Abstract
Immunocytochemical (ICC) techniques are frequently used in basic and clinical research. Here, we focus on the importance of using antisera/antibodies at optimal dilutions to achieve specificity and reduce costs. Unfortunately, the basic principle, the necessity to test method specificity of the staining by a series of increasing dilutions of primary antiserum/antibodies, is only occasionally seen in papers using ICC. Many researchers rely on the company’s information or others’ published data. In this study, we show examples with monoclonal antibodies used in the peroxidase-based ICC technique in mouse and guinea pig brain sections. We show images of ICC staining of phospho-S129 alpha-synuclein in A53T mice and NeuN in guinea pig brains and demonstrate that optimal staining with them can be achieved at least at two to three orders of magnitude higher dilutions than generally used in the literature. We strongly recommend that when antisera/antibodies are used for the first time in any laboratory, independent of what the manufacturer or vendor recommends or are found in the literature, a dilution curve should be set up to identify the optimal dilution. This practice provides not only the highest specificity but is also an economic approach.
Precis
This study tested the optimal dilution of alpha-synuclein antibodies in A53T transgenic mice and found that the antibodies used at two to three orders of magnitude higher dilutions than those published earlier provide comparable results.
Introduction
Large numbers of publications serve as a testament to the popularity, simplicity, specificity, and accessibility of immunocytochemical (ICC) techniques in basic and clinical research. PubMed search lists almost 100,000 articles that use ICC for examining the central nervous system. The recent mandate to demonstrate rigor includes the authentication of biological and/or chemical resources.1–4 Inherent in validating antibody/antiserum use (we will use the terms “antibody” or “antibodies” for monoclonal antibodies and “antiserum” or “antisera” for polyclonal antiserum/antisera) is providing evidence that the technique accomplishes the stated goal. Although most studies use ICC for demonstrating where molecules are found in a tissue, some works use ICC for assessments of protein content as well. The latter is beyond the scope of this article.
In this study, we focus on the importance of using antisera/antibodies at optimal dilution (optimal concentration/titer) to achieve specificity and reduce costs associated with these ICC reagents. Unfortunately, the basic principle, the necessity to test method specificity of the staining by a series of increasing dilutions of primary antiserum/antibodies, resulting in a gradual decrease and eventual disappearance of the immunostaining, is only occasionally seen in papers using ICC. Many researchers rely on the company’s information or others’ published data. Most of the ICC studies use antibodies/antisera at the commonly suggested 1:500–1:2000 dilutions without testing higher dilutions (lower concentrations), although the use of high concentrations can provoke undesirable cross-reactivity and nonspecific staining.
Although testing method specificity is the most important step in ICC, other steps include testing the possible interference with endogenous peroxidase, if peroxidase-based techniques are used. This can be easily achieved by staining sections starting with the chromogen, most frequently diaminobenzidine (DAB) step, or treating the sections with absolute methanol containing 1% H2O2. 5 The specificity of the primary antiserum can also be tested by staining adjacent sections using antibodies that had been absorbed with increasing quantities of the antigen in question.
Testing specificity is not as critical when monoclonal antibodies are used, but particularly important when polyclonal antibodies are used. In the latter case, the goal is to dilute out nonspecific antibodies present in polyclonal antisera in lower concentrations than those raised against the epitopes in question.
In this study, we show examples with monoclonal antibodies used in peroxidase-based ICC techniques in mouse, rat, and guinea pig brain sections. First, we show images of ICC staining using rabbit monoclonal antibodies against phospho-S129 alpha-synuclein (α-Syn) (Abcam; Ab51253, lot# GR285618-6) following proteinase-K (PK) digestion of endogenous, non-toxic α-Syn and show that the antibodies can be used at least at two to three orders of magnitude higher dilutions than generally reported in the literature.
A dominantly inherited missense mutation (A53T) in SNCA, the α-Syn gene, was found to cause a familial form of Parkinson’s disease (PD).6–8 In our studies, we used the A53T mouse model that overexpresses the human SNCA gene. Moreover, α-Syn may fold into a β-sheet-rich structure and is able to polymerize into highly toxic amyloid fibrils and aggregates.6,8 The misprocessed, aggregated α-Syn is the major component of filamentous neuronal inclusions known as Lewy bodies, a characteristic feature of PD and other α-synucleinopathies.9,10 A classical Lewy body is a cytoplasmic inclusion of aggregated α-Syn in the nucleus and perikaryon, and a Lewy neurite is the inclusion of aggregated α-Syn in the nerve processes. 7
In these studies, we used PK digestion of non-toxic, endogenous α-Syn and as a result show PK-resistant, toxic α-Syn aggregates. Then, we used mouse monoclonal antibodies against NeuN, a neuronal marker, in guinea pig brain tissue. We demonstrate that optimal staining with this antiserum can be achieved when the antibodies are used at 1:500,000 dilution in contrast to 1:100–1:1000 dilutions frequently found in the literature.
Immunohistochemistry
For investigating the importance of optimal dilution of the antibodies, female A53T mice expressing the human α-Syn gene and female guinea pigs were used. Three-month-old A53T mice were treated with lipopolysaccharide (LPS; 2.0 mg/kg) intraperitoneally to induce chronic neuroinflammation. 11 The guinea pigs were intact without any treatment. The animal use was approved by the Institutional Animal Care and Use Committee (IACUC approval number: 1216001). The experimental animals were euthanized with CO2 asphyxiation and they were perfusion-fixed with 1% followed by 4% phosphate-buffered paraformaldehyde. The brains were immersion-fixed in the same fixative for 1–2 days and then washed in phosphate buffer (0.01 M) for a day and placed into 30% sucrose in phosphate buffer. Once the brains sank, they were sectioned on a cryostat and 40 µm sections were prepared. The sections were collected in 12-well plates containing phosphate-buffered saline (PBS, 0.01 M).
Brain sections from A53T were treated with PK (50 µg/ml in PK buffer for 30 min at room temperature 12 ) to abolish endogenous, PK-sensitive α-Syn in transgenic mice. Sections treated with PK from the A53T mice and untreated sections from guinea pigs underwent a mild ethyl alcohol shock (50% for 10 min, 75% for 30 min, 50% for 10 min 13 ) and then they were rinsed in PBS. Before exposing the sections to a series of dilutions of primary antibodies, they were treated with 2% normal horse serum for 10 min. The free-floating sections were incubated with α-synuclein antibodies and antibodies against NeuN, starting with 1:100 dilution, and then doubling the dilutions up to 1:4,000,000 at 4C for 24 hr. Following three washes in PBS (5 min each), the sections were treated with secondary antibodies (biotinylated donkey anti-rabbit and anti-mouse IgG; 1:2000 dilution for an hour at room temperature; Jackson Immunocytochemicals). Following a wash in PBS (3× 5 min), the sections were incubated in streptavidin-peroxidase (Jackson Immunocytochemicals) for an hour at room temperature. The sections then were washed in PBS (2 × 5 min) and Tris-HCl (0.05 M, 5 min). During these steps, DAB was dissolved in Tris buffer, filtered, and 8 µl of 30% H2O2/100 ml of Tris buffer was added. Each section was incubated in DAB solution for 10 min. Then, the sections were washed in Tris, followed by PBS for 10 min, each. The free-floating sections were mounted on Superfrost Plus microscopic slides from 5% polyvinyl alcohol. Once the sections were dry, they were dipped in 100% ethanol followed by xylene treatment for 5 min each and then cover-slipped with DePex. Images were taken with a Nikon microscope.
Although α-Syn immunoreactivity was present in many brain areas, we show only the hippocampus, the cerebral cortex, and the substantia nigra. Images of sections from the same brain area stained with rabbit monoclonal antibodies at dilutions of 1:4000 (A), 1:16,000 (B), 1:64,000 (C) are depicted in Fig. 1. Figure 2 shows images of sections from the same brain area stained with these antibodies at dilutions of 1:256,000 (A), 1:1,000,000 (B), and 1:4,000,000 (C). The immunostaining method was the standard ABC protocol without antigen retrieval or other intensification procedures. Figure 3 demonstrates that the misfolded α-Syn inclusions are present in the entire neuron producing Golgi-like images (Fig. 3A–C) in LPS-treated A53T mice.
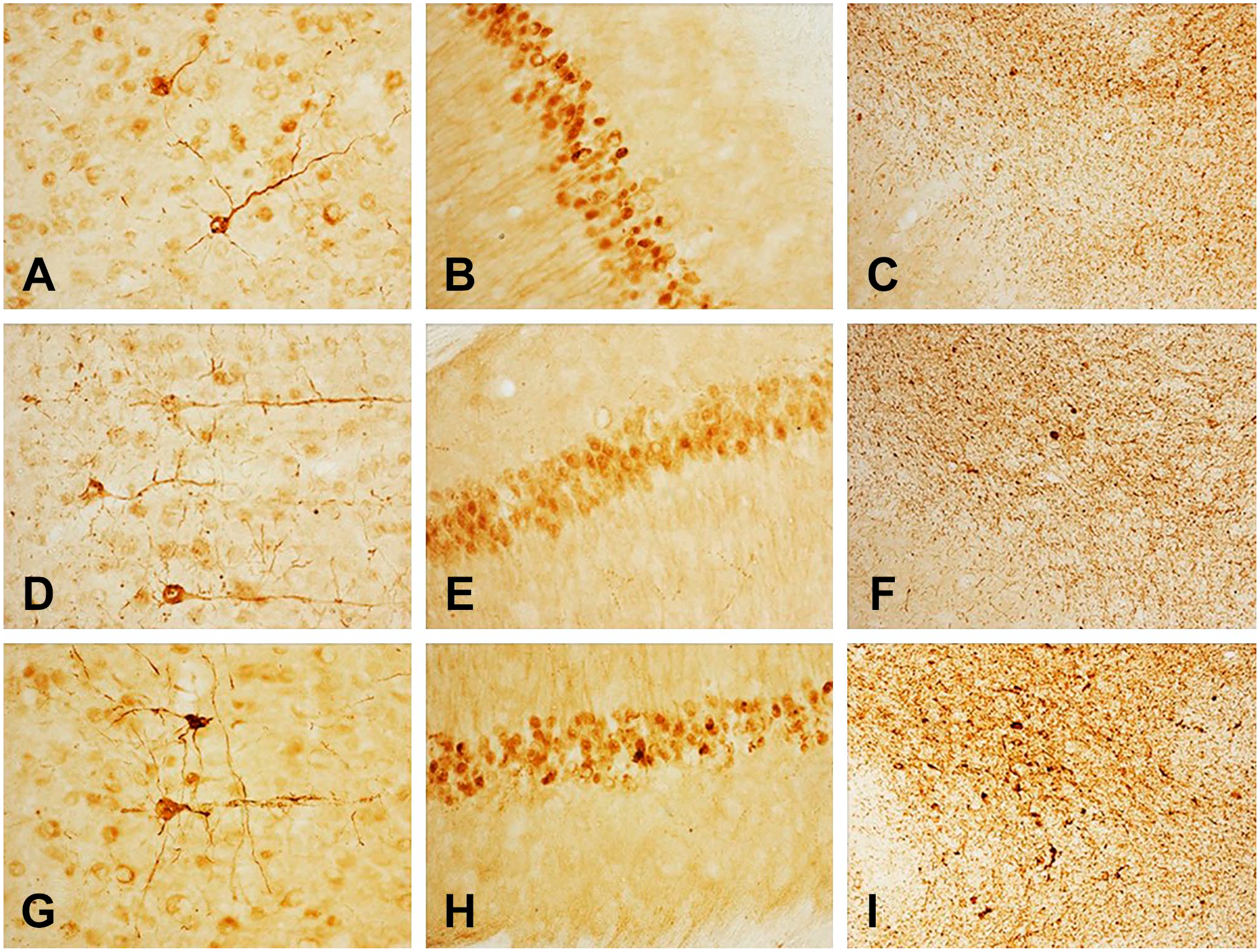
α-p129-synuclein immunoreactivity in the cerebral cortex (left column), hippocampus (middle column), and substantia nigra area of the brainstem (right column) in LPS-treated A53T mice following treatment of sections with PK to abolish endogenous, PK-sensitive α-Syn in transgenic mice. Dilutions of the monoclonal antibodies: 1:4000 (A–C), 1:16,000 (D–F), 1:64,000 (G–I). Immunostaining was achieved using the standard Avidin-Biotin Complex (ABC) protocol without antigen retrieval or other intensification procedures. Abbreviations: LPS, lipopolysaccharide; PK, proteinase-K.
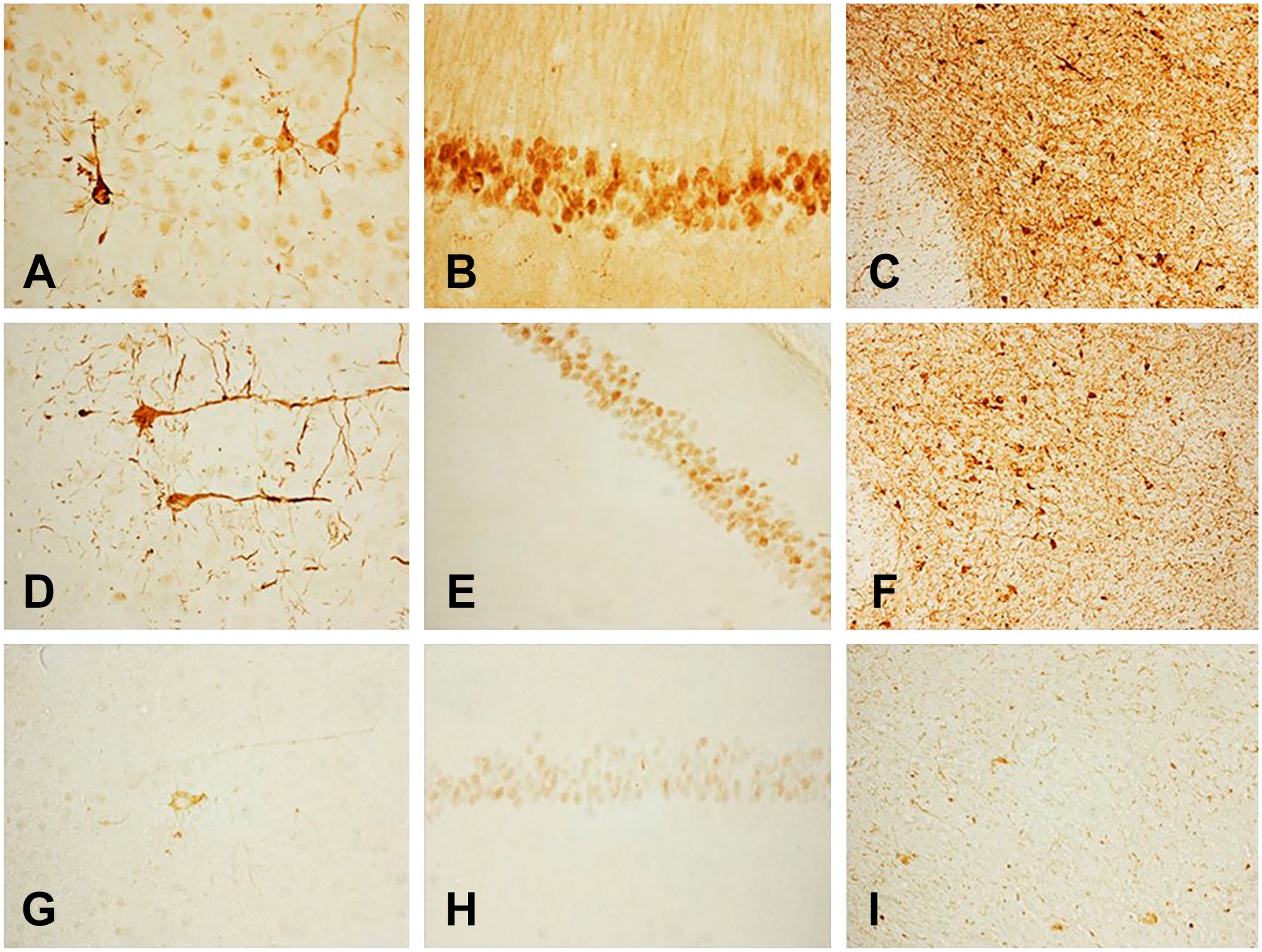
α-p129-synuclein immunoreactivity in the cerebral cortex (left column), hippocampus (middle column), and substantia nigra area of the brainstem (right column) in LPS-treated A53T mice. Dilutions of the monoclonal antibodies: 1:256,000 (A–C), 1:1,000,000 (D–F), and 1:4,000,000 (G–I). Immunostaining was achieved using the standard Avidin-Biotin Complex (ABC) protocol without antigen retrieval or other intensification procedures. Abbreviation: LPS, lipopolysaccharide.
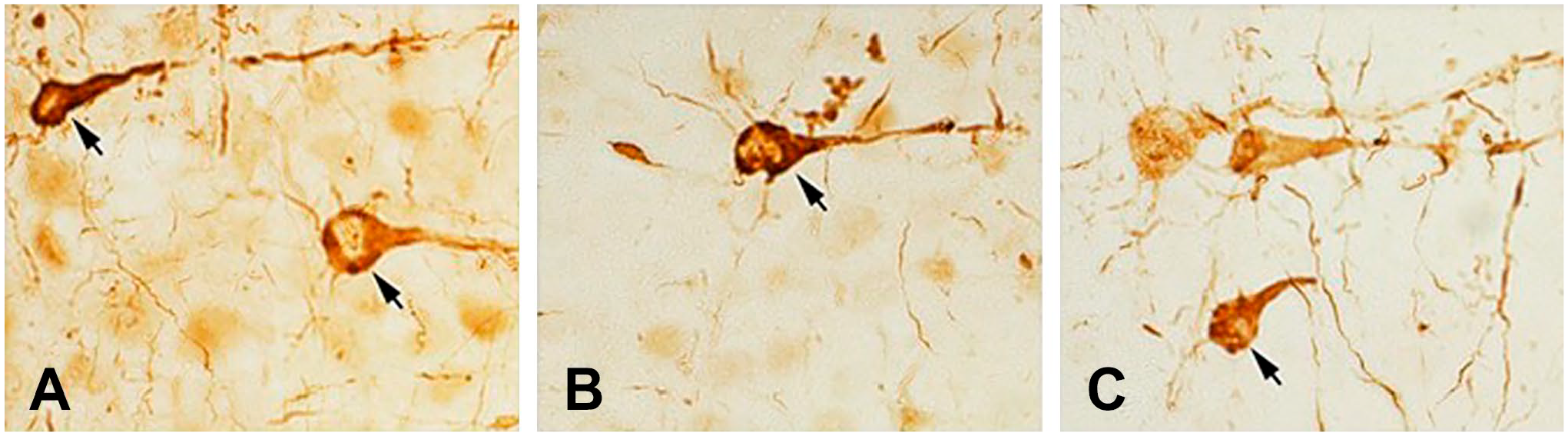
High-magnification images of α-p129-synuclein immunoreactivity staining in the cerebral cortex show that the misfolded α-Syn inclusions (arrows) are present in the entire neuron producing Golgi-like images in LPS-treated A53T mice. Dilutions: (A): 1:4000; (B): 1:32,000; (C): 128,000. Abbreviation: LPS, lipopolysaccharide.
Increasing the dilution results in reduction of the intensity of both the specific and background staining; nevertheless, the specific immunoreaction was still visible at a dilution of 1:1,000,000. These data are contradictory to those obtained by several laboratories using this antiserum at 1:2000 dilutions (one even at 1:00 dilution 14 ) with the exception of Henderson et al.15,16 who used these antibodies at a higher 1:20,000 dilutions. Unfortunately, none of the large number of papers using this or any other antibodies mentioned whether the antibodies were tested at higher dilutions and whether the optimal dilution was selected to conduct the study.
Interestingly, the morphology of α-Syn-immunoreactive neurons in the cortex was better when the antibodies were used at very high dilutions [cf. Fig. 3A (1:4000) with Fig. 3B (1: 32,000) or Fig. 3C (1:128,000)]. This phenomenon might be explained by the better contrast between the lower background staining and a still definite signal at higher antibody dilutions, or with the hindrance among antibodies used at high concentrations.
Images of sections from the same brain area of female guinea pigs stained with NeuN antibodies at dilutions of 1:64,000 (A), 1:128,000 (B), 1:256,000 (C), and 1:512,000 (D) are shown in Fig. 4. Each section has NeuN immunostaining in the cerebral cortex and the hippocampus, including the CA1 area and the dentate gyrus.
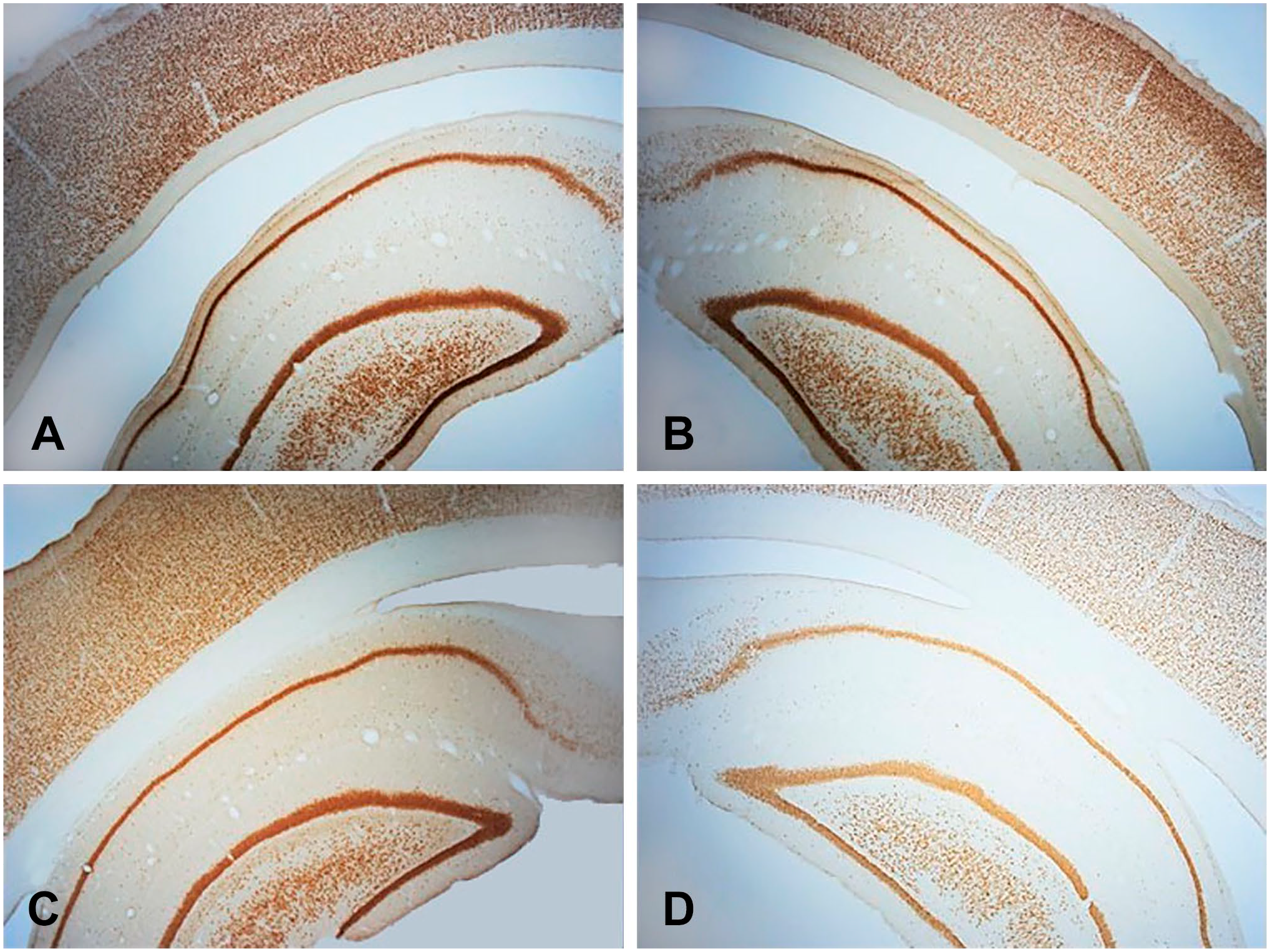
NeuN immunoreactivity in the cerebral cortex and hippocampus of an intact, female guinea pig. Dilutions: (A) 1:64,000, (B) 1:128,000, (C) 1:256,000, and (D) 1:532,000.
The Optimal Dilution: Significance
The immunostaining of any antigen with optimal dilution is of paramount importance for specificity, particularly when polyclonal antisera are used.17,18 These antisera contain, in addition to the specific antibodies with high titer (high concentration), antibodies against the same epitopes present in many antigens with lower titers (lower concentrations). Diluting the primary antiserum reduces nonspecific binding resulting from low-affinity interactions such as binding to Fc receptors. Although this is not a critical issue when monoclonal antibodies are used, the typically short sequences (epitopes) recognized by monoclonal immunoglobulins (antibodies) may also be present in other related or unrelated molecules. If so, these antibodies will recognize these sequences and bind to them, resulting in nonspecific staining. As these nonspecific sites, including alternative epitopes and Fc receptors, as well as sites with ionic interactions have generally lower affinity to specific antibodies, diluting the primary antibodies retains binding primarily to high-affinity binding sites, which are more likely the target of specific, monoclonal, or polyclonal antibodies. Nonspecific staining cannot be solved easily with the traditional specificity tests (preabsorption of the antisera with the antigen used for immunization), but preabsorption of the antisera with known, structurally related antigens may be beneficial. However, it is still possible that the antisera recognize sequences in unknown molecules. This is the situation when other approaches, such as in situ hybridization histochemistry, can be used to show the presence of the specific RNA in the cells. Although various parameters, including using frozen vs fixed tissues, optimal fixation and antigen retrieval methods, and different postmortem and post-fixation times, often shift the best possible dilution of the antibodies,2,3 in principle, these variables do not affect the importance of setting up the dilution curve and pinpointing the optimal dilution that remains a central focus with the evolution of multiplex staining protocols and complex assay validation techniques. 19 Legitimate examples in which diverse optimal antibody concentrations/dilutions are provided by various laboratories include differences in the application of the antibody (IHC, Western Blot), the tissue used, the chosen antigen retrieval protocol (variables include buffer, temperature, and pH), 1 and the applied detection system. 2
The use of antibodies at the optimal dilution is not only a specificity issue but also a pivotal economic concern. In our studies, we have used the α-Syn antibodies and NeuN antibodies at two to three orders of magnitude higher dilution than most of the published studies, and doing so we have significantly reduced the cost of the study.
In conclusion, we strongly recommend that when antisera/antibodies are used for the first time in any laboratory, independent of what the manufacturer or vendor recommends or are found in the publications by others, a dilution curve should be achieved to identify the highest dilution which gives acceptable signal intensity with the lowest possible background staining. For practical use, when doubling the dilutions, we recommend using a dilution one step below the one which shows a small reduction in staining intensity. This practice must also be appreciated by the reviewers, and only ICC data based on these principles should be recommended for publication unless specificity is confirmed by other means.
Footnotes
Competing Interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
Dr. Dudás planned and executed some of the immunocytochemical experiments. He has also prepared the final figures and participated in writing the manuscript. Malcolm Lane prepared the tissue for immunocytochemical analysis and supervised the activity of the students. The students, Nikitha Mupparaju and Hye Min Kim performed the immunostainings. Dr. Merchenthaler planned the experiments, prepared the initial figures and together with Dr. Dudás wrote the manuscript.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
