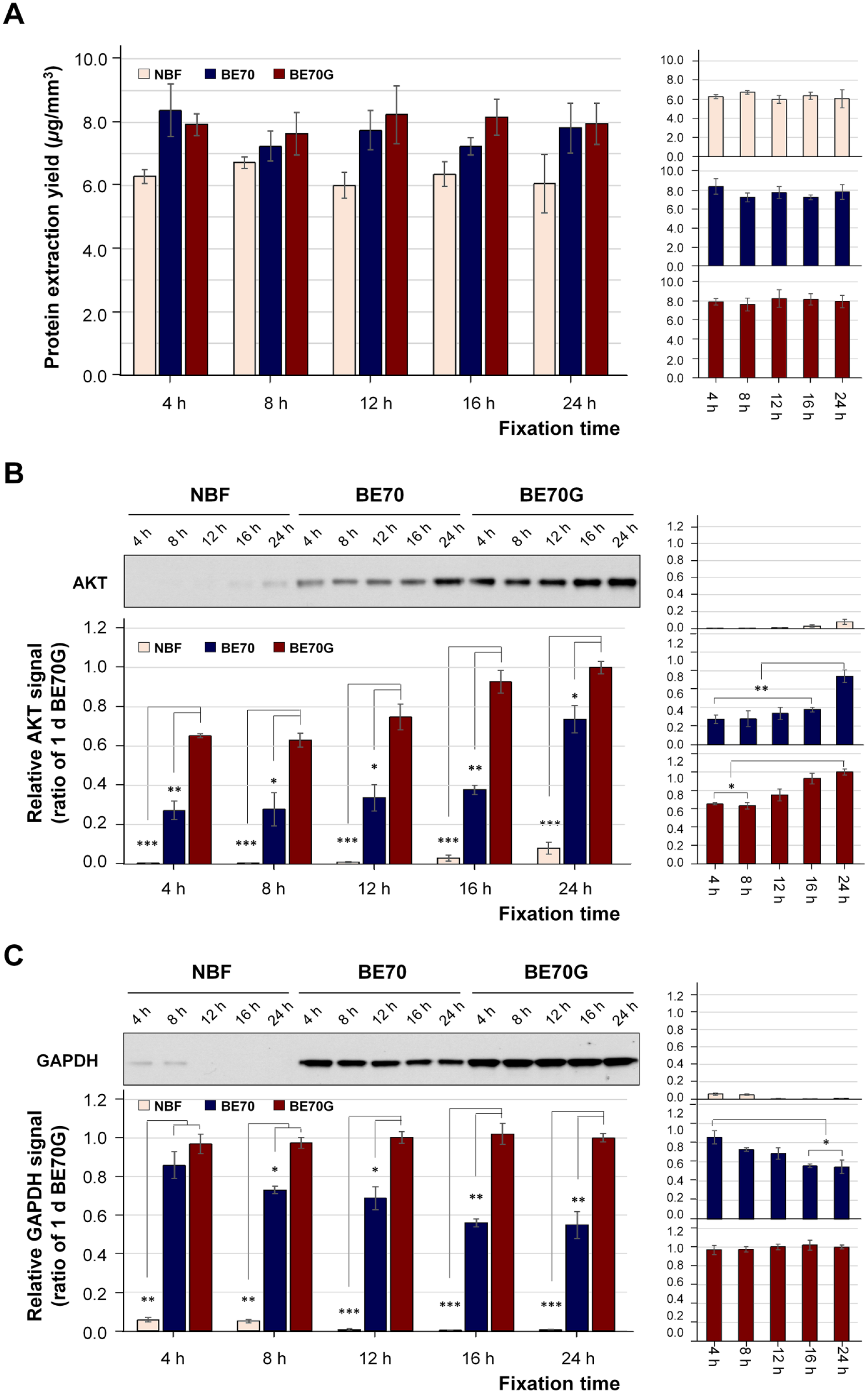
Protein quantity and quality according to the fixation time in NBF, BE70, and BE70G. (A) The protein was extracted from mouse liver in NBF-, BE70-, or BE70G-fixed and paraffin-embedded tissue cores. The bar graph shows the extraction yields. (B) Relative serine/threonine protein kinase (AKT) expression signal under each condition. The quality of proteins in different fixatives according to fixation time was assessed by Western blotting. Quantitative analyses were performed using ImageQuant version 5.2. Protein quantity and quality according to the fixation time in a single fixative are presented as miniature graphs in the right panel. The relative protein extraction yield and expression signal of each entity was normalized to 1-day BE70G-fixed tissue (1.00). Data are presented as the mean ± standard deviation (SD) from three independent experiments. Abbreviations: NBF, neutral buffered formalin; GAPDH, glyceraldehyde 3-phosphate dehydrogenase. *p<0.05, **p<0.01, ***p<0.001.
Effects of Storage on the Detection of Protein Immunoreactivity in Different Fixatives
Unstained 5 µm-thick sections of NBF-, BE70-, or BE70G-fixed and paraffin-embedded mouse liver tissues were stored for 3 months at room temperature either in the presence of desiccant (VD) or in a HC. To examine the storage impact according to the fixatives, we assessed the quality of proteins isolated from NBF-, BE70-, or BE70G-fixed tissues by Western blotting. AKT and GAPDH expression were detectable at all stored times in BE70- and BE70G-fixed tissues except for 3 months stored in a HC, while the AKT and GAPDH signal in the NBF-fixed tissues were detected minimal signals in all VD conditions and only 1-day storage in HC. Detailed evaluation demonstrates, the AKT immunoreactive signal was significantly stronger in fresh-cut BE70G-fixed tissue (5.0-fold increase, p<0.001) and fresh-cut BE70-fixed tissue (3.2-fold increase, p<0.001) than in NBF-fixed tissue (Fig. 3A). The AKT immunoreactive signal of 3 months stored in BE70G-fixed tissue section in VD (43.1% decrease compare with the fresh-cut BE70G-fixed tissue, p<0.001) was relatively well preserved than that of BE70- (81.2% decrease) and NBF-fixed (94.5% decrease) tissue section (Fig. 3A). In contrast, for tissue sections stored in HC, AKT expressions of BE70G-fixed tissues showed less decrease in intensity until 1 month (60.5% decrease compared with the fresh-cut BE70G), while the signal of BE70-fixed tissue rapidly decreased as time passed, signal intensity 64.6% decrease at 1 day, 79.3% decrease at 1 week, and 92.8% decrease at 1 month. In addition, we found that the AKT signal of the NBF-fixed tissues was only detected in 1-day storage in HC (Fig. 3A).
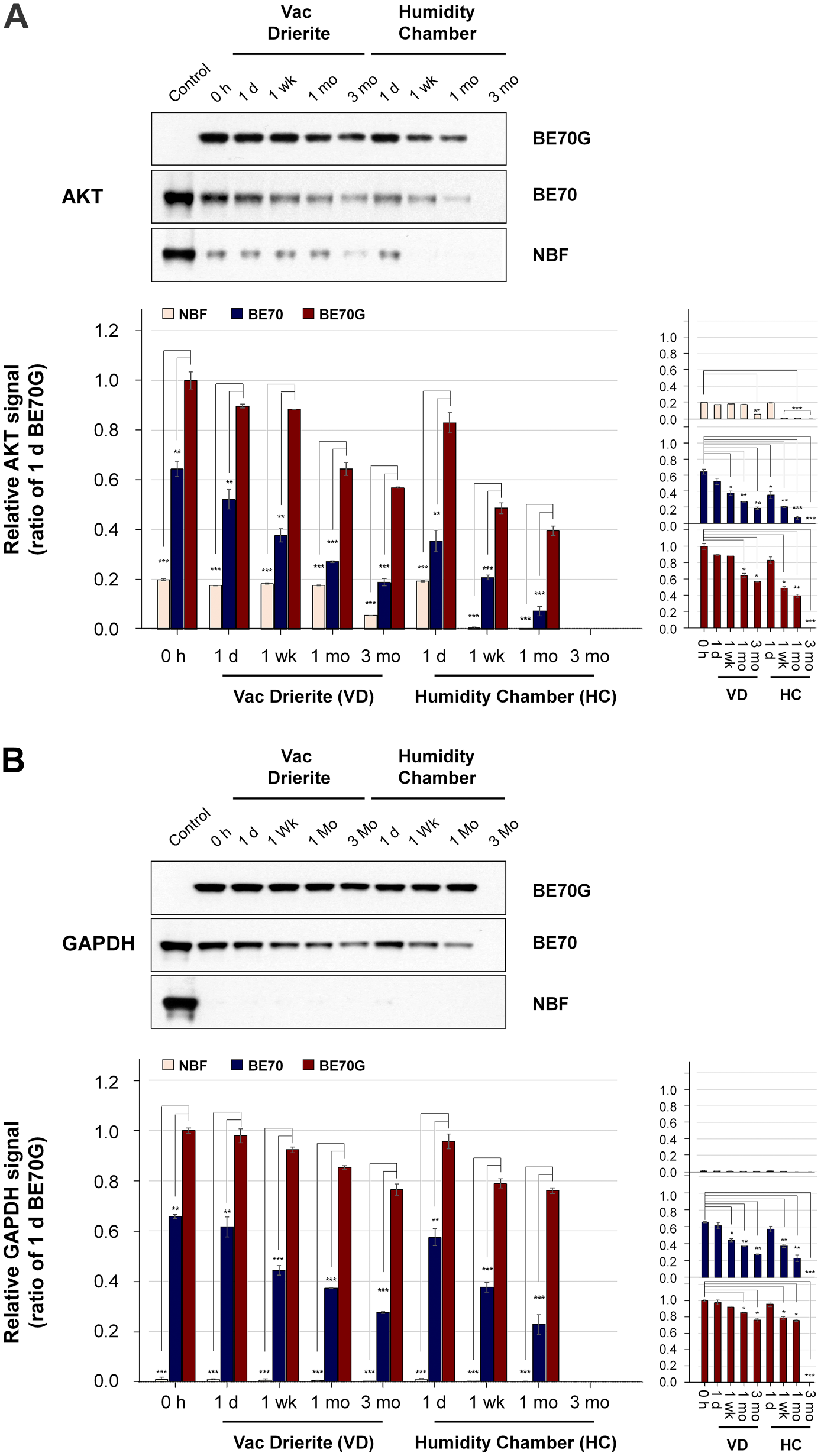
The quality of protein immunoreactivity in NBF-, BE70-, or BE70G-fixed tissue sections under different storage conditions. The protein immunoreactivity in different fixatives according to fixation time was assessed by Western blotting. (A) Relative AKT expression signal according to the fixation time in NBF, BE70, and BE70G. (B) Relative GAPDH expression signal according to the fixation time in NBF, BE70, and BE70G. Protein quantity and quality according to the fixation time in a single fixative are presented as miniature graphs in the right panel. The relative protein extraction yield and expression signal of each entity were normalized to 1-day BE70G-fixed tissue (1.00). Data are presented as the mean ± standard deviation (SD) from three independent experiments. Control, BE70G (0 h); Abbreviations: NBF, neutral buffered formalin; GAPDH, glyceraldehyde 3-phosphate dehydrogenase; VD, vacuum + drierite; HC, humidity chamber. *p<0.05, **p<0.01, ***p<0.001.
Evaluation of GAPDH replicated the same trends. The GAPDH signals of BE70G-fixed tissues were little less at 3 months of storage in the VD condition than they were the fresh-cut (23.6% diminution, p<0.01, Fig. 3B, right panel). BE70-fixed tissues gradually decrease in GAPDH signal at 3 months storage in the VD (72.4% less than fresh-cut BE70G, p<0.01, Fig. 3B), while NBF-fixed tissues showed a traceable band at all tested storage times. Surprisingly, the GAPDH signal of BE70G-fixed tissue was well-preserved until 1-month storage in a HC (23.9% less than fresh-cut BE70G, p<0.01, Fig. 3B). As expected, BE70-fixed tissue significantly decreased in GAPDH signal (77.2% less than fresh-cut BE70G, p<0.01, Fig. 3B), while the GAPDH signal of NBF-fixed tissue was detected traceable signal at 1-day storage in a HC (Fig. 3B). These data suggest that the AKT and GAPDH immunoreactivities of BE70G-fixed tissues are better preserved for prolonged storage times in VD condition, in contrast to the moderate and fast degradation observed in BE70- and NBF-fixed tissue sections.
RNA Quantity and Quality According to the Fixation Time in Different Fixatives
RNA was successfully extracted from fixed mouse liver tissues in NBF, BE70, or BE70G fixatives according to fixation times. The RNA recovery yield from BE70G-fixed tissues (mean: 6.69 ± 0.292 µg/mm3) was slightly higher than both BE70 (mean: 5.94 ± 0.339 µg/mm3) and NBF (mean: 4.67 ± 0.228 µg/mm3) from 12- to 24-hr fixation, while there was no significant difference at 4- and 8-hr fixation with all three fixatives due to incomplete fixation (Fig. 4A). Furthermore, we compared the quality of ACTB transcripts extracted from the fixative-fixed and paraffin-embedded tissues using quantitative real-time reverse transcriptase (RT)-PCR. As expected, the Ct values of ACTB in BE70G and BE70 were significantly lower than that of the NBF for all tested fixation times. The Ct values of ACTB in BE70G were slightly decreased in standard fixation times (16 and 24 hr; mean: 27.99 ± 0.048) compared with a short-term stored period (from 4 to 12 hr; mean: 28.48 ± 0.241), while there were no substantial differences in both BE70 (28.54 ± 0.165) and NBF (32.29 ± 0.141) according to the fixation periods (Fig. 4B). These data suggest that RNA integrity was more preserved in optimal fixation time with BE70G than tested other two fixatives.
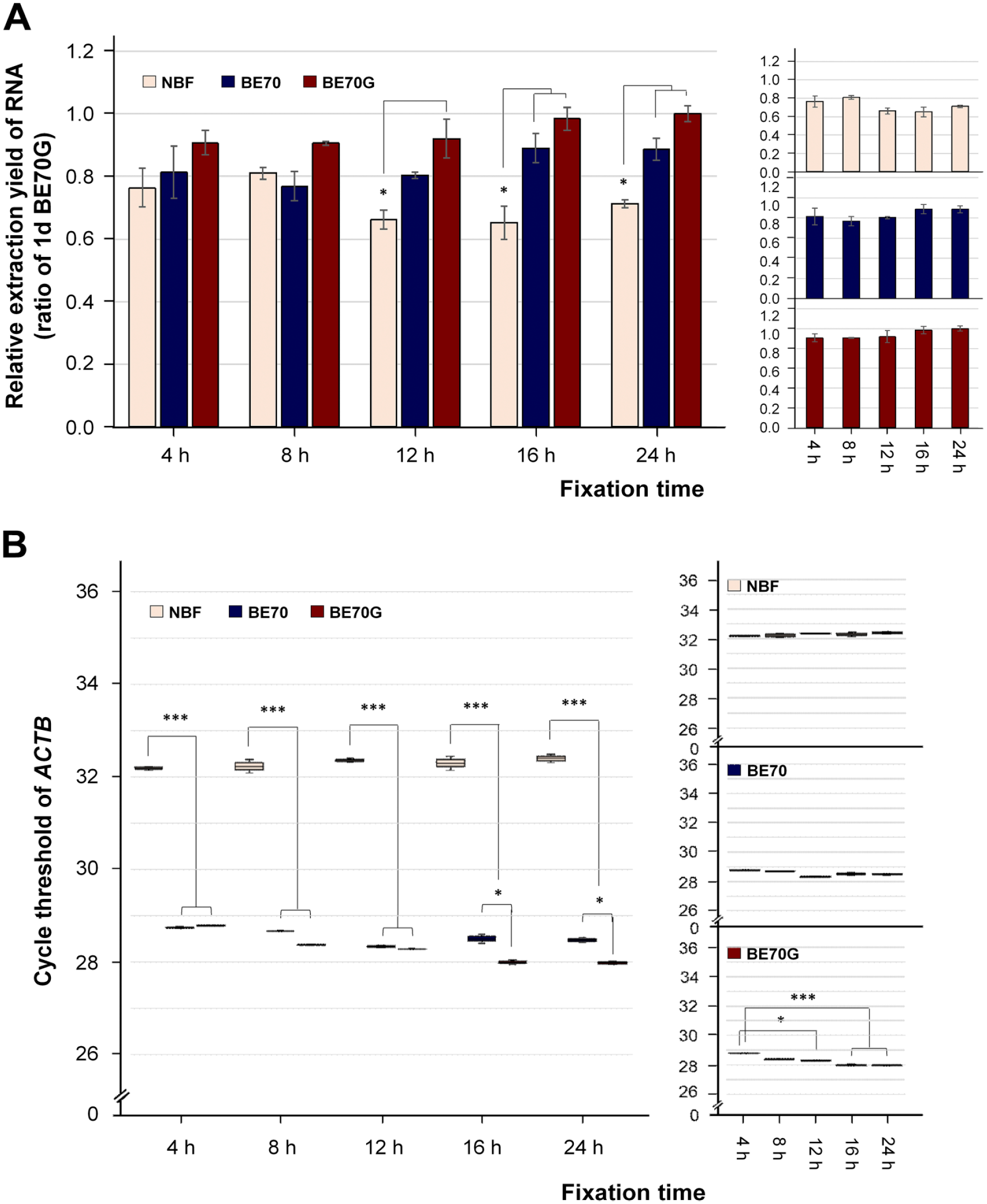
RNA quantity and quality according to the fixation time in NBF, BE70, and BE70G. (A) RNA extraction yield under each condition. The RNA was extracted from mouse liver in NBF-, BE70-, or BE70G-fixed and paraffin-embedded tissue cores. The relative RNA extraction yield of each entity was normalized to 1-day BE70G-fixed tissue (1.00). (B) RNA integrity derived from each fixative condition is presented as the mean cycle threshold (Ct) value of the beta-Actin (ACTB). Gene expression levels are shown as box plots. Relative RNA extraction yield and Ct values according to fixation time in a single fixative are presented as miniature graphs in the right panel. Data are the mean ± SD from three independent experiments. Abbreviations: NBF, neutral buffered formalin; ACTB, beta-Actin. *p<0.05, **p<0.01, ***p<0.001.
Nucleic Acids Quantity and Quality in Different Fixatives
We next performed similar analyses for DNA quantity and quality assessment after DNA extracted from NBF-, BE70-, and BE70G-fixed and paraffin-embedded mouse kidney and liver tissues. The A260/A280 ratio of DNA extracted from BE70G-fixed kidney and liver tissues was similar to that of BE70- and NBF-fixatives, ranging from 1.77 to 1.91 (data not shown). The DNA recovery yield from the BE70G-fixed kidney (mean: 4.68 ± 0.389 µg/mm3) and liver (mean: 3.41 ± 0.077 µg/mm3) tissues was similar to that of BE70 (kidney and liver; mean: 4.96 ± 0.394 and 3.70 ± 0.309, respectively) and NBF (kidney and liver; mean: 4.96 ± 0.193 and 3.40 ± 0.34693, respectively) fixatives (Fig. 5A). DNAs extracted from BE70G-fixed kidney (mean: 12.34 ± 0.064 µg) and liver (mean: 11.52 ± 0.338 µg) tissues were amplified high-quality DNA for nucleic acid array analysis. Similarly, DNAs recovered from BE70-fixed kidney (mean: 12.80 ± 0.389 µg) and liver (mean: 11.78 ± 0.267 µg) tissues were also successfully amplified. However, the DNA prepared from NBF-fixed samples was showed intermediate quality for the microarray application in both kidney (mean: 6.01 ± 0.221 µg) and liver (mean: 5.77 ± 0.396 µg) tissues (Fig. 5B). These data suggest that the nucleic acids extracted from BE70G- and BE70-fixed tissues are compatible for PCR and real-time PCR.
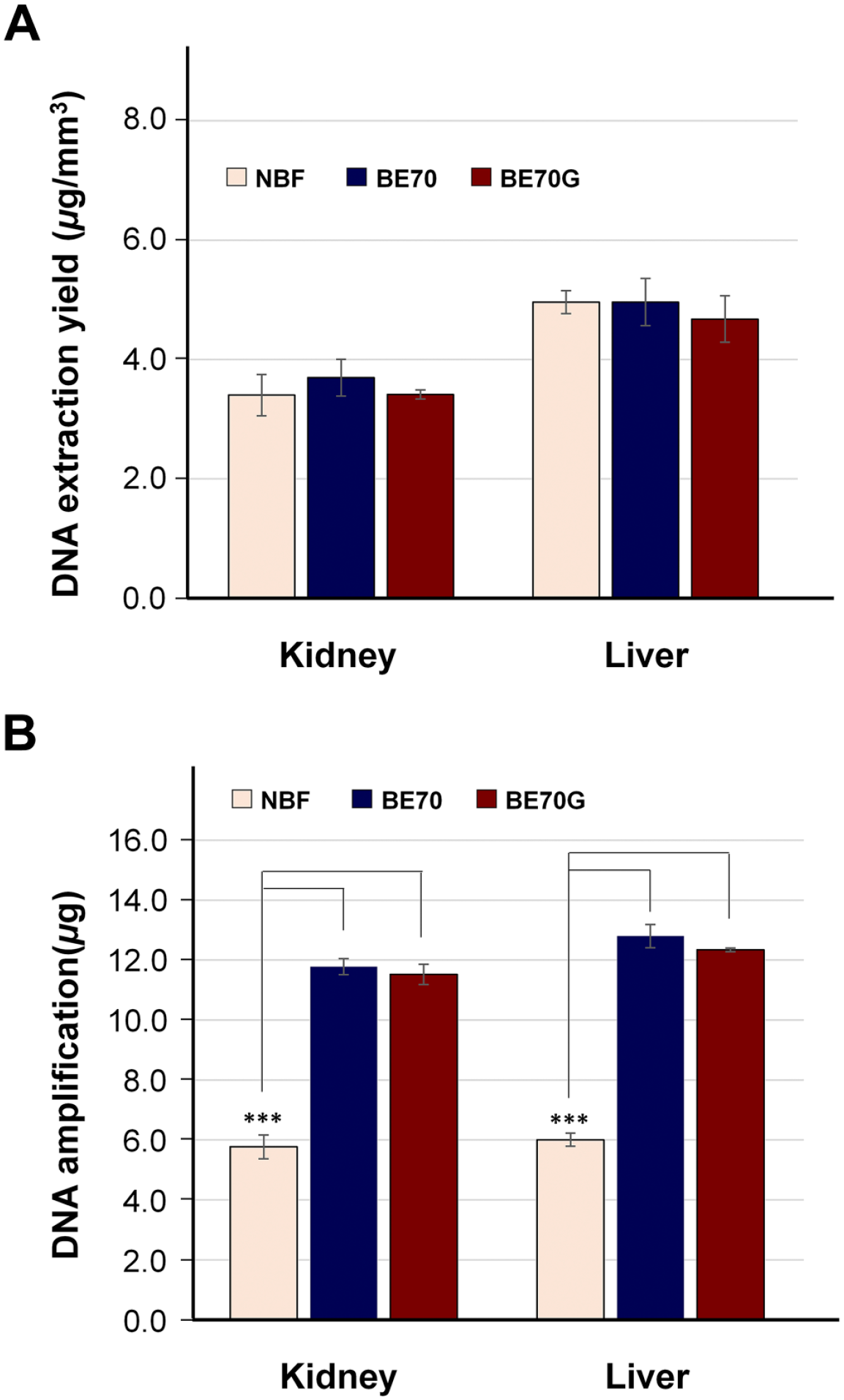
DNA quantity and quality according to the fixatives. (A) The DNA was extracted from mouse kidney and liver in NBF-, BE70-, or BE70G-fixed and paraffin-embedded tissue cores. (B) Amplification of DNA derived from each condition was assessed using the BioScore Screening and amplification kit. Data are the mean ± SD from three independent experiments. Abbreviation: NBF, neutral buffered formalin. *p<0.05, **p<0.01, ***p<0.001.
Histology and Immunoreactivity in Different Fixatives
To compare the staining quality, the various mouse tissues were fixed in NBF, BE70, and BE70G solutions. Figure 6 revealed the representative H&E staining with stomach, kidney, brain, and heart tissue according to fixatives. The overall staining quality of BE70G was similar to that of NBF, although the tissues fixed in BE70 showed the most obvious contrast between nucleus and cytoplasm (Fig. 6). In tissues fixed in BE70G, the cytoplasm was stained without major differences with other fixatives. However, the nuclei showed mild shrinkage of nuclei and smudged nuclear chromatin pattern, which was most noticeable in renal tubule cells (Fig. 6B). In case of immunohistochemical stainings, cytokeratin 14, Ki-67, and E-cadherin were stained with mouse ear skin, mouse spleen, and mouse small intestine tissue, respectively (Fig. 7).
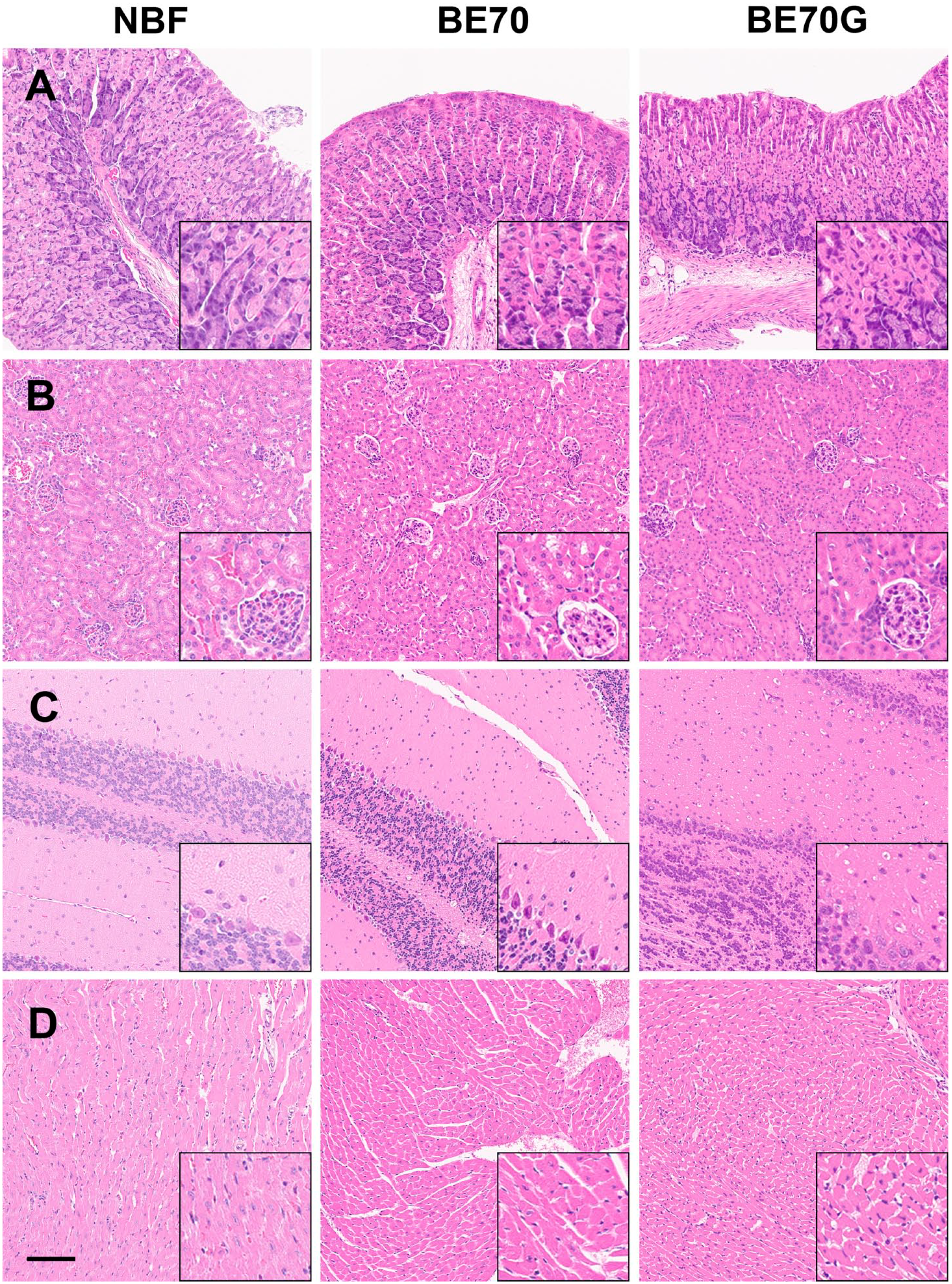
Comparison between BE70G, BE70, and NBF fixation with representative mouse tissues obtained from stomach (A), kidney (B), brain (C), and heart (D). The overall histomorphological features of BE70G are similar to those of NBF. But kidney tissue fixed in BE70G shows blurred nuclear chromatin details and perinuclear halo due to nuclear shrinkage, which is observed in especially renal tubular cells (inlet of B). Scale bar, 100 µm. Abbreviation: NBF, neutral buffered formalin.
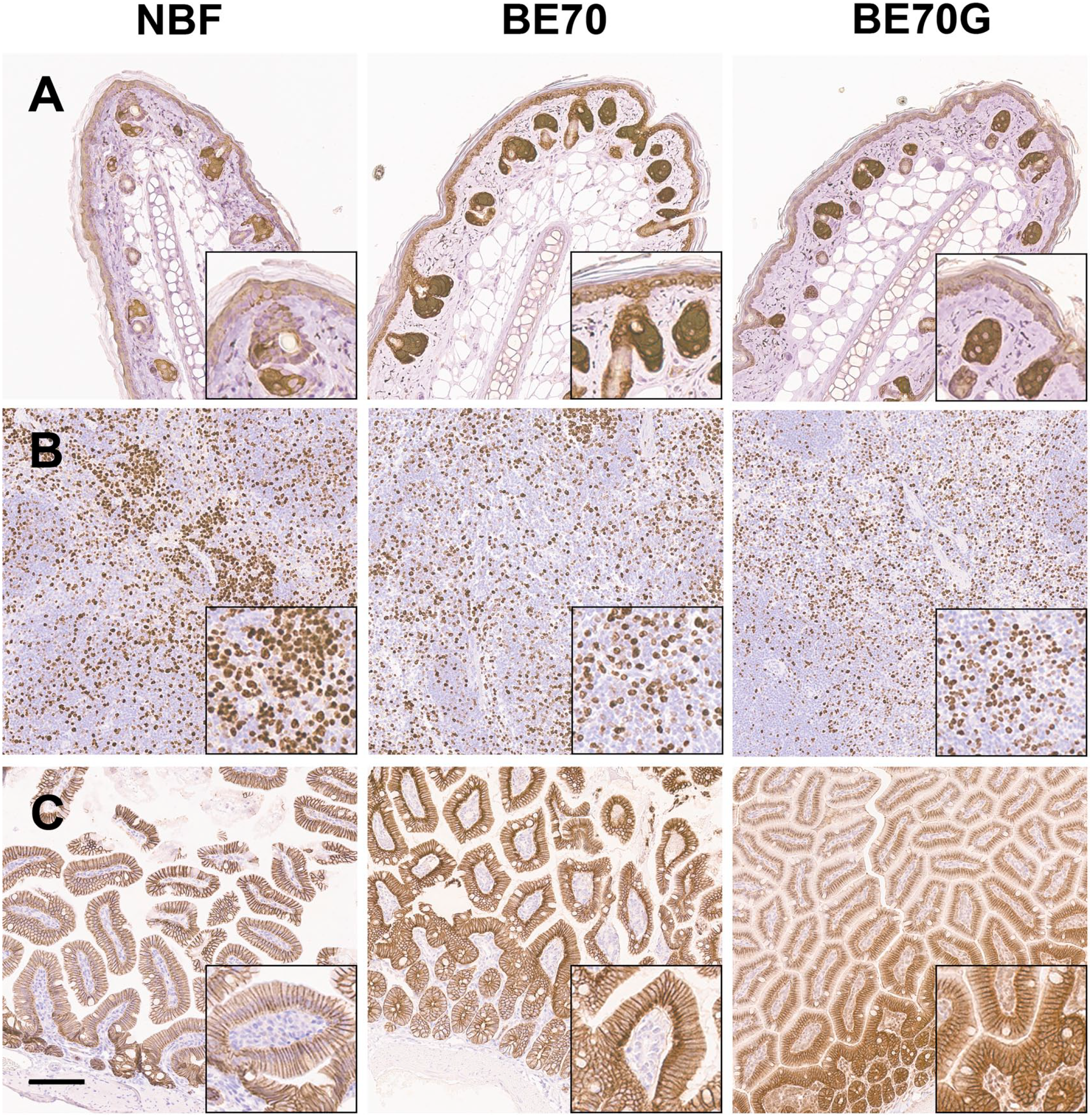
Immunohistochemical stainings for cytokeratin 14 with mouse ear (A), Ki-67 with mouse spleen (B), and E-cadherin with mouse small intestine (C). The cytoplasmic, nuclear, and membranous staining patterns of BE70G are definite and show similar intensity of BE70 or NBF. Scale bar, 100 µm. Abbreviation: NBF, neutral buffered formalin.
Discussion
The addition of guanidinium salt to BE70 improves the quality of both proteins and nucleic acids extracted from the paraffin tissue block. Notably, protein isolated from tissue fixed in BE70G proved significantly higher quality than that recovered from BE70-fixed tissue based on Western blot analysis results. We recently demonstrated coagulative fixatives provide a broader window for fixative time including shorter optimal fixation times. 21 Consistent with the previous data, proteins and RNAs extracted from relatively short-term fixation in BE70G couple with paraffin-embedded tissue is compatible with Western blot and real-time RT-PCR analyses, respectively. Difference in performance with BE70G is more prominent at fixation times at 12 hr and beyond, when the fixation process is complete.
A particularly challenging issue with chemical fixation, paraffin impregnated tissue is the documented degradation of biomolecules. This problem has been appreciated for a number of years27,28; however, the mechanism was unknown. In Xie et al., we demonstrate that antigenicity loss of FFPE tissue slide depends on humidity and temperature during storage. 22 This study confirms our previous finding that the presence of water at storage condition resulted in significant protein degradation and reduced immunoreactivity. More importantly, we demonstrate that the addition of guanidinium salt into BE70 fixative impedes the protein degradation and antigenicity loss during storage at room temperature. The long-term storage of paraffin blocks or slides is one of a keystone for translational research that rely on the collection and storage of specimens for future assays. Unfortunately, tissue sections resulted in a considerable loss of antigenicity after a short-term storage.3,27,29 The lack of an appropriate storage methodology remains a challenge in basic and clinical studies exploration of biomarkers because the loss of antigenicity likely contributes to misleading test results. 29 It has been assumed that the oxidation is a major mechanism for the degradation of antigenicity. Prior studies addressed the same benefits of storing slides at low temperature and also appreciated advantages and disadvantages through the blocking of oxidation by means of paraffin coating and nitrogen desiccator.28,29 We previously demonstrate that endogenous and exogenous water originated from inappropriate tissue fixation and processing or storage condition resulted in significant biomolecule degradation through hydrolysis.5,8,22
The mechanism by which guanidinium improves biomolecule quality in the setting for chemical fixation and paraffin impregnation appears to be through its mechanism of action as a primary denaturing agent for proteins and chaotropic action in the protection of nucleic acids. Guanidinium, known as carbamimidoylazanium by the International Union of Pure and Applied Chemistry (IUPAC) naming convention has a molecular formula of [CH6N3]+ in the cation state and is available as salt, commonly with thiocyanate, but also with X and Y. The activities of denaturation and chaotropic activity are functionally identical to the disruption of hydrogen bonding. We came to evaluate guanidinium due to the performance, but noted limitations of two similar applications. RNAlater, which is an ammonium salt, has been used extensively for the protection of RNA in frozen tissue. Although there are protocols for its application of RNAlater with subsequent formaldehyde fixation, the process is complicate and the histomorphology is impaired. This model does not account for the improvements in biomolecule recovery and performance during shorter fixation times. In this setting, guanidinium may be enhancing the penetration of the fixative through the lipid bilayer.
In 2016, Boi et al. 30 described the use of disaccharides in antigen retrieval buffers to prevent the damage resultant from drying artifact. It was clear that neither chemical was compatible with the entire process of fixation and impregnation. We subsequently noted Bolognesi et al. 31 used guanidinium hydrochloride for stripping antibodies in multiplex/multi-round immunohistochemistry. Based on the biomolecular characteristics, we evaluated guanidinium salts as a constituent of BE70. Our initial studies evaluated different salt formulations and found them equal in performance (data not shown), however carried these studies out due to cost issues. The biomolecular protective features of guanidinium require that it be present during fixation. We have conducted additional studies, evaluating the removal of dissolved oxygens and other reactive gases in the fixative and solutions used in impregnation, including the paraffins, by means of sparging with Argon (Patent Number US9,714,409 B2) 32 support a model whereby the disruption of the hydrogen bonds, presumably with “unfreezable water” (Bolognesi) are capable of protecting the molecules from degradation by means of hydrolysis. We note that alternative concentrations of guanidinium in ethanol-based, as well as formaldehyde-based, fixatives can be formulated; however, the tissues become hardened and cannot be evaluated.
Tissue handling and processing steps directly impact on tissue block quality and its susceptibility for subsequent genomic and proteomic analysis.3,5,21 Gillespie et al. 9 demonstrated that ethanol-based fixative allows better quality of biomolecules for prolonged periods of time compared with NBF. We recently confirmed and expanded that coagulative fixatives have wide range of fixation time from 4 hr to 3 months at room temperature, while NBF has narrow window from 12 hr to 1 week. 21 Prior studies have demonstrated that a minimum of 6 hr in NBF allows reliable results for estrogen receptor, progesterone receptor, and HER-2 expression in invasive breast cancer.33,34 However, prior studies of shorter fixation methods exclusively focused on histomorphological and immunohistochemical analysis and did not assess the quantity and quality of protein and nucleic acids in molecular assays. Our results show that AKT and GAPDH immunoreactivities of proteins extracted from tissue fixed in BE70G for a minimum of 4 hr higher than those of BE70 fixative (Fig. 2), suggesting guanidinium salt could accelerate the process.
In this study, we demonstrate that the guanidinium salt supplemented fixative retained higher quality of proteins than that of BE70 fixative based on Western blotting (Fig. 3), whereas the quality of RNA slightly improved (Fig. 4). Furthermore, guanidinium salt contributed protection of protein immunoreactivity in both dry and wet storage conditions (data not shown). Guanidinium salt is strong chaotropic agent that can disrupt hydrogen bonding network between water molecules and cause protein denaturation. 30 The working hypothesis is that guanidinium replaces non-freezable water, and prevents hydrolysis and oxidation of the molecules. It is reasonable that the guanidinium protect diminution of protein quality in the paraffin blocks and tissue sections, presumably through prevention of hydrolysis and oxidation. Although we have noted the potential mechanism of antigenicity protection by addition of guanidinium salt, further studies will be required to define maintaining of protein integrity.
The overall staining quality of BE70G fixed tissue was similar to that of NBF on H&E staining. The blur of nuclear chromatin and mild nuclear shrinkage in size were observed, especially renal tubular cells (Fig. 6B). This difference in nuclear morphology may represent a difference in nuclear penetration of BE70G compared with BE70. These nuclear changes could adversely effect on routine pathology diagnosis, because the detailed chromatin pattern can provide evidences to diagnose cancers. 35 However, there was no significant difference in staining pattern and intensity between NBF, BE70, and BE70G on immunohistochemical staining (Fig. 7). In case of delayed staining after tissue sectioning, the tissue fixed in BE70G showed more preserved stainability and antigenicity compared with NBF or BE70 on routine H&E and immunohistochemical staining, especially storing more than 3 months in humid condition (data not shown). Therefore, BE70G fixative are less suitable for routine pathology diagnosis than NBF or BE70, but may be more useful for long-time storage of unstained slides.
In conclusion, the addition of guanidinium salt to BE70 fixative enhanced protein and RNA quality based on Western blot and real-time polymerase chain reaction analysis results, respectively. The excellent protein preservation of BE70G fixative might be relevance in proteomic analysis, which is a highly sensitive molecular test combined with small amount of clinical tissue specimens, with keeping the quality of histopathological analysis. Further analysis of proteomic and genomic investigation needs to fully appreciate the benefits of BE70G as a potential substitute for NBF in the research and clinical setting.
Supplemental Material
sj-tif-1-jhc-10.1369_00221554231159451 – Supplemental material for The Application of Guanidinium to Improve Biomolecule Quality in Fixed, Paraffin-embedded Tissue
Supplemental material, sj-tif-1-jhc-10.1369_00221554231159451 for The Application of Guanidinium to Improve Biomolecule Quality in Fixed, Paraffin-embedded Tissue by Joon-Yong Chung, Kyungeun Kim, Kris Ylaya, Katharine E. Walker-Bawa, Candice Perry, Robert A. Star and Stephen M. Hewitt in Journal of Histochemistry & Cytochemistry
Footnotes
Competing Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The National Institutes of Health have filed a patent application (WO2017083729A3) for the fixative described herein. Ms. Perry, Dr. Chung, Dr. Star, and Dr. Hewitt are listed as inventors on this patent application; however, the application is assigned to the U.S. Department of Health and Human Services, as the work was performed under official duty (J.-Y.C., R.A.S., and S.M.H.).
Author Contributions
J-YC, and SMH conceived the study and devised the experimental design. J-YC, KY, KEW-B, and CP performed the experiments. J-YC, KK, RAS, and SMH contributed to the interpretation of the results. J-YC and KK wrote the manuscript consulting with SMH. All authors provided critical feedback and helped the research and manuscript and reviewed the final manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the Intramural Research Program of the National Institutes of Health, National Cancer Institute, Center for Cancer Research.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
