Abstract
Nerves in the renal parenchyma comprise sympathetic nerves that act on renal arteries and tubules to decrease blood flow and increase primary urine reabsorption, respectively. Synaptic vesicles release neurotransmitters that activate their effector tissues. However, the mechanisms by which neurotransmitters exert individual responses to renal effector cells remain unknown. Here, we investigated the spatial and molecular compositional associations of renal Schwann cells (SC) supporting the nerve terminals in male rats. The nerve terminals of vascular smooth muscle cells (SMCs) enclosed by renal SC processes were exposed through windows facing the effectors with presynaptic specializations. We found that the adrenergic receptors (ARs) α2A, α2C, and β2 were localized in the SMC and the basal side of the tubules, where the nerve terminals were attached, whereas the other subtypes of ARs were distributed in the glomerular and luminal side, where the norepinephrine released from nerve endings may have indirect access to ARs. In addition, integrins α4 and β1 were coexpressed in the nerve terminals. Thus, renal nerve terminals could contact their effectors via integrins and may have a structure, covered by SC processes, suitable for intensive and directional release of neurotransmitters into the blood, rather than specialized structures in the postsynaptic region.
Keywords
Introduction
Renal sympathetic nerves innervate the renal vasculature and nephrons,1–4 and they can increase blood pressure by modulating vascular contractility and tubular reabsorption.5,6 Norepinephrine (NE), a sympathetic neurotransmitter, stimulates adrenergic receptors (ARs) in numerous organs. Many AR subtypes exhibit their pharmacological activity in the kidneys.5,6 The ARs in the kidney are expressed in arterial smooth muscles, tubule epithelial cells, and juxtaglomerular renin cells, where they reduce renal blood flow, increase reabsorption, and stimulate renin secretion, respectively. Although in vivo and in vitro pharmacological studies have revealed diverse effects of ARs in the kidney5,7–12 and the vasculature and nephron segments have different types of ARs,13–16 the positional relationship of ARs to sympathetic nerve terminals and the pathways through which NE stimulates them remains unknown.
Postganglionic neurons of sympathetic nerves, which pass along the arteries to reach targets, appear non-synaptic and innervate various autonomic target cells. Detailed morphological studies have revealed neuroeffector junctions made using various sympathetic nerve terminals in the kidney, iris, mesenteric veins, and vas deferens.17–20 They do not contain highly constructed synaptic structures such as endplates of somatic motor nerves but only swollen varicosities containing synaptic vesicles with prejunctional membrane specializations and a single basal lamina layer.21–23 In the kidney, renal nerve axons are divided into two distinct types of ultrastructures based on their structural features as well as the presence or absence of a bundle of microtubules running through the varicosities. Moreover, specialized neuroeffector junctions are present on juxtaglomerular arterioles in rabbits and rats.24,25 However, little is known about the molecular compositions that effectively stabilize nerve terminals of renal sympathetic nerves and the relationship among nerve terminals, supporting cells, and ARs.
There are many forms of Schwann cells, and they not only insulate axons but also support synaptic transmission at nerve endings, known as the tripartite synapse.26–28 Non-myelinating Schwann cell sheaths envelop the peripheral nerves, including afferent C-nerve fibers and postganglionic sympathetic nerve fibers (known as Remak bundles), and motor nerve terminals of neuromuscular junctions (NMJs).29–32 Moreover, they act as insulators for nerve conduction, physically support axons, and could be involved in axonal outgrowth and maintenance with respect to development and reinnervation. However, the role of Schwann cells in sensory and autonomic efferent bundles remains unknown.30,31 Darlot et al. 33 examined the topology of Schwann cells and sympathetic nerves in mice through confocal microscopy and found that Schwann cells are distributed in apposition to adrenergic nerves. However, the relationship between renal varices and Schwann cell sheath is unclear. On the contrary, close associations between sympathetic nerve terminals and P2X purinergic receptors have been reported in the renal arteries 34 and urinary bladder. 35 Although these structures indicate the multifunctional properties of Schwann cells, the functional relationships among nerve terminals, effectors, and Schwann cells in the renal sympathetic nervous system remain unclear. Thus, this study aimed to describe the spatial and molecular compositional associations of renal Schwann cells supporting nerve terminal varicosities that release neurotransmitters and effectors.
Materials and Methods
Animals
Male Sprague-Dawley rats (age, 7–9 weeks; weight, 250–350 g) were purchased from CLEA Japan Inc. (Tokyo, Japan). The animals were housed in isolated cages in a light-controlled room with a 12-hr light–dark cycle. Food and water were provided ad libitum. Twenty rats were included in the experiments, and they were examined under deep anesthesia using 2% isoflurane (Intervet K. K.; Osaka, Japan) followed by pentobarbital sodium (100 mg/kg) (Kyoritsu Seiyaku; Tokyo, Japan).
Immunofluorescent Histochemistry
Deeply anesthetized rats underwent intracardiac perfusion with physiological saline followed by 4% paraformaldehyde (Merck KGaA; Darmstadt, Germany) in 0.1 M phosphate buffer (PB, pH 7.4). The kidneys were removed and sectioned into 2- to 3-mm-thick pieces, and the sections were fixed using 4% paraformaldehyde in PB fixative at 4C overnight. Next, the specimens were immersed in 20% sucrose (Fujifilm Wako Pure Chemicals; Osaka, Japan) in 0.1 M PB for 5 hr, and they were quickly frozen using an optimal cutting temperature compound (OCT compound; Sakura Finetek, Tokyo, Japan) under liquid nitrogen. Finally, the specimens were sliced into 10-μm-thick sections on glass slides or 40-μm-thick sections for free-floating staining using a cryostat (CM3050S III; Leica, Wetzlar, Germany), and they were subjected to immunohistochemistry (IHC). Twelve rats were used in a series of IHC experiments with primary antibodies and their respective controls.
All immunoreactive experiments, except for the cooling reaction, were performed at room temperature (approximate range, 23C–25C). The obtained sections were immersed in 5% normal donkey serum in PB saline containing 0.025% Tween 20 (PBST), and they were incubated with diluted specific antibodies (Table 1) at 4C overnight. Multifluorescent IHC was performed using primary antibodies obtained from different host animals, including rabbits, mice, and guinea pigs (Table 1). The sections were washed with PBST, incubated with fluorescent-conjugated secondary antibodies (Table 2), mounted onto glass slides, and examined under a confocal laser microscope LSM-780 (Carl Zeiss; Dresden, Germany) equipped with argon (488 nm), HeNe (633 nm), and DPSS (561 nm). The captured images were processed using software Imaris (Carl Zeiss) for surface deconvolution.
Details of the Primary Antibodies Used in This Study.
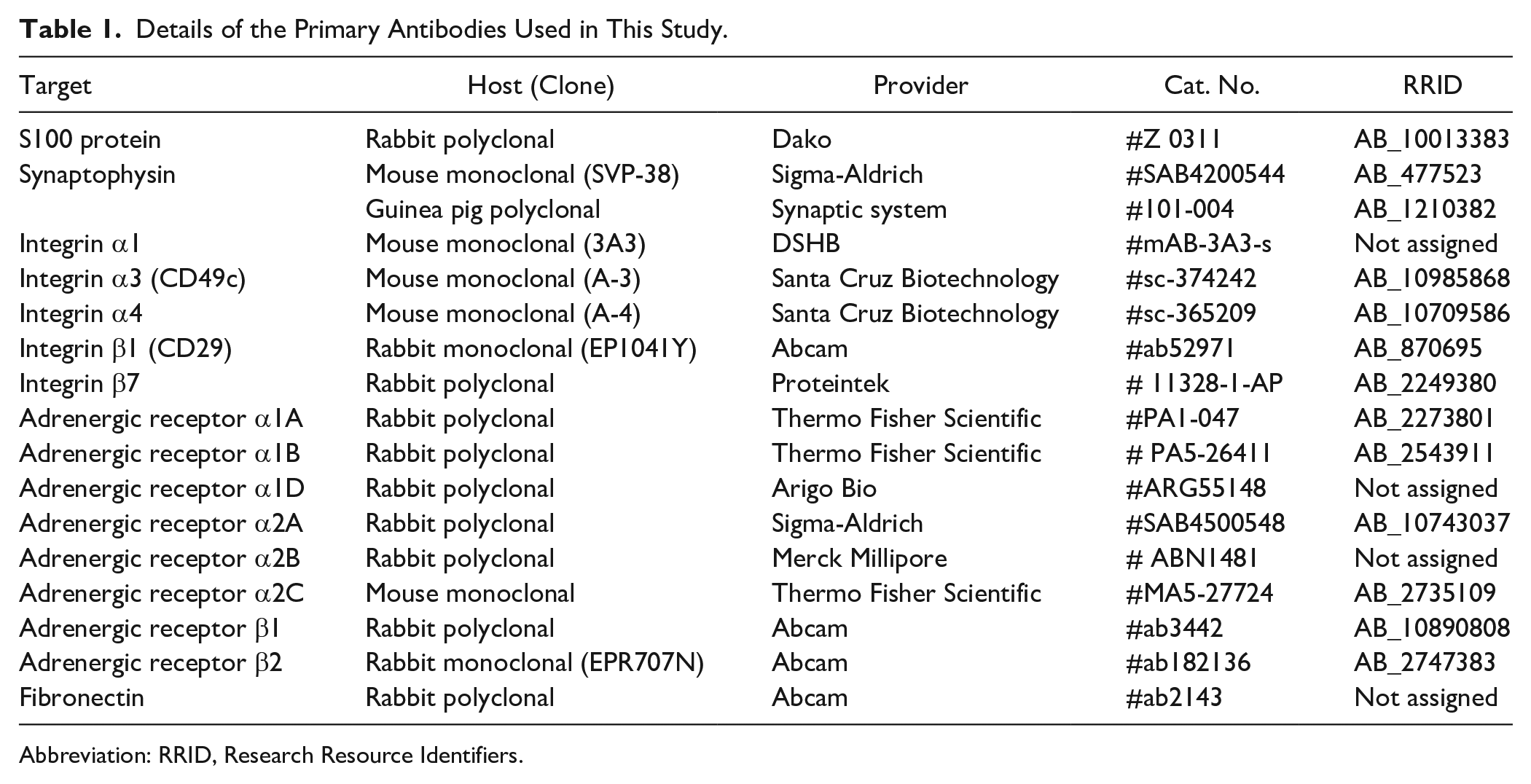
Abbreviation: RRID, Research Resource Identifiers.
Details of the Secondary Antibodies Used in This Study.

Abbreviation: RRID, Research Resource Identifiers.
Regarding multiple fluorescent labeling and IHC of ARs using S100 and synaptophysin antibodies, anti-rabbit antibody IHC was conducted using the Fab fragment of secondary antibody (Table 2). Briefly, rabbit anti-AR (Table 1) and mouse anti-synaptophysin antibodies were diluted and mixed as a primary antibody solution. Following incubation with primary antibody solution, renal tissues were incubated with Alexa Fluor 488–conjugated donkey anti-rabbit IgG Fab fragment and Alexa Fluor 647–conjugated donkey anti-mouse IgG. After washing with PBST, the tissues were incubated in a blocking solution containing diluted donkey anti-rabbit IgG unconjugated Fab fragments. The sections were incubated with diluted S100 antibody and donkey anti-rabbit IgG biotinylated Fab fragment, and subsequently, they were washed and mixed with Alexa Fluor 555–conjugated streptavidin (#S21381; Thermo Fisher, Waltham, MA). After confirming the absence of overlapping contaminations, the tissues were examined under a confocal microscope (LSM780, with objective Plan-Apochromat 20×/0.8 M27 or alpha Plan-Apochromat 100×/1.46 Oil DIC M27). Alexa Fluor 488 was detected using a filter set with 488 nm excitation and 490–552 nm emission, Cy3 and Alexa Fluor 555 were detected using 561 nm excitation and 570–632 nm emission, and Cy5 and Alexa Fluor 647 were detected using 633 nm excitation and 640–759 nm emission. Images (1024 × 1024 pixels) were first acquired using the ZEN 2012 SP2 (black) software version 11.0.0.0 (Carl Zeiss), and multi-color images were examined using ImageJ software Fiji 36 (NIH; Bethesda, MD).
Electron Microscopy
Three deeply anesthetized rats underwent intracardiac perfusion with physiological saline, followed by a fixative containing 2.5% glutaraldehyde (Fujifilm Wako) and 4% paraformaldehyde in 0.1 M PB (pH 7.4). The kidneys were removed and sectioned into 2- to 3-mm-thick pieces, and they were fixed in the same fixative at 4C overnight. The specimens were slightly washed with PB, and they were then re-fixed using 1% osmium tetroxide (TAAB; Berkshire, UK) in PB at 4C for 3 hr. Furthermore, the specimens were dehydrated with graded ethanol and embedded in Spurr resin (Polysciences; Warrington, PA). Ultrathin sections (70 nm) were cut using an ultramicrotome (Ultracut UC6rt; Leica) for observation under a transmission electron microscope (TEM) (JEM-1400 plus; Jeol Ltd., Tokyo, Japan).
Immunoelectron Microscopy
Specimens for immunoelectron microscopy were prepared using the aforementioned immunohistochemical procedure, with the exception of detergent-free PBS. Tissue sections were blocked using 5% normal donkey serum in PBS, and this was followed by incubation with diluted rabbit anti-S100 or mouse anti-synaptophysin antibody at 4C overnight (Table 1). After washing with PBS, the sections were incubated with horseradish peroxidase–conjugated secondary antibody, and subsequently, diaminobenzidine colorization was performed. The colorized specimens were re-fixed using 5% glutaraldehyde, followed by 1% osmium tetroxide at 4C. They were dehydrated with graded ethanol and embedded in Spurr resin (PolySciences) by sandwiching them with an Aclar film (Nissin EM; Tokyo, Japan). Next, the sections were observed using light microscope BX51 (Olympus; Tokyo, Japan) with regions of interest being cut and mounted on epoxy columns. Ultrathin sections (70 nm) were cut for observation under a TEM. Electron microscopy images were obtained at an accelerating voltage of 80 kV. TEM images were an 8-bit grayscale bitmap with dimensions of 3296 × 2472 pixels. Five rats were used to collect the renal tissues.
Specimens for scanning electron microscopy (SEM) were prepared using the same procedure used for TEM. A total of 337 serial ultrathin sections, aligned in sequence, were broadly scanned (9 × 9 tiling images per section) through field emission SEM using a JSM-7800F (Jeol Ltd.). The SEM images were an 8-bit grayscale TIFF with dimensions of 20,600 × 16,000 pixels. To obtain Z-stack immunoreactivity images, a region of interest was cropped, and arrayed sections were captured with the axis of each image adjusted using the Image J software Fiji.
Results
IHC revealed predominant synaptophysin immunoreactivity in perivascular regions of the kidney, which were distributed along the arterioles (Fig. 1A, left panel), followed by branching. The vascular poles of renal corpuscles or those next to renal tubules appeared to be synaptophysin-negative. Furthermore, renal nerve fibers along the renal arteries were positive for S100 protein, which was indicative of Schwann cells (Fig. 1A, middle panel). Moreover, distal tubules were slightly reactive to S100 antibody. 37 Synaptophysin-positive nerve terminals forming pearl necklace–like varicosities were covered with S100-positive Schwann cell sheaths following the neural trajectory (Fig. 1A, right panel). Schwann cell sheaths revealed window-like openings facing synaptophysin-positive terminal varicosities. The varicosities of the renal cortex were sheathed with Schwann cell processes, and they exposed their surfaces at the windows (Fig. 1B and C). In the negative control experiment, in which the primary antibody was omitted, no crossreactivity with secondary antibodies or other types of antibodies was observed (Supplemental Figs. 1–3).
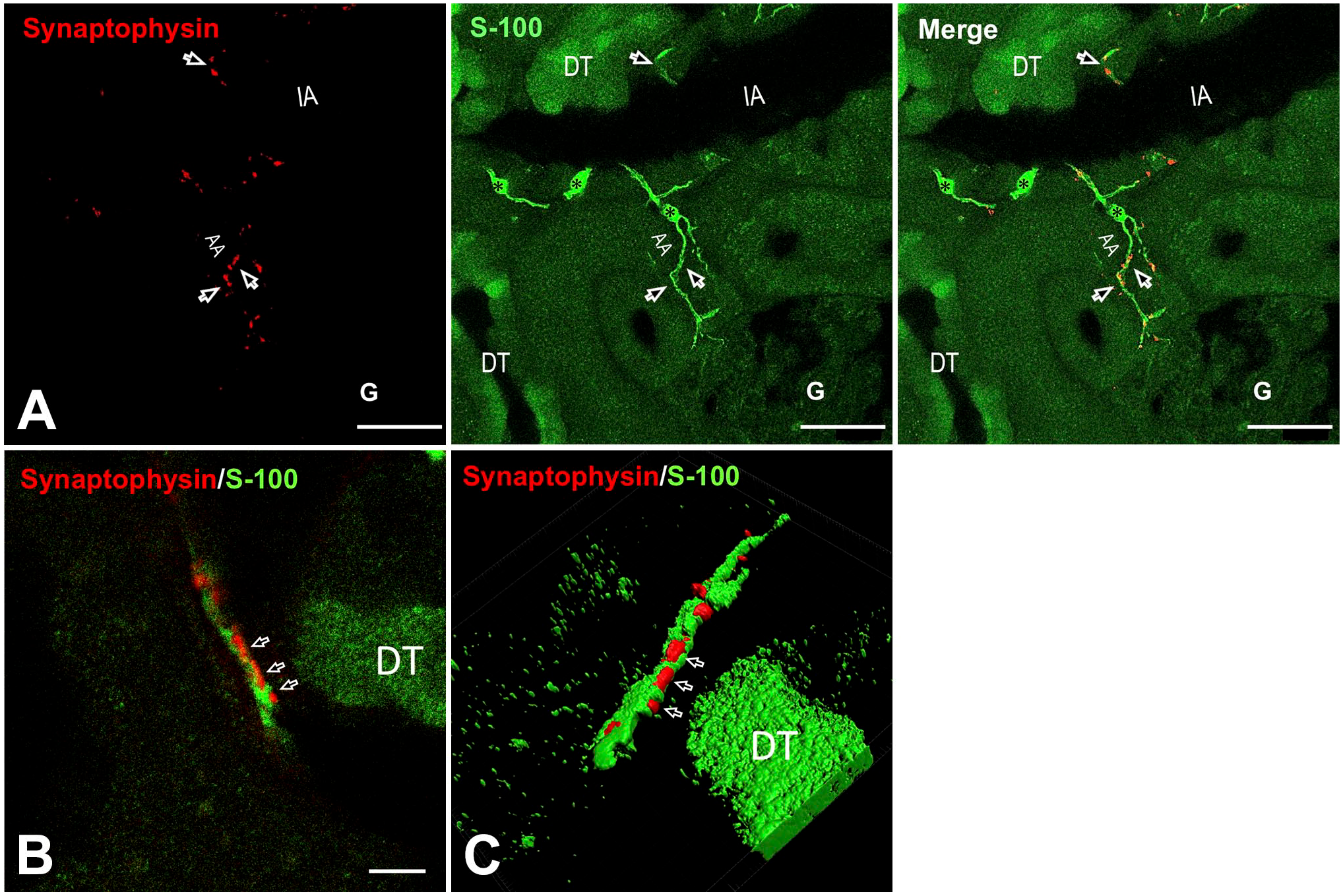
Immunohistochemical analysis of synaptophysin and S100 protein in rat kidneys. (A) Left panel: Synaptophysin is mostly distributed in the dots around renal cortex arteries (arrows), which pass along the interlobar artery to AA. Middle panel: S100-positive Schwann cells (asterisks) were found fibrously in the peri-arterioles surrounding synaptophysin-positive neurons (merged in the right panel). The DT was also positive for anti-S100 protein antibodies. Bar = 20 μm. (B) A synaptophysin-positive nerve (red) enveloped by Schwann cell sheath (green) passing along the interstitial space. Synaptophysin-positive varicosities were found in Schwann cell windows (arrows). (C) Three-dimensional reconstruction of nerve fibers in (B) varicosities that were exposed through the windows of Schwann cell sheaths (arrows) was observed from the front of the windows at different angles. Bar = 5 μm. Abbreviations: IA, interlobular artery; AA, afferent arterioles; DT, distal tubule.
Electron microscopy revealed that most varicosities tightly adhered to the basal lamina on the vascular side, and Schwann cells elongated their processes, wrapping the axons to the adventitia of arteries (Fig. 2A). In addition to Schwann cells, many fibroblast processes, which were more abundant than Schwann cell processes, were distributed in the adventitia (Fig. 2A). The Schwann cell–covered varicosities were in close contact with the basal lamina of arterial smooth muscle cells, with surfaces exposed through the windows of Schwann cell process gaps (Fig. 2B). Several nerve terminals were covered with a single Schwann cell process, and they contained a 70- to 100-nm synaptic gap at the interface with smooth muscle cells across a single layer of basal lamina. Synaptic vesicles accumulated in the varicosities and showed an asymmetric distribution toward open windows and prejunctional membrane specializations, indicating synaptic terminals of sympathetic nerves where the transmitter is released (Fig. 2C).
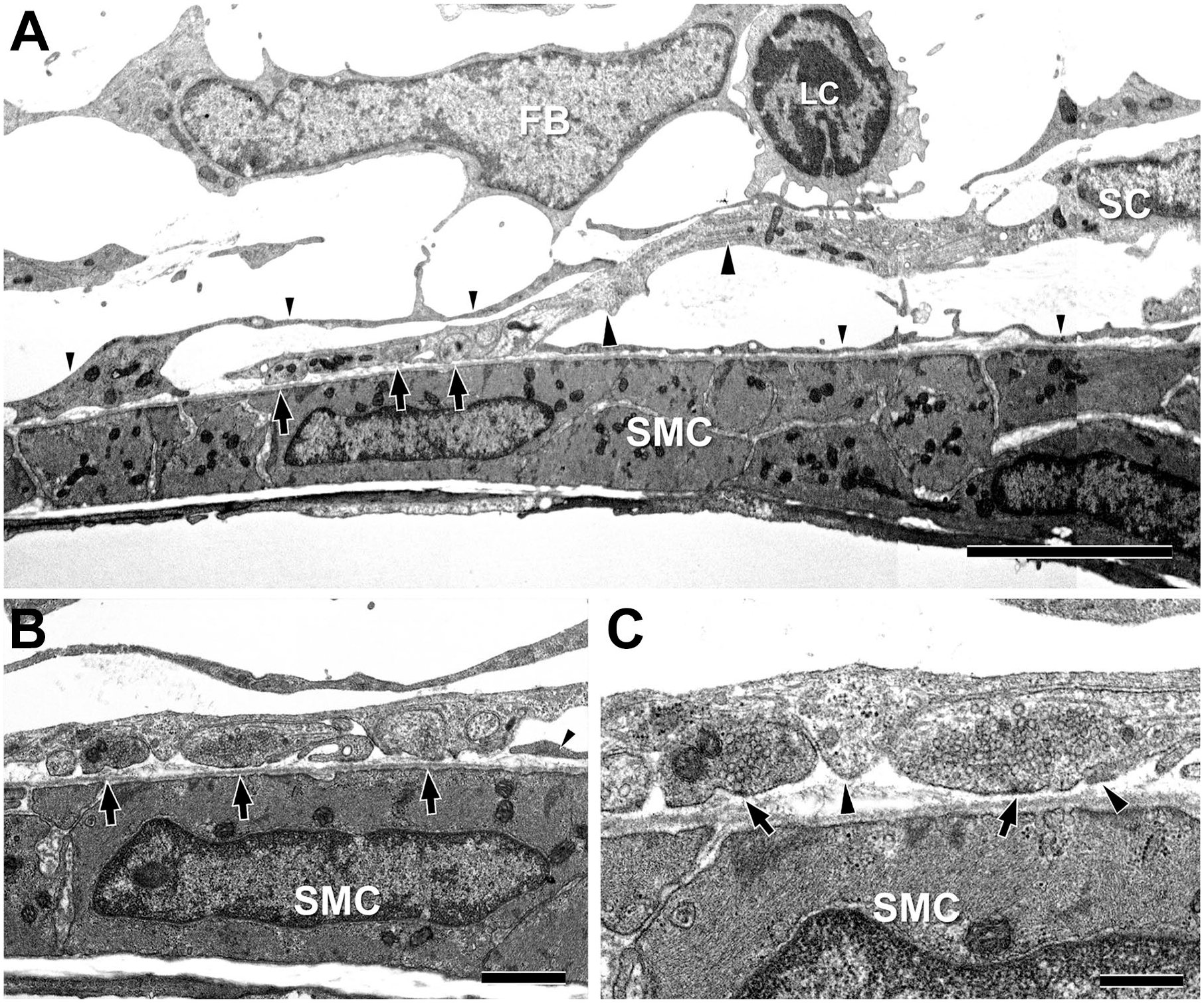
Electron microscopy of multiple nerve terminals covered with a Schwann cell process in the renal arteriole. (A) A Schwann cell elongating its process (large arrowheads) to the nerve terminal varicosities (large arrows) adhering to arterial SMC observed under lower magnification. Arteriole surface areas not covered by Schwann cell processes are covered by processes presumably derived from fibroblasts (small arrowheads). Bar = 5 μm. (B) High magnification of the terminal varicosities (arrows) to SMC and multiple open Schwann cell windows, exposing a concentration of varicosities to SMCs. Bar = 1 μm. (C) Further magnification shows that the terminal vesicles appear to assemble toward the window of the gap in the Schwann cell process (large arrowheads). Arrows indicate the presynaptic membrane specializations. Bar = 0.5 μm. Abbreviations: SMC, smooth muscle cell; FB, fibroblast; LC, leukocyte.
In the adventitia, most of the outer surface of arteries was covered with fibroblast processes, which contribute to the scaffold of perivascular constructions, whereas the axon terminals were enclosed directly by Schwann cell processes (Fig. 2). To confirm whether cell processes around the axons were Schwann cell–derived, immunoelectron microscopy was used. In most nerve terminal regions, Schwann cell–derived S100 antibody–positive processes covered the axons, and there was no contribution from S100-negative fibroblast processes (Fig. 3A and B). S100-positive Schwann cell sheaths in the arteries revealed that the nerve endings, exposed through the window of Schwann cell sheath, contained one or more varicosities (Fig. 3A). In contrast, nerve terminals distributed between the renal tubules showed no Schwann cell sheaths and were almost bare, clinging to a single layer of basal lamina (Fig. 3C). Sequential images of immunoelectron microscopy showed that fibroblasts occupied a large area around the adventitia, whereas Schwann cell processes were attached to the adventitia crowding out the fibroblast processes (Fig. 3D and E). We found that the processes supporting the renal nerve terminals to effector cells were Schwann cell–derived but not fibroblast-derived. Furthermore, using gaps in the adventitia supported by fibroblast-derived processes, Schwann cell–derived processes mediated contact of the nerve terminals to effector cells (Fig. 2A).
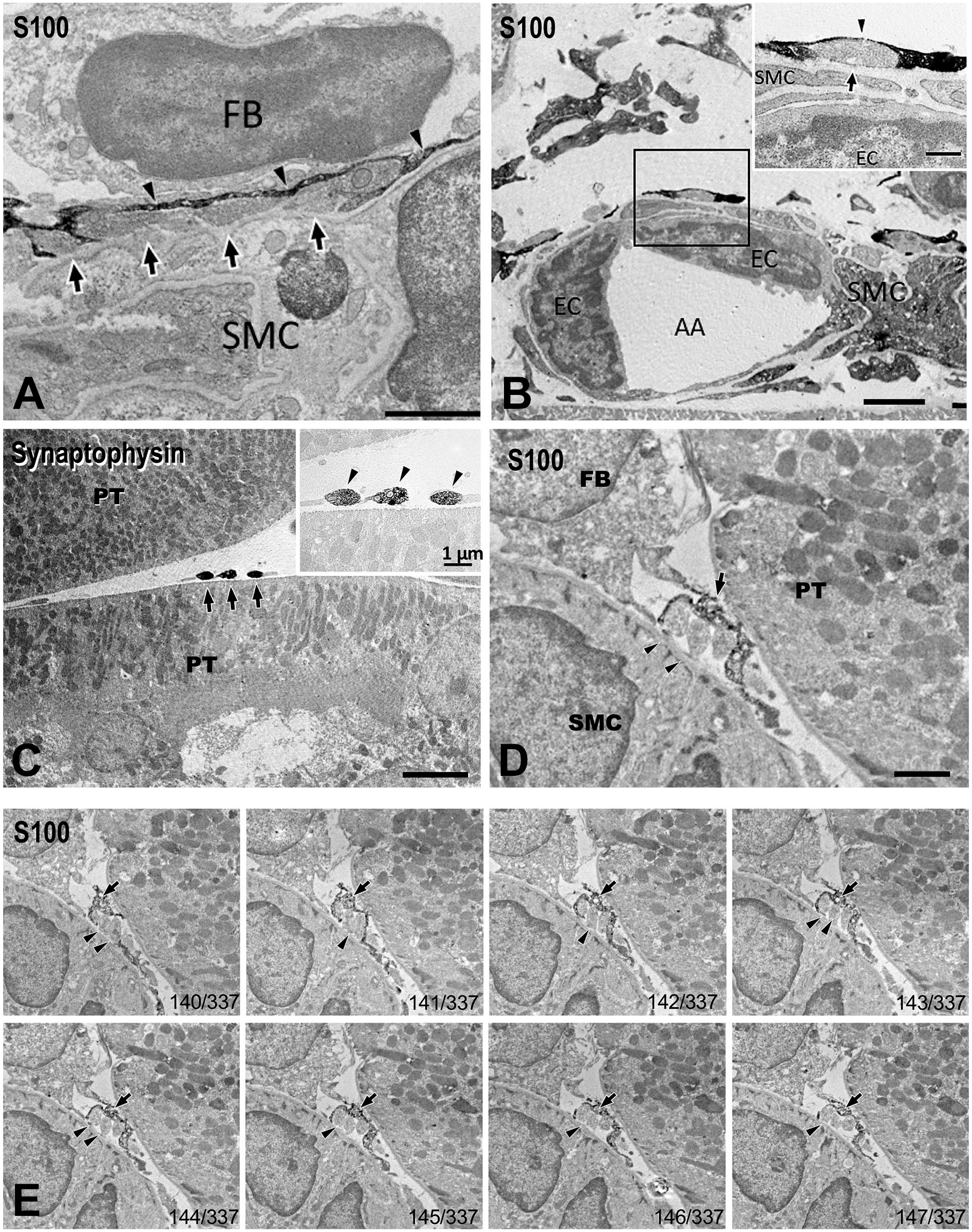
Immunoelectron microscopic analysis of synaptophysin and S100 protein in rat renal nerves. (A) S100-positive Schwann cell process (arrowheads) covering the nerve varicosities (arrows) against a fibroblast in the interstitial space. Bar = 2 μm. (B) A terminal varicosity on the small arteriole (AA) covered by S100-positive Schwann cell processes (arrowhead). A nerve terminal oriented toward the thin SMC surface (arrow). Bar = 2 μm (inset bar = 0.5 μm). (C) Axon endings attached to PT (arrows). Nerve terminals show no supporting cell process, and the terminals are bare (insert, arrowheads). Bar = 2 μm (inset bar = 1 μm). (D) In the narrow interstitial space between SMC and PT, nerve fibers in the adventitia and interstitium contain multiple varicosities (arrowheads) surrounded by Schwann cell processes (arrow). Bar = 2 μm. Sequential images of Z-stack are shown in (E). The numbers at the bottom right of each figure are in the order of sequential tissue sections. Abbreviations: FB, fibroblast; SMC, smooth muscle cell; EC, endothelial cell; AA, afferent arterioles; PT, proximal tubules.
Although the physiological effects of many AR subtypes in the kidney, such as the glomerular filtration rate and reabsorption by tubular epithelial cells, are known, there is limited information on the positional relationship between sympathetic nerve terminals distributed in the vasculatures and nephrons and specific localization of ARs in effector cells. In this study, we examined the immunohistochemical localization of eight AR subtypes, namely, α1A, α1B, α1D, α2A, α2B, α2C, β1, and β2-AR, in rat renal tissues. α1A-AR immunoreactivity was observed in the basal side of distal tubules; however, it was not closely localized with the synaptophysin-positive nerve terminals (Fig. 4A). Moreover, α1A-AR immunoreactions were strongly expressed in the collecting ducts of the medulla where no nerve terminals were seen (Fig. 4B). α1B-AR was localized to the luminal membrane of proximal tubules, where the nerve terminals were not directly distributed (Fig. 4C). Among the group of α1-ARs, α1D-AR immunoreactivity was the most widespread, with predominant reactions occurring within the glomeruli and luminal side of distal tubular epithelial cells (Fig. 4D). A barely discernible degree of immunoreactivity was observed in the arterioles. Each subtype of α2-AR showed a diverse distribution pattern. α2A-AR immunoreactions were observed in arterial smooth muscles where the nerve terminals were densely distributed (Fig. 4E), and their reactivities were closely localized to the synaptophysin-positive terminals, which were covered with S100-positive Schwann cell processes (Fig. 5A). Immunoreaction of α2B-AR was observed in the basal side of distal tubules; however, there might not be a correlation between the localization of nerve terminals and AR immunoreactivity (Fig. 4F). α2C-AR was exclusively observed at the nerve terminals colocalized with synaptophysin (Fig. 4G), where it was surrounded by Schwann cell processes (Fig. 5B). β1-AR immunoreactions were observed toward the luminal side of proximal tubules and slightly toward the side of glomeruli (Fig. 4H). β2-AR was found to have the most widespread distribution among the examined AR subtypes; its expression was strong in the arteries, glomeruli, and luminal side of proximal and distal tubules, whereas it was weak in the basal side of tubules (Fig. 4I). Although the arterial distribution of β2-AR was closely localized with Schwann cell–covered nerve terminals, β2-AR facing the nerve terminals did not have a specific clustering localization compared with the receptors on postsynaptic membranes (Fig. 5C). The comparison of vascular localization with α2B-AR showed that α2B-AR was localized throughout the SMC layer of arteries, whereas β2-AR was localized more in the inner layer or endothelia (Figs. 4I and 5C). In the negative control experiment, where the primary antibodies were omitted, no crossreactivity was observed (Supplemental Figs. 4 and 5). As mentioned above, the distribution of these receptors was diverse, and they were not only observed at the base of epithelial cells and vascular walls, where they are relatively close to the nerve endings, but also in the glomeruli, luminal side of tubules, and/or medullary tubules where the nerves are not projected. This suggests that most of the ARs distributed in the renal effector cells receive catecholamines without direct synaptic connections with sympathetic nerve terminals. Although receptors expressed in relatively proximal effectors to nerve terminals may directly receive NE release associated with nerve firing, other ARs, which are not in close proximity to the nerve terminals, may be affected by catecholamines, including adrenaline and NE, from other sources, such as plasma and urine.
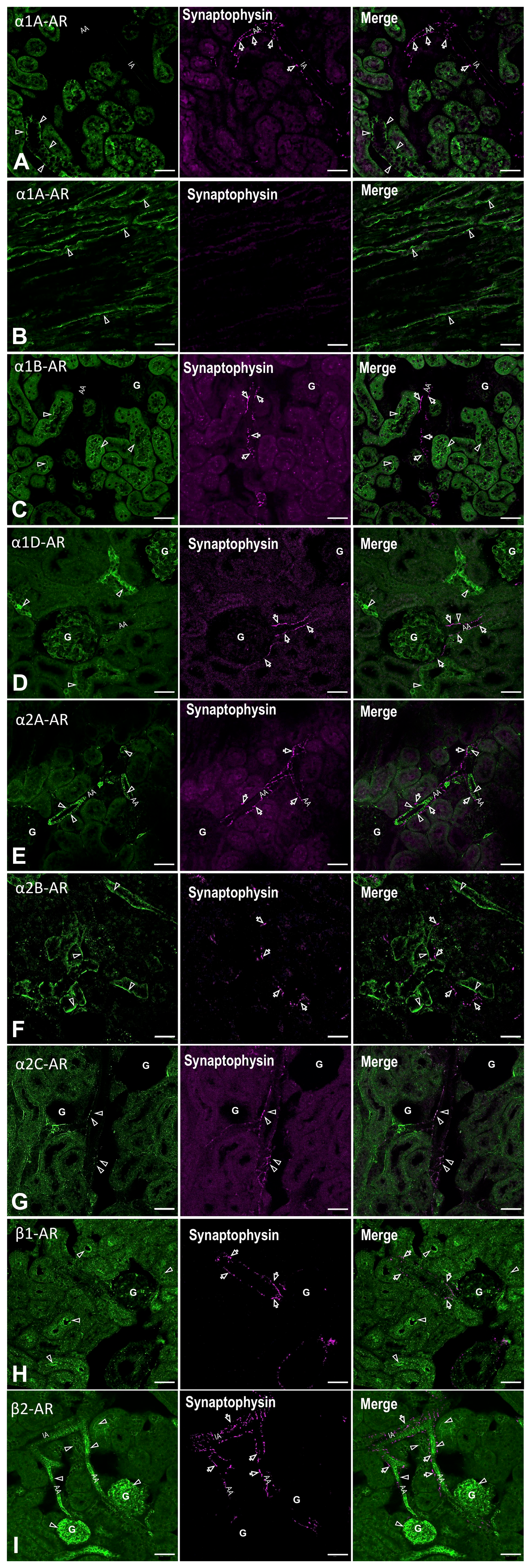
Localization of AR (green) in the rat kidneys (arrowheads). ARs are expressed in the arterioles (AA), glomeruli (G), and tubule segments with subtype-specific localization. α2A-AR (E) and β2-AR (I) are expressed in the arterioles, which are localized to synaptophysin-positive nerve terminals (magenta, arrows), whereas α1A-AR is expressed in the cortex (A) and medulla (B); α1B-AR (C), α1D-AR (D), α2B-AR (F), and β1-AR (H), which are expressed in the nephron segments and/or glomeruli, are not in direct contact with renal nerve terminals. α2C-AR (F) is colocalized with synaptophysin-positive nerve terminals. Bar = 50 μm. Abbreviations: AR, adrenergic receptor; AA, afferent arterioles; IA, interlobular artery.
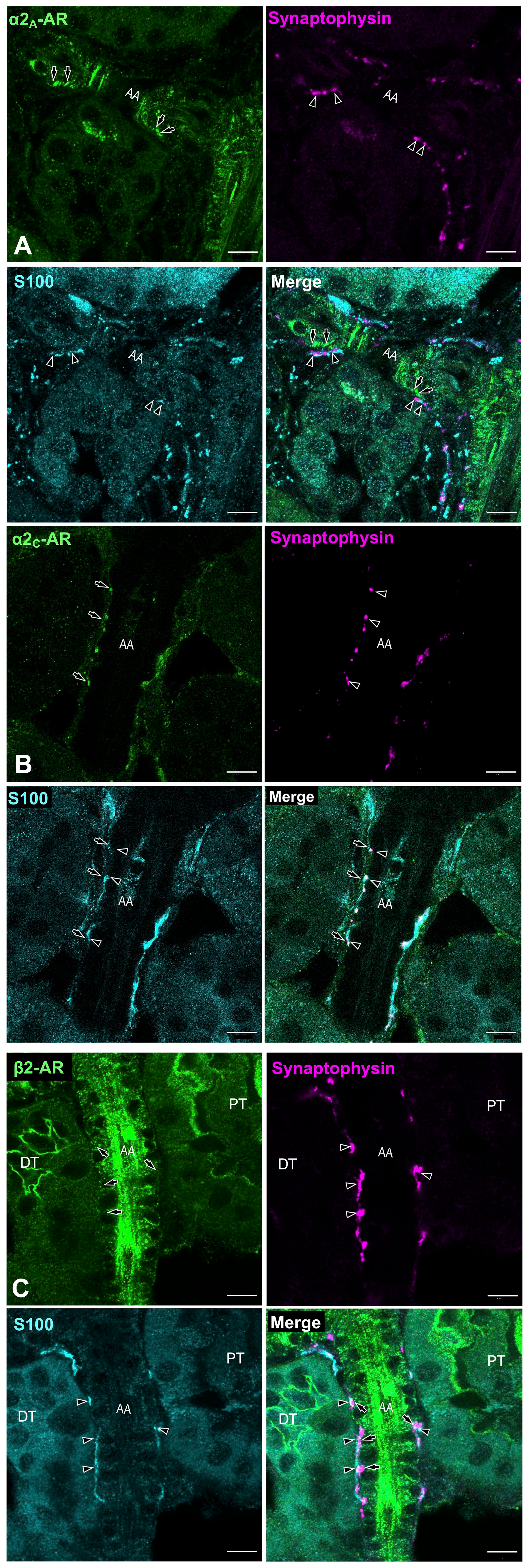
A highly magnified image of α2A-AR, α2C-AR, and β2-AR immunohistochemistry. (A) α2A-AR immunolocalization was strongly observed in arterial smooth muscles (arrow). Many synaptophysin-positive nerve endings, surrounded by S100-positive Schwann cells, are exposed through the synaptic window (arrowheads) toward the receptor expressing SMCs. (B) Highly magnified image of the localization of α2C-AR with synaptophysin immunoreactivity. α2C-AR immunolocalization (arrows) is colocalized with synaptophysin-positive varicosities covered with Schwann cell processes (arrowheads). Bar = 10 μm. (C) Higher magnification image of β2-AR immunoreactivity in the arteriole (arrows). Several synaptophysin-positive nerve terminals exposed from gaps in Schwann cell processes (arrowheads) were in close proximity to receptor-positive arteriole. β2-ARs were also found in the luminal membranes of PT and DT, which were not directly attached to the nerve terminals. Bar = 10 μm. ARs facing the nerve varicosities do not have a specific clustering localization compared with those at postsynaptic membranes. Abbreviations: AR, adrenergic receptor; SMC, smooth muscle cell; PT, proximal tubules; DT, distal tubule; AA, afferent arterioles.
Renal nerve varicosities on the arteries adhered to the basal lamina surface of smooth muscle cells, and they were sheathed with Schwann cell processes. Conversely, the varicosities next to proximal tubules, which are presumably the distal fiber terminal, were partially covered by a process; however, the terminal portion was attached to the basal lamina without any cellular support (Fig. 3B). The bare terminals were presumably attached to the basal lamina of the arteriole. Therefore, considering the mechanism of determination of the directionality of NE release from terminals, these neuroeffector junctions may bond using adhesion molecules. This suggests that the basal lamina components, including fibronectin (FN) and laminin, are involved in the formation of various neuroeffector junctions. Next, we examined the expression and localization of integrin subunits, which comprise the primary candidate adhesion molecules in rat renal tissues, via IHC. We examined integrin α subunits (α1, α2, α3, α4, and αv), and we found that only the α4 subunit was found in the renal nerves around arteries (Fig. 6; Supplemental Figs. 6 and 7). The distribution of the integrin α4 subunit was observed with respect to nerve fibers in the renal arteries (Fig. 6). The integrin α4 subunit colocalized with the reactivities of tyrosine hydroxylase in arterioles (Fig. 6A and B), suggesting that they were present on sympathetic efferent nerves. It is known that the integrin α4 subunit is coupled to form a heterodimer with integrin β1 or β7 subunits in mammals; therefore, we examined the immunohistochemical expression of these integrin β subunits in renal nerves. Integrin β1 immunoreactivity has been broadly observed in numerous renal tissue cells, including renal tubules, glomeruli, interstitium, and vasculature. In the vasculature, integrin β1 immunoreactivity was colocalized with that of integrin α4 in the adventitia (Fig. 6C). Integrin β7 immunoreactivity was primarily observed in the glomeruli, and it was slightly observed in the tubules (Supplemental Fig. 7E). No crossreactivity was observed in the negative control experiment (Supplemental Fig. 8).
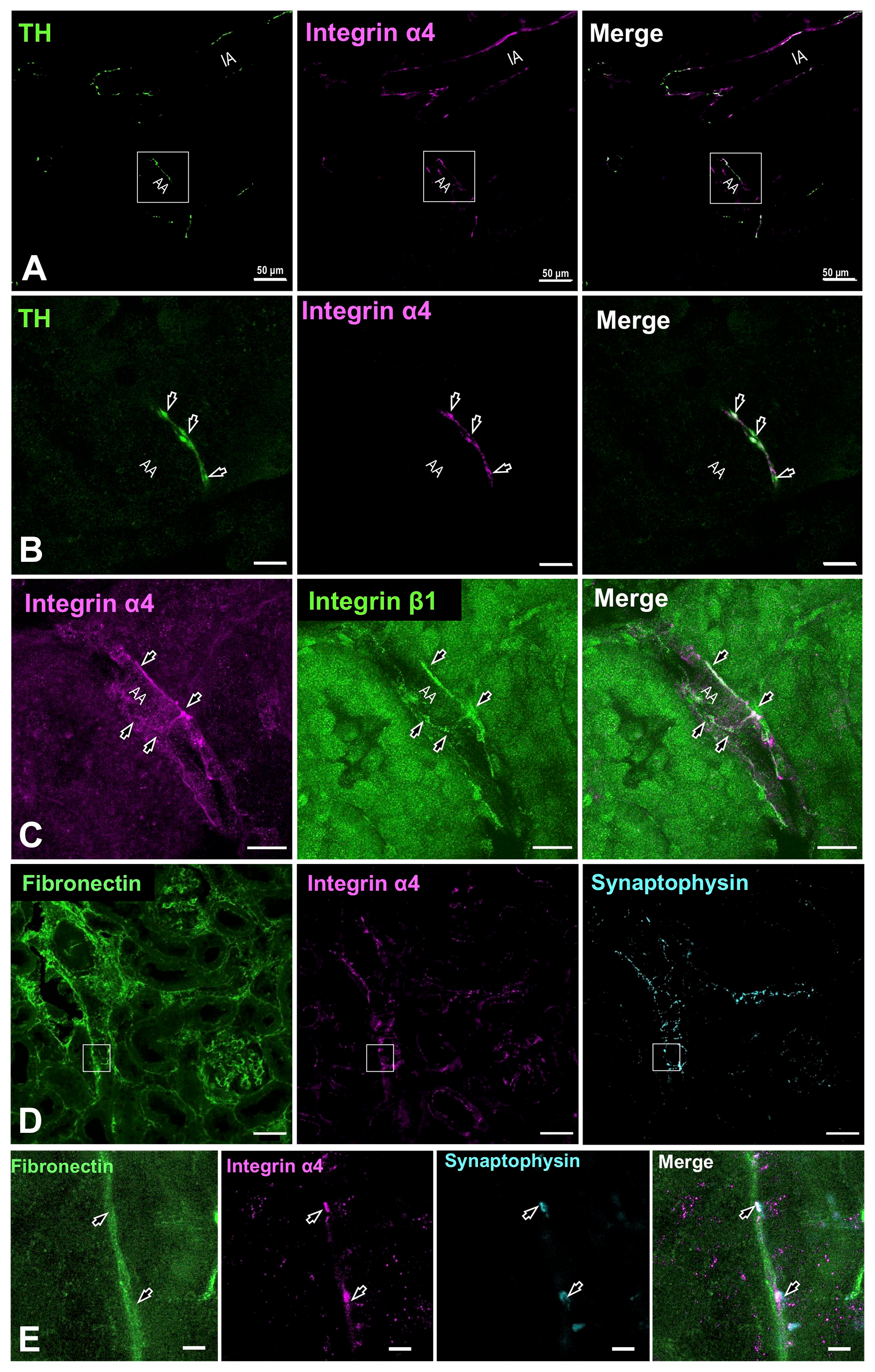
Integrin expression in rat renal nerves. (A) Integrin α4 immunoreactivity was localized to TH-positive nerve fibers. Bar = 50 μm. (B) Higher magnification of the square site in (A). TH-positive terminals were also positive to integrin α4 (arrows). Bar = 10 μm. (C) Integrin α4 was colocalized with integrin β1, which should theoretically yield a heterodimer, in synaptophysin-positive nerve varicosities (arrowheads). Bar = 20 μm. (D and E) Localization of integrin α4 and fibronectin in the renal nerve varicosities at lower (D) and higher (E) magnifications. Renal tissues were immunohistochemically stained using antibodies against fibronectin (green), integrin α4 (Magenta), and synaptophysin (cyan). Fibronectin was distributed in interstitial spaces; furthermore, integrin α4 and synaptophysin reactivities were closely localized with those of fibronectin (arrows). Bar = 50 μm (D) and 5 μm (E). Abbreviations: TH, tyrosine hydroxylase; IA, interlobular artery; AA, arteriole.
Considering that FN and vascular cell adhesion molecule-1 (VCAM-1) are known ligands for α4β1 (also known as VLA-4), we examined FN and VCAM-1 localization with integrin α4 and synaptophysin using immunohistochemistry. FN immunoreactivity was localized to the basal lamina of the interstitium of nephrons and vasculature (Fig. 6D). Triple immunohistochemical staining of FN, integrin α4, and synaptophysin revealed their close localization to the renal vasculature (Fig. 6E). In addition, VCAM-1 immunoreactivity was observed in the fibroblasts and endothelia of the arteries; however, it was not in close proximity to the nerve fibers, smooth muscle cells, or renal tubules (data not shown). These results indicate that FN is a candidate ligand of the integrin for maintaining attachment of the renal nerve terminals to the basal lamina of renal effector cells. Furthermore, these findings suggest that α4 and β1 integrins, a couple known as VLA-4, could be involved in the adhesion of renal sympathetic nerve terminals to the basal lamina of effector cells.
Discussion
Sympathetic activities such as NE release and AR responses decrease the renal blood flow and glomerular filtration rate and increase tubular reabsorption and renin secretion in juxtaglomerular cells; thus, they help regulate blood pressure in the kidneys in a wide range of effector cells.5,6 In this study, we investigated the relationship between specialized sympathetic nerve terminals compartmentalized by Schwann cell processes and the distribution of ARs expressed on effector cells activated by NE, and we examined the tripartite relationship between Schwann cells, nerve terminals, and effector cells. In the nerve terminal varicosities, synaptic vesicles and presynaptic membrane specializations were observed; however, no notable specialization was observed in the postsynaptic membrane of effector cells in skeletal muscle motor endplates.21,23,38 This study describes Schwann cell sheath, which opens windows at the attachment of nerve terminal sites facing the effector cells. These windows enable close adherence of the basal lamina of terminals to the basal lamina of effector cells. In addition, synaptic terminals appearing from the windows might attach to the basal lamina of effector cells while the terminals could attach to integrins with extracellular matrices. Therefore, the release of neurotransmitters from terminals appears to be directed toward a particular effector, especially in the renal vasculature, with scattering in other directions being avoided to a certain extent. This suggests the directional specificity of effector cells rather than simply affecting the surroundings through non-directional diffusion from the terminals.39,40
Schwann cells insulate neural activity in many types of nerve fibers. Specifically, myelinated Schwann cells are strong insulators, and they are the mainstay of fast nerve conduction. Although non-myelinated Schwann cells are thought to have an insulating effect, their function remains unclear. 31 While Remak bundles are found in postganglionic autonomic neurons and sensory neurons in the renal nerve, which is primarily composed of sympathetic neurons only, nerve fibers are formed with several axons coordinated around the cell periphery in more proximal forming mesaxons; however, they form more sparse fiber bundles in distal fibers and have a different morphology from Schwann cells in sensory fibers and NMJ terminals. 31 This axon bundle of Schwann cells in the sympathetic nerve terminals is held in place from the outside. As this is similar to radial sorting during Schwann cell development,41–44 it is suggested that the structure of sympathetic Schwann cells is somewhat primitive in appearance. Schwann cells in the autonomic postganglionic nerves have not been mentioned much in terms of functions other than axonal scaffolding. In the same efferent nerves, motor neurons and perisynaptic Schwann cells in NMJs are not involved in the myelination process. However, they could be involved in regulating transmission through the expression of various transmitter receptors rather than having only a structural role.45,46 It is necessary to discuss the possibility that sympathetic Schwann cells, which form the windows of nerve-ending varicosities, may be involved in the modulation of synaptic transmission.
In this study, we examined the distribution of AR subtypes along renal nerve terminals. Several AR subtypes, such as α2A, α2C, and β2-AR, are localized in regions where the nerve terminals are accessible, such as arterial smooth muscles and tubular basolateral membranes. However, the distribution of these ARs was not concentrated in the postsynaptic membrane facing the nerve terminals. This supports the idea that, unlike highly completed synapses, NE in the basement membranes could be fluidly traced to ARs on the effectors. 39 In vascular smooth muscles, α2A-AR and β2-AR localization was observed; however, they were coupled to G proteins with opposite functions, that is, inhibitory Gi/o and active Gs proteins for adenylate cyclase, respectively. These appear to complicate the response to NE in the kidney; however, there also appears to be an ordinal affinity for the type of ligand, catecholamine, which may modulate the response of vascular smooth muscle by varying the affinity and concentration of NE derived from intrarenal sympathetic nerve endings and systemic epinephrine from the adrenal glands. 47
On the other hand, some ARs were localized to regions without nerve distribution, such as the glomeruli and tubular luminal membrane. These results suggest that most of the ARs distributed in the renal effector cells receive catecholamines without direct synaptic connections with sympathetic nerve terminals. Although the receptors expressed in relatively proximal effectors to nerve terminals may directly receive NE release associated with nerve firing, other ARs, which are not in close proximity to the nerve terminals, may be affected by catecholamines, including adrenaline and NE, from other sources such as plasma and urine. The circulatory system of the kidney is distinctive, with primary capillaries located in the glomerulus and secondary capillaries located around the tubules. While the blood circulating in the kidney contains catecholamines, epinephrine from the adrenal glands and NE from the sympathetic nerves, which is mainly released from intrarenal sympathetic nerve terminals, act on the kidney.48,49 Thus, the amount of NE released from nerve terminals distributed among afferent arterioles would affect the stimulation of ARs within the plasma and primary urine. To supply an effective amount of NE to the glomeruli and luminal membrane of tubular epithelial cells, the neurotransmitter released from nerve terminals must flow through the arterial wall into the blood. 49 It may be possible that the nerve terminals supported by Schwann cells distributed in the arterioles ensure the provision of the necessary amount of plasma NE to access the glomerular cells and tubular luminal surface. The fact that Schwann cells, which form the release window for NE in the arterioles, are more firmly supported than varicosities, which attach to other effectors, and this may be important to ensure the amount of NE released into the plasma and primary urine to access the glomeruli and tubular luminal membrane. Another characteristic localization of ARs is that α2C-AR is locally expressed in nerve terminal varicosities with synaptophysin covered by the Schwann cell sheath (see Fig. 5). α2C-AR is a Gi/o protein that inhibits adenylate cyclase activation to regulate NE release from presynaptic terminals, and it is responsible for provision of relatively low-frequency stimuli.50,51 NE released from nerve terminals that are covered with Schwann cell processes may efficiently stimulate α2C-AR at the presynaptic membrane in a relatively closed space. In addition neurotransmitters, not only NE but also adenosine triphosphate (ATP) and neuropeptide Y, are released from the nerve terminals in sympathetic nerves.34,52–54 The expression of receptors for these transmitters in Schwann cells suggests the importance of the interaction between nerve terminal activity and Schwann cells. Notably, Schwann cell interactions with neuroeffector junctions may be required to modulate renal function. To prove this hypothesis, further studies are required to elucidate the expression and function of receptors and ion channels in renal nerve terminals and Schwann cells.
Considering that periarterial terminals are organized by Schwann cells while renal tubular terminals are free ends, the intensity of the activity of released transmitters on specific effectors in the relevant direction remains unclear. However, the direction of neurotransmitter release may be secured by several molecules assembled at the site adjacent to the synaptic cleft and actively attached to the basement membrane due to presynaptic membrane specialization. 55 Integrin is an adhesion molecule that binds to extracellular matrix molecules to determine cell alignment and construction. Numerous integrins are expressed in the kidney during nephrogenesis and renal morphogenesis56–58; however, there is limited evidence regarding mature renal nerves. In this study, integrin α4 and β1 subunits were localized to the renal sympathetic nerve fibers. VLA-4, which results from the dimerization of integrins α4 and β1, is involved in the adhesion of T- and B-lymphocytes to endothelial cells.59,60 In the peripheral nervous system, integrin α4 is involved in nerve regeneration and neurite outgrowth.16,61 Similarly, VLA-4 expression has been reported in sympathetic nerves in the heart, where it could be involved in maintaining the innervation of cardiomyocytes. 62 Therefore, integrins α4 and β1, dimerized as VLA-4, may be partly involved in terminal construction in the sympathetic nerves to the basal lamina of renal effector cells. This asymmetrical attachment may determine the direction and concentration of neurotransmitter release rather than allowing non-directional diffusion. Although there were no morphologically notable synaptic structures in the postjunctional effector membranes, various adhesion molecules and receptors may comprise unique aggregates, including those that maintain the terminals or control neurotransmitter flowability. In addition to the morphological features, including the windows of Schwann cells, the information of these molecular bases would indicate the mechanism of the activities of renal sympathetic nerves, which stimulate the complex effector responding to various ARs.
A limitation of this study is the lack of molecular evidence regarding the direct association between Schwann cells and sympathetic nerve varicosities in the kidneys. Schwann cells in sympathetic nerves accept signals from neuronal varicosities35,52,53,63,64; however, there is no evidence of physical reinforcement between Schwann cells and sympathetic axons. Furthermore, the molecular architecture involved in the formation of Schwann cell windows is unknown. Although it is conceivable that these are the primitive forms of mesaxons with extended gaps, these elegant window structures in renal sympathetic nerves indicate that Schwann cells may actively participate in nerve terminal construction. Therefore, further studies are warranted to understand the tripartite synapse structure comprising a prototypic construction between the renal nerve terminal, effector cells, and supporting Schwann cells in the kidney.
Supplemental Material
sj-docx-1-jhc-10.1369_00221554221106812 – Supplemental material for Morphology of Schwann Cell Processes Supports Renal Sympathetic Nerve Terminals With Local Distribution of Adrenoceptors
Supplemental material, sj-docx-1-jhc-10.1369_00221554221106812 for Morphology of Schwann Cell Processes Supports Renal Sympathetic Nerve Terminals With Local Distribution of Adrenoceptors by Seishi Maeda, Yusuke Minato, Sachi Kuwahara-Otani, Hiroki Yamanaka, Mitsuyo Maeda, Yosky Kataoka and Hideshi Yagi in Journal of Histochemistry & Cytochemistry
Footnotes
Acknowledgements
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
SM and H Yagi conceived and designed the research; SM, YM, and SK-O performed histochemical and TEM experiments; H Yamanaka analyzed three-dimensional data using Imaris; MM and YK examined with field emission SEM. All authors approved the final version of the manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported in part by a Grant-in-Aid for Science Research (C) from the Japan Society for Promotion of Science KAKENHI (17K08501 and 21K06415).
Compliance With Ethical Standards
All rodent experiments were performed in accordance with the animal protocols approved by the Institutional Animal Care and Use Committee of Hyogo College of Medicine (#16-001 and #16-079).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
