Abstract
The antigen retrieval (AR) technique, which is predominantly based on high-temperature heating of tissues, is used as a non-enzymatic pretreatment for immunohistochemical staining of formalin-fixed, paraffin-embedded tissue sections. It has been widely applied in pathology and analytical morphology. The existence of a growing body of literature on the AR technique raises a number of interesting issues for the further development of AR. These issues include the use of a “test battery” and the concept of “maximal retrieval” applied to the selection of optimal test protocols for the standardization of AR.
Keywords
FORMALDEHYDE, originally discovered to be a tissue fixative by Ferdinand Blum in 1893, has been used widely to the present day (Fox et al. 1985; Puchtler and Meloan 1985). As a fixative, formalin is made up of commercial concentrated formalin (37–40% solution of formaldehyde) diluted to a 10% solution (3.7–4% formaldehyde). Formalin has several advantages over alcohol, particularly the superior preservation of morphological detail. Because of the long history of the use of formalin as the standard fixative for tissue processing in histopathology, most of the criteria for pathological diagnosis have been established by the observation of formalin-fixed, paraffin-embedded tissue sections stained with hematoxylin and eosin. For this reason, although many other fixatives exist and some may be superior for immunohistochemistry (IHC), none has supplanted formalin in general use.
The recent development of IHC has provided a revolutionary approach to histopathology in terms of analytical and functional morphology. However, wide-spread application of IHC has raised a critical issue concerning traditional formalin fixation because most antigens under investigation are influenced significantly and adversely by formalin fixation (Taylor 1979, 1980, 1994b; Taylor and Cote 1994; Leong and Gilham 1989; Battifora and Kopinski 1986). This problem has proved to be a great challenge to histopathologists attempting to apply immunohistochemical staining to formalin paraffin sections that constitute the daily practice of pathology. A series of technical developments have helped to improve the immunohistochemical method (Taylor and Cote 1994; Elias 1990; Pinkus 1982; Hsu et al. 1981).
The development of the hybridoma technique was a major contribution, serving to facilitate the large-scale manufacture of many highly specific monoclonal antibodies (MAbs) (Kohler and Milstein 1975). The critical importance of rendering the IHC technique suitable for routine paraffin sections was pointed out by Taylor and Burns (1974). The utility of IHC in surgical pathology increased with the use of enzyme digestion (Huang 1975), other pretreatments for unmasking antigens (Kitamoto et al. 1987; Hausen and Dreyer 1982; Elias and Johnsen 1979), and the development of antibodies against formalin-modified antigens (Taylor and Cote 1994; Harrach and Robenek 1990). However, the use of many antibodies for routinely processed paraffin sections continued to be limited in spite of these enhancement techniques and improvements in the detection system (Leong et al. 1988).
With the recognition that formalin fixation causes major chemical changes in antigens by unknown mechanisms, several attempts were made to modify the methods of fixation, and a variety of modified fixatives have been proposed for preservation of certain antigens (Taylor and Cote 1994; Dapson 1993; Elias 1990; Colvin et al. 1988; DeLellis 1988; Larsson 1988; Hopwood 1985). However, no ideal fixative has been found that can be used universally in IHC. In addition, the modified fixatives do not closely reproduce the preservation of morphology achieved by formalin fixation. A change of fixative, therefore, may also mean changing the histopathological criteria employed for diagnosis. A direct consequence would be loss of the benefit of the rich collection of archival paraffin tissues the world over. These were the primary thoughts behind the development of a novel antigen retrieval (AR) technique, to maintain formalin as the standard fixative for both morphology and immunohistochemistry. A key question, in solving this dilemma was whether formalin fixation modified antigens in a reversible or an irreversible manner. Biochemical studies of the chemical reaction between protein and formalin by Fraenkel—Conrat and co-workers in the 1940s (1947,1948a,b) indicated that hydrolysis of crosslinkages between formalin and protein is limited by certain amino acid side chains, such as imidazol and indol, but that these crosslinkages can be reversed by high-temperature heating (120C) or strong alkaline treatment. This observation formed the basis for the development of AR techniques in 1991 (Shi et al. 1991,1992a). A simple method of either heating the routinely fixed paraffin-embedded tissue sections in water or immersing the routinely formalin-fixed, acid-decalcified, celloidin-embedded tissue sections in an NaOH-methanol solution yielded dramatic retrieval results (Shi et al. 1991,1992a,b,1993a). The AR technique has been used increasingly and has demonstrated its value in many studies for over 200 antibodies (Taylor et al. 1996; Werner et al. 1996; Boon and Kok 1995; Brown and Chirala 1995; Cattoretti and Suumeijer 1995; Shi et al. 1995b; Cuevas et al. 1994; Gu 1994; Gu et al. 1994a,b; Igarashi et al. 1994; Kawai et al. 1994a; Swanson 1994; Taylor and Cote 1994; Cattoretti et al. 1993; Gown et al. 1993a; Leong and Milios 1993; McKee et al. 1993; Norton 1993; Suurmeijer and Boon 1993a,b; Shi et al. 1991).
The worldwide use of the AR-IHC has, however, raised some questions concerning the methodology of heating, the factors that influence the AR-IHC, the variety of AR solutions used for heating the tissue sections, limitations and possible false positive staining, and the mechanism of AR. The purpose of this review article is to address several critical issues concerning AR-IHC, including the factors that influence the AR-IHC. Proposals for standardization of AR-IHC through the use of a “test battery” with the concept of “maximal retrieval” are also described, together with our hypothesis of the mechanism of AR.
Recent Developments
Methodology
Recently, we conducted a test to demonstrate that an optimal result of AR-IHC is correlated with the product of heating temperature (T) X time of AR heating treatment (t); “T × t” means the heating condition. For example, the lower the temperature of heating used for AR, the longer the time required to reach the same intensity of AR-IHC compared with that obtained by higher-temperature heating. Our test was based on AR-IHC of MAb MIB1 in archival paraffin sections, and obtained the same strong intensity by the following heating conditions: 100C for 20 min, 90C for 30 min, 80C for 50 min, and 70C for 10 hr (Shi et al. 1995c; Taylor et al. 1995).
We conducted a study concerning the influence of pH on AR-IHC and observed three distinct patterns of staining: stable type (which changed slightly with pH); V-form type [which gave the best results at extremes (high or low) of pH], and ascending type (which improved with increasing pH). This study was performed using seven buffer solutions. The pH value achieved proved more valuable than the chemical composition of the buffer solution (Shi et al. 1995a). On the basis of this study, we recommended the use of AR solution at higher pH value for general application, giving results that are marginally superior to the pH value of 6 in widespread use today. Although the low-pH AR solution may yield the strongest intensity for some antibodies such as ER, PR, Rb, thrombospondin, and MIB1, weak false-positive nuclear staining may also be found in focal areas for some antibodies tested, giving rise to background staining. In these cases, a negative control slide must always be reviewed, and caution must be taken in interpreting the results of the low-pH AR solution.
Application
The use of the MW heating AR technique for successful immunodetection of androgen receptor was developed in our laboratory (Taylor et al. 1994a; Shi et al. 1993b) and was subsequently demonstrated by Iwamura et al. (1994), Janssen et al. (1994), and Loda et al. (1994). This has led to further studies on the assessment of potential androgen responsiveness by studying the androgen receptor status in metastatic prostate carcinoma and in prostate needle biopsy specimens and in monitoring the response to anti-androgen therapy. In addition, a series of retrospective studies has been performed on archival tissues to demonstrate the prognostic significance or the potential utility of androgen receptor as a specific marker in the differential diagnosis of metastatic adenocarcinoma with an unknown primary site (Loda et al. 1994). Bremner et al. (1994) studied immunolocalization of the androgen receptor in rat testis by the AR technique and demonstrated that androgen receptor immunolocalization in Sertoli cells is stage-dependent. The use of AR-IHC in analytical morpholgy may create a wide field for retrospective studies designed to understand function and pathogenesis. For example, Gown et al. (1993b) performed a retrospective study on archival tissue of nasopharyngeal angiofibroma by using androgen receptor immunolocalization after AR treatment. They demonstrated a correlation of androgen receptor with the pathogenesis of angiofibroma, confirming the hypothesis concerning the etiology of male angiofibroma (Toomey 1980).
For immunoelectron microscopy, the AR technique has successfully been applied by using the MW heating AR method either after etching the grids with a 10% fresh saturated solution of sodium ethoxide diluted with anhydrous ethanol for 2 min or with a saturated aqueous solution of sodium metaperiodate for 1 hr (Stirling and Graff 1995), or by directly heating the grid and followed by some washing procedures including 50 mM NH4Cl and 1% Tween 20 (Wilson et al. 1996).
The MW heating method was also used for enhancement of mRNA in situ hybridization (Lan et al. 1996; McMahon and McQuaid 1996; Sibony et al. 1995) and for the TUNEL method with the use of short MW heating for 1 min (Strater et al. 1995). However, for the TUNEL method it may have some limitations, as recently reported by Lucassen et al. (1995). They found that the heating method used for enhancement of in situ end-labeling yields strong non-specific labeling for some briefly postfixed perfused brain or immersion-fixed postmortem brain tissues, although the heating method may benefit other conditions of tissue processing, such as a prolonged postfixation of perfusion-fixed rat brain. McMahon and McQuaid (1996) emphasized a combination of enzyme and microwave pretratment for detection of measles virus by in situ hybridization, particularly in long-term formalin-fixed paraffin tissues.
Recently, the AR technique has also been successfully applied to circumvent the loss of p53 antigenicity in stored paraffin tissue sections (Kato et al. 1995).
Current Issues
Issues of Standardization
The standardization of IHC is an important issue that involves three areas: antibodies and reagents, technical procedures, and interpretation and reports (Taylor 1994a). Perhaps the most difficult issue is the largely unknown but clearly adverse influence of fixation and processing of the paraffin-embedded tissue. Many studies concerning formalin fixation of tissues have shown that the influence of formalin on tissue antigenicity is dependent on the total time of fixation (Battifora and Kopinski 1986; Fox et al. 1985). This raises the next question: How can the immunostaining results on hundreds of paraffin blocks that are fixed in formalin for an unkown time be standardized? As mentioned above, a variety of modified fixatives and processing procedures have been used in an attempt to standardize IHC, but have failed. The AR technique can be used to help standardize IHC on archival paraffin sections (Deluque and Luque 1995; von Wasielewski et al. 1994; Taylor 1994a; Suurmeijer and Boon 1993a,b; Swanson 1993). On the basis of the maximal retrieval level, it may also be possible to equalize the immunoreactivity in a variety of archival paraffin tissue sections that are fixed in formalin for various periods of time. In fact, the results of immunostaining on routinely processed tissue using AR are usually equal to (and occasionally better than) the frozen tissue. Several studies have suggested that increased heating may reverse the effects of longer fixation (Munakata and Hendricks 1993).
What is the optimal heating condition and optimal pH value of the AR solution to obtain the maximal retrieval level for AR-IHC with the most commonly used antibodies in pathology? Further studies are necessary to address these questions. The potential effect of the AR technique in standardization of IHC has been indicated by some studies regarding ER-AR-IHC based on comparison between frozen, archival paraffin tissue sections, biochemical assay, and clinical behavior. As an example, a comparison of AR-IHC using antibodies to p53 has been summarized in Table 1. First of all, the necessity of adopting AR-IHC routinely for detection of p53 in archival tissue sections has been demonstrated by all the data shown in Table 1, which demonstrated that the intensity of p53 with the use of AR treatment was much stronger than that obtained by regular immunostaining without AR, the increasing rate ranging from 13% to 42% for PAb 1801 and 34% to 56.4% for DO7. The discrepancies in the increasing rate may be caused by the use of different AR protocols. Our recent experience also indicated the same conclusion, and we have established an optimal protocol for detection of p53 using AR-IHC in diagnostic pathology. Second, the possibility of standardizing the AR protocol was indicated by data reported from three independent research groups (Binks et al. 1994; Lambkin et al. 1994; Terada et al. 1994), showing very similar values of increasing rate, 50%, 56.4%, and 54% for the MAb to p53, DO7, respectively, when using either PBS or citrate buffer solutions at pH 6 to 7 as the AR solutions with MW or conventional heating under similar heating conditions (T X t), although the specimens are different from each group. Third, reevaluation of the cut-off point of p53 is a necessary issue in comparing the increasing rate between the two groups of AR and without AR, with the increasing rate between the frozen tissue section and archival paraffin section. In addition, this increasing rate generated by AR-IHC was larger than that generated by a comparison between frozen and paraffin sections. For example, Barton et al. (1991) reported the increasing rate of frozen vs archival paraffin sections as 37% with the use of the polyclonal antibody to p53, CM1, while a higher increasing rate representing the differences between AR and without AR groups by using the same antibody CM1 on archival paraffin sections could be indicated as 47% (Terada et al. 1994). Therefore, the cut-off point of p53, based on different conditions of IHC, may indicate different clinical significances, which need reevaluation as described below. This interesting phenomenon can also be demonstrated when AR-IHC is performed on archival paraffin sections with the use of other antibodies such as ER, PR, MIB1, PCNA, and RB (Shi et al. 1996; Gee et al. 1995; Barbareschi et al. 1994; Battifora 1994a). It has been suggested that frozen tissue sections can be replaced by archival paraffin tissue sections with the use of AR-IHC (Battifora 1994a). To a degree this is true. However, because the protocols of the AR method are not yet unified, caution should be taken in interpreting the intensity of AR-IHC for qualitative as well as quantitative purposes. To reach the goal of standardization of IHC on routine archival paraffin sections using the AR technique, the following issues may be important.
Increasing rate of p53-immunostaining obtained by the AR technique
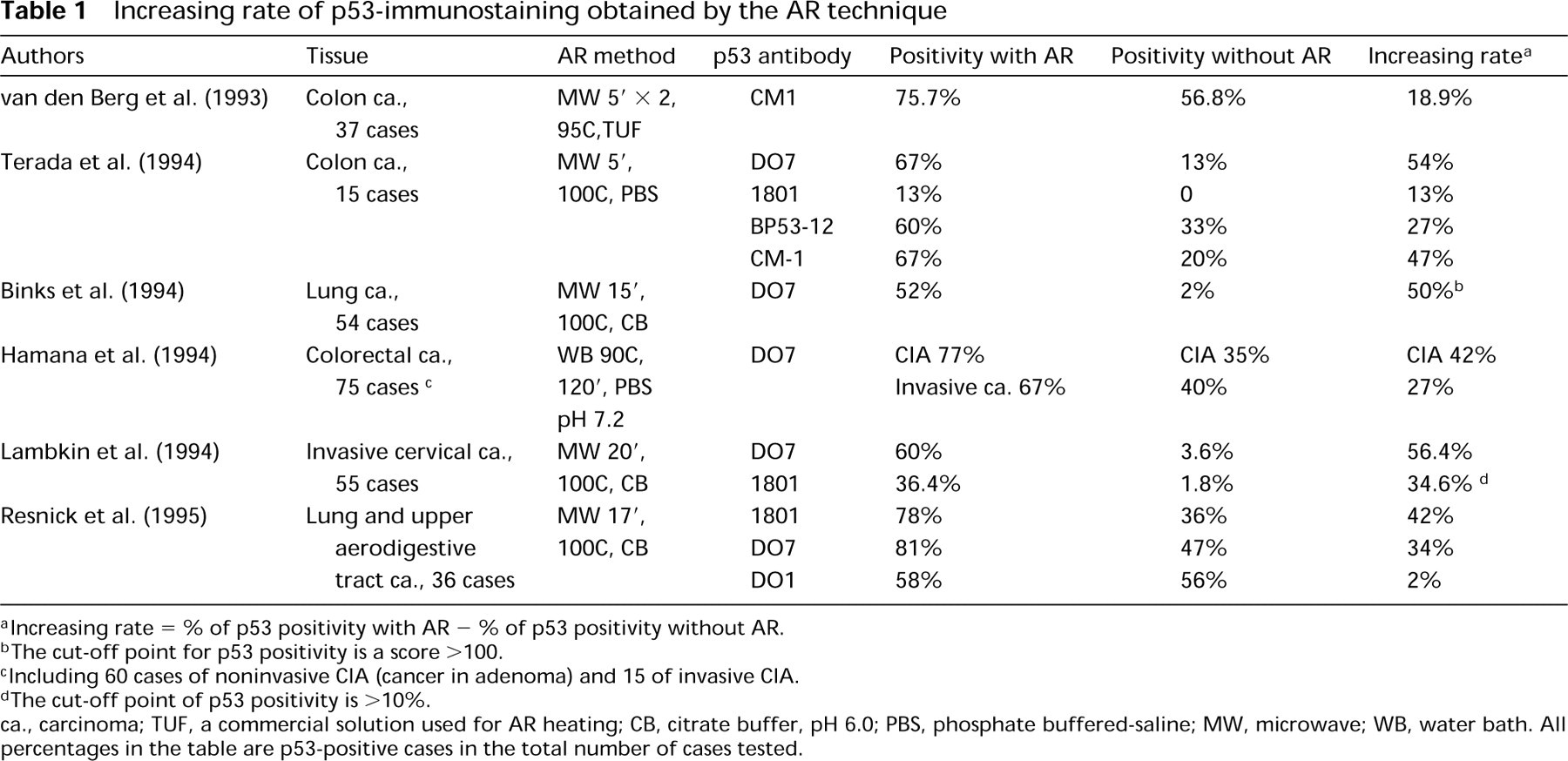
aIncreasing rate = % of p53 positivity with AR - % of p53 positivity without AR.
bThe cut-off point for p53 positivity is a score <100.
cIncluding 60 cases of noninvasive CIA (cancer in adenoma) and 15 of invasive CIA.
dThe cut-off point of p53 positivity is <10%.
ca., carcinoma; TUF, a commercial solution used for AR heating; CB, citrate buffer, pH 6.0; PBS, phosphate buffered-saline; MW, microwave; WB, water bath. All percentages in the table are p53-positive cases in the total number of cases tested.
“Test Battery”
This concept for the development of optimal protocols for AR-IHC is based on previous studies concerning the two major factors that influence the effect of AR-IHC, i.e., heating condition and the pH value of the AR solution (Shi et al. 1995a,b,c; Taylor et al. 1995). Three levels of heating times and three pH values of the AR solution, ranging from low (pH 1–2), middle (pH 6–8), and high (pH 10–11) can be used, as illustrated in Table 2. Recently, we have demonstrated that different heating methods (including MW, MW and pressure cooker, steam, and autoclave heating methods) (Taylor et al. 1995), can be evaluated in a similar fashion and adjusted to yield similar intensities of staining by AR-IHC. In general, there is a correlation between the heating condition and the pH value of the AR solution: When a middle or high-pH solution is used, the higher the temperature, the better the result for most nuclear and cytoplasmic antibodies tested. In contrast, when a low-pH solution is used, intense heating (such as in a pressure cooker) may yield a poor result. For example, the strongest intensity of MIB1 or RB on archival paraffin sections by AR-IHC was generated by either regular MW heating with low-pH solution or by high-pH solution with intense heating (pressure cooker or prolonged regular MW heating method), but not low pH with intense heating (Shi et al. 1996).
Test battery suggested for screening an optimal AR protocol a
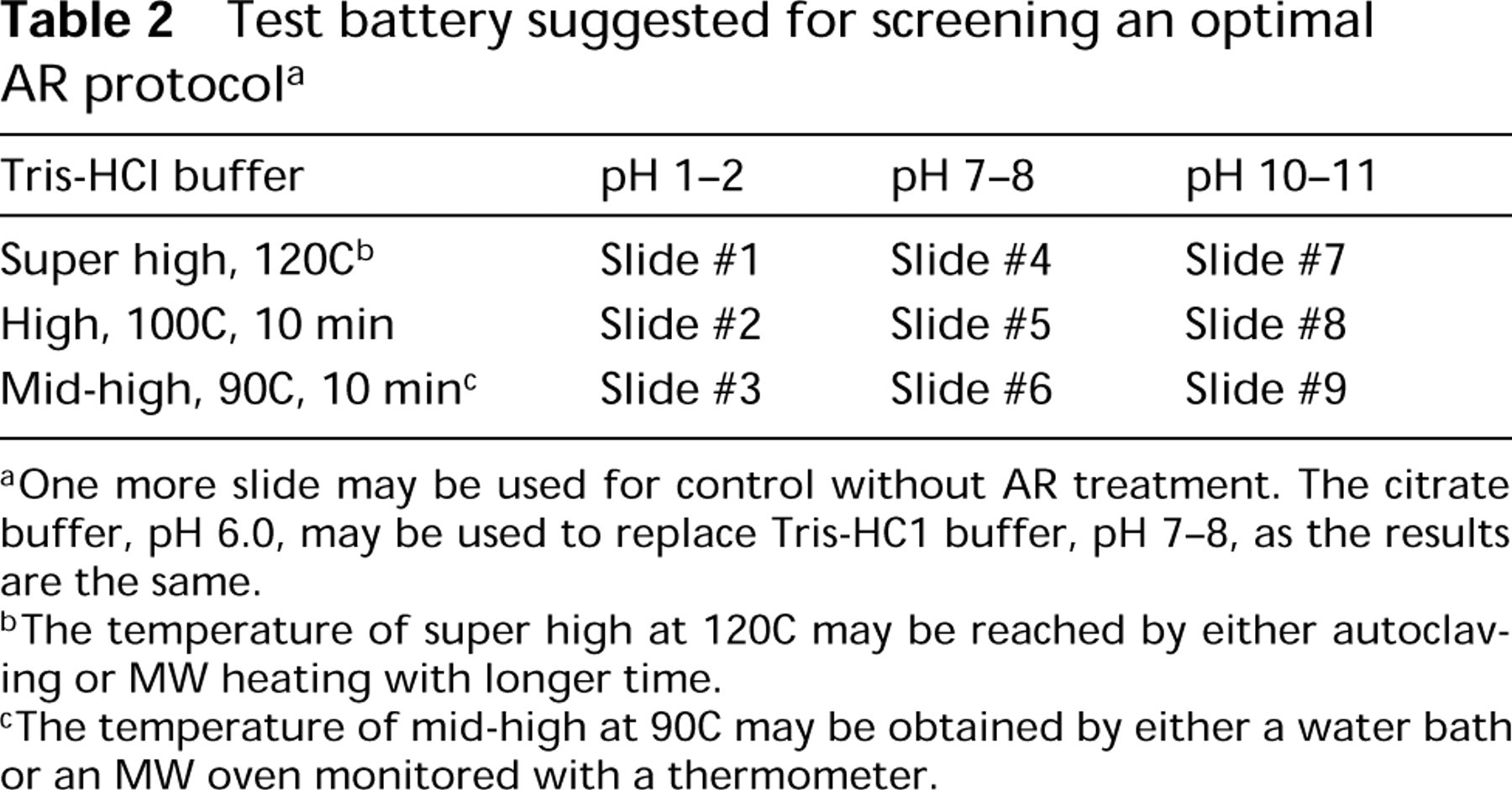
aOne more slide may be used for control without AR treatment. The citrate buffer, pH 6.0, may be used to replace Tris-HC1 buffer, pH 7–8, as the results are the same.
b The temperature of super high at 120C may be reached by either autoclaving or MW heating with longer time.
c The temperature of mid-high at 90C may be obtained by either a water bath or an MW oven monitored with a thermometer.
The test battery is a rapid screening approach to develop an optimal protocol for any antibody to be tested. It is an important step in establishing the maximal retrieval level for formalin-masked antigens with a variety of fixation times, to standardize immuno-staining results. In our recent study of 14 antibodies using the test battery method, we demonstrated that the strongest intensity of AR-IHC for most antibodies tested was achieved either using low-pH buffer as the AR solution with regular MW heating conditions, or high pH with intense heating conditions (such as autoclave heating or MW heating for a prolonged heating time) (Figure 1) (Shi et al. 1995c,1996). In addition, the use of the test battery may reduce some false-negative AR-IHC staining results. As indicated in Figure 1, positive immunostaining of thrombospondin can be obtained only with the use of an AR solution at low pH.
Standardization of AR Technique
Reevaluation of the Clinical Interpretation of AR-IHC
To demonstrate the clinical significance of AR-IHC, it is necessary to compare the intensity of AR-IHC with that obtained by routine IHC (without AR) of frozen and paraffin sections and to study the correlations among the three conditions. This type of comparison was, in fact, done in a recent study on Ki-67, MIB1, and PCNA expression on frozen and paraffin sections of breast cancer (Gee et al. 1995). On the basis of the results of AR-IHC, the authors reevaluated the clinical significance, and suggested that a new cut-off point should be applied. Linden et al. (1994,1995) compared the p53 immunoreactivity of three antibodies (listed according to the intensity of staining): DO7 (Dako; Carpinteria, CA), BP53-12 (BioGenex; San Ramon, CA) and 1801 (Oncogene Science; Uniondale, NY), by using the MW AR method. They concluded that wild-type p53 may be detectable in normal cells after using AR, and pointed out the importance of technical conditions as well as the evaluation of normal tissue controls in the clinical application of p53 immunostaining. Goulding et al. (1995) performed a comparative study based on two MAbs to ER, 1D5 (Dako) and H222 (Abbott; North Chicago, IL) for both fresh cryosections and archival formalin-fixed, paraffin-embedded tissue sections of 90 cases of breast carcinoma. For the archival paraffin tissue sections with the use of MAb 1D5, the MW AR method was used before immunostaining. The comparison of H score between 1D5 (archival tissue + AR), H222 (archival tissue + enzyme digestion), and H222 (frozen section) showed a significant correlation, although the group of 1D5-AR-IHC showed the highest H score. These authors further calculated the specificity and sensitivity of the ER status in predicting response to tamoxifen therapy based on the UICC (Union International Contre le Cancer) criteria for evaluation of clinical reponse to tamoxifen therapy. By comparing the three different “cut-off” H scores of 30, 50, and 100, they found that 1D5 was more sensitive but slightly less specific than H222. They concluded that it was necessary to reevaluate the clinical data with the ER status assessed by a new antibody with the use of the AR technique to establish a valid cut-off point. In a similar study, Pertschuk et al. (1996) concluded that paraffin section IHC (with ER1D5) is now the method of choice for evaluation of ER status in predicting prognosis and response to therapy.
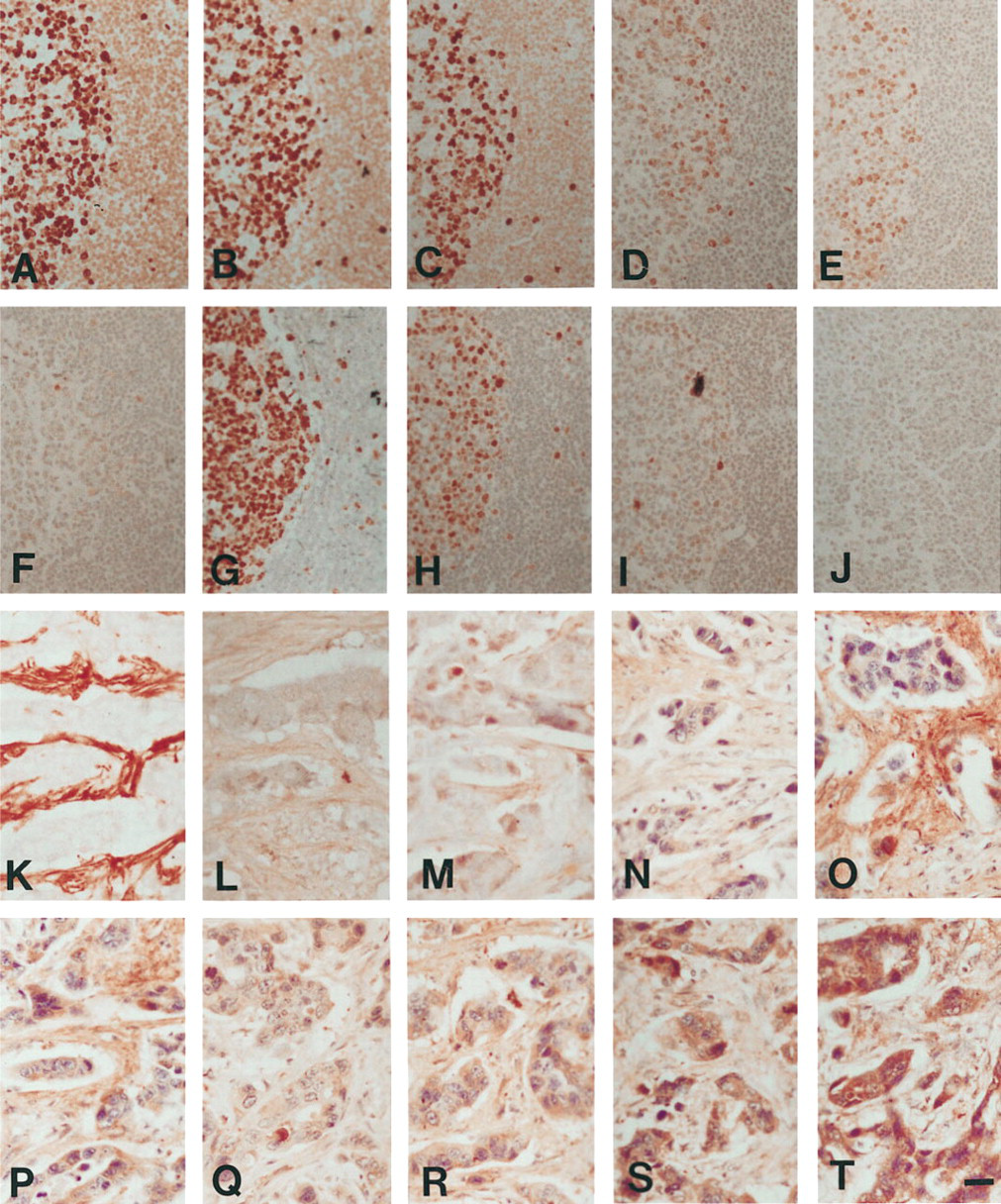
Comparison of intensity of AR-IHC by using the test battery for MAb MIB1 on sections of tonsil
One issue that must be addressed is the threshold of p53 nuclear reactivity which should be considered as prognostically significant in cancers, as pointed out by Battifora (1994b) and by Dowell and Hall (1995). As indicated in Table 1, variable results of AR-IHC for p53 may be obtained with different reagents, methods, and interpretation of AR-IHC used by different authors. What gold standard can be applied to demonstrate the optimal score of p53 or other prognostic markers? Increased intensity of AR-IHC may not correlate with gene status or tumor behavior, and it is therefore extremely important to continually re-assess the results obtained by AR-IHC with the biological parameters and with the clinical outcome.
In summary, using the test battery may help to establish the optimal protocol for the immunostaining intensity of any antibody at a maximal retrieval level. The development of optimal protocols will make it possible to compare results among different laboratories and may provide a meaninful cut-off point for clinical significance after comparison with standard techniques and clinical follow-up.
Remaining Problems and Artifacts
Is there any false positivity of immunostaining after the AR technique is used for immunostaining? This is a critical issue in interpreting the results of AR-IHC. Thus far, most studies have reported satisfactory results, without false positivity, for a variety of antibodies tested (Brown and Chirala 1995; Lavieille et al. 1995; Cuevas et al. 1994; Igarashi et al. 1994; Taylor et al. 1994a; Tenaud et al. 1994; von Wasielewski et al. 1994; Cattoretti et al. 1993; Dookhan et al. 1993; Gown et al. 1993a; Leong and Milios 1993; Shi et al. 1991). Most recently, Baas et al. (1996) questioned whether a potential false-positive result of AR-IHC using the MAb DO7 might be obtained after extreme AR treatment. However, this requires further demonstration by both frozen section and other biochemical assay, since the immunolocalization of p53 in paraffin sections after AR treatment is comparable to the pattern obtained in frozen sections. Some studies have reported nonspecific staining and unwanted immunoreactivity with AR-IHC, using a broad panel of antibodies, particularly by use of different heating conditions and/or different AR solutions with a variety of pH values. For example, some weakly positive nuclear staining was found when a low-pH AR solution was used for the MAb to MIB1 (Shi et al. 1995a); Sebenik and Wieczorek (1995) showed that the nonspecific nuclear staining after AR was caused by the secondary antibody, because exclusion of the second antibody resulted in no nonspecific nuclear staining in their test. Therefore, re-titration of the second antibody may be necessary if nonspecific staining occurs when AR-IHC is used. Focal keratin staining could be found in archival paraffin sections of malignant melanoma and plasma cell tumor when the MW AR-IHC was used, which may have been caused by the detection of low levels of keratins in these tumors (Guiter et al. 1995). One potential cause of increased background after AR heating may be the unmasking of endogenous biotin, which is also found in frozen tissue sections. This effect can be obviated by use of a routine blocking procedure (Macrea and Towne Poster presentation; 1995 Natl Soc Histotech Ann Symp). It is important to keep in mind that any background staining that may be found in frozen tissue sections can also be found in the MW-heated archival paraffin sections, because the paraffin sections after AR treatment have the same sensitivity or even higher sensitivity than frozen sections.
Mighell et al. (1995) recently reported interesting variable patterns of immunoreactivity to an anti-fibronectin polyclonal antibody in routinely processed oral tissues. They indicated that the immunostaining pattern of fibronectin in frozen tissue sections was localized in both connective tissue and around vascular endothelial cells, but was immunolabeled in the vascular region only by the enzymatic digestion method, in connective tissue after MW AR treatment, and in both connective tissue and an unwanted nuclear staining of the epithelium after autoclave AR treatment for archival paraffin sections. It should be noted that the use of a test battery to select an optimal AR protocol is based on a comparison with the immunostaining pattern obtained in frozen sections to avoid any false-positivity or unwanted change of pattern.
Further Development of AR
Study of the Mechanism of AR
AR-IHC has shed some light on our understanding of the mechanism(s) of protein-formalin interactions. First of all, it showed that the modification of protein structure by formalin is reversible under certain conditions, such as high-temperature heating or strong alkaline treatment. We have suggested a possible mechanism of the AR technique, i.e., loosening or breaking of the crosslinkages caused by formalin fixation (Shi et al. 1991,1992a). A recent study by Mason and O'Leary (1991) has demonstrated that the process of crosslinking does not result in discernible alteration of protein secondary stucture. Therefore, the AR technique may take advantage of the fact that the crosslinking of protein produced by formalin fixation may “protect” formalin-modified epitopes from denaturation during the heating procedure. Suurmeijer and Boon (1993a) summarized the possible mechanism of AR as follows: (a) breaking of the formalin-induced crosslinkage between epitopes and unrelated proteins; (b) extraction of diffusible blocking proteins; (c) precipitation of protein; and (d) rehydration of the tissue sections, allowing better penetration of antibody and increasing accessibility of epitopes. In contrast to the “breaking crosslinking” theory, Cattoretti et al. (1993) proposed a “protein denaturation” theory on the basis of their observation that some antigens or endogenous enzymatic activities might be lost after heating AR treatment. To understand the mechanism(s) of AR, the first step may be to study the exact alterations in the three-dimensional structure of proteins caused by formalin fixation. Because antigen-antibody recognition is often dependent on the three-dimensional structure of protein, it is likely that the mechanism of AR-IHC may be based on a re-modification of the formalin-modified protein structure.
Crosslinking is a special form of chemical modification of protein (Wong 1991), which can be defined as a process involving the joining of two molecular components by covalent bonds achieved by the use of crosslinking reagents, such as tissue fixatives. Traditionally, the process of tissue fixation has been divided into two stages: the primary reaction is an addition reaction between amine and an aldehyde (e.g., formalin) or ketone (e.g., acetone), R-NH2 + HCHO→R-NH-CH2OH, followed by a secondary condensation reaction: R-NH-CH2OH + H2N-CO-R'→R-NH-CH2-NH-CO-R' + H2O. Both aldehyde and ketone are carbonyl compounds that can react with amino groups of proteins to form Schiff bases (Wong 1991). This addition reaction of acetone may explain some of the enhancement of AR-IHC for tissues fixed in fixatives other than formalin (Oyaizu et al. 1996; Gown et al. 1993a).
The secondary, tertiary, and quaternary structures of proteins are dependent on the primary structure or the amino acid sequence. In general, if the primary structure of protein is still intact it may be possible to recover its original three-dimensional conformation under an optimal retrieval system. Because crosslinkages are complicated processes that depend on a variety of conditions (e.g., pH, temperature, conditions of tissue and fixation), they may lead to a variety of protein alterations (Fraenkel-Conrat et al. 1947,1948a,b; Olcott and Fraenkel-Conrat 1947). Therefore, the use of a test battery is recommended to solve this complicated situation.
Antibodies recognize specific epitopes localized in a particular spatial configuration within the protein molecule. This is particularly true for discontinuous antigenic determinants, which are composed of residues from different parts of the amino acid sequence (Barlow et al. 1986; Atassi 1975). A change in protein conformation caused by formalin fixation may mask the tissue antigenicity. The AR method may lead to a re-modification of the protein structure (induced by high-temperature heating or by non-heating procedures, such as strong alkaline treatment) and reestablish the three-dimensional structure of protein in its native condition, or very close to that state (Figure 2).
This “modification-re-modification” hypothesis has been supported by recent studies. Using MW heating followed by trypsin digestion, Szekeres et al. (1994a,b, 1995) found a decrease in the nuclear staining of ER and MIB1, which may correspond to a sensitivity of the nuclear Ki-67 to enzyme digestion, as the antigenic structure was modified by MW AR heating in such a way that the epitope was exposed for the subsequent alteration by enzyme treatment. Some studies have demonstrated that if a fixed tissue section shows satisfactory immunostaining without AR it may become worse after AR treatment; the converse may also be true (MacDonald et al. 1995; Tesch et al. 1995). This might mean that if an antigenic structure is formalin-resistant, i.e., without modification after fixation, it may be modified by heating. Therefore, the immunoreactivity may be worse after AR heating unless the antigenic structure is also heat-resistant.
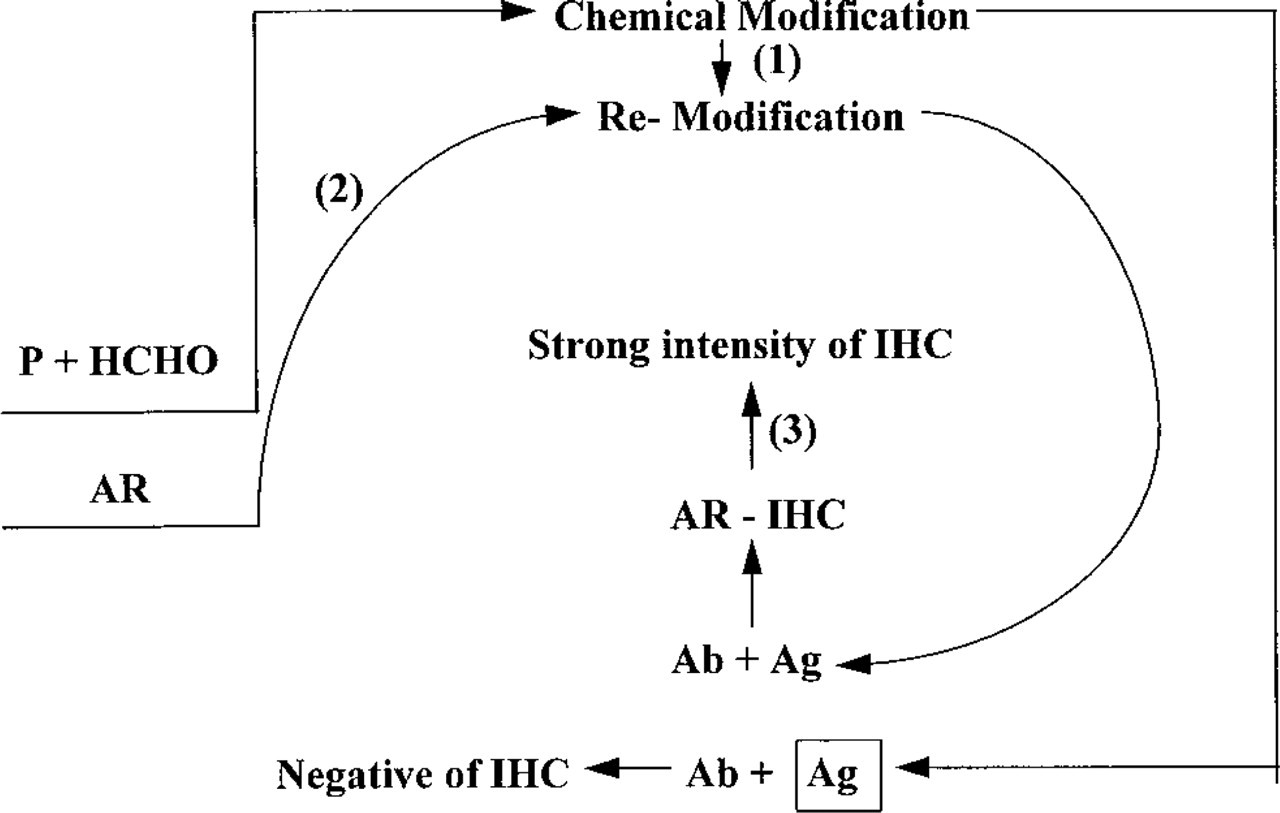
Diagram of our suggested mechanism of AR-IHC. P, protein in the tissue; HCHO, formalin; AR, antigen retrieval; IHC, immunohistochemistry; Ag, antigen; Ag in square, formalin-modified antigen. (
Recently, Morgan et al. (1994) suggested a hypothesis concerning the possible mechanism of AR, focusing on tissue-bound calcium ions. These authors suggested that the calcium complex formation with protein in their formalin-fixed state may mask the antigen, which may need a considerable amount of energy, i.e., high-temperature heating and calcium chelation (citrate), to release the calcium from this cage-like calcium complex. To test this hypothesis, they added excess calcium ions in a mixed solution of 50 mM CaCl2 with either EDTA, EGTA, or citrate buffer (calcium chelators) as the AR solution for autoclave heating treatment, and obtained a negative staining for MIB1. The same negative result could also be obtained by a sequential use of AR solution and 50 mM CaCl2 solution in heating the tissue sections. They suggest that there is inhibition of AR by either competitive (mixed CaCl2 with AR solution) or noncompetitive (sequential use of AR solution and CaCl2 solution) inhibition. They also tried to repeat the AR procedure after inhibition of CaCl2 and obtained partially recovered immunostaining intensity. When the concentration of CaCl2 was reduced to 10 mM for inhibition, the inhibition of MIB1 staining was completely reversed.
Test for demonstration of the effect of CaCl2 in AR-IHC a , b

aAvidin-biotin complex (ABC; Vector, Burlingame, CA) method was used for immunohistochemical staining for all primary antibodies listed above.
bThe inhibition of AR effect by using CaCl2 can be recovered completely by Test (5). Stronger intensity of AR-IHC can be obtained as illustrated in Figures 3A and 3B. All are archival paraffin sections for tissues tested. Citrate buffer solution is pH 6.0.
cKeratin cocktail = monoclonal antibodies AE1 + CAM 5.2.
Ca., carcinoma.
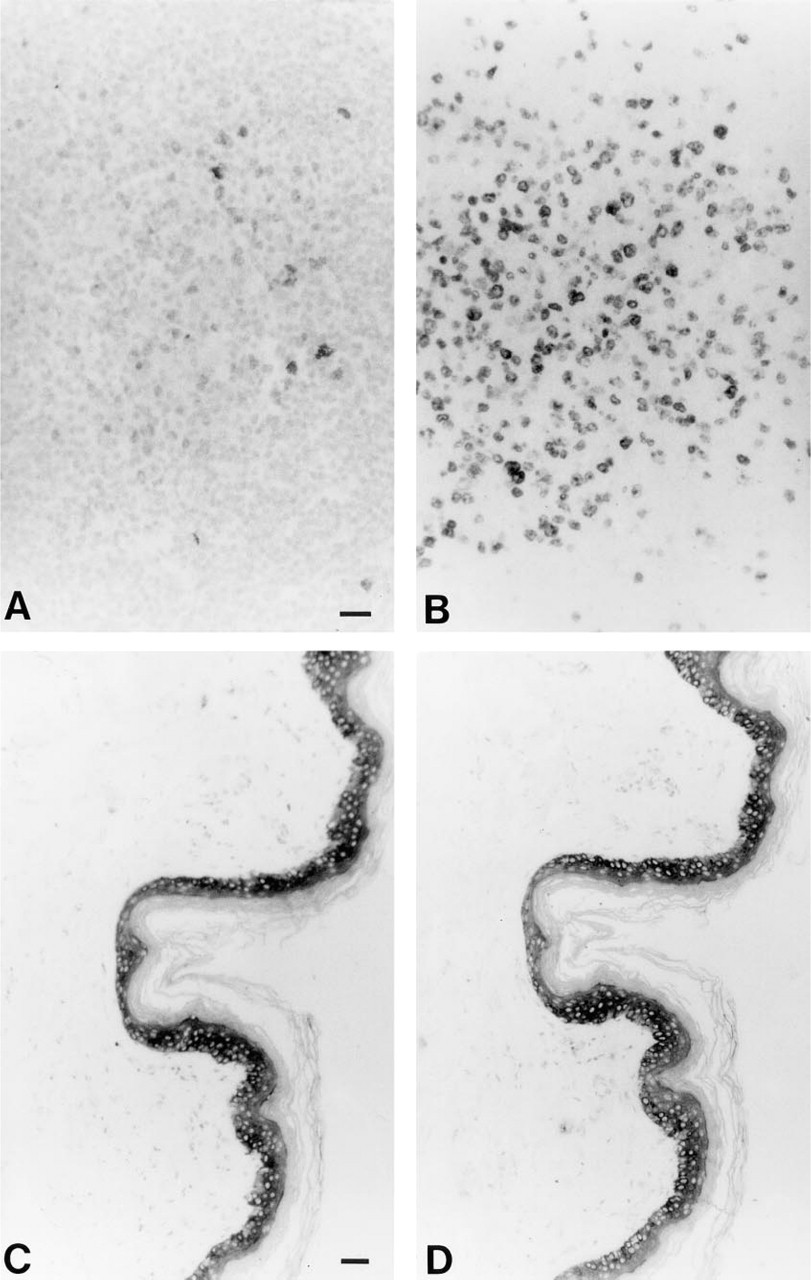
Results of AR-IHC with the effect of CaCl2, as indicated in Table 3. (
We conducted a test of AR-IHC in the presence of CaCl2, as summarized in Table 3. We found that (a) The same negative staining of MIB1 could be obtained by using CaCl2 alone as the AR solution; (b) not all antibodies showed a negative result, e.g., the intensity of AR-IHC for keratin remained the same after use of CaCl2 (Figure 3); and (c) re-heating the slides in Tris-HCl buffer, pH 1.5, restored a stronger positive staining for both MIB1 and p53. Although the fact that calcium interferes with AR (at least for some antigens) is consistent with the hypothesis that calcium-induced modification of protein may play a role in the retrieval process, it does not prove that binding of endogenous calcium is responsible for widespread loss of antigenicity in formalin fixation. Another issue that arouses suspicion of the calcium-binding hypothesis is the fact that the same intensity of AR-IHC with MIB1 can be obtained by a variety of AR solutions other than citrate buffer or with any calcium chelating agents. In particular, a stronger intensity can be produced by using a low pH or a higher pH of Tris-HCl buffer for AR-IHC of MIB1 (Shi et al. 1995a,b). It is necessary to study the superstructure of protein, particularly the antigenic structure, under various conditions, such as normal (frozen), after formalin fixation, and after AR treatment, to understand the exact mechanism of AR. On the basis of this study, more effective AR techniques may be developed.
Conclusion
Although the AR technique is still in the developmental stage, it has been widely used in immunohistochemical studies. Studies on the mechanism of AR-IHC are ongoing, as are attempts to standardize the AR procedure. Widespread application of standardized protocols for AR may lead to valuable retrospective studies in various fields of pathology. Further studies may examine the practical use of AR-IHC in both diagnostic pathology and analytical morphology. Studies on the mechanism of AR-IHC may create a new field in molecular morphology.
Footnotes
Acknowledgements
Supported by the University Pathology Associates of the University of Southern California.
We greatly appreciate Dr Denis G. Baskin's valuable comments and suggestions for this manuscript. We thank Ms Lillian Young and Elisabeth Yee and Mr Moung Win for technical assistance, and Ms Christina Yang for drawing illustrations and composition of the microphotographs.
